Abstract
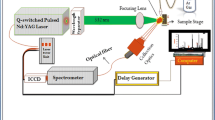
Drug safety has become a very important subject, and more countries have joined in the fight against counterfeit drugs. This study demonstrated a non-invasive Raman spectroscopy method that could be utilized for screening liquid injectable drugs for spurious/falsely-labeled/falsified/counterfeit medical products (SFFCs). Two problems were solved to remove the blocks in identification and quantitation: one problem was the weak API signal extraction from the non-invasive Raman spectra and the other was the problem of Raman absolute measurement. Principal component analysis (PCA) and classical least square (CLS) algorithms were performed to establish the models. Water was chosen as the “internal standard” to normalize the spectra to solve the problem of Raman absolute measurement. The results showed that the 11 positive samples and 66 negative samples were all well identified with a threshold of 0.95. One of the positive samples contained the excipient propylene glycol, which was identified successfully at the same time. The accuracy of quantitative results was approximately 5% for doxofylline liquid injectables and about 10% for the low-concentration and big glass bottle-containers of Levofloxacin Lactate and Sodium Chloride Injections as compared to the results using an HPLC method, this is satisfactory for fast screening of SFFCs. In conclusion, with the development of a database of identification and quantitation models, this method may determine liquid injectable drugs in a fast and non-invasive way and become one of the most powerful weapons against SFFCs.

ᅟ





Similar content being viewed by others
REFERENCES
International Medical Products Anti-Counterfeiting Taskfore(IMPACT). Counterfeit Drugs Kill! Geneva: World Health Organization; 2008.
Attaran A, Bate R, Kendall M. Why and how to make an international crime of medicine counterfeiting. J Int Crim Justice. 2011;9:325–54.
Leung D, Kang SO, Anslyn EV. Rapid determination of enantiomeric excess: a focus on optical approaches. R Soc Chem. 2012;41:448–79. doi:10.1039/C1CS15135E.
Scafi SHF, Pasquini C. Identification of counterfeit drugs using near-infrared spectroscopy. Analyst. 2001;126:2218–24. doi:10.1039/b106744n.
Hu CQ, Zou WB, Hu WS, et al. Establishment of a Fast Chemical Identification System for screening of counterfeit drugs of macrolide antibiotics. J Pharm Biomed Anal. 2006;40:68–74. doi:10.1016/j.jpba.2005.06.026.
Räsänen E, Sandler N. Near infrared spectroscopy in the development of solid dosage forms. J Pharm Pharmacol. 2007;59:147–59. doi:10.1211/jpp.59.2.0002.
Chen B, Zhang SM, Lin YY, et al. Rapid Raman spectra identification and determination of levofloxzcin hydrochloride injection. Chin J Pharm Anal. 2011;31:1715–9.
Wu LH, Liu HJ, Chen R, et al. Rapid identification determination of Ribavirin injection by Raman Spectra. China Pharm. 2011;14:1465–7.
Zhang R, Shi BJ, Wang Y. Quantitative determination of Tiopronin injection by Raman Spectroscopy. Chin Pharm J. 2012;47(3):232–5.
Jackson R. Patent 13,905,385, US. 2012;5.
Zhao Y, Yin LH. Patent application No.201310048251.2, China. 2013;6.
China Pharmacopeia, China Medical Science Press, Beijing, Volume 2(2010), p. 106–107.
Yin LH, Zhao Y. Patent application No.201210593760.9, China. 2013;5.
Smith TEL, Wooster MJ, Tattaris M, Griffith DWT. Atmos Meas Tech. 2011;4:97–116. doi:10.5194/amt-4-97-2011
Esler MB, Griffith DWT, Wilson SR, et al. Precision trace gas analysis by FT-IR spectroscopy. 1. Simultaneous analysis of CO2, CH4, N2O, and CO in air. Anal Chem. 2000;72:206–15. doi:10.1021/ac9905625.
ACKNOWLEDGMENTS
This research was supported by the national science and technology support program “Research of prohibited substances in health food detection technology Subjects” (2011BAZ02015-2) for P. R. China. We would like to show appreciation to our partners for sample collection and spectra measurements: Donglai XU and Rui LI from Nanning Institute for Food and Drug Control; Weijiao WANG from Hunan Institute for Food and Drug Control; Xiaohua ZHOU from Jiangsu Institute for Food and Drug Control; Tianyang LIU and Liqun LIU from Heilongjiang Institute for Food and Drug Control; Yong GAO from Xinjiang Institute for Food and Drug Control; Yanjia GAO from Dongying Institute for Food and Drug Control; Yi LEI and Bibo HU from Guangdong Institute for Food and Drug Control.
Disclaimer
The findings and conclusion of this article have not been formally disseminated by the National Institutes of Food and Drug Control (NIFDC) and should not be construed to represent Agency determination or policy.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(XLSX 11 kb)
Rights and permissions
About this article
Cite this article
Zhao, Y., Ji, N., Yin, L. et al. A Non-invasive Method for the Determination of Liquid Injectables by Raman Spectroscopy. AAPS PharmSciTech 16, 914–921 (2015). https://doi.org/10.1208/s12249-015-0286-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-015-0286-0