Abstract
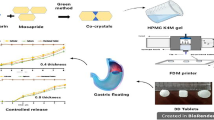
The high-drug-loaded sustained-release gastric-floating clarithromycin (CAM) tablets were proposed and manufactured via semisolid extrusion (SSE)-based 3D printing. The physical and mechanical properties, such as dimensions, weight variation, friability, and hardness, were accessed according to the quality standards of Chinese Pharmacopoeia (Ch.P). The interactions among the drug-excipients were evaluated via differential scanning calorimetry (DSC), Fourier-transform infrared spectroscopy (FTIR), and X-ray diffraction (XRD) techniques. Next, the rheological properties of the paste and the effect of the excipients and solvents were evaluated. Finally, a very high drug-loading of up to 81.7% (w/w) with the sustain release time of 8 h (125 mg) and 12 h (250 mg) was achieved. The results revealed the potential of SSE for achieving a high drug loading and identified the suitable properties of the paste for SSE-based 3D printing.







Similar content being viewed by others
References
Zhu X, Li H, Huang L, Zhang M, Fan W, Cui L. 3D printing promotes the development of drugs. Biomed Pharmacother. 2020;131:110644.
Elkasabgy NA, Mahmoud AA, Maged A. 3D printing: An appealing route for customized drug delivery systems. Int J Pharm. 2020;588:119732.
S. K, A. N, N. A. 3D printing technology in drug delivery: Recent progress and application. Curr Pharm Des. 2018;24:5039–48.
Herrada-Manchón H, Rodríguez-González D, Alejandro Fernández M, Suñé-Pou M, Pérez-Lozano P, García-Montoya E, et al. 3D printed gummies: Personalized drug dosage in a safe and appealing way. Int J Pharm. 2020;587:119687.
Lim SH, Kathuria H, Tan JJY, Kang L. 3D printed drug delivery and testing systems — a passing fad or the future? Adv Drug Deliv Rev. 2018;132:139–68.
Awad A, Trenfield SJ, Goyanes A, Gaisford S, Basit AW. Reshaping drug development using 3D printing. Drug Discov Today. 2018;23:1547–55.
Alhnan MA, Okwuosa TC, Sadia M, Wan KW, Ahmed W, Arafat B. Emergence of 3D Printed Dosage Forms: Opportunities and Challenges. Pharm Res. 2016;33:1817–32.
Pietrzak K, Isreb A, Alhnan MA. A flexible-dose dispenser for immediate and extended release 3D printed tablets. Eur J Pharm Biopharm. 2015;96:380–7.
Cui M, Pan H, Fang D, Qiao S, Wang S, Pan W. Fabrication of high drug loading levetiracetam tablets using semi-solid extrusion 3D printing. J Drug Deliv Sci Technol. 2020;57:101683.
Goyanes A, Buanz ABM, Hatton GB, Gaisford S, Basit AW. 3D printing of modified-release aminosalicylate (4-ASA and 5-ASA) tablets. Eur J Pharm Biopharm. 2015;89:157–62.
Khaled SA, Alexander MR, Wildman RD, Wallace MJ, Sharpe S, Yoo J, et al. 3D extrusion printing of high drug loading immediate release paracetamol tablets. Int J Pharm. 2018;538:223–30.
Tagami T, Ando M, Nagata N, Goto E, Yoshimura N, Takeuchi T, et al. Fabrication of Naftopidil-Loaded Tablets Using a Semisolid Extrusion-Type 3D Printer and the Characteristics of the Printed Hydrogel and Resulting Tablets. J Pharm Sci. 2019;108:907–13.
Krause J, Bogdahn M, Schneider F, Koziolek M, Weitschies W. Design and characterization of a novel 3D printed pressure-controlled drug delivery system. Eur J Pharm Sci. 2019;140:105060.
Fu J, Yin H, Yu X, Xie C, Jiang H, Jin Y, et al. Combination of 3D printing technologies and compressed tablets for preparation of riboflavin floating tablet-in-device (TiD) systems. Int J Pharm. 2018;549:370–9.
Chen N, Niu J, Li Q, Li J, Chen X, Ren Y, et al. Development and evaluation of a new gastroretentive drug delivery system: Nanomicelles-loaded floating mucoadhesive beads. J Drug Deliv Sci Technol. 2019;51:485–92.
