Abstract
Background
Broad applications of nanoparticles have invoked the concerns about their impacts on living organisms. This study aimed to evaluate the possible changes that could take place in the liver of feral pigeons (Columba livia domestica) after oral ingestion of zinc oxide nanoparticles (nano-ZnO).
Methods
Pigeons were acclimatized to the laboratory conditions with a photoperiod of 12:12 h at 20 ± 2 ˚C for 14 days and maintained on seed mixture and clean tap water ad libitum. The birds were randomly assigned to five groups of 10, including one control group and four experimental groups orally receiving 0, 30, 50, and 75 mg/kg b.w. of nano-ZnO through oral gavage for 7 and 14 consecutive days.
Results
The oxidative stress (OS) biomarkers, namely lipid peroxidation content and catalase activity in the liver samples, and the level of hepatic necrosis markers (ALT, AST, and ALP) in the blood sera were increased in a concentration-dependent manner. Meanwhile, the total antioxidant capacity of liver samples measured by FRAP test was reduced. Histopathological changes revealed inflammatory cell infiltration, swelling area, vacuolization, and expansion of interstitial spaces in liver samples exposed to 75 mg/kg nano-ZnO.
Conclusions
Nano-ZnO induced obvious hepatotoxicity in the liver of pigeons, where the OS pathway may be the potential mechanism underlying this toxicity.
Similar content being viewed by others
Background
For many years, birds because of their phylogenetic distance from man have not been used as suitable models for toxicological evaluations; however, these animals and man have a virtual food relationship, and therefore, environmental factors affecting the health of bird populations have the potential of an impact on the food safety of man [1]. Nowadays, there is a growing need to use nanoparticles (NPs) in biomedical and industrial applications. More studies have indicated that manufactured NPs due to high surface area to volume ratio and abundant reactive sites on the surface are toxic to organisms [2]. The NPs may leak into the environment in their life cycles including production, consumption, and disposal processes. Of all the NPs, metallic NPs (MNPs) have considerable commercial interest due to unique properties such as small size and greater surface area to volume; entering the body via the skin, inhalation, and gastrointestinal tract; and circulating into the body, eventually accumulating in various organs [3]. There is a lack of information regarding the magnitude of released MNPs and their impact on living organisms in the terrestrial systems. Therefore, there is an urgent need for information on the ecological risks of MNPs [4].
Zinc oxide nanoparticles (nano-ZnO) as MNPs have attracted attention owing to their applications in veterinary sciences due to their wound healing, antibacterial, and angiogenic properties, etc. [3]. Once nano-ZnO is absorbed, the elevated level of Zn2+ can be recorded in the liver, adipose tissue, and pancreas [2]. Common provenances of zinc toxicity in birds include the coating on galvanized wire cages, food, and water dishes, etc. [5]. In Aves, plasma zinc concentrations above 4 ppm are suggestive of toxicosis. However, birds may be clinically affected without high plasma zinc concentrations [6]. The MNPs may stimulate the reactive oxidative species (ROS) generation by modifying the cellular metabolism [6] or damaging the antioxidant defense system, resulting in oxidative damage to the proteins, lipids, and DNA [7, 8].
The biomarkers are determined as molecular, biochemical, physiological, or histological indicators of contaminant effects [9, 10]. The use of biomarkers can be a complementary approach to standard toxicity tests because it provides more information about the organism’s stress response to individual and/or combined toxicants [10]. The antioxidant systems, enzymatic and non-enzymatic antioxidants, play a vital role in defense against cellular oxidative damage caused by ROS [11, 12]. Oxidative stress (OS) is defined as an imbalance between the excessive ROS generation and its quenching by antioxidant defense systems. Once OS occurs, the lipid peroxidation (LPO) process and its by-product of malondialdehyde (MDA) are the initial steps of cellular membrane damage and are considered to be valuable indicators of oxidative damage of cellular components. Excessive accumulation of MDA changes the permeability of cell membranes and disrupts DNA and proteins resulting in cell death [12, 13].
Biochemical methods of monitoring chemical pollution have been widely used with avian models, and some of the methods may have direct application to health effects [1]. The feral pigeons (Columba livia domestica) are appropriate animal models in ecotoxicology studies. Due to their commercial importance, these animals must be protected from environmental pollutants [14]. Birds exposed to NPs show adverse alterations in the function of certain tissues [15, 16], but the ecotoxicological data on nano-ZnO are just emerging and scanty.
