Abstract
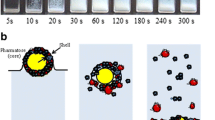
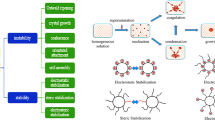
Characterization of wet-milled drug suspensions containing neutral polymer–anionic surfactant as stabilizers poses unique challenges in terms of assessing the aggregation state and examining the stabilization mechanisms. Using a multi-faceted characterization method, this study aims to assess the aggregation state of wet-milled griseofulvin (GF) nanosuspensions and elucidate the stabilization mechanisms and impact of stabilizers. Two grades, SSL and L, of hydroxypropyl cellulose (HPC) with molecular weights of 40 and 140 kg/mol, respectively, were used as a neutral stabilizer at concentrations varying from 0 to 7.5% (w/w) without and with 0.05% (w/w) sodium dodecyl sulfate (SDS). The aggregation state was examined via laser diffraction, scanning electron microscope (SEM) imaging, and rheometry. Zeta potential, stabilizer adsorption, surface tension, and drug wettability were used to elucidate the stabilization mechanisms. The results suggest that deviation from a uni-modal PSD and pronounced pseudoplasticity with power–law index lower than one signify severe aggregation. Polymer or surfactant alone was not able to prevent GF nanoparticle aggregation, whereas HPC–SDS combination led to synergistic stabilization. The effect of polymer concentration was explained mainly by the stabilizer adsorption and partly by surface tension. The synergistic stabilization afforded by HPC–SDS, traditionally explained by electrosteric mechanism, was attributed to steric stabilization provided by HPC and enhanced GF wettability/reduced surface tension provided by SDS. Zeta potential results could not explain the mitigation of aggregation by HPC–SDS. Overall, this study has demonstrated that the elucidation of the complex effects of HPC–SDS on GF nanosuspension stability entails a multi-faceted and comprehensive characterization approach.







Similar content being viewed by others
References
Merisko-Liversidge E, Liversidge GG. Nanosizing for oral and parenteral drug delivery: a perspective on formulating poorly-water soluble compounds using wet media milling technology. Adv Drug Deliv Rev. 2011;63(6):427–40.
Li M, Azad M, Davé R, Bilgili E. Nanomilling of drugs for bioavailability enhancement: a holistic formulation-process perspective. Pharmaceutics. 2016;8(2):17.
Afolabi A, Akinlabi O, Bilgili E. Impact of process parameters on the breakage kinetics of poorly water-soluble drugs during wet stirred media milling: a microhydrodynamic view. Eur J Pharm Sci. 2014;51:75–86.
Merisko-Liversidge EM, Liversidge GG. Drug nanoparticles: formulating poorly water-soluble compounds. Toxicol Pathol. 2008;36(1):43–8.
Kesisoglou F, Panmai S, Wu Y. Nanosizing—oral formulation development and biopharmaceutical evaluation. Adv Drug Deliv Rev. 2007;59(7):631–44.
Monteiro A, Afolabi A, Bilgili E. Continuous production of drug nanoparticle suspensions via wet stirred media milling: a fresh look at the Rehbinder effect. Drug Dev Ind Pharm. 2013;39(2):266–83.
Müller RH, Jacobs C, Kayser O. Nanosuspensions as particulate drug formulations in therapy: rationale for development and what we can expect for the future. Adv Drug Deliv Rev. 2001;47(1):3–19.
Cooper ER. Nanoparticles: a personal experience for formulating poorly water soluble drugs. J Control Release. 2010;141(3):300–2.
Smyth H. Nanoparticles and nanosuspensions: physical and biological properties. Drug Dev Ind Pharm. 2016;42(5):685.
Jiang X, Cheng Y-S, Smyth HD. Nanostructured aerosol particles: fabrication, pulmonary drug delivery, and controlled release. J Nanomater. 2011;2011:1–2.
Basa S, Muniyappan T, Karatgi P, Prabhu R, Pillai R. Production and in vitro characterization of solid dosage form incorporating drug nanoparticles. Drug Dev Ind Pharm. 2008;34(11):1209–18.
Lee J. Drug nano- and microparticles processed into solid dosage forms: physical properties. J Pharm Sci. 2003;92(10):2057–68.
Van Eerdenbrugh B, Van den Mooter G, Augustijns P. Top-down production of drug nanocrystals: nanosuspension stabilization, miniaturization and transformation into solid products. Int J Pharm. 2008;364(1):64–75.
Peltonen L, Hirvonen J. Pharmaceutical nanocrystals by nanomilling: critical process parameters, particle fracturing and stabilization methods. J Pharm Pharmacol. 2010;62(11):1569–79.
Bilgili E, Afolabi A. A combined microhydrodynamics–polymer adsorption analysis for elucidation of the roles of stabilizers in wet stirred media milling. Int J Pharm. 2012;439(1):193–206.
