Abstract
Background
Synchronous bilateral breast cancers frequently share the same estrogen receptor (ER) status, yet may differ in other histopathologic features. We sought to examine concordance rates of Oncotype DX recurrence score (RS) testing in women with synchronous bilateral ER-positive breast cancer.
Methods
Institutional databases were reviewed to identify patients with synchronous (within 6 months) bilateral primary invasive breast cancer and multiple RSs. RSs were stratified by risk group (RS < 18, low; RS 18–30, intermediate; RS ≥ 31, high) and considered discordant if they reflected different risk groups.
Results
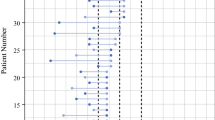
From 2005–2014, a total of 115 patients presented with synchronous bilateral invasive breast cancer; 43 (37 %) had two RSs available. Median patient age was 60 years (42–84), median tumor size was 1.2 cm (0.5–3.7), and all cases were HER2 negative and node negative. Of 86 RSs, 63 (73 %) were low risk, 20 (23 %) were intermediate risk, and 3 (3 %) were high risk. RSs were concordant in 29 (67 %) patients. Patients with concordant RSs were older (62 years vs. 56 years) and had median levels of progesterone receptor (PR) expression that were higher and more similar—80 and 85 % for bilateral cancers, respectively, compared with 55 and 75 % for bilateral cancers in discordant cases. Discordant RS led to a treatment change in 8/14 (57 %) cases.
Conclusions
Among women with synchronous bilateral ER-positive HER2-negative breast cancer, RSs were concordant in 67 % of cases. Concordance rates may be higher in older women or among those with comparable levels of PR expression. These data suggest that testing of both tumors should be considered in patients who are candidates for adjuvant chemotherapy.
Similar content being viewed by others
References
Goldstein LJ, Gray R, Badve S, et al. Prognostic utility of the 21-gene assay in hormone receptor-positive operable breast cancer compared with classical clinicopathologic features. J Clin Oncol. 2008;26(25):4063–71.
Harris L, Fritsche H, Mennel R, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol. 2007;25(33):5287–312.
Paik S, Shak S, Tang G, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351(27):2817–26.
Albain KS, Paik S, van’tVeer L. Prediction of adjuvant chemotherapy benefit in endocrine responsive, early breast cancer using multigene assays. Breast. 2009;18(Suppl 3):S141–5.
Carlson JJ, Roth JA. The impact of the Oncotype Dx breast cancer assay in clinical practice: a systematic review and meta-analysis. Breast Cancer Res Treat. 2013;141(1):13–22.
Tang G, Shak S, Paik S, et al. Comparison of the prognostic and predictive utilities of the 21-gene Recurrence Score assay and Adjuvant! for women with node-negative, ER-positive breast cancer: results from NSABP B-14 and NSABP B-20. Breast Cancer Res Treat. 2011;127(1):133–42.
Gollamudi SV, Gelman RS, Peiro G, et al. Breast-conserving therapy for stage I–II synchronous bilateral breast carcinoma. Cancer. 1997;79(7):1362–9.
Kollias J, Ellis IO, Elston CW, et al. Clinical and histological predictors of contralateral breast cancer. Eur J Surg Oncol. 1999;25(6):584–9.
Toole MJ, Kidwell KM, Van Poznak C. Oncotype dx results in multiple primary breast cancers. Breast Cancer (Auckl). 2014;8:1–6.
Kilgore AR. The incidence of cancer in the second breast. JAMA. 1921;77:454–7.
Haagensen CD. The physiology of the breast as it concerns the clinician. Am J Obstet Gynecol. 1971;109(2):206–9.
Schwentner L, Wolters R, Wischnewsky M, et al. Survival of patients with bilateral versus unilateral breast cancer and impact of guideline adherent adjuvant treatment: a multi-centre cohort study of 5292 patients. Breast. 2012;21(2):171–7.
Hartman M, Czene K, Reilly M, et al. Incidence and prognosis of synchronous and metachronous bilateral breast cancer. J Clin Oncol. 2007;25(27):4210–6.
Holm M, Tjonneland A, Balslev E, et al. Prognosis of synchronous bilateral breast cancer: a review and meta-analysis of observational studies. Breast Cancer Res Treat. 2014;146(3):461–75.
de la Rochefordiere A, Asselain B, Scholl S, et al. Simultaneous bilateral breast carcinomas: a retrospective review of 149 cases. Int J Radiat Oncol Biol Phys. 1994;30(1):35–41.
Irvine T, Allen DS, Gillett C, et al. Prognosis of synchronous bilateral breast cancer. Br J Surg. 2009;96(4):376–80.
Nichol AM, Yerushalmi R, Tyldesley S, et al. A case-match study comparing unilateral with synchronous bilateral breast cancer outcomes. J Clin Oncol. 2011;29(36):4763–8.
Schmid SM, Pfefferkorn C, Myrick ME, et al. Prognosis of early-stage synchronous bilateral invasive breast cancer. Eur J Surg Oncol. 2011;37(7):623–8.
Imyanitov EN, Suspitsin EN, Grigoriev MY, et al. Concordance of allelic imbalance profiles in synchronous and metachronous bilateral breast carcinomas. Int J Cancer. 2002;100(5):557–64.
Janschek E, Kandioler-Eckersberger D, Ludwig C, et al. Contralateral breast cancer: molecular differentiation between metastasis and second primary cancer. Breast Cancer Res Treat. 2001;67(1):1–8.
Shibata A, Tsai YC, Press MF, et al. Clonal analysis of bilateral breast cancer. Clin Cancer Res. 1996;2(4):743–8.
Chen JJ, Wang Y, Xue JY, et al. A clinicopathological study of early-stage synchronous bilateral breast cancer: a retrospective evaluation and prospective validation of potential risk factors. PLoS One. 2014;9(4):e95185.
Hartman M, Czene K, Reilly M, et al. Genetic implications of bilateral breast cancer: a population based cohort study. Lancet Oncol. 2005;6(6):377–82.
Jobsen JJ, van der Palen J, Ong F, et al. Synchronous, bilateral breast cancer: prognostic value and incidence. Breast. 2003;12(2):83–8.
Buggi F, Folli S, Curcio A, et al. Multicentric/multifocal breast cancer with a single histotype: is the biological characterization of all individual foci justified? Ann Oncol. 2012;23(8):2042–6.
Saad RS, Denning KL, Finkelstein SD, et al. Diagnostic and prognostic utility of molecular markers in synchronous bilateral breast carcinoma. Mod Pathol. 2008;21(10):1200–7.
Coradini D, Oriana S, Mariani L, et al. Is steroid receptor profile in contralateral breast cancer a marker of independence of the corresponding primary tumour? Eur J Cancer. 1998;34(6):825–30.
Hungness ES, Safa M, Shaughnessy EA, et al. Bilateral synchronous breast cancer: mode of detection and comparison of histologic features between the 2 breasts. Surgery. 2000;128(4):702–7.
Acknowledgment
This study was funded in part through National Institutes of Health/National Cancer Institute Cancer Center Support Grant P30 CA008748, and supported in part by the Breast Cancer Alliance.
Disclosure
Maria Karsten, Michelle Stempel, Julia Radosa, Sujata Patil, and Tari A. King have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karsten, M., Stempel, M., Radosa, J. et al. Oncotype DX in Bilateral Synchronous Primary Invasive Breast Cancer. Ann Surg Oncol 23, 471–476 (2016). https://doi.org/10.1245/s10434-015-4841-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4841-4