Abstract
Background
Polycystic ovarian syndrome (PCOS) is a reproductive disorder associated with several endocrine and metabolic alterations. The mechanism underlying this syndrome is controversial. On the other hand, drugs used for the treatment are associated with several side effects and poor in controlling PCOS phenotype. l-Carnitine (LC) has been reported to have a significant regulatory function on the redox and metabolic status of female reproductive system. Nevertheless, its regulatory pathways to regulate PCOS are still under investigation. Therefore, this study aimed to evaluate the effects of LC on the steroidogenic pathways, oxidative stress markers and metabolic profile in letrozole (LTZ)-induced PCOS rat model.
Methods
For this aim, animals were divided into four groups (n = 6). Control group, untreated letrozole-induced PCOS group (1 mg/kg bwt) for 21 days, PCOS group treated with l-Carnitine (100 mg/kg bwt) for 14 days and PCOS group treated with clomiphene citrate (2 mg/kg bwt) for 14 days. Finally, body and ovarian weight, metabolic state(glucose and lipid profile), hormonal assays (testosterone, 17 β estradiol, LH and FSH levels), intraovarian relative gene expression (CYP17A1, StAR, CYP11A1 and CYP19A1 genes), ovarian redox state (malondialdehyde (MDA), reduced glutathione content (GSH) and catalase enzyme activity (CAT)) as well as serum total antioxidant capacity (TAC) were detected. Also, histomorphometric ovarian evaluation (number and diameter of cystic follicles, granulosa cell thickness and theca cell thickness) as well as immune expression of caspase-3 of granulosa cells of cystic follicles were determined.
Results
LC significantly improved ovarian redox state (GSH, MDA and CAT), steroidogenic pathways gene expression (CYP17A1, StAR, CYP11A1 and CYP19A1 genes), hormonal profile (Follicle stimulating hormone (FSH) and luteinizing hormone (LH), testosterone and estradiol), metabolic state (Glucose and lipid profile) histomorphometric alterations and decreased caspase 3 immune reaction of granulosa cells.
Conclusion
l-Carnitine supplementation can ameliorate the PCOS phenotype through its energetic, antioxidant and antiapoptotic functions as well as steroidogenesis regulatory role. This protocol could be modified to produce the best therapeutic benefits, and it could be regarded as a prospective therapeutic intervention for PCOS.
Similar content being viewed by others
1 Background
Women during reproductive age are subjected to continuous hormonal changes during their reproductive life and 4 to 21% of them develop polycystic ovarian syndrome (PCOS) [1]. PCOS is a reproductive disorder associated with several endocrine and metabolic alterations [1]. PCOS's clinical characteristics are ovulatory dysfunction with polycystic ovarian features, amenorrhea, hirsutism and infertility [2]. In addition, it is complex disorder that is characterized by several complications, such as hyperinsulinemia, obesity, cardiovascular diseases as well as ovarian, endometrial and breast tumors [3, 4] Also, imbalance in the main steroidogenic enzymes and hormones are the main features of PCOS, that result in disturbances in estrogen and androgen production leading to excess androgen secretion [5,6,7]. Despite the fact that genetic disorders are thought to predispose to PCOS, the genetic makeup which could be correlated to PCOS phenotypes is elusive owing to the trait's complexity [8].
On the other hand, antioxidants and free radicals are key regulators of the ovarian normal physiology [9]. Previous studies have reported the ROS/antioxidants interplay that greatly influences the reproductive activities in female mammalian animals [9, 10]. It has been previously reported that oxidative stress (OS) biomarkers such as malondialdehyde (MDA), catalase (CAT), and glutathione content are abnormal in patients with PCOS [11].
However, the exact etiological mechanism underlying this syndrome is still controversial and unclear. Till now, there is no cure for PCOS, and in the absence of mechanistic understanding, the medical management is poor [7]. Several therapeutic approaches, including metformin and clomiphene citrate, have been used to treat PCOS, although they are frequently linked with substantial adverse effects [12,13,14,15,16,17]. These provoke the need for more researches and studies that reveal PCOS's possible mechanistic pathways, and biomarkers as well as searching for alternative therapeutic approaches to improve the PCOS together and decrease its side effects that impair quality of life. l-Carnitine (LC) is a cofactor that can be obtained from diet or it was endogenously synthesized. It is crucial for fatty acid metabolism through aiding the long-chain free fatty acids to be transported into mitochondria which then undergoes beta-oxidation [18].LC was previously reported to have a significant regulatory function on the redox status and metabolism of the female reproduction[19]. Nevertheless, its mechanistic pathways to regulate PCOS are still under investigation. Therefore, the current study aimed to assess the impact of l-Carnitine on the steroidogenic pathways, oxidative stress biomarkers and metabolic profile of LTZ-induced PCOS rat model, giving new molecular, biochemical and histological insights into the possible mechanistic pathway of PCOS and the impact of l-Carnitine as therapeutic potential.
2 Methods
2.1 Experimental design
Animal procedures and experimental protocols were approved under the consent of the Institutional Animal Care and Use Committee (IACUC), Cairo University, Egypt (CUIIF921) and in line with the guidelines for Guide for the Care and Use of Laboratory Animals: Eighth Edition [20].
In the current experiment, 24 adult female Sprague–Dawley rats (8–12 weeks old) of average bodyweight 200–250 g. with 4–5 days estrus cycle were chosen and randomly allocated into four groups (n = 6). Animals were kept in the Physiology Department animal house, Faculty of Veterinary Medicine, Cairo University, Egypt. Rats were acclimatized to a 12-h light–dark cycle at 22 ± 2 °C and 55–65% humidity. All of animals had unrestricted access to feed and water.
All animals were divided into 2 groups. The first group (control group, n = 6) received orally a vehicle carboxymethylcellulose (CMC) (1 ml) once daily for 21 successive days. The second group is used to induce PCOS (PCOS group) (n = 18) received letrozole (1 mg/kg bwt/day) dissolved in a vehicle carboxymethyl cellulose (CMC) (1 ml) orally to induce PCOS daily for 21 successive days [21]. The estrus staging of the animals was done daily using vaginal smear technique as previously mentioned [11]. After PCOS induction, blood samples were taken from all animals for testosterone evaluation. Female rats showing significantly higher testosterone than control rats as well as prolonged diestrus phase in their estrous cycle were chosen to simulate the PCOS phenotype. After PCOS induction, animals were assigned to the treatment schedule for 14 days according to previous studies [22, 23] as follows:
(1) Control group (n = 6): | Normal control (animals without PCOS) received normal saline (1 ml) for 14 days |
(2) PCOS group (n = 6): | Letrozole-induced PCOS animal model using oral letrozole (1 mg/kg bwt) |
(3) PCOS + l-Carnitine (n = 6): | PCOS female rats treated orally with l-Carnitine (100 mg/kg bwt) for 14 days dissolved in normal saline (1 ml) [24] |
(4) PCOS + Clomiphene citrate(n = 6): | PCOS female rats treated with Clomiphene citrate (2 mg/kg bwt) for 14 days orally dissolved in normal saline (1 ml) as a standard drug for ovulation induction [11] |
Body weight was evaluated weekly. Finally, animals were euthanized on the 35th day of the experiment.
l-Carnitine was obtained form from MEPACO, Egypt. Letrozole (Femara® non-steroidal aromatase inhibitor), 2.5 mg tablets, was purchased from Novartis Pharma AG, Basel, Switzerland. Clomiphene citrate (Clomid ®) was purchased from Sanofi, Aventis Pharma, France. Carboxy methyl cellulose was purchased from Merck, India.