The liver as a main organ in the body possesses abilities of detoxification, storage, secretion, synthesis, etc. MNP intoxication may lead to liver structural changes and dysfunction. Markers such as a significant increase of alkaline phosphatase (ALP) with aspartate aminotransferase (AST) in the blood serum indicate liver injury. These markers mainly manifested in liver structural changes causing metabolic dysfunction. MNPs could cause liver toxicity via mechanisms including inflammation, OS, and different types of cell death [17]. Given their diffusion, there is insufficient information in the consulted literature regarding the toxicity of nano-ZnO in birds and requires further investigation. Therefore, this study aimed to evaluate the hepatic function and structure in feral pigeons (Columba livia domestica) exposed to zinc oxide nanoparticles under laboratory conditions.
Methods
Chemicals and animal model
The nano-ZnO white powder with a diameter size of ≤ 20 nm was purchased from Nano-shop Company, Tehran, Iran. According to the company brochure, the shape and purity of tested nano-ZnO were spherical and 99%, respectively. Other chemicals in reagent grade were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). The nano-ZnO suspension was made using deionized water to minimize ROS generation during sonication, and the cold water bath was used to minimize the heating of the particles. Then, the suspension was sonicated for 20 min in a bath sonicator (SONOPULS HD 2070.2, BANDELIN electronic GmbH & Co. KG, Germany) and was vortexed for 1 min before every administration [1].
Healthy feral pigeons (Columba livia domistica), with a mean weight of about 300–350 g, were purchased from a local bird market in Shahrekord, Iran. Birds reared in clean wooden cages with metal nets in the Animal Physiology Laboratory, Department of Animal Sciences, University of Shahrekord, Iran. Thereafter, all birds were acclimatized to the laboratory conditions with a photoperiod of 12:12 h at 20 ± 2˚C for 14 days. Seed mixture and tap water were provided ad libitum.
Experimental design
After acclimatization, birds were randomly divided into five groups having 10 pigeons each, including one control and four experimental groups (in triplicate). The experimental groups received 30, 50, and 75 mg/kg b.w. of nano-ZnO solutions for 7 and 14 consecutive days (1 ml/bird) through an oral gavage cannula (Fig. 1). The control group received tap water for the same time periods. During the experimental period, pigeons were monitored for clinical and physical symptoms.
Biochemical assays
The blood samples, about 2–3 ml, were collected via puncturing from the brachial vein. The samples were then transferred to the test tubes without any anticoagulant agent and centrifuged at 3000 rpm for 15 min [18]. The resulting blood sera were used to determine the activities of the hepatic necrosis markers AST, alanine aminotransferase (ALT), and ALP by related commercial kits. Then, the pigeons were euthanized by cervical dislocation. The liver samples were taken out and, after perfusion with normal saline, were homogenized in chilled 50-mM potassium phosphate buffer (1:8, w/v; pH 7.0) containing 0.5 mM EDTA. The resulting homogenate was divided into two parts: (i) for measuring MDA and protein content and (ii) for centrifugation at 10,000 rpm for 10 min at 4˚C to obtain the post-mitochondrial supernatant for CAT assay [12].
MDA level
The TBARS assay [19] was performed to determine the MDA/LPO level in the supernatant of liver homogenates. In brief, 500 µl of aliquots was mixed with 1 ml of chilled 30% trichloroacetic acid (TCA) and centrifuged at 5000 × g for 10 min. The resulting aliquot reacted with thiobarbituric acid (TBA) and MDA absorbance was recorded at 532 nm, using a UV-1700 spectrophotometer (Shimadzu, Japan). The MDA level was expressed as n moles MDA per mg protein min.
CAT activity
The changes in CAT activity in the assay mixture containing H2O2 phosphate buffer and supernatant of liver homogenate were recorded at 240 nm. The activity was expressed as IU H2O2 decomposed per mg protein min [20].
Total protein
To measure total protein content, the Bradford method was used using bovine serum albumin (BSA) as standard at an absorbance of 595 nm. Results were expressed as milligram protein per milliliter of whole organ extract [21].
Hepatic necrosis markers
The commercial standard kits were obtained from Sigma-Aldrich Company to measure the level of hepatic necrosis markers namely ALT (#MAK052), AST (#ERMAD457IFCC), and ALP (#AP0100) in the pigeon blood sera.
