Abstract
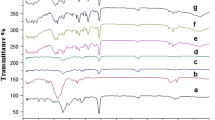
The objective of this study was to compare a novel controlled release tablet formulation based on interpolyelectrolyte complex (PEC). Interpolymer interactions between the countercharged polymers like Eudragit® EPO (polycation) and hypromellose acetate succinate (polyanion) and Eudragit® EPO and hypromellose phthalate (polyanion) were investigated with a view to their use in per oral controlled release drug delivery systems. The formation of inter-macromolecular ionic bonds between cationic polymer and anionic polymer was investigated using Fourier transform infrared (FT-IR) spectroscopy and differential scanning calorimetry. The FT-IR spectra of the tested polymeric matrices are characterized by visible changes in the observed IR region indicating the interaction between chains of two oppositely charged copolymers. The performance of the in situ formed PEC as a matrix for controlled release of drugs was evaluated, using acetaminophen as a model drug. The dissolution data of these matrices were fitted to different dissolution models. It was found that drug release followed zero-order kinetics and was controlled by the superposition of the diffusion and erosion. These profiles could be controlled by conveniently modifying the proportion of the polymer ratio, polymer type, and polymer concentration the in the tablets.











Similar content being viewed by others
REFERENCES
Bani-Jaber AK, Alkawareek MY, Al-Gousous JJ, Abu Helwa AY. Floating and sustained-release characteristics of effervescent tablets prepared with a mixed matrix of Eudragit L-100-55 and Eudragit E PO. Chem Pharm Bull. 2011;59:155–60.
Mustafin RI, Bukhovets AV, Sitenkov AY, Garipova VR, Kemenova VA, Rombaut P, et al. Synthesis and characterization of a new carrier based on Eudragit® EPO/S100 interpolyelectrolyte complex for controlled colon-specific drug delivery. Pharm Chem J. 2011;45:568–74.
Bani-Jaber A, Al-Aani L, Alkhatib H, Al-Khalidi B. Prolonged intragastric drug delivery mediated by Eudragit® E-Carrageenan polyelectrolyte matrix tablets. AAPS PharmSciTech. 2011;12:354–61.
Obeidat WM, Abu Znait AH, Sallam AS. Novel combination of anionic and cationic polymethacrylate polymers for sustained release tablet preparation. Drug Dev Ind Pharm. 2008;34:650–60.
Prado HJ, Matulewicz MC, Bonelli P, Cukierman AL. Basic butylated methacrylate copolymer/kappa-carrageenan interpolyelectrolyte complex: preparation, characterization and drug release behaviour. Eur J Pharm Biopharm. 2008;70:171–8.
Bawa P, Pillay V, Choonara YE, du Toit LC, Ndesendo VM, Kumar P. A composite polyelectrolytic matrix for controlled oral drug delivery. AAPS PharmSciTech. 2011;12:227–38.
Saleem MA, Kotadia DR, Kulkarni RV. Effect of formulation variables on dissolution of water-soluble drug from polyelectrolyte complex beads. Dissolut Technol. 2012;19:21–8.
Devi N, Maji TK. Preparation and evaluation of gelatin/sodium carboxymethyl cellulose polyelectrolyte complex microparticles for controlled delivery of isoniazid. AAPS PharmSciTech. 2009;10:1412–9.
Arora S, Gupta S, Narang RK, Budhiraja RD. Amoxicillin loaded chitosan-alginate polyelectrolyte complex nanoparticles as mucopenetrating delivery system for h. Pylori Sci Pharm. 2011;79:673–94.
Wang L, Khor E, Wee A, Lim LY. Chitosan-alginate PEC membrane as a wound dressing: assessment of incisional wound healing. J Biomed Mater Res. 2002;63:610–8.
Calvo P, Remuñán-lopez C, Vila-Jato JL, Alonso MJ. Novel hydrophilic chitosan–polyethylene oxide nanoparticles as protein carriers. J Appl Polym Sci. 1997;63:125–32.
Leclercq L, Boustta M, Vert M. A physico-chemical approach of polyanion-polycation interactions aimed at better understanding the in vivo behaviour of polyelectrolyte-based drug delivery and gene transfection. J Drug Target. 2003;11:129–38.
