Abstract
Background
Inflammatory breast cancer (IBC) is an aggressive form of breast cancer characterized by rapid progression and early metastatic dissemination. The purpose of this study was to assess contemporary rates of local regional recurrence (LRR) in the era of trimodality therapy for nonmetastatic IBC and identify risk factors leading to local failure.
Methods
A total of 114 patients with nonmetastatic IBC receiving trimodality therapy (neoadjuvant chemotherapy, surgery, and radiation therapy) were identified from a prospectively collected database from 2007 to 2015 and outcomes analyzed.
Results
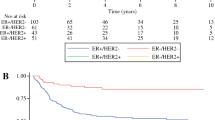
Median age at diagnosis was 52 years, and the median follow-up was 3.6 years. Sixty-three (55%) patients presented with N2 IBC, and 52 patients (45%) presented with N3 IBC. Local regional recurrence was observed during follow-up for four patients; 25 died, and 85 were censored at last follow-up. Surgical margins were negative in 99% of patients (n = 113). The 2-year probability of LRR was 3.19% (95% confidence interval 1.03–9.90%). Five-year overall survival for this cohort was 69.14%. Improvement in disease-free survival was seen among patients with HER2+ subtype, clinical stage IIIB, complete or partial radiologic response to neoadjuvant therapy, pathologic complete response, and lower nodal burden on presentation.
Conclusions
Locoregional recurrences were rare at a median of 3.6 years follow-up in a contemporary cohort of IBC patients treated with trimodality therapy. Although longer follow-up is needed, aggressive surgical resection to negative margins in the frame of trimodality therapy with curative intent can lead to LRR rates that mirror non-IBC rates.

Similar content being viewed by others
References
Dawood S, Merajver SD, Viens P, et al. International expert panel on inflammatory breast cancer: consensus statement for standardized diagnosis and treatment. Ann Oncol. 2011;22(3):515–23. doi:10.1093/annonc/mdq345.
National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (version 2.2017). www.nccn.org. Accessed 15 Jan 2017.
Wingo PA, Jamison PM, Young JL, et al. Population-based statistics for women diagnosed with inflammatory breast cancer (United States). Cancer Causes Control. 2004;15(3):321–8.
Hance KW, Anderson WF, Devesa SS, et al. Trends in inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program at the National Cancer Institute. J Natl Cancer Inst. 2005;97(13):966–75.
Droulias CA, Sewell CW, McSweeney MB, et al. Inflammatory carcinoma of the breast: a correlation of clinical, radiologic and pathologic findings. Ann Surg. 1976;184(2):217–22.
Fastenberg NA, Martin RG, Buzdar AU, et al. Management of inflammatory carcinoma of the breast. A combined modality approach. Am J Clin Oncol. 1985;8(2):134–41.
Low JA, Berman AW, Steinberg SM, et al. Long-term follow-up for locally advanced and inflammatory breast cancer patients treated with multimodality therapy. J Clin Oncol. 2004;22(20):4067–74.
Verges R, Felip E, Alastuey I, et al. Combined chemotherapy, radiotherapy and surgery in inflammatory breast carcinoma. Acta Oncol. 1995;34(1):123–4.
Fields JN, Kuske RR, Perez CA, et al. Prognostic factors in inflammatory breast cancer. Univariate and multivariate analysis. Cancer. 1989;63(6):1225–32.
Perez CA, Fields JN. Role of radiation therapy for locally advanced and inflammatory carcinoma of the breast. Oncology (Williston Park). 1987;1(1):81–94.
Rueth NM, Lin HY, Bedrosian I, et al. Underuse of trimodality treatment affects survival for patients with inflammatory breast cancer: an analysis of treatment and survival trends from the National Cancer Database. J Clin Oncol. 2014; 32(19):2018–24. doi:10.1200/JCO.2014.55.1978. Epub 2014 Jun 2.
Akay CL, Ueno NT, Chisholm GB, et al. Primary tumor resection as a component of multimodality treatment may improve local control and survival in patients with stage IV inflammatory breast cancer. Cancer. 2014;120(9):1319–28. doi:10.1002/cncr.28550. Epub 2014 Feb 7.
Miller A, Hoogstraten B, Staquet M, et al. Reporting results of cancer treatment. Cancer. 1981;47(1):207–14.
Fine JP, R.J.G. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–09.
Mordenti C, Peris K, Concetta Fargnoli M, et al. Cutaneous metastatic breast carcinoma: a study of 164 patients. Acta Dermatoven. 2000;9(4):143–8.
Northouse LL, Mood D, Kershaw T, et al. Quality of life of women with recurrent breast cancer and their family members. J Clin Oncol. 2002;20(19):4050-64.
Anscher MS, Jones P, Prosnitz LR, et al. Local failure and margin status in early-stage breast carcinoma treated with conservation surgery and radiation therapy. Ann Surg. 1993;218(1):22–8.
Huang EH, Tucker SL, Strom EA, et al. Predictors of locoregional recurrence in patients with locally advanced breast cancer treated with neoadjuvant chemotherapy, mastectomy, and radiotherapy. Int J Radiat Oncol Biol Phys. 2005;62(2):351–7.