2.2 Sampling procedures
Animals fasted for 10 h and then blood samples were collected from all animals at the 5th week. Special serum separating tubes (IVAC ™) were used to collect blood, then left to stand for 15 min and eventually centrifuged at 4000 rpm for 15 min. Collected serum samples were kept at − 20 °C for further metabolic analysis and hormones evaluation. Before dissection, phosphate-buffered (PH 7.4) saline (PBS) was used to wash ovaries, then they were weighed and dissected into two sets. The one set was kept at − 80 °C to determine intraovarian redox status and RT-PCR analysis, and the second set was fixed in 10% neutral buffered formalin (10% NBF) for histomorphometry and immunohistochemistry analysis.
2.3 Serum metabolic profile
Fasting blood glucose (FBG), and lipid profile (Total cholesterol (TC), Triglycerides (TG), High density lipoprotein cholesterol (HDL-C) were evaluated using commercial kits purchased from spectrum according to constructions of manufacturer. Estimation of very low-density lipoprotein (VLDL-C), Low-density lipoprotein cholesterol (LDL-C) concentrations, coronary risk indices, atherogenic index and triglycerides-glucose index were calculated as following:
-
VLDL-C (mg/dl) = triglycerides/5 [25]
-
LDL-C (mg/dl) = total cholesterol − (HDL-C + VLDL-C) [25]
-
The coronary risk indices (TC/HDL-C and TG/HDL-C ratios) were calculated [26].
-
Triglyceride-glucose index (TyG index) = Ln [TG (mg/dl) × FPG (mg/dl)/2] [27]
2.4 Oxidative stress/antioxidant evaluation
Weight of the Ovarian tissues were determined. Homogenization of ovarian tissues was done using cold phosphate buffer (pH 7.4) and then tissue samples went for centrifugation at 6000 rpm for 20 min at 4 °C. The supernatant was used to evaluate the intraovarian redox state though the following assays: CAT (catalase assay) [28], Reduced glutathione (GSH) [29], Malondialdehyde (MDA) [30] were assessed as well as measuring serum total antioxidant capacity (TAC) was determined [31]. All measures were done using special highly standardized kits purchased from Bio-diagnostic company, Egypt.
2.5 Hormonal assays
Evaluating Serum testosterone serum levels using ELISA kit (Diagnostic Biochem Canada). Follicle stimulating hormone (FSH) and luteinizing hormone (LH) were evaluated by ELISA method along with the instructions of the manufacturer using rat specific ELISA kits (SunLong Biotech Co., LTD). LH/FSH ratio was evaluated. Serum 17β Estradiol was measured using ELISA kit (Diagnostic Biochem Canada).
2.6 RT-PCR analysis
2.6.1 Quantitative real-time RT-PCR analysis for CYP17A1, CYP11A1, StAR and CYP19A1 genes
The relative expression of the ovarian mRNA levels of CYP17A1, CYP11A1, StAR and CYP19A1 genes were determined by real-time RT-PCR, guided with GAPDH gene as a reference [32]. About, 100 mg of ovarian tissue was used to extract total RNA by total RNA Extraction Kit (Vivantis, Malaysia). Purified total RNA (1 µg) was reverse transcribed using M-MuLV Reverse Transcriptase (NEB#M0253). Real-time RT-PCR for each gene was performed using the primers shown in Table 1 by a fluorescence based real-time detection method with a fluorescent SYBR Green dye (Thermo Scientific, Cat. No. K0221). The real-time PCR conditioning were performed as follows: 95 °C for 5 min (initial denaturation) and then 40 cycles at 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 30 s in each experiment. Each experiment involved all samples in triplicate, a No Template Control (NTC) and a “Melting Curve” analysis included for all samples. The relative transcription levels was calculated using The comparative 2−ΔΔCT method [33].
2.7 Histopathological examination
2.7.1 Histopathological and histomorphometric analysis
Dissection of Right and left ovaries were done and then fixation in 10% neutral buffered formalin (10%NBF), embedded in paraffin wax, cut into sections with 3–5 μm thickness, and staining with hematoxylin and eosin (H&E) for the histological evaluation according to Bancroft, and Gamble [34]. For histomorphometric studies, the number, diameter and thickness of granulosa and theca cells of the ovarian cysts were measured. Stained sections were visualized using Leica Quin 500 analyzer computer system (Leica Microsystems, Switzerland) in the Faculty of Veterinary Medicine, Cairo University. Calibration of the image analyzer was done automatically to convert the measurement units (pixels) formed by the image analyzer program into actual micrometer units. The images of each section were captured for each sample with a final magnification of 400 ×.
2.7.2 Immunohistochemistry for activated caspase 3
Caspase 3 (active form) was used to evaluate apoptotic progression in granulosa cells. The Avidin–biotin–peroxidase technique [35] was used for activated caspase 3 determination which is an apoptotic marker. Brown cytoplasmic or nuclear staining is considered a positive reaction. After deparaffinization and rehydration of the ovarian sections, antigen recovery was accomplished via boiling of the sections in citrate buffer using a microwave. Endogenous peroxidase blockage was done by H2O2. Then non-specific background was blocked with 10% serum-tris buffer for 20 min at room temperature, after that these sections were then incubated with the primary antibody; anti-caspase-3 rabbit polyclonal antibody (Catalogue No.: RB-1197 from Thermo Fisher Scientific) diluted 1/100 at room temperature for 120 min. The slides were subsequently incubated with a biotinylated polyvalent secondary antibody and then incubated with an avidin–biotin–peroxidase complex solution (LSAB2 Kit; Dako). Visualization of the reaction was done via addition of 3,3′-diaminobenzidine tetrachloride to these sections. Sections counterstaining was done using Hematoxylin stain. Negative control slides were considered using slides stained with 2ry antibody IgG only, whereas palatine tonsils sections were used as positive controls.
2.8 Statistical analysis
Statistical analysis was done using SPSS (SPSS Inc.: Chicago, IL, USA, 2017) [36], including the calculations of Means ± standard error (SE) using one-way analysis of variance (ANOVA) accompanied with Tukey as a post-hoc test. Significance was set at P < 0.05 level.
3 Results
3.1 Estimation of physiological parameters
Data illustrated in Fig. 1 showed no significant difference in the final body weight of animals of different groups. While relative ovarian weight showed significant elevation in the PCOS group when compared to the control one at P < 0.05. Otherwise LC and CLO intake for 2 weeks led to significant reduction in the relative ovarian weight in comparison to PCOS group at P < 0.05.
