Abstract
Background
Breast cancer subtype, as determined by the expression of estrogen receptor (ER) and progesterone receptor (PR), together defined as hormone receptor (HR) status, and the HER2/neu receptor (HER2), is important in predicting prognosis and guiding therapy. Knowledge regarding how tumors evolve during treatment and whether subtype is influenced by neoadjuvant chemotherapy (nCT) is limited. The purpose of this study was to compare the HR and HER2 status between core needle biopsy and residual tumor after surgery of breast cancer patients treated with nCT and to evaluate the impact of status change on therapeutic management.
Methods
After institutional review board approval, we performed a retrospective review of all patients with a diagnosis of breast cancer who received nCT and had their initial biopsy and post-nCT surgical specimens evaluated for tumor subtype between January 2009 and December 2014 at our institution. Immunohistochemistry (IHC) of ER, PR, HER2, and fluorescence in situ hybridization for HER2 expression, when indicated, was performed using identical technique and measured by a single pathologist who specializes in breast pathology. Pre- and post-nCT subtype was cross-tabulated to assess change. Standard diagnostic metrics were computed.
Results
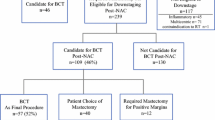
Fifty-two patients with 54 cancers were identified to have their initial biopsy and post-nCT surgical specimens evaluated for tumor subtype in identical fashion. There was a complete pathologic response after nCT in 23 cancers (42.6%). Residual disease was noted in 31 cancers (57.4%). Five of these (16.1%) had a change in tumor subtype, of which four changes were based on IHC. HR status changed from positive to negative in two cases and from negative to positive in one case. HER2 status changed from positive to negative in one case and from negative to positive in one case. Subtype change led to treatment change in all five cases, with either the addition or discontinuation of adjuvant therapies.
Conclusions
Patients with breast cancer may experience alterations in their tumor subtype after nCT. At our institution, this led to a change in adjuvant treatment in 100% of such patients. This implies that retesting receptor status of residual tumors after nCT should be routinely performed to tailor adjuvant therapy after nCT.

Similar content being viewed by others
References
Fisher B, Bryant J, Wolmark N, Mamounas E, Brown A, Fisher ER. Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol. 1998;16(8):2672–85.
Bartlett JM, Brookes CL, Robson T, van de Velde CJ, Billingham LJ, Campbell FM. Estrogen receptor and progesterone receptor as predictive biomarkers of response to endocrine therapy: a prospectively powered pathology study in the Tamoxifen and Exemestane Adjuvant Multinational trial. J Clin Oncol. 2011;29(12):1531–8.
van der Hage JA, van de Velde CJ, Julien JP, Tubiana-Hulin M, Vandervelden C, Duchateau L. Preoperative chemotherapy in primary operable breast cancer: results from the European Organization for Research and Treatment of Cancer trial 10902. J Clin Oncol. 2001;19(22):4224–37.
Mauri D, Pavlidis N, Ioannidis JP. Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst. 2005;97(3):188–94.
Chen S, Chen CM, Yu KD, Zhou RJ, Shao ZM. Prognostic value of a positive-to-negative change in hormone receptor status after neoadjuvant chemotherapy in patients with hormone receptor-positive breast cancer. Ann Surg Oncol. 2012;19(9):3002–11.
Lee SH, Chung MA, Quddus MR, Steinhoff MM, Cady B. The effect of neoadjuvant chemotherapy on estrogen and progesterone receptor expression and hormone receptor status in breast cancer. Am J Surg. 2003;186:348–50.
van de Ven S, Smit VT, Dekker TJ, Nortier JW, Kroep JR. Discordances in ER, PR, and HER2 receptors after neoadjuvant chemotherapy in breast cancer. Cancer Treat Rev. 2011;37:422–30.
Zhang N, Moran MS, Huo Q, Haffty BG, Yang Q. The hormonal receptor status in breast cancer can be altered by neoadjuvant chemotherapy: a meta-analysis. Cancer Invest. 2011;29:594–8.
