Abstract
Background
Limited data exist on the prognosis of preoperative Helicobacter pylori (H. pylori) infection in gastric adenocarcinoma (GAC).
Methods
Patients who underwent curative-intent resection for GAC from 2000 to 2012 at seven academic institutions comprising the United States Gastric Cancer Collaborative were included in the study. The primary end points of the study were overall survival (OS), recurrence-free survival (RFS), and disease-specific survival (DSS).
Results
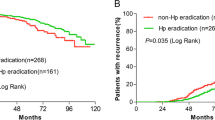
Of 559 patients, 104 (18.6 %) who tested positive for H. pylori were younger (62.1 vs 65.1 years; p = 0.041), had a higher frequency of distal tumors (82.7 vs 71.9 %; p = 0.033), and had higher rates of adjuvant radiation therapy (47.0 vs 34.9 %; p = 0.032). There were no differences in American Society of Anesthesiology (ASA) class, margin status, grade, perineural invasion, lymphovascular invasion, nodal metastases, or tumor-node-metastasis (TNM) stage. H. pylori positivity was associated with longer OS (84.3 vs 44.2 months; p = 0.008) for all patients. This relationship with OS persisted in the multivariable analysis (HR 0.54; 95 % CI 0.30–0.99; p = 0.046). H. pylori was not associated with RFS or DSS in all patients. In the stage 3 patients, H. pylori was associated with longer OS (44.5 vs 24.7 months; p = 0.018), a trend of longer RFS (31.4 vs 21.6 months; p = 0.232), and longer DSS (44.8 vs 27.2 months; p = 0.034).
Conclusions
Patients with and without preoperative H. pylori infection had few differences in adverse pathologic features at the time of gastric adenocarcinoma resection. Despite similar disease presentations, preoperative H. pylori infection was independently associated with improved OS. Further studies examining the interaction between H. pylori and tumor immunology and genetics are merited.


Similar content being viewed by others
References
Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12:354–62.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Patel SH, Kooby DA. Gastric adenocarcinoma surgery and adjuvant therapy. Surg Clin N Am. 2011;91:1039–77.
Jain VK, Cunningham D, Chau I. Preoperative and postoperative chemotherapy for gastric cancer. Surg Oncol Clin N Am. 2012;21:99–112.
Guggenheim DE, Shah MA. Gastric cancer epidemiology and risk factors. J Surg Oncol. 2013;107:230–6.
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–9.
Warren JR, Marshall BJ. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;1:1273–5.
Forman D, Newell DG, Fullerton F, et al. Association between infection with Helicobacter pylori and risk of gastric cancer: evidence from a prospective investigation. BMJ. 1991;302:1302–5.
Helicobacter and Cancer Collaborative Group. Gastric cancer and Helicobacter pylori: a combined analysis of 12 case–control studies nested within prospective cohorts. Gut. 2001;49:347–53.
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Schistosomes, liver flukes, and Helicobacter pylori. Lyon, 7–14 June 1994. IARC Monogr Eval Carcinog Risks Hum. 1994;61:1–241.
Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030–44.
Edge SB, American Joint Committee on Cancer. AJCC Cancer Staging Manual. 7th ed. New York: Springer; 2010.
Correa P. Human gastric carcinogenesis: a multistep and multifactorial process. First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992;52:6735–40.
Correa P. A human model of gastric carcinogenesis. Cancer Res. 1988;48:3554–60.
Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci USA. 1993;90:7915–22.
Lee WJ, Lin JT, Shun CT, et al. Comparison between resectable gastric adenocarcinomas seropositive and seronegative for Helicobacter pylori. Br J Surg. 1995;82:802–5.
Marrelli D, Pedrazzani C, Berardi A, et al. Negative Helicobacter pylori status is associated with poor prognosis in patients with gastric cancer. Cancer. 2009;115:2071–80.
Meimarakis G, Winter H, Assmann I, et al. Helicobacter pylori as a prognostic indicator after curative resection of gastric carcinoma: a prospective study. Lancet Oncol. 2006;7:211–22.
Kang SY, Han JH, Ahn MS, et al. Helicobacter pylori infection as an independent prognostic factor for locally advanced gastric cancer patients treated with adjuvant chemotherapy after curative resection. Int J Cancer. 2012;130:948–58.
Wang F, Sun GP, Zou YF, et al. Helicobacter pylori infection predicts favorable outcome in patients with gastric cancer. Curr Oncol. 2013;20:e388–95.
Qiu HB, Zhang LY, Keshari RP, et al. Relationship between H. pylori infection and clinicopathological features and prognosis of gastric cancer. BMC Cancer. 2010;10:374.