Effect of l-Carnitine (LC) and Clomiphene citrate (CLO) on Ovarian weight (A) and Final body weight (B) in different groups. Values presented as mean ± SE (n = 6). Means denoted with different superscript letters are statistically significant at P < 0.05. C Control group, PCOS letrozole-induced polycystic ovarian syndrome, PCOS + LC letrozole induced polycystic ovarian syndrome treated with l-Carnitine, PCOS + CLO letrozole induced polycystic ovarian syndrome treated with Clomiphene Citrate
3.2 Serum metabolic profile
Data illustrated in Table 2 showed significant elevation in levels of blood glucose level in the PCOS group compared to the control group at P < 0.05 while LC as well as CLO treated groups showed that blood glucose level was significantly reduced when compared to the PCOS group at (P < 0.05). Also, lipid profile in PCOS group showed significant alterations as serum total cholesterol (TC), triglycerides (TG), LDL-C and VLDL-C were significantly elevated, while HDL-C showed significant decrease (P < 0.05) in contrast to the control group. Moreover, LC and CLO treated groups showed significant mitigating effect of the lipid profile compared with the PCOS group at P < 0.05.Coronary risk indices (CRI) and triglycerides glucose index (TyG) were significantly deteriorated in the PCOS group when compared with the control group at P < 0.05. CRI and TyG were significantly improved in LC and CLO treated groups comparable with PCOS group at P < 0.05. Upon comparing LC and CLO in their lipid modulatory role, LC treatment showed significant improvement at the levels of TC, HDL-C, LDL-C, TC/HDL-C, and TG/HDL-C when compared to CLO PCOS treated group.
3.3 Oxidative stress/antioxidant assessment
Data illustrated in Fig. 2 showed the antioxidant assessment of LC and CLO in letrozole-induced PCOS rat model. Serum total antioxidant capacity (TAC) (Fig. 2A) and ovarian reduced glutathione (GSH) (Fig. 2C) content as well as catalase (CAT) activity (Fig. 2D) were significantly diminished in PCOS group, while malondialdehyde (MDA) (Fig. 2B) was elevated significantly in comparison with control group at P < 0.05. LC and CLO PCOS treated groups showed significant improvement when compared with PCOS group at P < 0.05. Regarding CAT activity, CLO PCOS treated group did not show significant improvement in the ovarian CAT activity in comparison with PCOS untreated group.
Effect of l-Carnitine and Clomiphene citrate on Levels of MDA, GSH and CAT in ovarian tissues and serum TAC in letrozole-induced PCOS rat model. Values presented as mean ± SE (n = 6). Means denoted with different superscript letters are statistically significant at P < 0.05. A TAC: Total antioxidant capacity, B MDA: Malondialdehyde, C GSH: Glutathione reduced and D CAT: Catalase. C: Control group, PCOS: letrozole induced polycystic ovarian syndrome, PCOS + LC: letrozole induced polycystic ovarian syndrome treated with l-Carnitine, PCOS + CLO: letrozole induced polycystic ovarian syndrome treated with Clomiphene Citrate
3.4 Hormonal assays
LC effect and CLO effect on the different hormonal assays in letrozole-induced PCOS animal model was demonstrated in Table 3. LC significantly modulated serum testosterone levels when compared to the PCOS animal model at P < 0.05 and there is no statistical significance between LC treated group and CLO treated group at P < 0.05. Regarding serum 17β estradiol level, PCOS induction using letrozole led to significant estradiol reduction in the PCOS untreated group. On the other hand, both LC and CLO interventions significantly restored serum estradiol in comparison with PCOS untreated group.
At the gonadotrophic hormonal levels (FSH and LH), letrozole administration significantly elevated serum LH levels when compared to the control group and upon treatment with LC and CLO, LH levels were significantly diminished at P < 0.05 when compared to PCOS group. In addition, LC and CLO treated group showed significantly elevated FSH levels when compared with PCOS group at P < 0.05. Furthermore, LH/FSH ratio showed significant elevation in PCOS group when compared to the control one at P < 0.05.
3.5 RT.PCR
Data presented in Fig. 3 showed that the PCOS group exhibited significantly elevated expression of the ovarian CYP17A1 gene by 2 folds compared to the normal group. Moreover, the ovaries of the PCOS group exhibited a significant decrease in the gene expression of CYP11A1 by 0.43 fold when compared to the control group. On the same line, there was a significant decline in StAR and CYP19A1 genes expression levels by 0.06 and 0.34 fold respectively in the ovaries of the PCOS group when compared to the control one.
Effect of l-Carnitine and Clomiphene citrate on mRNA relative gene expression of Cyp17a1, Cyp11a1 and STAR genes in ovarian tissue of different groups in letrozole-induced PCOS rat models. A CYP17A: Cytochrome P450 17 A1, B CYP11A1: Cytochrome P450 11 A1, C StAR: Steroidogenic Acute Regulatory protein, D CYP19A1: Aromatase in different groups. Values presented as mean ± SE (n = 6). Means denoted with different superscript letter are statistically significant at P < 0.05. C: Control group, PCOS: letrozole induced polycystic ovarian syndrome, PCOS + LC: letrozole induced polycystic ovarian syndrome treated with l-Carnitine, PCOS + CLO: letrozole induced polycystic ovarian syndrome treated with Clomiphene Citrate
Following the treatment of the PCOS group with LC, our results showed that the ovarian expression level of CYP17A1 gene was significantly decreased to 0.95-fold among LC treated group compared to the untreated PCOS ones. Moreover, treatment with LC was able to significantly increase the gene expression of CYP11A1 in the ovaries of the treated group to 0.95-fold compared to the untreated PCOS group. As for StAR gene, LC induced a significant increase in the ovarian mRNA relative expression to be 0.3-fold instead of 0.056-fold in the untreated PCOS group. On the same line, the treatment of the PCOS group with LC revealed that mRNA relative expression of CYP19A1 gene was significantly increased to 0.70 compared to the untreated PCOS ones.
On the same line, the treatment of the PCOS group with CLO revealed that CYP17A1 gene ovarian expression was significantly decreased to 0.90 compared to the untreated PCOS ones. On the other hand, the ovarian expression level of CYP11A1 was significantly increased to 0.77 fold among LC treated group compared to untreated PCOS ones. Meanwhile, CLO could not statistically increase the ovarian StAR mRNA expression compared to the untreated PCOS group. CLO could not statistically increase the ovarian StAR, and CYP19A1mRNA expression compared to the untreated PCOS group.
3.6 Histological and histomorphometric evaluation
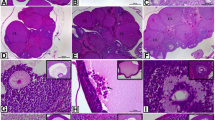
Histological observation of the ovary of the control group showed normal ovarian follicles at different stages of development and corpus luteum (Fig. 4A). The ovarian follicles obtained in the control group were 3.33 ± 1.53 in number, with an average 244.42 ± 38.97 µm in diameter. Granulosa cells of the ovarian follicles of the control group appeared with an average 70.42 ± 29.72 µm thickness, while the thickness of the theca layer of the same group was recorded with an average 21.93 ± 5.65 µm (Table 4). On the other hand, PCOS group showed numerous ovarian cysts that were recorded as 17.67 ± 1.45 in number. These ovarian cysts appeared thin walled with an average diameter 453.25 ± 80.61 µm. Moreover, granulosa cells of the ovarian cysts in PCOS group appeared degraded with an average thickness 35.22 ± 4.83 µm, while the thickness of theca cells in the same group was observed with an average 16.02 ± 0.63 µm (Table 4). Degenerated oocyte and pale acidophilic homogenous substance filling the lumen of the ovarian cysts were also noticed (Fig. 4B). Interestingly, the LC treated group revealed a decrease in the number of the ovarian cysts (7.00 ± 1.00 in number), when compared with the PCOS group. The ovarian cysts in PCOS + LC treated group appeared smaller in size with an average diameter 239.90 ± 44.10 µm. The thickness of the granulosa layer measured 36.30 ± 6.15 µm, while that of the theca cells was 13.41 ± 0.77 µm (Table 5, Fig. 4C). CLO exposed group showed slight decrease in the number of the ovarian cysts (8.00 ± 1.00). Some of these ovarian cysts appeared thin walled as those in the PCOS group, while others appeared with thickened granulosa layer. The recorded diameter of the ovarian cysts in CLO exposed group was 438.55 ± 168.24 µm. Moreover, the thickness of the granulosa cell layer was 31.36 ± 1.04 µm, while that of the theca layer was 14.83 ± 0.24 µm (Table 4, Fig. 4D).
