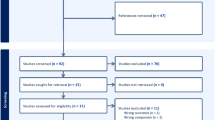
Abstract
Subcutaneous (SC) administration of therapeutic proteins is perceived to pose higher risk of immunogenicity when compared with intravenous (IV) route of administration (RoA). However, systematic evaluations of clinical data to support this claim are lacking. This meta-analysis was conducted to compare the immunogenicity of the same therapeutic protein by IV and SC RoA. Anti-drug antibody (ADA) data and controlling variables for 7 therapeutic proteins administered by both IV and SC routes across 48 treatment groups were analyzed. RoA was the primary independent variable of interest while therapeutic protein, patient population, adjusted dose, and number of ADA samples were controlling variables. Analysis of variance was used to compare the ADA incidence between IV and SC RoA, while accounting for controlling variables and potential interactions. Subsequently, 10 additional therapeutic proteins with ADA data published for both IV and SC administration were added to the above 7 therapeutic proteins and were evaluated for ADA incidence. RoA had no statistically significant effect on ADA incidence for the initial dataset of 7 therapeutic proteins (p = 0.55). The only variable with a significant effect on ADA incidence was the therapeutic protein. None of the other controlling variables, including their interactions with RoA, was significant. When all data from the 17 therapeutic proteins were pooled, there was no statistically significant effect of RoA on ADA incidence (p = 0.81). In conclusion, there is no significant difference in ADA incidence between the IV and SC RoA, based on analysis of clinical ADA data from 17 therapeutic proteins.
Graphical Abstract



Similar content being viewed by others
Data Availability
Data are available upon request.
References
National Cancer Institute at the National Institute of Health. Definition of biotherapy. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/biotherapy. Accessed 17 Feb 2023.
United States Food and Drug Administration. Center for Drug Evaluation and Research. 21 CFR 600. 2020–03505. Definition of the term biological product. 2020;85(35):10057–63. https://www.federalregister.gov/documents/2020/02/21/2020-03505/definition-of-the-term-biological-product. Accessed 17 Feb 2023.
Shankar G, Arkin S, Cocea L, Devanarayan V, Kirshner S, Kromminga A, et al. Assessment and reporting of the clinical immunogenicity of therapeutic proteins and peptides – harmonized terminology and tactical recommendations. AAPS J. 2014;16(4):658–73. https://doi.org/10.1208/s12248-014-9599-2.
Hamuro L, Kijanka G, Kinderman F, Kropshofer H, Bu DX, Zepeda M, et al. Perspectives on subcutaneous route of administration as an immunogenicity risk factor for therapeutic proteins. J Pharm Sci. 2017;106(10):2946–54. https://doi.org/10.1016/j.xphs.2017.05.030.
United States Food and Drug Administration, Center for Drug Evaluation and Research. Guidance document: Immunogenicity assessment for therapeutic protein products. 2014. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/immunogenicity-assessment-therapeutic-protein-products. Accessed 17 Feb 2023.
United States Food and Drug Administration, Center for Drug Evaluation and Research. Guidance document: Immunogenicity testing of therapeutic protein products – developing and validating assays for anti-drug antibody detection. 2019. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/immunogenicity-testing-therapeutic-protein-products-developing-and-validating-assays-anti-drug. Accessed 17 Feb 2023.
Pitiot A, Heuzé-Vourc’h N, Sécher T. Alternative routes of administration for therapeutic antibodies-state of the art. Antibodies (Basel). 2022;11(3):56. https://doi.org/10.3390/antib11030056.
Davis JD, Padros MB, Conrado DJ, Ganguly S, Guan X, Hassan HE, et al. Subcutaneous administration of monoclonal antibodies: pharmacology, delivery, immunogenicity, and learnings from applications to clinical development. Clin Pharmacol Ther. 2024;115(3):422–39. https://doi.org/10.1002/cpt.3150.
Davda J, Declerck P, Hu-Lieskovan S, Hickling TP, Jacobs IA, Chou J, et al. Immunogenicity of immunomodulatory, antibody-based, oncology therapeutics. J Immunother Cancer. 2019;7:105. https://doi.org/10.1186/s40425-019-0586-0.
Genovese MC, Covarrubias A, Leon G, Mysler E, Keiserman M, Valente R, et al. Subcutaneous abatacept versus intravenous abatacept: a phase IIIb noninferiority study in patients with an inadequate response to methotrexate. Arthritis Rheum. 2011;63(10):2854–64. https://doi.org/10.1002/art.30463.
Mateos MV, Nahi H, Legiec W, Grosicki S, Vorobyev V, Spicka I, et al. Subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma (Columba): a multicentre, open-label, non-inferiority, randomised, phase 3 trial. Lancet Haematol. 2020;7(5):e370–80. https://doi.org/10.1016/s2352-3026(20)30070-3.
Simponi (golimumab). Highlights of prescribing information. Janssen Biotech, Inc. 2009. https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/125289s000lbl.pdf. Accessed 17 Feb 2023.
Simponi Aria (golimumab). Highlights of prescribing information. Janssen Biotech, Inc. 2009. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125433s032lbl.pdf. Accessed 17 Feb 2023.
