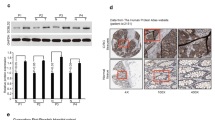
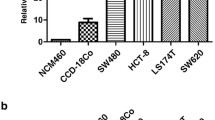
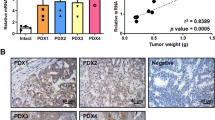
Abstract
Background
To assess the correlations and functions of complement C1r/C1s, Uegf, Bmp1 domain-containing protein-1 (CDCP1) in identifying colorectal cancer (CRC) patients who are at high risk for metastasis.
Methods
Tumor specimens from 101 patients were analyzed by real-time polymerase chain reaction to detect CDCP1 expression. CDCP1 expression plasmids and shRNA were used to knock down CDCP1 expression in this study to investigate migratory and invasive abilities by Boyden chambers. The mRNA expression profiles in shCDCP1 transfectants were compared to those in control cells by conducting microarray analysis. Its downstream effectors were also invested in this study.
Results
CRC patients with a high CDCP1 expression had a statistically significant lower overall survival and disease-free survival compared to those exhibiting low CDCP1 expression. In vitro, knock-down CDCP1 expression significantly decreased migratory and invasive abilities in HCT116. Aberrant expression of CDCP1 increased cancer cell migration and invasion. By using integrated genomics, we identified ROCK1 (rho-associated, coiled-coil-containing protein kinase 1 pseudogene 1) as a downstream effector in CDCP1-mediated migration and as an invasion mediator. Clinically, ROCK1 and CDCP1 mRNA expression exhibited a strong positive correlation in CRC patient samples.
Conclusions
Our results implicated CDCP1 as a key regulator of CRC migration and invasion, and suggest that it is a useful prognostic factor for patients with CRC. Improved identification of a high-risk subset of early metastatic patients may guide indications of individualized treatment in clinical practice.




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics. CA Cancer J Clin. 2009;59:225–49.
Cheung ST, Chen X, Guan XY, et al. Identify metastasis-associated genes in hepatocellular carcinoma through clonality delineation for multinodular tumor. Cancer Res. 2002;62:4711–21.
Uekita T, Sakai R. Roles of CUB domain-containing protein 1 signaling in cancer invasion and metastasis. Cancer Sci. 2011;102:1943–8.
Scherl-Mostageer M, Sommergruber W, Abseher R, Hauptmann R, Ambros P, Schweifer N. Identification of a novel gene, CDCP1, overexpressed in human colorectal cancer. Oncogene. 2001;20:4402–8.
Wortmann A, He Y, Deryugina EI, Quigley JP, Hooper JD. The cell surface glycoprotein CDCP1 in cancer—insights, opportuniites, and challenges. IUBMB Life. 2009;61:723–30.
Bühring HJ, Kuçi S, Conze T, et al. CDCP1 identifies a broad spectrum of normal and malignant stem/progenitor cell subsets of hematopoietic and nonhematopoietic origin. Stem Cells. 2004;22:334–43.
Hooper JD, Zijlstra A, Aimes RT, et al. Subtractive immunization using highly metastatic human tumor cells identifies SIMA135/CDCP1, a 135 kDa cell surface phosphorylated glycoprotein antigen. Oncogene. 2003;22:1783–94.
Brown TA, Yang TM, Zaitsevskaia T, et al. Adhesion or plasmin regulates tyrosine phosphorylation of a novel membrane glycoprotein p80/gp140/CUB domain-containing protein 1 in epithelia. J Biol Chem. 2004;279:14772–83.
Bhatt AS, Erdjument-Bromage H, Tempst P, Craik CS, Moasser MM. Adhesion signaling by a novel mitotic substrate of src kinases. Oncogene. 2005;24:5333–43.
Spassov DS, Baehner FL, Wong CH, McDonough S, Moasser MM. The transmembrane src substrate trask is an epithelial protein that signals during anchorage deprivation. Am J Pathol. 2009;174:1756–65.
Spassov DS, Ahuja D, Wong CH, and Moasser MM. The structural features of trask that mediate its anti-adhesive functions. PLoS One. 2011;6:e19154.
Wong CH, Baehner FL, Spassov DS, et al. Phosphorylation of the src epithelial substrate trask is tightly regulated in normal epithelia but widespread in many human epithelial cancers. Clin Cancer Res. 2009;15:2311–22.
Ikeda JI, Oda T, Inoue M, et al. Expression of CUB domain containing protein (CDCP1) is correlated with prognosis and survival of patients with adenocarcinoma of lung. Cancer Sci. 2009;100:429–33.
Uekita T, Jia L, Narisawa-Saito M, Yokota J, Sakai R. CUB domain-containing protein 1 is a novel regulator of anoikis resistance in lung adenocarcinoma. Mol Cell Biol. 2007;27:7649–60.
Perry SE, Robinson P, Melcher A, et al. Expression of the CUB domain containing protein 1 (CDCP1) gene in colorectal tumour cells. FEBS Lett. 2007;581:1137–42.
Awakura Y, Nakamura E, Takahashi T, et al. Microarray-based identification of CUB-domain containing protein 1 as a potential prognostic marker in conventional renal cell carcinoma. J Cancer Res Clin Oncol. 2008;134:1363–9.