A photomicrograph of H&E-stained sections (100 ×) of the ovary of female albino rat showing: A control ovary with normal ovarian follicles and corpus luteum in different stages of development. B PCOS group with numerous ovarian cysts. The cysts appeared thin walled with degraded granulosa cells, degenerated oocyte and pale acidophilic residues. C PCOS + LC group with ovarian cysts. The cysts appeared smaller in size. Notice the increase in the granulosa cell layer. D PCOS + CLO group nearly the same as LTZ group. C: Control group, PCOS: letrozole induced polycystic ovarian syndrome, PCOS + LC: letrozole induced polycystic ovarian syndrome treated with l-Carnitine, PCOS + CLO: letrozole induced polycystic ovarian syndrome treated with Clomiphene Citrate
3.7 Immunohistochemistry for activated caspase 3
Immunohistochemical analysis of the ovary of the control group showed low expression of caspase 3 (Fig. 5A), while PCOS group exhibited strong positive immune reactivity to caspase 3 (Fig. 5B). Moreover, mild expression of caspase 3 was observed in the PCOS group treated with LC (Fig. 5C), while PCOS group showed moderate positive immune reaction to caspase-3 when treated with CLO (Fig. 5D). The recorded mean area % was 5.12 ± 0.88 for the control group, 27.53 ± 3.92 for PCOS group, 12.85 ± 3.01 for PCOS group treated with LC and 15.47 ± 0.50 for PCOS group treated by CLO (Table 5).
A photomicrograph of caspase 3-stained sections (400 ×) of the ovary of female albino rat showing: A control ovary with low expression of caspase 3. B PCOS group showing intense positive immune reaction to caspase 3. C PCOS + LC group with mild expression of caspase 3. D PCOS + CLO showing moderate positive immune reaction to caspase 3. C: Control group, PCOS: letrozole induced polycystic ovarian syndrome, PCOS + LC: letrozole induced polycystic ovarian syndrome treated with l-Carnitine, PCOS + CLO: letrozole induced polycystic ovarian syndrome treated with Clomiphene Citrate
4 Discussion
Polycystic ovary syndrome (PCOS) is a multifaceted reproductive disorder linked to several metabolic and hormonal changes. [37]. The symptoms of this disorder include irregular ovulation and hyperandrogenism. [11]. PCOS has been linked to several sorts of dyslipidemia, including low HDL-C levels, high triglycerides, total cholesterol, and LDL-C levels. [38]. Several studies have shown the onset of earlier cardiovascular risk profile abnormalities associated with PCOS phenotype together with development of abnormal glucose and lipid metabolism, obesity, insulin resistance and other hall marks of metabolic syndrome [39,40,41]. In parallel with our results, we reported significant elevation of blood glucose, total cholesterol, LDL-C, triglycerides and worsening of coronary risk indices in PCOS group. LTZ alters lipid profile in PCOS animal models [42]. Such metabolic alterations were associated with worsening of triglycerides-glucose index (TyG), a good indicator of insulin resistance (IR). [43] and a good predictor of diabetes type 2 and coronary heart diseases [44]. We used TyG index as surrogate marker for IR in letrozole-induced PCOS animal model. Recent studies have recognized TyG as a practical surrogate of IR [45,46,47]. In addition, TyG, TG/HDL-C, and TC/HDL-C indices were found to be substantially associated with IR in PCOS reproductive disorder [48]. This dysregulated glucose metabolism and insulin resistance are suggested to be due to dysfunction of pancreatic β cells, reduction in hepatic clearance of insulin associated with PCOS phenotype [49, 50]. LC is used to increase energy consumption, regulate the lipid profile and play a pivotal role in the glucose metabolism as well as redox state regulation [51].
Despite the pivotal physiological regulatory role of free radicals within the ovary, excessive production of ROS may lead to an elevated cumulative risk of ovarian pathology that would be aggravated under altered intraovarian redox state [52]. Growing evidence reported that oxidative stress was implicated in the pathophysiology of PCOS [53]. Previous studies documented significant alteration in the oxidative stress biomarkers such as total antioxidant capacity (TAC), MDA, glutathione content and antioxidant enzymes [49, 50]. In parallel to those findings, we reported a significant deterioration of serum TAC, and ovarian reduced glutathione (GSH) content as well as catalase (CAT) activity in letrozole-induced PCOS groups, in addition to significant elevation of malondialdehyde (MDA) in the ovary of PCOS induced animals. There is a close relation and crosstalk between oxidative stress and altered steroidogenesis pathways in the ovaries, thus contributing to disturbed follicular development, hyperandrogenemia and, ultimately, infertility [54]. This altered ovarian redox state can be attributed to the elevation of biomolecules oxidation and activation of redox-sensitive transcription factors in PCOS [53, 55] Hyperandrogenism can be caused by low-grade inflammation in the ovarian tissues, and hyperandrogenism causes OS through IR. Antioxidant defense systems scavenge excess ROS to overcome cell damage induced by excessive ROS production [56]. Antioxidants maintain a tight balance between beneficial oxidant production (acts in cell signaling) and destructive OS [56]. The present study showed significant improvement of OS markers upon LC supplementation, as LC has a free radical scavenging property and interferes with ROS formation as well as ferrous ions chelation [57]. The carbonyl group in the LC molecule can stabilize free radicals generated on the α-carbon with conjugation. As a result, it protects against the adverse effects of reactive oxygen and nitrogen species [57,58,59].
In this study, LC significantly increased FSH levels and decreased LH and LH/FSH levels in the serum when compared to levels in rats with letrozole-induced PCOS. In addition, LC administration dramatically reduced serum testosterone levels in rats with PCOS. Increased testosterone was found to be a crucial factor to PCOS pathogenesis in a prior study, and its reduction was demonstrated to be beneficial in PCOS disorders. [60]. Furthermore, raised androgen and LH serum levels, as well as decreased FSH and an increased LH:FSH ratio, can be used as biomarker for PCOS identification [61]. To uncover the underlying molecular mechanism of LC steroidogenic regulatory role, we assessed ovarian expression of genes regulating steroidogenesis. Our candidate genes involved in steroidogenesis are CYP17A1, CYP11A1 StAR and CYP19A1 genes. CYP17A1 encodes the rate-liming enzyme of androgen biosynthesis (17-α-hydroxylase/17–20 lyase (P450 17α)), through its hydroxylase activity, it catalyzes the hydroxylation of pregnenolone together with progesterone to 17-hydroxypregnenolone and 17-hydroxyprogesterone, respectively as well as its lyase activity is responsible for their conversion to androstenedione and dehydroepiandrosterone (DHEA), respectively [62]. CYP11A1 gene encodes the cytochrome P450 side chain cleavage enzyme (P450scc) that is responsible for the cholesterol side-chain cleavage with subsequent formation of pregnenolone; the first step in steroid hormone synthesis [63]. StAR gene encodes the rate-limiting step in the de novo synthesis of all steroidal hormones by cholesterol transport via the steroidogenic acute regulatory (StAR) protein from outer to the inner mitochondrial membrane [64]. Aromatase (Cyp19A1) is a steroidogenic enzyme that catalyzes the conversion of testosterone to estradiol and estrone independently. The Cyp19A1 gene encodes aromatase [65]. Surprisingly, the Cyp19A1 encoding gene has been identified as a substantial risk factor for PCOS [66].