Mamounas EP, Anderson SJ, Dignam JJ, et al. Predictors of locoregional recurrence after neoadjuvant chemotherapy: results from combined analysis of National Surgical Adjuvant Breast and Bowel Project B-18 and B-27. J Clin Oncol. 2012;30(32):3960–6. doi:10.1200/JCO.2011.40.8369. Epub 2012 Oct 1.
Curcio LD, Rupp E, Williams WL, et al. Beyond palliative mastectomy in inflammatory breast cancer: a reassessment of margin status. Ann Surg Oncol. 1999;6(3):249–54.
Boughey JC, Suman VJ, Mittendorf EA, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA. 2013;310(14):1455–61. doi:10.1001/jama.2013.278932.
Hidar S, Bibi M, Gharbi O, et al. Sentinel lymph node biopsy after neoadjuvant chemotherapy in inflammatory breast cancer. Int J Surg. 2009;7(3):272–5. doi:10.1016/j.ijsu.2009.04.012. Epub 2009 May 3.
Stearns V, Ewing CA, Slack R, et al. Sentinel lymphadenectomy after neoadjuvant chemotherapy for breast cancer may reliably represent the axilla except for inflammatory breast cancer. Ann Surg Oncol. 2002;9(3):235–42.
Sharma R, Bedrosian I, Lucci A, et al. Present-day locoregional control in patients with t1 or t2 breast cancer with 0 and 1 to 3 positive lymph nodes after mastectomy without radiotherapy. Ann Surg Oncol. 2010;17(11):2899–08. doi:10.1245/s10434-010-1089-x. Epub 2010 May 5.
Greenbaum MP, Strom EA, Allen PK, et al. Low locoregional recurrence rates in patients treated after 2000 with doxorubicin based chemotherapy, modified radical mastectomy, and post-mastectomy radiation. Radiother Oncol. 2010;95(3):312–6. doi:10.1016/j.radonc.2010.02.013. Epub 2010 Mar 11.
Warren LE, Guo H, Regan MM, et al. Inflammatory breast cancer: patterns of failure and the case for aggressive locoregional management. Ann Surg Oncol. 2015;22(8):2483–91. doi:10.1245/s10434-015-4469-4. Epub 2015 Mar 20.
Bristol IJ, Woodward WA, Strom EA, et al. Locoregional treatment outcomes after multimodality management of inflammatory breast cancer. Int J Radiat Oncol Biol Phys. 2008;72(2):474–84. doi:10.1016/j.ijrobp.2008.01.039. Epub 2008 Apr 24.
Gianni L, Eiermann W, Semiglazov V, et al. Neoadjuvant and adjuvant trastuzumab in patients with HER2-positive locally advanced breast cancer (NOAH): follow-up of a randomised controlled superiority trial with a parallel HER2-negative cohort. Lancet Oncol. 2014;15(6):640–7. doi:10.1016/S1470-2045(14)70080-4. Epub 2014 Mar 20.
Tsai CJ, Li J, Gonzalez-Angulo AM, et al. Outcomes after multidisciplinary treatment of inflammatory breast cancer in the era of neoadjuvant HER2-directed therapy. Am J Clin Oncol. 2015;38(3):242–7. doi:10.1097/COC.0b013e3182937921.
Fong Y, Gonen M, Rubin D, et al. Long-term survival is superior after resection for cancer in high-volume centers. Ann Surg. 2005;242(4):5404
Lucci A, Hall CS, Lodhi AK, et al. Circulating tumour cells in non-metastatic breast cancer: a prospective study. Lancet Oncol. 2012;13(7):688–95. doi:10.1016/S1470-2045(12)70209-7. Epub 2012 Jun 6.
Pierga JY, Bidard FC, Autret A, et al. Circulating tumour cells and pathological complete response: independent prognostic factors in inflammatory breast cancer in a pooled analysis of two multicentre phase II trials (BEVERLY-1 and -2) of neoadjuvant chemotherapy combined with bevacizumab. Ann Oncol. 2017; 28(1):103–109. doi:10.1093/annonc/mdw535.
Hall CS, Karhade M, Laubacher BA, et al. (2015) Circulating tumor cells and recurrence after primary systemic therapy in stage III inflammatory breast cancer. J Natl Cancer Inst. doi:10.1093/jnci/djv250.
Acknowledgements
This work was supported in part by the University of Texas Morgan Welch Inflammatory Breast Cancer Research Program and Clinic.
Author information
Authors and Affiliations
Corresponding author
Additional information
Wendy A. Woodward is a Co-senior author.
Rights and permissions
About this article
Cite this article
Rosso, K.J., Tadros, A.B., Weiss, A. et al. Improved Locoregional Control in a Contemporary Cohort of Nonmetastatic Inflammatory Breast Cancer Patients Undergoing Surgery. Ann Surg Oncol 24, 2981–2988 (2017). https://doi.org/10.1245/s10434-017-5952-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-5952-x