Mandal UK, Chatterjee B, Senjoti FG. Gastro-retentive drug delivery systems and their in vivo success: A recent update. Asian J Pharm Sci. 2016;11:575–84.
Ghosh Dastidar D, Saha S, Chowdhury M. Porous microspheres: Synthesis, characterisation and applications in pharmaceutical & medical fields. Int J Pharm. 2018;548:34–48.
Murphy C, Pillay V, Choonara Y, du Toit L. Gastroretentive Drug Delivery Systems: Current Developments in Novel System Design and Evaluation. Curr Drug Deliv. 2009;6:451–60.
Devi P, Rathor S, Sharma P, Sen J, Kaur H, Singh J. Development of novel gastroretentive salbutamol sulfate-loaded sodium alginate-pectin bubble beads prepared by co-axial needle air-injection method and in vivo clinical evaluation by ultrasound studies. Eur J Pharm Sci. 2018;122:359–73.
Abou Youssef NAH, Kassem AA, El-Massik MAE, Boraie NA. Development of gastroretentive metronidazole floating raft system for targeting Helicobacter pylori. Int J Pharm. 2015;486:297–305.
Hao S, Wang Y, Wang B, Zou Q, Zeng H, Chen X, et al. A novel gastroretentive porous microparticle for anti-Helicobacter pylori therapy: Preparation, in vitro and in vivo evaluation. Int J Pharm. 2014;463:10–21.
Yao X, Smolka AJ. Gastric Parietal Cell Physiology and Helicobacter pylori–Induced Disease. Gastroenterology. 2019;156:2158–73.
Malladi M, Jukanti R. Formulation development and evaluation of a novel bi-dependent clarithromycin gastroretentive drug delivery system using Box-Behnken design. J Drug Deliv Sci Technol. 2016;35:134–45.
Smoke T. & S. IARC monographs on the evaluation of carcinogenic risks to humans. IARC Monogr Eval Carcinog Risks to Humans. 2010;93:9–38.
Fallone CA, Moss SF, Malfertheiner P. Reconciliation of Recent Helicobacter pylori Treatment Guidelines in a Time of Increasing Resistance to Antibiotics. Gastroenterology. 2019;157:44–53.
Eslami M, Yousefi B, Kokhaei P, Jazayeri Moghadas A, Sadighi Moghadam B, Arabkari V, et al. Are probiotics useful for therapy of Helicobacter pylori diseases? Comp Immunol Microbiol Infect Dis. 2019;64:99–108.
Alkim H, Koksal AR, Boga S, Sen I, Alkim C. Role of bismuth in the eradication of helicobacter pylori. Am J Ther. 2016;24:751–7.
Peng C, Hu Y, Ge Z-M, Zou Q-M, Lyu N-H. Diagnosis and treatment of Helicobacter pylori infections in children and elderly populations. Chronic Dis Transl Med. 2019;5:243–51.
Lopes CM, Bettencourt C, Rossi A, Buttini F, Barata P. Overview on gastroretentive drug delivery systems for improving drug bioavailability. Int J Pharm. 2016;510:144–58.
Chueh HR, Zia H, Rhodes CT. Optimization of sotalol floating and bioadhesive extended release tablet formulations. Drug Dev Ind Pharm. 1995;21:1725–47.
Huanbutta K, Sangnim T. Design and development of zero-order drug release gastroretentive floating tablets fabricated by 3D printing technology. J Drug Deliv Sci Technol. 2019;52:831–7.
Rapolu K, Sanka K, Vemula PK, Aatipamula V, Mohd AB, Diwan PV. Optimization and characterization of gastroretentive floating drug delivery system using Box-Behnken design. Drug Dev Ind Pharm. 2013;39:1928–35.
Prasanthi CH, Prasanthi NL, Manikiran SS, Rama RN. Focus on current trends in the treatment of helicobacter pylori infection: An update. Int J Pharm Sci Rev Res. 2011;9:42–51.
Kumar R, Philip A. Gastroretentive dosage forms for prolonging gastric residence time. Int J Pharm Med. 2007;21:157–71.
Kim JY, Bae HJ, Choi J, Lim JR, Kim SW, Lee SH, et al. Efficacy of gastro-retentive forms of ecabet sodium in the treatment of gastric ulcer in rats. Arch Pharm Res. 2014;37:1053–62.