The current study revealed that letrozole induced PCOS led to elevation of serum testosterone and reduction of intraovarian mRNA expression of steroidogenesis genes StAR, CYP11A1, and CYP19A1, with the exception of Cyp17A1, which was significantly up-regulated in rats receiving letrozole when compared to the control group. Down regulation of mRNA expression of StAR and CYP11A1 in letrozole induced PCOS model could be a direct consequence of androgen dependent negative feedback inhibition on the steroidogenesis pathway suggesting that androgens can regulate steroidogenesis at the rate-limiting step of cholesterol transport to the inner mitochondrial membrane [62]. Furthermore, estrogen has been shown to upregulate mRNA expression of StAR gene [67], suggesting that the estrogen deprivation induced by letrozole could be linked to the reduction in StAR mRNA expression. Thus, these results suggested that negative feedback control exploited by steroid hormones on steroidogenic enzymes can provoke auto/paracrine mechanisms through which steroid hormones may be auto-regulated by the ovarian tissue [62].On the other side, CYP11A1 mRNA expression has shown down regulation was previously reported in letrozole-induced PCOS rat model [61, 62, 68]. The CYP11A1 gene has been shown to be a biomarker for pre-ovulatory follicles such as antral follicles, and its transcriptional and/or translational expression is controlled by FSH production [68]. We speculated that letrozole induced FSH reduction led to down regulation of CYP11A1 gene expression together with the presence of several cystic preovulatory follicles. In addition, our findings showed a significant decline in Cyp19A1 mRNA expression in PCOS ovarian tissues. These findings were consistent with previous studies [66, 69]. Cyp19A1 expression may be inhibited on a regular basis in PCOS ovaries due to transcriptional regulation, such as Cyp19A1 promoter hypermethylation, which may play a role in PCOS pathophysiology [66].
The current study showed a significant increase in testosterone and LH levels, as well as an increase in Cyp17A1 mRNA expression, in the letrozole-induced PCOS group. Previously, it was demonstrated that granulosa cell-derived estrogens regulates theca cell steroidogenesis via a short estrogen derived granulosa cells negative-feedback loop within the follicles of rodents [70]. Furthermore, it has been speculated that the letrozole-induced rise in androgen levels could be due to the release of the negative-feedback loop of estrogens on theca cell steroidogenesis as well as the direct blockage of androgen aromatization [62]. Letrozole-induced estrogen reduction stimulates theca cell androgen production via a paracrine mechanism by nullification of estrogen negative feedback on Cyp17A1 expression [62]. In addition, it has been recently reported that ROS attenuates steroidogenesis pathway in mouse ovarian granulosa cells [71]. As the suppressed antioxidant mechanisms within ovarian compartments are responsible for follicular regression and steroidogenic transcriptional machinery alterations [72].
Our study showed that LC was successfully able to restore the expression levels of both CYP17A1, CYP11A1, StAR and Cyp19A1 genes to their normal. To the best of our knowledge, no previous studies investigated the impact of LC supplementation on the expression levels of the investigated genes in letrozole-induced PCOS rats model have been published. It has been previously reported that LC was able to induce FSH and LH in streptozotocin diabetic rats through its antioxidant mechanism on the basophilic cells of the pituitary gland [73]. Also, LC was able to offset endocrine disruption in carbendazim treated male rats through its antioxidant and anti-inflammatory signaling pathways regulation [74]. Accumulating evidences have stated that the low-grade inflammation progression and oxidative stress (OS) production in response to hyperandrogenism might be a major contributor to PCOS phenotype induction in letrozole-induced models [37, 75]. The current study's findings imply that OS is a major contributor to PCOS formation in this model via intra-ovarian estrogen deprivation via letrozole aromatase enzyme suppression [62], as estrogen plays an important role in antioxidant defense mechanisms via intracellular signaling pathways [76]. As a result, we hypothesized that the regulatory role of LC steroidogenesis through its antioxidant processes could lead to modification of the hypothalamic-pituitary–gonadal (HPG) axis, which regulates gonadotropin production as well as boosting estrogen synthesis [60].
Consequently, LC administration significantly modulates gonadotrophic hormones secretion that enhances follicular maturation together with estrogen production [77]. Therefore, LC supplementation upregulates intraovarian CYP11A1, StAR and CYP19A1 mRNA gene expression. Thereby, it can regulate intraovarian estrogen negative feedback impact on ovarian steroidogenesis pathway and inhibit elevated androgen production as well as CYP17A1 gene expression. This anti-polycystic syndrome action of LC could be attributed to its ability to regulate steroidogenesis pathways, as well as its favorable intraovarian redox state modulation.
Our results were confirmed by histological and histomorphometry evaluation of the ovarian tissues. LC supplementation showed significant improvement in the ovarian architecture (Number of ovarian cysts, Diameter of ovarian cysts, Granulosa cell layer thickness and Theca layer thickness) in comparison with PCOS animal model. Shen et al. [78] considered the presence of follicular cysts with a reduction of the granulosa cell layer an indicator of the successful induction of PCOS phenotype. This comes in the same line with the findings of the current study that revealed many ovarian cysts in the LTZ exposed group with a reduction of the granulosa cell layer and theca cell layer. The LC anti-polycystic effect was supported by Kalhori et al. [79] as they reported the ability of LC supplementation to improve follicular development and endocrine dysfunction in mice after induction of PCOS.
Apoptosis in the ovarian tissues is critical in PCOS [80]. PCOS is related to an inflammatory response and cell apoptosis [81, 82]. A study by Salehi et al. [83] as well as Shen et al. [78] reported that Caspase 3,the apoptotic executer, was highly expressed in PCOS, and its overexpression may result in apoptosis overexpression [83]. In this study, caspase-3 immune expression was higher in LTZ-induced PCOS rats compared to control rats, whereas LC downregulated caspase 3 expression and thus inhibited ovarian cell apoptosis. Similarly to our findings, data reported by previous studies documented the antiapoptotic action of LC [84]. LC, as a free radical scavenger, inhibits follicular atresia by oxidative stress reduction and mitochondrial dysfunction regulation [79]. Apoptosis in granulosa cells reduce androgen to estrogen aromatization, resulting in hyperandrogenemia; thus, inhibition of granulosa cell apoptosis prevents hyperandrogenemia [85]. Our findings newly support that l-Carnitine was able to regulate expression of genes involved in steroidogenic pathway. Besides its antioxidant antiapoptotic, and metabolic regulatory role. Our reported data can advocate the use of l-Carnitine as an adjunctive therapy in the management of hormonal imbalance in PCOS syndrome and other female infertility problems associated with hormonal dysregulation. However, further investigation is required to evaluate its long-term use and its impact when synchronized with other fertility enhancing drugs.