Knieke C, Azad M, Davé R, Bilgili E. A study of the physical stability of wet media-milled fenofibrate suspensions using dynamic equilibrium curves. Chem Eng Res Des. 2013;91(7):1245–58.
Li M, Lopez N, Bilgili E. A study of the impact of polymer–surfactant in drug nanoparticle coated pharmatose composites on dissolution performance. Adv Powder Technol. 2016;27(4):1625–36.
Knieke C, Azad MA, To D, Bilgili E, Davé RN. Sub-100 micron fast dissolving nanocomposite drug powders. Powder Technol. 2015;271:49–60.
Bhakay A, Merwade M, Bilgili E, Dave RN. Novel aspects of wet milling for the production of microsuspensions and nanosuspensions of poorly water-soluble drugs. Drug Dev Ind Pharm. 2011;37(8):963–76.
Bilgili E, Li M, Afolabi AI. The combination of cellulosic polymers and anionic surfactants a good strategy for ensuring physical stability of BCS class II drug nanosuspensions? Pharm Dev Technol. 2016;21(4):499–510.
Susarla R, Afolabi A, Patel D, Bilgili E, Davé RN. Novel use of superdisintegrants as viscosity enhancing agents in biocompatible polymer films containing griseofulvin nanoparticles. Powder Technol. 2015;285:25–33.
Verma S, Kumar S, Gokhale R, Burgess DJ. Physical stability of nanosuspensions: investigation of the role of stabilizers on Ostwald ripening. Int J Pharm. 2011;406(1):145–52.
Lebhardt T, Roesler S, Uusitalo HP, Kissel T. Surfactant-free redispersible nanoparticles in fast-dissolving composite microcarriers for dry-powder inhalation. Eur J Pharm Biopharm. 2011;78(1):90–6.
Choi J-Y, Park CH, Lee J. Effect of polymer molecular weight on nanocomminution of poorly soluble drug. Drug Deliv. 2008;15(5):347–53.
Sepassi S, Goodwin DJ, Drake AF, Holland S, Leonard G, Martini L, et al. Effect of polymer molecular weight on the production of drug nanoparticles. J Pharm Sci. 2007;96(10):2655–66.
Duro R, Alvarez C, Martı́nez-Pacheco R, Gómez-Amoza JL, Concheiro A, Souto C. The adsorption of cellulose ethers in aqueous suspensions of pyrantel pamoate: effects on zeta potential and stability. Eur J Pharm Biopharm. 1998;45(2):181–8.
Li M, Yaragudi N, Afolabi A, Dave R, Bilgili E. Sub-100nm drug particle suspensions prepared via wet milling with low bead contamination through novel process intensification. Chem Eng Sci. 2015;130:207–20.
Schubert MA, Harms M, Müller-Goymann CC. Structural investigations on lipid nanoparticles containing high amounts of lecithin. Eur J Pharm Sci. 2006;27(2):226–36.
Knieke C, Sommer M, Peukert W. Identifying the apparent and true grinding limit. Powder Technol. 2009;195(1):25–30.
Azad M, Afolabi A, Bhakay A, Leonardi J, Davé R, Bilgili E. Enhanced physical stabilization of fenofibrate nanosuspensions via wet co-milling with a superdisintegrant and an adsorbing polymer. Eur J Pharm Biopharm. 2015;94(0):372–85.
Müller R. Zetapotential und partikelladung—kurze theorie, praktische meßdurchführung, dateninterpretation. Stuttgart: Wissenschaftliche; 1996.
Riddick TM. Control of colloid stability through zeta potential. Zeta-Meter Inc. via Livingston Publishing Company; 1968.
Lakshmi P, Kumar GA. Nanosuspension technology: a review. Int J Pharm Pharm Sci. 2010;2(4):35–40.
Mishra PR, Shaal LA, Müller RH, Keck CM. Production and characterization of Hesperetin nanosuspensions for dermal delivery. Int J Pharm. 2009;371(1):182–9.
Müller R, Jacobs C. Buparvaquone mucoadhesive nanosuspension: preparation, optimisation and long-term stability. Int J Pharm. 2002;237(1):151–61.
Verma S, Huey BD, Burgess DJ. Scanning probe microscopy method for nanosuspension stabilizer selection. Langmuir. 2009;25(21):12481–7.
Goodwin DJ, Sepassi S, King SM, Holland SJ, Martini LG, Lawrence MJ. Characterization of polymer adsorption onto drug nanoparticles using depletion measurements and small-angle neutron scattering. Mol Pharm. 2013;10(11):4146–58.
Alghunaim A, Kirdponpattara S, Newby B-mZ. Techniques for determining contact angle and wettability of powders. Powder Technol. 2016;287:201–15.