Liu F, Lizio R, Meier C, Petereit HU, Blakey P, Basit AW. A novel concept in enteric coating: a double-coating system providing rapid drug release in the proximal small intestine. J Control Release. 2009;133:119–24.
Ho C, Hwang GC. Development of extended-release solid dispersions of nonsteroidal antiinflammatory drugs with aqueous polymeric dispersions: optimization of drug release via a curve-fitting technique. Pharm Res. 1992;9:206–10.
Schilling SU, Shah NH, Waseem Malick A, McGinity JW. Properties of melt extruded enteric matrix pellets. Eur J Pharm Biopharm. 2010;74:352–61.
Rowe RC. Handbook of pharmaceutical excipients. 5th ed. Washington DC: American Pharmacists Association; 2005.
Hilton AK, Deasy PB. Use of hydroxypropyl methylcellulose acetate succinate in an enteric polymer matrix to design controlled-release tablets of amoxicillin trihydrate. J Pharm Sci. 1993;82:737–43.
Tirpude RN, Puranik PK. Rabeprazole sodium delayed-release multiparticulates: effect of enteric coating layers on product performance. J Adv Pharm Technol Res. 2011;2:184–91.
Kim MS, Kim JS, Kang SH, Yoo YH, Lee S, Park JS, et al. Influence of water soluble additives and HPMCP on drug release from Surelease-coated pellets containing tamsulosin hydrochloride. Arch Pharm Res. 2007;30:1008–13.
Siepmann F, Siepmann J, Walther M, MacRae RJ, Bodmeier R. Blends of aqueous polymer dispersions used for pellet coating: importance of the particle size. J Control Release. 2005;105:226–39.
Lankalapalli L, Kolapalli VRM. Polyelectrolyte complexes: a review of their applicability in drug delivery technology. Indian J Pharm Sci. 2009;71:481–7.
Philipp B, Dautzenberg H, Linow K, Kotz J, Dawydoff W. Polyelectrolyte complexes-recent developments and open problems. Prog Polym Sci. 1989;14:91–172.
Moustafine RI, Zaharov IM, Kemenova VA. Physicochemical characterization and drug release properties of Eudragit E PO/Eudragit L 100-55 interpolyelectrolyte complexes. Eur J Pharm Biopharm. 2006;63:26–36.
Margulis EB, Moustafine RI. Swellability testing of chitosan/Eudragit L100-55 interpolyelectrolyte complexes for colonic drug delivery. J Control Release. 2006;116:e36–7.
Korsmeyer RW, Gurnya R, Doelkera E, Buria P, Peppas NA. Mechanism of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15:25–35.
Costa P, Lobo JMS. Modeling and comparison of dissolution profiles. Eur J Pharm Sci. 2001;13:123–33.
Evonik Pharma polymers 2012. Eudragit® Technical information. Available at: http://eudragit.evonik.com/product/eudragit/Documents/evonik-specification-eudragit-e-100,eudragit-epo-and-eudragit-e-12,5.pdf. Accessed on 12 April 2014.
Sharma M, Sharma V, Panda AK, Majumdar DK. Enteric microsphere formulations of papain for oral delivery. Yakugaku Zasshi. 2011;131:697–709.
Evonik Pharma polymers 2012. Eudragit® Technical information. Available at: http://eudragit.evonik.com/product/eudragit/Documents/evonik-specification-eudragit-l-100-55.pdf. Accessed on 12 April 2014.
Acknowledgments
The authors are grateful to Mylan laboratories Limited, Hyderabad, India, for the generous gift samples of acetaminophen and excipients. The authors wish to thank Dr. Abhijit Deshmukh for supporting the project.
Conflict of Interest
The authors report no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeganathan, B., Prakya, V. Interpolyelectrolyte Complexes of Eudragit® EPO with Hypromellose Acetate Succinate and Eudragit® EPO with Hypromellose Phthalate as Potential Carriers for Oral Controlled Drug Delivery. AAPS PharmSciTech 16, 878–888 (2015). https://doi.org/10.1208/s12249-014-0252-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-014-0252-2