5 Conclusion
L-Carnitine (LC) supplementation ameliorates the expression levels of the three studied genes (CYP17A1, StAR, CYP11A1 and CYP19A1 genes and improves oxidative stress markers in the ovarian microenvironment as well as it modulates metabolic state in PCOS rat model. LC regulates folliculogenesis and reduces the expression of caspase 3 in the granulosa cells. It can be concluded that LC supplementation is able to ameliorate the PCOS phenotype and alleviate the associated ovarian dysfunction, not only by its known biological energetic, antioxidant and antiapoptotic functions but also through modulations of genes involved in steroidogenesis. In conclusion, our reported data can advocate the use of LC as an adjunctive therapy in the management of hormonal imbalance in PCOS syndrome.
Availability of data and materials
This published article presents all of the data generated or assessed during this study.
Abbreviations
- PCOS:
-
Polycystic ovarian syndrome
- MDA:
-
Malondialdehyde
- GSH:
-
Reduced Glutathione
- CAT:
-
Catalase enzyme
- CYP17A1 :
-
Cytochrome P450 17A1
- CYP11A1 :
-
Cytochrome P450 11A1
- StAR :
-
Steroidogenic acute regulatory protein
- CYP19A1 :
-
Aromatase
- OS:
-
Oxidative stress
- LC:
-
L-Carnitine
- CLO:
-
Clomiphene citrate
- ROS:
-
Reactive oxygen species
- CRI:
-
Coronary risk indices
- TyG index:
-
Triglyceride-glucose index
- LTZ:
-
Letrozole
References
Trikudanathan S (2015) Polycystic ovarian syndrome. Med Clin N Am 99:221–235
Papadakis G, Kandaraki EA, Tseniklidi E, Papalou O, Diamanti-Kandarakis E (2019) Polycystic ovary syndrome and NC-CAH: distinct characteristics and common findings. A systematic review. Front. Endocrinol. (Lausanne) 10:388
Barry JA, Azizia MM, Hardiman PJ (2014) Risk of endometrial, ovarian and breast cancer in women with polycystic ovary syndrome: A systematic review and meta-analysis. Hum Reprod Update 20:748–758
Ladrón De Guevara A, Fux-Otta C, Crisosto N, Szafryk De Mereshian P, Echiburú B, Iraci G et al (2014) Metabolic profile of the different phenotypes of polycystic ovary syndrome in two Latin American populations. Fertil Steril 101:1732–1739
Dunaif A (1998) Polycystic ovary syndrome. Polycystic ovary syndrome. Health Prog 4:4
Escobar-Morreale HF (2018) Polycystic ovary syndrome: Definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol 14:270–284
Walters KA (2020) Polycystic ovary syndrome: is it androgen or estrogen receptor? Curr Opin Endocr Metab Res 12:1–7
Salilew-Wondim D, Wang Q, Tesfaye D, Schellander K, Hoelker M, Hossain MM et al (2015) Polycystic ovarian syndrome is accompanied by repression of gene signatures associated with biosynthesis and metabolism of steroids, cholesterol and lipids. J Ovarian Res 8:1–14. https://doi.org/10.1186/s13048-015-0151-5
Wang S, He G, Chen M, Zuo T, Xu W, Liu X (2017) The role of antioxidant enzymes in the ovaries. Oxid Med Cell Longev. https://doi.org/10.1155/2017/4371714
Al-Gubory KH, Fowler PA, Garrel C (2010) The roles of cellular reactive oxygen species, oxidative stress and antioxidants in pregnancy outcomes. Int J Biochem Cell Biol 42:1634–1650
Ndeingang EC, Defo Deeh PB, Watcho P, Kamanyi A (2019) Phyllanthus muellerianus (Euphorbiaceae) restores ovarian functions in letrozole-induced polycystic ovarian syndrome in rats. Evid-Based Complement Altern Med. https://doi.org/10.1155/2019/2965821
Jones T, Ho JR, Gualtieri M, Bruno-Gaston J, Chung K, Paulson RJ et al (2018) Clomiphene stair-step protocol for women with polycystic ovary syndrome. Obstet Gynecol 131:91–95
Wheeler KM, Sharma D, Kavoussi PK, Smith RP, Costabile R (2019) Clomiphene citrate for the treatment of hypogonadism. Sex Med Rev 7:272–276
Del Pup L, Peccatori FA, Levi-Setti PE, Codacci-Pisanelli G, Patrizio P (2018) Risk of cancer after assisted reproduction: a review of the available evidences and guidance to fertility counselors. Eur Rev Med Pharmacol Sci 22:8042–8059
Farzinvash Z (2018) A young woman with sudden visual field shimmering: a case report. Indian J Ophthalmol 66:1504. https://doi.org/10.4103/ijo.IJO_515_18
Berk-Krauss J, Bieber AK, Criscito MC, Grant-Kels JM, Driscoll MS, Keltz M et al (2018) Melanoma risk after in vitro fertilization: a review of the literature. J Am Acad Dermatol 79:1133-1140.e3
Quaile MP, Melich DH, Jordan HL, Nold JB, Chism JP, Polli JW et al (2010) Toxicity and toxicokinetics of metformin in rats. Toxicol Appl Pharmacol 243:340–347
Longo N, Frigeni M, Pasquali M (2016) Carnitine transport and fatty acid oxidation. Biochim Biophys Acta - Mol Cell Res 1863:2422–2435. https://doi.org/10.1016/j.bbamcr.2016.01.023
Agarwal A, Sengupta P, Durairajanayagam D (2018) Role of l-Carnitine in female infertility. Reprod Biol Endocrinol 16(1):1–18
National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals (2011) Guide for the care and use of laboratory animals. National Academies Press, Washington, DC. http://www.nap.edu/catalog/12910
Kafali H, Iriadam M, Ozardali I, Demir N (2004) Letrozole-induced polycystic ovaries in the rat: a new model for cystic ovarian disease. Arch Med Res 35:103–108
Ibrahim YF, Alorabi M, Abdelzaher WY, Toni ND, Thabet K, Hegazy A et al (2022) Diacerein ameliorates letrozole-induced polycystic ovarian syndrome in rats. Biomed Pharmacother 149:112870
Haslan MA, Samsulrizal N, Hashim N, Zin NSNM, Shirazi FH, Goh YM (2021) Ficus deltoidea ameliorates biochemical, hormonal, and histomorphometric changes in letrozole-induced polycystic ovarian syndrome rats. BMC Complement Med Ther 21:291. https://doi.org/10.1186/s12906-021-03452-6
Şiktar E, Ekinci D, Şiktar E, Beydemir Ş, Gülçin I, Günay M (2011) Protective role of l-Carnitine supplementation against exhaustive exercise induced oxidative stress in rats. Eur J Pharmacol 668:407–413
Friedewald WT, Levy RIFD (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Baldissera MD, Souza CF, Grando TH, Stefani LM, Monteiro SG (2017) β-Caryophyllene reduces atherogenic index and coronary risk index in hypercholesterolemic rats: the involvement of cardiac oxidative damage. Chem Biol Interact 270:9–14
Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F (2008) The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord 6:299–304. https://doi.org/10.1089/met.2008.0034