Kipp JE. The role of solid nanoparticle technology in the parenteral delivery of poorly water-soluble drugs. Int J Pharm. 2004;284(1–2):109–22.
Kuentz M, Egloff P, Röthlisberger D. A technical feasibility study of surfactant-free drug suspensions using octenyl succinate-modified starches. Eur J Pharm Biopharm. 2006;63(1):37–43.
Van Eerdenbrugh B, Vermant J, Martens JA, Froyen L, Van Humbeeck J, Augustijns P, et al. A screening study of surface stabilization during the production of drug nanocrystals. J Pharm Sci. 2009;98(6):2091–103.
Washburn EW. The dynamics of capillary flow. Phys Rev. 1921;17(3):273–83.
Hołownia D, Kwiatkowska I, Hupka J. An investigation on wetting of porous materials. Physicochem Probl Miner. 2008;42:251–62.
Li M, Ioannidis N, Gogos C, Bilgili E. A comparative assessment of nanocomposites vs. amorphous solid dispersions prepared via nanoextrusion for drug dissolution enhancement. Eur J Pharm Biopharm. 2017;119:68–80.
Ain-Ai A, Gupta PK. Effect of arginine hydrochloride and hydroxypropyl cellulose as stabilizers on the physical stability of high drug loading nanosuspensions of a poorly soluble compound. Int J Pharm. 2008;351(1–2):282–8.
Barthelmes G, Pratsinis S, Buggisch H. Particle size distributions and viscosity of suspensions undergoing shear-induced coagulation and fragmentation. Chem Eng Sci. 2003;58(13):2893–902.
Bernhardt C, Reinsch E, Husemann K. The influence of suspension properties on ultra-fine grinding in stirred ball mills. Powder Technol. 1999;105(1–3):357–61.
Knieke C, Steinborn C, Romeis S, Peukert W, Breitung-Faes S, Kwade A. Nanoparticle production with stirred media mills: opportunities and limits. Chem Eng Technol. 2010;33(9):1401–11.
Bhakay A, Azad M, Bilgili E, Dave R. Redispersible fast dissolving nanocomposite microparticles of poorly water-soluble drugs. Int J Pharm. 2014;461(1):367–79.
Yang H, Teng F, Wang P, Tian B, Lin X, Hu X, et al. Investigation of a nanosuspension stabilized by Soluplus® to improve bioavailability. Int J Pharm. 2014;477(1–2):88–95.
Patel CM, Chakraborty M, Murthy ZVP. Preparation of fenofibrate nanoparticles by combined stirred media milling and ultrasonication method. Ultrason Sonochem. 2014;21(3):1100–7.
George M, Ghosh I. Identifying the correlation between drug/stabilizer properties and critical quality attributes (CQAs) of nanosuspension formulation prepared by wet media milling technology. Eur J Pharm Sci. 2013;48(1–2, 142):–52.
Cerdeira AM, Mazzotti M, Gander B. Formulation and drying of miconazole and itraconazole nanosuspensions. Int J Pharm. 2013;443(1–2):209–20.
Cerdeira AM, Mazzotti M, Gander B. Miconazole nanosuspensions: influence of formulation variables on particle size reduction and physical stability. Int J Pharm. 2010;396(1):210–8.
Bilgili E, Hamey R, Scarlett B. Nano-milling of pigment agglomerates using a wet stirred media mill: elucidation of the kinetics and breakage mechanisms. Chem Eng Sci. 2006;61(1):149–57.
Sommer M, Stenger F, Peukert W, Wagner NJ. Agglomeration and breakage of nanoparticles in stirred media mills—a comparison of different methods and models. Chem Eng Sci. 2006;61(1):135–48.
Kissa E. Preparation and testing of dispersions. In: Dispersions: characterization, testing, and measurement. Boca Raton: CRC Press; 1999. p. 240–41.
Yasueda S-i, Inada K, Matsuhisa K, Terayama H, Ohtori A. Evaluation of ophthalmic suspensions using surface tension. Eur J Pharm Biopharm. 2004;57(2):377–82.
Korson L, Drost-Hansen W, Millero FJ. Viscosity of water at various temperatures. J Phys Chem. 1969;73(1):34–9.
Kushner LM, Duncan BC, Hoffman JI. A viscometric study of the micelles of sodium dodecyl sulfate in dilute solutions. J Res Natl Bur Stand. 1952;49:85–90.
Funding
The authors acknowledge financial support from the U.S. National Science Foundation (NSF) Engineering Research Center (ERC) for Structured Organic Systems (SOPS) through Grant EEC-0540855.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, M., Alvarez, P., Orbe, P. et al. Multi-faceted Characterization of Wet-milled Griseofulvin Nanosuspensions for Elucidation of Aggregation State and Stabilization Mechanisms. AAPS PharmSciTech 19, 1789–1801 (2018). https://doi.org/10.1208/s12249-018-0993-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-0993-4