Li G, Wang Z, Wang Z, et al. Gastric cancer patients with Helicobacter pylori infection have a poor prognosis. J Surg Oncol. 2013;108:421–6.
Lee WJ, Lin JT, Lee WC, et al. Clinicopathologic characteristics of Helicobacter pyloric seropositive gastric adenocarcinomas. J Clin Gastroenterol. 1995;21:203–7.
Dooley CP, Cohen H, Fitzgibbons PL, et al. Prevalence of Helicobacter pylori infection and histologic gastritis in asymptomatic persons. N Engl J Med. 1989;321:1562–6.
McJunkin B, Sissoko M, Levien J, Upchurch J, Ahmed A. Dramatic decline in prevalence of Helicobacter pylori and peptic ulcer disease in an endoscopy-referral population. Am J Med. 2011;124:260–4.
Sonnenberg A, Lash RH, Genta RM. A national study of Helicobactor pylori infection in gastric biopsy specimens. Gastroenterology. 2010;139:1894–901.e1892 (quiz e1812).
Ahn HS, Lee HJ, Yoo MW, et al. Changes in clinicopathological features and survival after gastrectomy for gastric cancer over a 20-year period. Br J Surg. 2011;98:255–60.
Moayyedi P, Axon AT, Feltbower R, et al. Relation of adult lifestyle and socioeconomic factors to the prevalence of Helicobacter pylori infection. Int J Epidemiol. 2002;31:624–31.
Dowd JB, Zajacova A, Aiello A. Early origins of health disparities: burden of infection, health, and socioeconomic status in U.S. children. Soc Sci Med. 2009;68:699–707.
Ottini L, Falchetti M, Lupi R, et al. Patterns of genomic instability in gastric cancer: clinical implications and perspectives. Ann Oncol. 2006;17(Suppl 7):vii97–102.
Gu M, Kim D, Bae Y, Choi J, Kim S, Song S. Analysis of microsatellite instability, protein expression, and methylation status of hMLH1 and hMSH2 genes in gastric carcinomas. Hepatogastroenterology. 2009;56:899–904.
Fang WL, Chang SC, Lan YT, et al. Microsatellite instability is associated with a better prognosis for gastric cancer patients after curative surgery. World J Surg. 2012;36:2131–8.
Lee HS, Choi SI, Lee HK, et al. Distinct clinical features and outcomes of gastric cancers with microsatellite instability. Mod Pathol. 2002;15:632–40.
Hayden JD, Cawkwell L, Dixon MF, et al. A comparison of microsatellite instability in early onset gastric carcinomas from relatively low- and high-incidence European populations. Int J Cancer. 2000;85:189–91.
Wu MS, Lee CW, Shun CT, et al. Clinicopathological significance of altered loci of replication error and microsatellite instability-associated mutations in gastric cancer. Cancer Res. 1998;58:1494–7.
Leung WK, Kim JJ, Kim JG, Graham DY, Sepulveda AR. Microsatellite instability in gastric intestinal metaplasia in patients with and without gastric cancer. Am J Pathol. 2000;156:537–43.
Kim JJ, Tao H, Carloni E, Leung WK, Graham DY, Sepulveda AR. Helicobacter pylori impairs DNA mismatch repair in gastric epithelial cells. Gastroenterology. 2002;123:542–53.
Xue LJ, Su QS, Yang JH, Lin Y. Autoimmune responses induced by Helicobacter pylori improve the prognosis of gastric carcinoma. Med Hypotheses. 2008;70:273–6.
Rad R, Prinz C, Schmid RM. Helicobacter pylori and prognosis of gastric carcinoma. Lancet Oncol. 2006;7:364–5.
Berger C, Riddell SR. Chapter 24. Principles of immune cell therapy. In: Lichtman MA, Kipps TJ, Seligsohn U, Kaushansky K, Prchal JT, editors. Williams Hematology. 8th ed. New York: The McGraw-Hill Companies; 2010.
Chen LJ, Zheng X, Shen YP, et al. Higher numbers of T-bet(+) intratumoral lymphoid cells correlate with better survival in gastric cancer. Cancer Immunol Immunother. 2013;62:553–61.
Bamford KB, Fan X, Crowe SE, et al. Lymphocytes in the human gastric mucosa during Helicobacter pylori have a T helper cell 1 phenotype. Gastroenterology. 1998;114:482–92.
Acknowledgment
This study was supported in part by the Katz Foundation.
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Postlewait, L.M., Squires, M.H., Kooby, D.A. et al. Preoperative Helicobacter pylori Infection is Associated with Increased Survival After Resection of Gastric Adenocarcinoma. Ann Surg Oncol 23, 1225–1233 (2016). https://doi.org/10.1245/s10434-015-4953-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4953-x