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Koracevic D, Koracevic G, Djordjevic V, Andrejevic S, Cosic V (2001) Method for the measurement of antioxidant activity in human fluids. J Clin Pathol 54:356–361
Ahmed YH, El-Naggar ME, Rashad MM, Youssef AM, Galal MK, Bashir DW (2022) Screening for polystyrene nanoparticle toxicity on kidneys of adult male albino rats using histopathological, biochemical, and molecular examination results. Cell Tissue Res 388:149–165. https://doi.org/10.1007/s00441-022-03581-5
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408
Bancroft JD, Gamble M (2007) Theory and practice of histological techniques, 6th edn. Elsevier Ltd, London
Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29:577–580. https://doi.org/10.1177/29.4.6166661
Corp IBM (2017) IBM SPSS statistics for windows. IBM Corp, Armonk, NY
Pandey V, Singh A, Singh A, Krishna A, Pandey U, Tripathi YB (2016) Role of oxidative stress and low-grade inflammation in letrozole-induced polycystic ovary syndrome in the rat. Reprod Biol 16:70–77
Karateke A, Dokuyucu R, Dogan H, Ozgur T, Tas ZA, Tutuk O et al (2018) Investigation of therapeutic effects of erdosteine on polycystic ovary syndrome in a rat model. Med Princ Pract 27:515–522
Kiranmayee D, Kavya K, Himabindu Y, Sriharibabu M, Madhuri GJ, Venu S (2017) Correlations between anthropometry and lipid profile in women with PCOS. J Hum Reprod Sci 10:167
Dokras A (2008) Cardiovascular disease risk factors in polycystic ovary syndrome. Semin Reprod Med 26:039–044. https://doi.org/10.1055/s-2007-992923
El-Mazny A, Abou-Salem N, El-Sherbiny W, El-Mazny A (2010) Insulin resistance, dyslipidemia, and metabolic syndrome in women with polycystic ovary syndrome. Int J Gynecol Obstet 109:239–241. https://doi.org/10.1016/j.ijgo.2010.01.014
Jahan S, Abid A, Khalid S, Afsar T, Qurat-Ul-Ain, Shaheen G et al (2018) Therapeutic potentials of Quercetin in management of polycystic ovarian syndrome using Letrozole induced rat model: a histological and a biochemical study. J Ovarian Res. 11:1–10
Unger G, Benozzi SF, Perruzza F, Pennacchiotti GL (2014) Triglycerides and glucose index: a useful indicator of insulin resistance. Endocrinol y Nutr (Engl Ed) 61:533–540
Hong S, Han K, Park CY (2020) The triglyceride glucose index is a simple and low-cost marker associated with atherosclerotic cardiovascular disease: a population-based study. BMC Med 18:1–8
Hameed EK (2019) TyG index a promising biomarker for glycemic control in type 2 diabetes mellitus. Diabetes Metab Syndr Clin Res Rev 13:560–563
Shi W, Xing L, Jing L, Tian Y, Yan H, Sun Q et al (2020) Value of triglyceride-glucose index for the estimation of ischemic stroke risk: Insights from a general population. Nutr Metab Cardiovasc Dis 30:245–253
Li S, Guo B, Chen H, Shi Z, Li Y, Tian Q et al (2019) The role of the triglyceride (triacylglycerol) glucose index in the development of cardiovascular events: a retrospective cohort analysis. Sci Rep 9:1–9
Kheirollahi A, Teimouri M, Karimi M, Vatannejad A, Moradi N, Borumandnia N et al (2020) Evaluation of lipid ratios and triglyceride-glucose index as risk markers of insulin resistance in Iranian polycystic ovary syndrome women. Lipids Health Dis 19:235. https://doi.org/10.1186/s12944-020-01410-8
Song DK, Lee H, Sung Y-A, Oh J-Y (2016) Triglycerides to high-density lipoprotein cholesterol ratio can predict impaired glucose tolerance in young women with polycystic ovary syndrome. Yonsei Med J 57:1404. https://doi.org/10.3349/ymj.2016.57.6.1404
Skarra DV, Hernández-Carretero A, Rivera AJ, Anvar AR, Thackray VG (2017) Hyperandrogenemia induced by letrozole treatment of pubertal female mice results in hyperinsulinemia prior to weight gain and insulin resistance. Endocrinology 158:2988–3003
Fenkci SM, Fenkci V, Oztekin O, Rota S, Karagenc N (2008) Serum total l-Carnitine levels in non-obese women with polycystic ovary syndrome. Hum Reprod 23:1602–1606
Behrman H (2001) Oxidative stress and the ovary*1. J Soc Gynecol Investig 8:S40–S42
Sulaiman M, Al-Farsi Y, Al-Khaduri M, Saleh J, Waly M (2018) Polycystic ovarian syndrome is linked to increased oxidative stress in Omani women. Int J Womens Health 10:763–771
Ilic MB, Dimitrijevic A, Ilic I. Roles of Oxidative Stress in Polycystic Ovary Syndrome. Serbian J Exp Clin Res [Internet]. 2019; 0:96–107. Available from: https://www.sciendo.com/article/10.2478/sjecr-2019-0059
Amato G (2003) Serum and follicular fluid cytokines in polycystic ovary syndrome during stimulated cycles. Obstet Gynecol 101:1177–1182
Fang Y-Z, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18:872–879
Kolodziejczyk J, Saluk-Juszczak J, Wachowicz B (2011) l-Carnitine protects plasma components against oxidative alterations. Nutrition 27:693–699
Gülçin I (2006) Antioxidant and antiradical activities of l-Carnitine. Life Sci Pergamon 78:803–811
Lee BJ, Lin JS, Lin YC, Lin PT (2014) Effects of l-Carnitine supplementation on oxidative stress and antioxidant enzymes activities in patients with coronary artery disease: a randomized, placebo-controlled trial. Nutr J 13:1–7
Di Emidio G, Rea F, Placidi M, Rossi G, Cocciolone D, Virmani A et al (2020) Regulatory functions of l-Carnitine, acetyl, and propionyl l-Carnitine in a PCOS mouse model: focus on antioxidant/antiglycative molecular pathways in the ovarian microenvironment. Antioxidants 9:1–17
Yang H, Lee SY, Lee SR, Pyun BJ, Kim HJ, Lee YH et al (2018) Therapeutic effect of Ecklonia cava extract in letrozole-induced polycystic ovary syndrome rats. Front Pharmacol 9:1325. https://doi.org/10.3389/fphar.2018.01325/full
Ortega I, Sokalska A, Villanueva JA, Cress AB, Wong DH, Stener-Victorin E et al (2013) Letrozole increases ovarian growth and Cyp17a1 gene expression in the rat ovary. Fertil Steril 99:889–896
Guo I-C, Shih M-C, Lan H-C, Hsu N-C, Hu M-C, Chung B (2007) Transcriptional regulation of human CYP11A1 in gonads and adrenals. J Biomed Sci 14:509–515. https://doi.org/10.1007/s11373-007-9177-z
Saddick SY (2020) Identifying genes associated with the development of human polycystic ovary syndrome. Saudi J Biol Sci 27:1271–1279
Warsy AS, Almukaynizi FB, AlDaihan S, Alam S, Daghastani M (2017) Genetic polymorphisms in aromatase (CYP19) gene and cancer. Genet Polymorphisms. InTech. http://www.intechopen.com/books/genetic-polymorphisms/genetic-polymorphisms-in-aromatase-cyp19-gene-and-cancer