Mamat S, Ikeda JI, Enomoto T, et al. Prognostic significance of CUB domain containing protein expression in endometrioid adenocarcinoma. Oncol Rep. 2010;23:1221–7.
Lin BR, Huang MT, Chen ST, et al. Prognostic significance of TWEAK expression in colorectal cancer and effect of its inhibition on invasion. Ann Surg Oncol. 2012;19:S385–94.
Marsters SA, Sheridan JP, Pitti RM, Brush J, Goddard A, Ashkenazi A. Identification of a ligand for the death-domain-containing receptor Apo3. Curr Biol. 1998;8:525–8.
Bühring HJ, Kuçi S, Conze T, et al. CDCP1 identifies a broad spectrum of normal and malignant stem/progenitor cell subsets of hematopoietic and nonhematopoietic origin. J Biol Chem. 2011;286:42303–15.
Uekita T, Tanaka M, Takigahira M, et al. CUB domain–containing protein 1 regulates peritoneal dissemination of gastric scirrhous carcinoma. Am J Pathol. 2008;172:1729–39.
Miyazawa Y, Uekita T, Hiraoka N, et al. CUB domain–containing protein 1, a prognostic factor for human pancreatic cancers, promotes cell migration and extracellular matrix degradation. Cancer Res. 2010;70:5136–46.
Liu H, Ong SE, Badu-Nkansah K, Schindler J, White FM, Hynes RO. CUB-domain-containing protein 1 (CDCP1) activates Src to promote melanoma metastasis. Proc Natl Acad Sci U S A. 2011;108:1379–84.
Gao W, Chen L, Ma Z, et al. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology. 2013;145:636–46.
Gupta GP, Nguyen DX, Chiang AC, et al. Mediators of vascular remodelling co-opted for sequential steps in lung metastasis. Nature. 2007;446:765–70.
Ding Q, Chang CJ, Xie X, et al. APOBEC3G promotes liver metastasis in an orthotopic mouse model of colorectal cancer and predicts human hepatic metastasis. J Clin Invest. 2011;121:4526–36.
Satoh K, Fukumoto Y, Shimokawa H. Rho-kinase: important new therapeutic target in cardiovascular diseases. Am J Physiol Heart Circ Physiol. 2011;301:H287–96.
Shimokawa H, Rashid M. Development of Rho-kinase inhibitors for cardiovascular medicine. Trends Pharmacol Sci. 2007;28:296–302.
Lin G, Craig GP, Zhang L, et al. Acute inhibition of Rho-kinase improves cardiac contractile function in streptozotocin-diabetic rats. Cardiovasc Res. 2007;75:51–8.
Kobayashi N, Horinaka S, Mita S, et al. Critical role of Rho-kinase pathway for cardiac performance and remodeling in failing rat hearts. Cardiovasc Res. 2002;55:757–67.
Bao W, Hu E, Tao L, et al. Inhibition of Rho-kinase protects the heart against ischemia/reperfusion injury. Cardiovasc Res. 2004;61:548–58.
Wolfrum S, Dendorfer A, Rikitake Y, et al. Inhibition of Rho-kinase leads to rapid activation of phosphatidylinositol 3-kinase/protein kinase Akt and cardiovascular protection. Arterioscler Thromb Vasc Biol. 2004;24:1842–7.
Noma K, Oyama N, Liao JK. Physiological role of ROCKs in the cardiovascular system. Am J Physiol Cell Physiol. 2006;290:C661–8.
Surma M, Wei L, Shi J. Rho kinase as a therapeutic target in cardiovascular disease. Future Cardiol. 2011;7:657–71.
Hall A. Rho GTPases and the actin cytoskeleton. Science. 1998;279:509–14.
Wan X, Cheng Q, Peng R, et al. ROCK1, a novel target of miR-145, promotes glioma cell invasion. Mol Med Rep. 2014;9:1877–82.
Brown M, Roulson JA, Hart CA, Tawadros T, Clarke NW, Arachidonic acid induction of Rho-mediated transendothelial migration in prostate cancer. Br J Cancer. 2014;110:2099–108.
Acknowledgment
This work was supported by Grants from National Science Council, Taiwan (NSC 99-2314-B-002-083-MY3), National Taiwan University Hospital (NTUH.100-S1508), and Chang Gung Medical Research Foundation, Taiwan (CMRPF6A0043, CMRPF6C0061 and CMRPF6C0062). We thank Dr. Zee-Fen Chang (Institute of Biochemistry and Molecular Biology, College of Medicine and National Yang-Ming University, Taipei, Taiwan, R.O.C.) for providing the ROCK1 plasmid and antibody.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Equal contribution
Yue-Ju Li and Cheng-Chi Chang contributed equally to this article
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chou, CT., Li, YJ., Chang, CC. et al. Prognostic Significance of CDCP1 Expression in Colorectal Cancer and Effect of Its Inhibition on Invasion and Migration. Ann Surg Oncol 22, 4335–4343 (2015). https://doi.org/10.1245/s10434-015-4505-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4505-4