Total Antioxidant Capacity (TAC) content
Ferric reducing ability of plasma (FRAP) was performed to measure changes in the TAC of liver samples. During the assay, yellow ferric tripyridyltriazine complex (Fe (III)-TPTZ) is reduced to blue ferrous complex (Fe (II)-TPTZ) by the action of electron-donating antioxidants in liver samples. At the end of the experiment, the resulting blue color is linearly related to the total reducing capacity of antioxidants in liver samples. Briefly, FRAP reagent (1.5 ml) was incubated at 37 ˚C for 5 min. Then, this solution was mixed with 0.1 ml of diluted liver sample (1:10) and incubated at 37 °C for 10 min. The end product ferrous tripyridyltriazine complex (Fe2+-TPTZ) had a blue color. The absorbance of the reaction mixture was measured at 593 nm for 4 min. The standard was FeSO4.7H2O in methanol. The absorbance values obtained were expressed as m moles Fe2+ produced per mL [22].
Histopathological examinations
For the histopathology of the liver samples, after the narcotization and decapitation, the pigeons were dissected. The liver tissues were taken and placed into 10% of formalin. Then, it was dehydrated in graded alcohol concentrations and embedded in paraffin. Sections of 5 microns were prepared, stained with hematoxylin and eosin (H&E) technique, and evaluated with a light microscope (Leica DM-2500 biosystem) for assessment of histopathological changes.
Statistical analyses
Data were presented as mean ± standard deviation (SD) (n = 10/group). The SPSS software (2019 v26) was used to perform statistical analyses using standard ANOVA techniques, followed by Tukey’s HSD post hoc test with p < 0.05 as a significant probability level. Pearson’s correlation coefficient analysis was conducted to determine the strength of association among parameters.
Results
During the experimental period, no clinical and physical symptoms were monitored in the tested pigeons.
MDA level
An increase was recorded in the MDA/LPO level over a time exposure of 7 and 14 days, compared with the controls (Fig. 2). The MDA level increased in a concentration-dependent manner. The highest MDA level was obtained in 75 mg/kg of nano-ZnO-treated groups after 14 days by 95.65% (p < 0.05). The elevated MDA level was greater in groups with 14 than 7-day exposures (Fig. 2). A positive correlation was observed between nano-ZnO increasing concentrations and elevated MDA level (r = 0.81, p < 0.05) after a 14-day exposure to nanoparticles.
CAT activity
According to Fig. 3, the CAT activity was elevated in all treated groups compared to controls in a concentration-dependent trend. The elevation was found to be significant only in 75 mg/kg groups after 14 days (24.14%, p < 0.05). The correlation between nano-ZnO increasing level and elevated activity of CAT was r = 0.87 (p < 0.05) (Fig. 3) following a 14-day exposure to nano-ZnO.
Hepatic necrosis markers
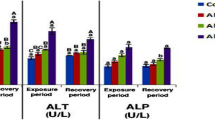
As shown in Fig. 4, the level of ALT increased concentration dependently in the blood sera samples. This elevation was significant (p < 0.05) in all treated groups except 30 mg/kg after 7 days. The highest ALT level was obtained in 75 mg/kg nano-ZnO-treated groups after 14 days by 58.04% (p < 0.05). A strong correlation (r = 0.91, p < 0.001) was observed between increased ALT levels and elevated nanoparticle concentrations after a 14-day exposure.
Following 7 and 14 days, the level of AST in all groups exposed to nano-ZnO was found to be increased significantly (p < 0.05). The highest level obtained for AST was in 75 mg/kg nano-ZnO-treated groups after 14 days by 39.31% (p < 0.05). A positive value of correlation between increasing nano-ZnO concentrations and elevated AST level was observed (r = 0.93, p < 0.001) after a 14-day exposure (Fig. 5).
The results showed that there was a clear concentration–response relationship between nano-ZnO treatments and elevated levels of ALP in the pigeon blood sera (Fig. 6). It was found that the increase in ALP level was greater in 14 than in 7-day exposed groups. In addition, the highest level of ALP was observed in the 75 mg/kg nano-ZnO-treated group after 14 days by 36.73% (p < 0.05). The correlation between MDA increasing level and elevated ALP level was r = 0.82 (p < 0.05) following a 14-day exposure (Fig. 6).
TAC content
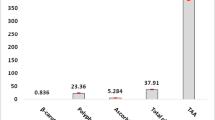
According to the results in Fig. 7, the total antioxidant capacity (TAC) values of liver samples in feral pigeons exposed to nano-ZnO after 7 and 14 days were found to be reduced upon an increase in the nanoparticle concentrations. It was found that the reduction in TAC values was greater in 14 than in 7-day exposed groups. In addition, the lowest values of TAC of liver samples were observed in 75 mg/kg nano-ZnO-treated groups after 14 days by 44.74% (p < 0.05) (Fig. 8). A negative correlation between TAC value and increasing nano-ZnO concentrations was observed in liver samples by r = -0.91 (p < 0.001) following a 14-day exposure (Fig. 7).