Azouz AA, Ali SE, Abd-Elsalam RM, Emam SR, Galal MK, Elmosalamy SH et al (2021) Modulation of steroidogenesis by Actaea racemosa and vitamin C combination, in letrozole induced polycystic ovarian syndrome rat model: promising activity without the risk of hepatic adverse effect. Chin Med 16:36. https://doi.org/10.1186/s13020-021-00444-z
Townson DH, Wang XJ, Keyes PL, Kostyo JL, Stocco DM (1996) Expression of the steroidogenic acute regulatory protein in the corpus luteum of the rabbit: dependence upon the luteotropic hormone, estradiol-17 beta. Biol Reprod 55:868–874
Yang H, Kim HJ, Pyun B-J, Lee HW (2018) Licorice ethanol extract improves symptoms of polycytic ovary syndrome in letrozole-induced female rats. Integr Med Res 7:264–270
Lee Y, Yang H, Lee S, Kwon S, Hong E-J, Lee H (2018) Welsh onion root (Allium fistulosum) restores ovarian functions from letrozole induced-polycystic ovary syndrome. Nutrients 10:1430
Erickson GF, Magoffin DA, Dyer CA, Hofeditz C (1985) The ovarian androgen producing cells: a review of structure/function relationships. Endocr Rev 6:371–399
Zaidi SK, Shen WJ, Cortez Y, Bittner S, Bittner A, Arshad S et al (2021) SOD2 deficiency-induced oxidative stress attenuates steroidogenesis in mouse ovarian granulosa cells. Mol Cell Endocrinol 519:110888
Pillai P, Pandya C, Gupta S, Gupta S (2010) Biochemical and molecular effects of gestational and lactational coexposure to lead and cadmium on ovarian steroidogenesis are associated with oxidative stress in F1 generation rats. J Biochem Mol Toxicol 24:384–394
Rezaei N, Mardanshahi T, Shafaroudi MM, Abedian S, Mohammadi H, Zare Z (2018) Effects of l-carnitine on the follicle-stimulating hormone, luteinizing hormone, testosterone, and testicular tissue oxidative stress levels in streptozotocin-induced diabetic rats. J Evid-Based Integr Med 23:1–10
Salem MA, Ismail RS, Zaki HF, Arafa HMM, El-Khatib ASN (2021) L-Carnitine extenuates endocrine disruption, inflammatory burst and oxidative stress in carbendazim-challenged male rats via upregulation of testicular StAR and FABP9, and downregulation of P38-MAPK pathways. Toxicology 457:152808
Zuo T, Zhu M, Xu W (2016) Roles of oxidative stress in polycystic ovary syndrome and cancers. Oxid Med Cell Longev. https://doi.org/10.1155/2016/8589318
Borrás C, Gambini J, López-Grueso R, Pallardó FV, Viña J (2010) Direct antioxidant and protective effect of estradiol on isolated mitochondria. Biochim Biophys Acta - Mol Basis Dis 1802:205–211
Ismail AM, Hamed AH, Saso S, Thabet HH (2014) Adding l-Carnitine to clomiphene resistant PCOS women improves the quality of ovulation and the pregnancy rate. A randomized clinical trial. Eur J Obstet Gynecol Reprod Biol 180:148–152
Shen HR, Xu X, Li XL (2021) Berberine exerts a protective effect on rats with polycystic ovary syndrome by inhibiting the inflammatory response and cell apoptosis. Reprod Biol Endocrinol 19:1–11
Kalhori Z, Mehranjani MS, Azadbakht M, Shariatzadeh MA (2019) L -Carnitine improves endocrine function and folliculogenesis by reducing inflammation, oxidative stress and apoptosis in mice following induction of polycystic ovary syndrome. Reprod Fertil Dev 31:282–293
Yang R, Chen J, Wang L, Deng A (2018) LncRNA BANCR participates in polycystic ovary syndrome by promoting cell apoptosis. Mol Med Rep 19:1581–1586. https://doi.org/10.3892/mmr.2018.9793
Tao T, Wu P, Wang Y, Liu W (2018) Comparison of glycemic control and β-cell function in new onset T2DM patients with PCOS of metformin and saxagliptin monotherapy or combination treatment. BMC Endocr Disord 18:14. https://doi.org/10.1186/s12902-018-0243-5
Shorakae S, Teede H, de Courten B, Lambert G, Boyle J, Moran L (2015) The emerging role of chronic low-grade inflammation in the pathophysiology of polycystic ovary syndrome. Semin Reprod Med 33:257–269. https://doi.org/10.1055/s-0035-1556568
Salehi E, Aflatoonian R, Moeini A, Yamini N, Asadi E, Khosravizadeh Z et al (2017) Apoptotic biomarkers in cumulus cells in relation to embryo quality in polycystic ovary syndrome. Arch Gynecol Obstet 296:1219–1227. https://doi.org/10.1007/s00404-017-4523-5
Hassan N, Rashad M, Elleithy E, Sabry Z, Ali G, Elmosalamy S (2022) l-Carnitine alleviates hepatic and renal mitochondrial-dependent apoptotic progression induced by letrozole in female rats through modulation of Nrf-2, Cyt c and CASP-3 signaling. Drug Chem Toxicol. https://doi.org/10.1080/01480545.2022.2039180
Yeh J, Kim HH (1996) Polycystic ovary syndrome (PCOS): the possible roles of apoptosis in human granulosa cells. In: Chang RJ (ed) Polycystic ovary syndrome. Serono Symposia USA. Springer, New York, NY, pp 51–70. https://doi.org/10.1007/978-1-4613-8483-0_4
Acknowledgements
Not applied for this manuscript.
Funding
This research didn’t receive any specific grant from funding agencies in the public, commercial, or nonprofit sectors.
Author information
Authors and Affiliations
Contributions
SHE and NHH conceived and designed the experiments, performed hormonal and metabolic profiles as well as antioxidant assays, analyzed the data, provided reagents/materials/analysis tools, and wrote the paper. EMME and ZSOA performed the histomorphometric and immunohistochemical analysis and provided reagents/materials/analysis tools required. MMR and GEA performed gene expression analysis and provided reagents/materials/analysis tools required. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal procedures and experiment were approved under the consent of the Institutional Animal Care and Use Committee (IACUC), Cairo University, Egypt (CUIIF921) and in line with the guidelines for Guide for the Care and Use of Laboratory Animals: Eighth Edition.
Consent for publication
Not applicable.
Competing interests
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elmosalamy, S.H., Elleithy, E.M.M., Ahmed, Z.S.O. et al. Dysregulation of intraovarian redox status and steroidogenesis pathway in letrozole-induced PCOS rat model: a possible modulatory role of l-Carnitine. Beni-Suef Univ J Basic Appl Sci 11, 146 (2022). https://doi.org/10.1186/s43088-022-00329-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43088-022-00329-6