Abstract
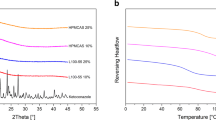
Preparation of amorphous solid dispersions using polymers is a commonly used formulation strategy for enhancing the solubility of poorly water-soluble drugs. However, often a single polymer may not bring about a significant enhancement in solubility or amorphous stability of a poorly water-soluble drug. This study describes application of a unique and novel binary polymeric blend in preparation of solid dispersions. The objective of this study was to investigate amorphous solid dispersions of glipizide, a BCS class II model drug, in a binary polymeric system of polyvinyl acetate phthalate (PVAP) and hypromellose (hydroxypropyl methylcellulose, HPMC). The solid dispersions were prepared using two different solvent methods: rotary evaporation (rotavap) and fluid bed drug layering on sugar spheres. The performance and physical stability of the dispersions were evaluated with non-sink dissolution testing, powder X-ray diffraction (PXRD), and modulated differential scanning calorimetry (mDSC). PXRD analysis demonstrated an amorphous state for glipizide, and mDSC showed no evidence of phase separation. Non-sink dissolution testing in pH 7.5 phosphate buffer indicated more than twofold increase in apparent solubility of the drug with PVAP–HPMC system. The glipizide solid dispersions demonstrated a high glass transition temperature (T g) and acceptable chemical and physical stability during the stability period irrespective of the manufacturing process. In conclusion, the polymeric blend of PVAP–HPMC offers a unique formulation approach for developing amorphous solid dispersions with the flexibility towards the use of these polymers in different ratios and combined quantities depending on drug properties.








Similar content being viewed by others
References
Serajuddin A. Solid dispersion of poorly water-soluble drugs: early promises, subsequent problems, and recent breakthroughs. J Pharm Sci. 1999;88(10):1058–66.
Brouwers J, Brewster ME, Augustijns P. Supersaturating drug delivery systems: the answer to solubility-limited oral bioavailability. J Pharm Sci. 2009;98(8):2549–72.
Yamashita K, Nakate T, Okimoto K, Ohike A, Tokunaga Y, Ibuki R, et al. Establishment of new preparation method for solid dispersion formulation of tacrolimus. Int J Pharm. 2003;267(1–2):79–91.
Ho HO, Su HL, Tsai T, Sheu MT. The preparation and characterization of solid dispersions on pellets using a fluidized-bed system. Int J Pharm. 1996;139:223–9.
Sun N, Wei X, Wu B, Chen J, Lu Y, Wu W. Enhanced dissolution of silymarin/polyvinylpyrrolidone solid dispersion pellets prepared by a one-step fluid-bed coating technique. Powder Technol. 2008;182:72–80.
Puri V, Dantuluri AK, Bansal A. Barrier coated drug layered particles for enhanced performance of amorphous solid dispersion dosage form. J Pharm Sci. 2011;101(1):342–53.
Gilis P, De Conde VFV, Vandecruys RPG. Beads having a core coated with an antifungal and a polymer. 1997. WO94/05263.
Leuner C, Dressman J. Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm. 2000;50(1):47–60.
Friesen DT, Shanker R, Crew M, Smithey DT, Curatolo WJ, Nightingale JA. Hydroxypropyl methylcellulose acetate succinate-based spray-dried dispersions: an overview. Mol Pharm. 2008;5(6):1003–19.
DiNunzio JC, Miller DA, Yang W, McGinity JW, Williams 3rd RO. Amorphous compositions using concentration enhancing polymers for improved bioavailability of itraconazole. Mol Pharm. 2008;5(6):968–80.
Gao P. Amorphous pharmaceutical solids: characterization, stabilization, and development of marketable formulations of poorly soluble drugs with improved oral absorption. Mol Pharm. 2008;5(6):903–4.
Chiang P, Ran Y, Chou K, Cui Y, Sambrone A, Chan C, et al. Evaluation of drug load and polymer by using a 96-well plate vacuum dry system for amorphous solid dispersion drug delivery. AAPS PharmSciTech. 2012;13(2):713–22.
Khougaz K, Clas SD. Crystallization inhibition in solid dispersions of MK-0591 and poly(vinylpyrrolidone) polymers. J Pharm Sci. 2000;89(10):1325–34.
Miller DA, DiNunzio JC, Yang W, McGinity JW, Williams 3rd RO. Enhanced in vivo absorption of itraconazole via stabilization of supersaturation following acidic to neutral pH transition. Drug Dev Ind Pharm. 2008;34(8):890–902.
Miller DA, DiNunzio JC, Yang W, McGinity JW, Williams 3rd RO. Targeted intestinal delivery of supersaturated itraconazole for improved oral absorption. Pharm Res. 2008;25(6):1450–9.
Konno H, Handa T, Alonzo DE, Taylor LS. Effect of polymer type on the dissolution profile of amorphous solid dispersions containing felodipine. Eur J Pharm Biopharm. 2008;70:493–9.
Kennedy M, Hu J, Gao P, Li L, Ali-Reynolds A, Chal B, et al. Enhanced bioavailability of a poorly soluble VR1 antagonist using an amorphous solid dispersion approach: a case study. Mol Pharm. 2008;5(6):981–93.
Taylor LS, Zografi G. Spectroscopic characterization of interactions between PVP and indomethacin in amorphous molecular dispersions. Pharm Res. 1997;14(12):1691–8.
Guidance for Industry: Dissolution testing of immediate release solid oral dosage forms. F.D.A. 1997: p. 17.
Guidance for Industry: Extended release oral dosage forms: development, evaluation, and application of in vitro/in vivo correlations. F.D.A. 1997; 27.
Murdande SB, Pikal MJ, Shanker RM, Bogner RH. Solubility advantage of amorphous pharmaceuticals: I. A thermodynamic analysis. J Pharm Sci. 2010;99(3):1254–64.
Konno H, Taylor LS. Influence of different polymers on the crystallization tendency of molecularly dispersed amorphous felodipine. J Pharm Sci. 2006;95(12):2692–705.
Konno H, Taylor LS. Ability of different polymers to inhibit the crystallization of amorphous felodipine in the presence of moisture. Pharm Res. 2008;25(4):969–78.
Ilevbare GA, Liu H, Edgar KJ, Taylor LS. Understanding polymer properties important for crystal growth inhibition: impact of chemically diverse polymers on solution crystal growth of ritonavir. Cryst Growth Des. 2012;12:3133–43.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Ping Gao and Lawrence Yu
Zahra N. Mahmoudi and Sampada B. Upadhye contributed equally to this project.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 108 kb)
Rights and permissions
About this article
Cite this article
Mahmoudi, Z.N., Upadhye, S.B., Ferrizzi, D. et al. In Vitro Characterization of a Novel Polymeric System for Preparation of Amorphous Solid Drug Dispersions. AAPS J 16, 685–697 (2014). https://doi.org/10.1208/s12248-014-9590-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-014-9590-y