Histopathological changes in microscopic view of the liver of feral pigeon in control group with normal structure (A, 10 ×) and treated with 75 mg/kg of nano-ZnO (B 10 × , C, D 40 ×) stained with H&E. CV central vein, L leukocyte infiltration (inflammation), P normal hepatic parenchyma, RBC red blood cells, S hepatocyte swelling area, VE vacuolization and expansion of interstitial spaces
Histopathological examinations
Figure 8A–D deals with the histopathological observations in pigeon liver samples. Normal architecture in parenchyma and the well-arranged nature of hepatocytes without any inflammatory cell (leukocytes) infiltrations in the interstitial spaces are depicted in Fig. 8A as the control group. Our results indicate that tissue damages in the pigeon liver samples were only seen in pigeons treated with higher concentrations of nano-ZnO viz. 75 mg/kg after 14 days of exposure including inflammatory cell infiltration, swelling area, vacuolization, and expansion of interstitial spaces (Fig. 8B, C, D). These tissue changes, somewhere less, and somewhere more are observed in all specimens of feral pigeons treated with a higher concentration of nano-ZnO compared to the control group where these changes were not present.
Discussion
It is inevitable that during their use, engineered MNPs will be released into the environment. Despite the greater attention on MNPs’ increased ecotoxicological risks for terrestrial organisms, evidence regarding their toxic impacts on feral pigeons (Columba livia domestica) is still insufficient, and in this research, we defined the hepatotoxicity of nano-ZnO in feral pigeons. NP-mediated ROS generation initiates a sequence of pathological events, including inflammation, genotoxicity, and carcinogenesis, and is modulated by physicochemical features of NPs, such as size, charge, surface area, and chemical structure [3, 23]. Interestingly, in bacteria, Streptococcus pyogenes, findings from Liang et al. [24] evidenced that nano-ZnO induces significant ROS generation, LPO propagation, and cell wall disruption with concomitant thiol depletion.
MDA level
The MDA/LPO level is used as a biomarker of OS which reflects indirectly the degree of damage from ROS insult [12]. It has been indicated that exposure to nano-ZnO leads to ROS generation and activation-specific signal transduction pathways in target cells. An imbalance in the ROS production and antioxidant defense system of cells results in interferences with normal cellular processes which cause several different outcomes leading to cell death [25]. There are some reports that attribute the toxicity of nano-ZnO to particle dissolution and subsequent release of Zn2+ ions that lead to ROS-mediated injuries such as lysosomal and mitochondria damage and ultimately cell death [26]. Our results also indicate an elevation in the MDA level, in a concentration-dependent manner. Augmented MDA level reflects the induction of LPO chain reaction followed by oxidative damages in pigeon liver samples exposed to nano-ZnO. Accumulation of MDA in organism’s tissues results in changes in the permeability of bio-membranes leading to cell dysfunction [11]. In agreement with our results, it has been demonstrated that nano-ZnO caused a significant increase in the splenic and thymic MDA levels in rats [1]. In addition, nano-ZnO consumed antioxidants in the liver and induced ROS to affect the structure and function of the endoplasmic reticulum of mouse hepatocytes, which is believed to be related to apoptosis and autophagy [27].
CAT activity
In the present study, the activity of the antioxidant enzyme CAT was found to be elevated which clearly indicated that nano-ZnO was able to induce OS and related damages. CAT is an enzymatic antioxidant that decomposes H2O2 and protects the tissue from highly reactive hydroxyl radicals and reduction of the cell membrane fluidity [28]. However, we can assume that such an increase in the CAT activity of liver samples may reflect the detoxification reactions in nano-ZnO-treated pigeons. El-Bahr et al. (2020) demonstrated that in Japanese quails upon stimulation with nano-ZnO to eliminate oxidative damage due to their toxic substances, the CAT activity was increased. They also found that the liver retained a higher nano-ZnO concentration than that of brain tissue [29]. On the basis of Mittler’s ROS theory, it can be postulated that nano-ZnO exposure resulted in the production of excessive H2O2 and lipid radicals in pigeons. In order to adapt to the oxidative status, the CAT activity was increased [30]. Other researchers also explained the role of nano-ZnO in enhancing the antioxidant status in chickens [31].