Aoki H, Iwao Y, Mizoguchi M, Noguchi S, Itai S. Clarithromycin highly-loaded gastro-floating fine granules prepared by high-shear melt granulation can enhance the efficacy of Helicobacter pylori eradication. Eur J Pharm Biopharm. 2015;92:22–7.
Adebisi AO, Laity PR, Conway BR. Formulation and evaluation of floating mucoadhesive alginate beads for targeting Helicobacter pylori. J Pharm Pharmacol. 2015;67:511–24.
Charoenying T, Patrojanasophon P, Ngawhirunpat T, Rojanarata T, Akkaramongkolporn P, Opanasopit P. Fabrication of floating capsule-in- 3D-printed devices as gastro-retentive delivery systems of amoxicillin. J Drug Deliv Sci Technol. 2020;55:101393.
Jiao D, Shi C, Yuan Q, An X, Liu Y, Li H. Effect of constituents on rheological properties of fresh concrete-A review. Cem Concr Compos. 2017;83:146–59.
Jeong GY, Bak JH, Yoo B. Physical and rheological properties of xanthan gum agglomerated in fluidized bed: Effect of HPMC as a binder. Int J Biol Macromol. 2019;121:424–8.
Englert C, Brendel JC, Majdanski TC, Yildirim T, Schubert S, Gottschaldt M, et al. Pharmapolymers in the 21st century: Synthetic polymers in drug delivery applications. Prog Polym Sci. 2018;87:107–64.
Bala R, Khanna S, Pawar P. Polymers in fast disintegrating tablets - A review. Asian J Pharm Clin Res. 2012;5:8–14.
Mohanachandran PS, Sindhumol PG, Kiran TS. Superdisintegrants: An overview. Int J Pharm Sci Rev Res. 2011;6:105–9.
Chang CC, Boland ED, Williams SK, Hoying JB. Direct-write bioprinting three-dimensional biohybrid systems for future regenerative therapies. J Biomed Mater Res - Part B Appl Biomater. 2011;98(B):160–70.
Skardal A, Atala A. Biomaterials for Integration with 3-D Bioprinting. Ann Biomed Eng. 2015;43:730–46.
Patra S, Young V. A Review of 3D Printing Techniques and the Future in Biofabrication of Bioprinted Tissue. Cell Biochem Biophys. 2016;74:93–8.
Elbadawi M, Gustaffson T, Gaisford S, Basit AW. 3D printing tablets: Predicting printability and drug dissolution from rheological data. Int J Pharm. 2020;590:119868.
Channer KS, Virjee JP. The effect of size and shape of tablets on their esophageal transit. J Clin Pharmacol. 1986;26:141–6.
Karst KR. A hard pill to swallow? in transit for a couple of years, FDA finally coughs up draft guidance on physical attributes of generic tablets and capsules. [Internet]. [cited 2018 Feb 28]. Available from: http://www.fdalawblog.net/2013/12/a-hard-pill-to-swallow-in-transit-for-a-couple-of-years-fda-finally-coughs-up-draft-guidance-on-phys/.
Inukai K, Takiyama K, Noguchi S, Iwao Y, Itai S. Effect of gel formation on the dissolution behavior of clarithromycin tablets. Int J Pharm. 2017;521:33–9.
Wu S, Ahmad Z, Li JS, Chang MW. Fabrication of flexible composite drug films via foldable linkages using electrohydrodynamic printing. Mater Sci Eng C. 2020;108:110393.
Acknowledgements
This work was supported by Guangdong “Climbing” Program for Undergraduates (Grant Number PDJH2020a0298); Guangdong Graduate Education Innovation Program: Project Funded by Guangdong Province Joint Training Graduate Demostration Base (Guangzhou General Pharmaceutical Research Institute Co., Ltd.) Guangdong Pharmaceutical University “Innovation and Enhancing Project”; and Guangzhou Science and Technology Program key projects(Grant Number 20170402199, 201903010002).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, P., Luo, H., Huang, S. et al. Preparation of High-Drug-Loaded Clarithromycin Gastric-Floating Sustained-Release Tablets Using 3D Printing. AAPS PharmSciTech 22, 131 (2021). https://doi.org/10.1208/s12249-021-01994-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-021-01994-z