Abstract
Background
The primary site of a neuroendocrine neoplasms (NEN) around the head of the pancreas is sometimes difficult to assess before resection, and the characteristics of the primary site around this region have not been elucidated for recurrence after curative resection. In this study, the clinicopathologic characteristics of pancreatic NEN (PanNEN) and duodenal NEN (DuNEN) were evaluated, and the risk factors as well as the recurrence types after resection were investigated.
Methods
Consecutively diagnosed NEN patients at the authors’ hospital from January 2000 to July 2016 were evaluated in this study. For 117 PanNEN patients and 31 non-ampullary DuNEN patients, R0 resection was achieved. The median follow-up period was 8.1 years.
Results
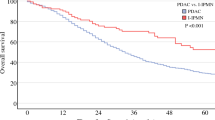
In this study, 27 PanNEN patients (23.1%) had recurrences, with a median disease-free survival (DFS) of 133 months, whereas 11 DuNEN patients (35.5%) had recurrences, with a median DFS of 156 months. The PanNEN patients tended to have primary recurrence in the liver (85.2%), followed by lymph nodes (11.1%). The independent risk factors for short DFS were lymph node metastasis at resection (p = 0.001) and microvascular invasion (p = 0.048). In contrast, the DuNEN patients were likely to have lymph node metastasis (81.8%). The independent risk factors for short DFS were lymph node metastasis at resection (p = 0.003) and large diameter (p = 0.013).
Conclusions
Most initial recurrences of PanNEN occurred in the liver, whereas those of DuNEN appeared in lymph nodes, suggesting that proper diagnosis of the primary site and appropriate imaging methods for surveillance after resection are necessary.

Similar content being viewed by others
References
Yao JC, Hassan M, Phan A, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26:3063–72.
Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97:934–59.
Pape UF, Jann H, Muller-Nordhorn J, et al. Prognostic relevance of a novel TNM classification system for upper gastroenteropancreatic neuroendocrine tumors. Cancer. 2008;113:256–65.
Jilesen AP, van Eijck CH, Busch OR, van Gulik TM, Gouma DJ, van Dijkum EJ. Postoperative outcomes of enucleation and standard resections in patients with a pancreatic neuroendocrine tumor. World J Surg. 2016;40:715–28.
Iwasaki T, Nara S, Kishi Y, Esaki M, Shimada K, Hiraoka N. Surgical treatment of neuroendocrine tumors in the second portion of the duodenum: a single-center experience and systematic review of the literature. Langenbeck’s Arch Surg. 2016.
Untch BR, Bonner KP, Roggin KK, et al. Pathologic grade and tumor size are associated with recurrence-free survival in patients with duodenal neuroendocrine tumors. J Gastrointest Surg. 2014;18:457–62; discussion 462–53.
Tsutsumi K, Ohtsuka T, Fujino M, et al. Analysis of risk factors for recurrence after curative resection of well-differentiated pancreatic neuroendocrine tumors based on the new grading classification. J Hepato-Biliary-Pancreatic Sci. 2014;21:418–25.
Verbeek WH, Korse CM, Tesselaar ME. GEP-NETs update: secreting gastro-enteropancreatic neuroendocrine tumours and biomarkers. Eur J Endocrinol. 2016;174:R1–7.
Isgro MA, Bottoni P, Scatena R. Neuron-specific enolase as a biomarker: biochemical and clinical aspects. Adv Exper Med Biol. 2015;867:125–43.
d’Herbomez M, Coppin L, Bauters C, Rouaix-Emery N, Carnaille B, Do Cao C. Biomarkers of neuroendocrine tumors. Ann Biol Clin. 2016;74:669–79.
Modlin IM, Frilling A, Salem RR, et al. Blood measurement of neuroendocrine gene transcripts defines the effectiveness of operative resection and ablation strategies. Surgery. 2016;159:336–47.
Dromain C, de Baere T, Lumbroso J, et al. Detection of liver metastases from endocrine tumors: a prospective comparison of somatostatin receptor scintigraphy, computed tomography, and magnetic resonance imaging. J Clin Oncol. 2005;23:70–8.
Scigliano S, Lebtahi R, Maire F, et al. Clinical and imaging follow-up after exhaustive liver resection of endocrine metastases: a 15-year monocentric experience. Endocrine-Related Cancer. 2009;16:977–90.
Casadei R, Ricci C, Pezzilli R, et al. Are there prognostic factors related to recurrence in pancreatic endocrine tumors? Pancreatology. 2010;10:33–8.
Yamamoto Y, Okamura Y, Uemura S, et al. Vascularity and tumor size are significant predictors for recurrence after resection of a pancreatic neuroendocrine tumor. Ann Surg Oncol. 2017.
Ge W, Zhou D, Xu S, Wang W, Zheng S. Surveillance and comparison of surgical prognosis for asymptomatic and symptomatic nonfunctioning pancreatic neuroendocrine tumors. Int J Surg. 2017;39:127–34.
Hoffmann KM, Furukawa M, Jensen RT. Duodenal neuroendocrine tumors: classification, functional syndromes, diagnosis, and medical treatment. Best Pract Res Clin Gastroenterol. 2005;19:675–97.
Hatta W, Koike T, Iijima K, et al. The risk factors for metastasis in non-ampullary duodenal neuroendocrine tumors measuring 20 mm or less in diameter. Digestion. 2017;95:201–9.
Rosentraeger MJ, Garbrecht N, Anlauf M, et al. Syndromic versus non-syndromic sporadic gastrin-producing neuroendocrine tumors of the duodenum: comparison of pathological features and biological behavior. Virchows Archiv. 2016;468:277–87.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Masui, T., Sato, A., Nakano, K. et al. Comparison of Recurrence Between Pancreatic and Duodenal Neuroendocrine Neoplasms After Curative Resection: A Single-Institution Analysis. Ann Surg Oncol 25, 528–534 (2018). https://doi.org/10.1245/s10434-017-6260-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6260-1