Schreiber S, Ben-Horin S, Leszczyszyn J, Dudkowiak R, Lahat A, Gawdis-Wojnarska B, et al. Randomized controlled trial: subcutaneous vs intravenous infliximab ct-p13 maintenance in inflammatory bowel disease. Gastroenterology. 2021;160(7):2340–53. https://doi.org/10.1053/j.gastro.2021.02.068.
Ortega HG, Liu MC, Pavord ID, Brusselle GG, Fitzgerald JM, Chetta A, et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med. 2014;371(13):1198–207. https://doi.org/10.1056/nejmoa1403290.
Bel EH, Wenzel SE, Thompson PJ, Prazma CM, Keene ON, Yancey SW, et al. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med. 2014;371(13):1189–97. https://doi.org/10.1056/nejmoa1403291.
Chupp GL, Bradford ES, Albers FC, Bratton DJ, Wang-Jairaj J, Nelsen LM, et al. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (Musca): a randomised, double-blind, placebo-controlled, parallel-group, multicentre, phase 3b trial. Lancet Respir Med. 2017;5(5):390–400. https://doi.org/10.1016/s2213-2600(17)30125-x.
Omontys (peginesatide). Highlights of prescribing information. Affymax, Inc. and Takeda Pharmaceuticals America, Inc. 2012. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202799s000lbl.pdf. Accessed 17 Feb 2023.
Neelakantan S, Oemar B, Johnson K, Rath N, Salganik M, Berman G, et al. Safety, tolerability, and pharmacokinetics of pf-06823859, an anti-interferon β monoclonal antibody: a randomized, phase I, single- and multiple-ascending-dose study. Clin Pharmacol Drug Dev. 2021;10(3):307–16. https://doi.org/10.1002/cpdd.887.
MabThera (rituximab). Summary of product characteristics. Roche Pharma. 2009. https://www.ema.europa.eu/en/documents/product-information/mabthera-epar-product-information_en.pdf. Accessed 17 Feb 2023.
Johnson ML, Braiteh F, Grilley-Olson JE, Chou J, Davda J, Forgie A, et al. Assessment of subcutaneous vs intravenous administration of anti-pd-1 antibody pf-06801591 in patients with advanced solid tumors: a phase 1 dose-escalation trial. JAMA Oncol. 2019;5(7):999–1007. https://doi.org/10.1001/jamaoncol.2019.0836.
Birbara C, Dabezies EJ, Burr AM, Fountaine RJ, Smith MD, Brown MT, et al. Safety and efficacy of subcutaneous tanezumab in patients with knee or hip osteoarthritis. J Pain Res. 2018;11:151–64. https://doi.org/10.2147/jpr.s135257.
Actemra (tocilizumab). Highlights of prescribing information Genentech, Inc. 2010. https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/125276lbl.pdf. Accessed 17 Feb 2023.
Ogata A, Tanimura K, Sugimoto T, Inoue H, Urata Y, Matsubara T, et al. Phase III study of the efficacy and safety of subcutaneous versus intravenous tocilizumab monotherapy in patients with rheumatoid arthritis. Arthritis Care Res. 2014;66(3):344–54. https://doi.org/10.1002/acr.22110.
Herceptin (trastuzumab). Summary of product characteristics. Genentech, Inc. 2010. https://www.ema.europa.eu/en/documents/product-information/herceptin-epar-product-information_en.pdf. Accessed 17 Feb 2023.
Sandborn WJ, Baert F, Danese S, Krznaric Z, Kobayashi T, Yao X, et al. Efficacy and safety of vedolizumab subcutaneous formulation in a randomized trial of patients with ulcerative colitis. Gastroenterology. 2020;158(3):562-572.e12. https://doi.org/10.1053/j.gastro.2019.08.027.
European Medicines Agency, Committee for Medicinal Products for Human Use (CHMP). Guideline on immunogenicity assessment of therapeutic proteins. 2017. https://www.ema.europa.eu/en/immunogenicity-assessment-biotechnology-derived-therapeutic-proteins-scientific-guideline.
Acknowledgements
Yazdi K. Pithavala, Pfizer, also contributed suggestions for the analyses. David Potter, Pfizer, advised on the optimal statistical analysis method and aided in explanation of statistical results.
Funding
This work was sponsored by Pfizer Inc. and funded by a grant from the University of California, San Diego Skaggs School of Pharmacy Summer Research Program.
Author information
Authors and Affiliations
Contributions
J.F. and Y.C. wrote the manuscript. J.F. collected and analyzed the data. L.R., M.D.V.A, D.M., and Y.C. provided the basis for the project, contributions to the analyses, as well as revisions to the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
J.F. is a student at the University of California, San Diego Skaggs School of Pharmacy and was a summer intern at Pfizer when this work was conducted who declared no competing interests for this work. M.D.V.A., D.M., and Y.C. are employees of Pfizer Inc. and own stock in Pfizer.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Felderman, J., Ramaiah, L., Vazquez-Abad, MD. et al. Anti-Drug Antibody Incidence Comparison of Therapeutic Proteins Administered Via Subcutaneous vs. Intravenous Route. AAPS J 26, 60 (2024). https://doi.org/10.1208/s12248-024-00930-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-024-00930-w