Burcombe RJ, Makris A, Richman PI, Daley FM, Noble S, Pittam M. Evaluation of ER, PgR, HER-2 and Ki-67 as predictors of response to neoadjuvant anthracycline chemotherapy for operable breast cancer. Br J Cancer. 2005;92:147–55.
Shet T, Agrawal A, Chinoy R, Havaldar R, Parmar V, Badwe R. Changes in the tumor grade and biological markers in locally advanced breast cancer after chemotherapy—implications for a pathologist. Breast J. 2007;13:457–64.
Balko JM, Giltnane JM, Wang K, Schwarz LJ, Young CD, Cook RS. Molecular profiling of the residual disease of triple-negative breast cancers after neoadjuvant chemotherapy identifies actionable therapeutic targets. Cancer Discov. 2014;4(2):232–45. https://doi.org/10.1158/2159-8290.cd-13-0286. Epub 2013 Dec 19.
Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, Gronroos E, Martinez P. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366(10):883–92.
Almendro V, Cheng YK, Randles A, Itzkovitz S, Marusyk A, Ametller E. Inference of tumor evolution during chemotherapy by computational modeling and in situ analysis of genetic and phenotypic cellular diversity. Cell Rep. 2014;6(3):514–27. https://doi.org/10.1016/j.celrep.2013.12.041. Epub 2014 Jan 23.
Qin Q, Gao F, Jiang W, Tan Q, Mo Q, Wei C. Effect of neoadjuvant chemotherapy on expressions of estrogen receptor, progesterone receptor, human epidermal growth factor receptor 2, and Ki-67 in breast cancer. Chin Med J (Engl). 2014;127(18):3272–7.
Lim SK, Lee MH, Park IH, You JY, Nam BH, Kim BN. Impact of molecular subtype conversion of breast cancers after neoadjuvant chemotherapy on clinical outcome. Cancer Res Treat. 2016;48(1):133–41. https://doi.org/10.4143/crt.2014.262. Epub 2015 Apr 7.
Tacca O, Penault-Llorca F, Abrial C, Mouret-Reynier MA, Raoelfils I, Durando X, et al. Changes in and prognostic value of hormone receptor status in a series of operable breast cancer patients treated with neoadjuvant chemotherapy. Oncologist. 2007;12:636–43.
Hirata T, Shimizu C, Yonemori K, Hirakawa A, Kouno T, Tamura K, et al. Change in the hormone receptor status following administration of neoadjuvant chemotherapy and its impact on the long-term outcome in patients with primary breast cancer. Br J Cancer. 2009;101:1529–36.
De La Cruz L, Blankenship SA, Chatterjee A, Geha R, Nocera N. Outcomes after oncoplastic breast-conserving surgery in breast cancer patients: a systematic literature review. Ann Surg Oncol. 2016;23(10):3247–58. https://doi.org/10.1245/s10434-016-5313-1. Epub 2016 Jun 29.
Chatterjee A Erban JK Neoadjuvant therapy for treatment of breast cancer: the way forward, or simply a convenient option for patients? Gland Surg. 2017;6(1):119–24. https://doi.org/10.21037/gs.2016.08.07.
Li ML, Dong Y, Luan SL, Zhao ZH, Ning FL. Changes of expression of estrogen and progesterone receptors, human epithelial growth factor receptor 2 and Ki-67 after neoadjuvant chemotherapy in the treatment of breast cancer. J Biol Regul Homeost Agents. 2016;30(4):1059–65.
Author information
Authors and Affiliations
Contributions
SU conception, design, and editing/revision of manuscript. LC and SU performed data acquisition. LC drafted the initial manuscript. MH performed the statistical analyses. PJZ reviewed slides. LC, MH, SU, and PJZ assisted in critical revision of the manuscript. All authors reviewed, revised, and approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Disclosures
The authors have no financial relationships or conflicts of interest relevant to this article to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
De La Cruz, L.M., Harhay, M.O., Zhang, P. et al. Impact of Neoadjuvant Chemotherapy on Breast Cancer Subtype: Does Subtype Change and, if so, How?. Ann Surg Oncol 25, 3535–3540 (2018). https://doi.org/10.1245/s10434-018-6608-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6608-1