Hepatic necrosis markers
Blood biochemistry parameters are important markers of physiological status in animal models. The current study demonstrated the increases in the level of ALT, AST, and ALP as pathological indicators. These are considered as hepatic necrosis markers, upsurge in their levels is a potential key indicator for diagnosing various hepatic injuries such as acute alcoholic hepatitis and cirrhosis [32]. Results from our study clearly indicate that the hepatotoxicity may be via necrotic changes induced by nano-ZnO, which in turn leads to increased cell membrane permeability and release of these cytoplasmic enzymes into the pigeon blood circulation. Knowing these enzymes as hepatic injury markers was confirmed by close histopathological examination of the hepatic organ that exhibited hepatic tissue injuries and enhanced serum enzyme activities linked with abnormal physiological ailment [33]. This depiction is met with our results and findings of Fazilati et al. [34] who observed the elevation in ALT and AST with the exposure to Zn2+ as a trace metal in the Japanese quail. It is reported that continuous exposure to nano-ZnO had toxicity in rats leading to an increase in liver enzymes, such as ALT, AST, and ALP at high doses [35]. On the other hand, the mRNA expression of proinflammatory cytokines such as IL-1β, IL-10, and TNF-α was found to be potentiated following nano-ZnO supplementation in rats [1]. Increased TNFα may be attributed to the OS induced by nano-ZnO poisoning which is evidenced by a significant increase in MDA level and decreased reduced glutathione (GSH) level in the blood serum of exposed birds [36]. Regarding nano-ZnO toxicity, it is also reported that these NPs dissolve in the lysosomes which leads to the reduction of pH level. Reduced pH affects lysosomal enzymes involved in protein digestion and increases intra-lysosomal Zn2+ level, which leads to lysosomal destabilization and cell membrane instability [37].
TAC content
The TAC value refers to the antioxidative status of an organism and includes the synergic and redox interactions between the different molecules present in the biological fluids [38]. Because the antioxidants do not work alone, the cooperation among the different antioxidants may provide greater protection against ROS insults. Thus, TAC may give more biologically relevant information than that obtained from individual antioxidants [39]. To counteract the oxidative damage, the organism’s body is well-equipped with antioxidant defense mechanisms [40]. Cellular non-enzymatic antioxidant defense potentially includes GSH, vitamins C and E, etc. Amongst them, the GSH plays a vital role in the protection of cells against oxidative injury. The depletion of GSH by toxicants could be due to the direct conjugation of their metabolites with GSH leading to OS [41]. Our results indicate that the TAC value in liver samples of treated pigeons with nanoparticles was decreased in a concentration-dependent manner which can be attributed to the nano-ZnO ability to lower the power of antioxidant defense systems in the liver samples. Hence, it can be concluded that in our study, induction of OS and excessive ROS production in the pigeon’s body might be the reason for the antioxidant power depletion (lowered TAC value) in the liver samples under the stress of nano-ZnO intoxication which come due to imbalance in homeostasis between pro-oxidants and antioxidants.
Histopathological examinations
The histologically based endpoints have been shown to be sensitive indicators of the sub-lethal toxicity of chemicals [42]. The liver has the critical job of maintaining the body’s metabolic homeostasis. Our results indicate that the liver samples of pigeons treated with higher concentrations of nano-ZnO viz. 75 mg/kg were found to have different tissue damages. Resulting damages may lead to hepatic dysfunction and the release of hepatic necrosis markers (ALT, AST, and ALP) into the blood circulation as well. Apparently, the histopathological changes in the liver samples were the result of tissue response to the induction of OS signaling pathways due to an imbalance between ROS production and antioxidant defense function [7, 43]. We assume that the liver is a sensitive organ to nano-ZnO toxicity, as evidenced here by elevated hepatic necrosis markers and alterations of other biochemical parameters, such as LPO and enzymatic antioxidant activities after nano-ZnO oral ingestion. Our findings about the histology of the liver of feral pigeons are in accordance with the results of Dashti et al. [44] and Wang et al. [45]. In broiler chicks, exposure to high doses of ZnO resulted in pronounced changes in the liver such as connective tissue hyperplasia, liver cell hypertrophy, and accumulation of inflammatory cells [46].
Conclusion
The findings of the present study call to reconsider the potential effects of nano-ZnO on pigeons. We showed that the nano-ZnO is injurious to the pigeon’s health at different concentration levels. Overall, our findings demonstrated that nano-ZnO could induce OS and have adverse effects on liver function and structure in the exposed pigeons. However, studies regarding the effect of nano-ZnO in pigeons are insufficient and require further investigation.
Availability of data and materials
Data are available with the corresponding author to be presented upon request.
References
Hill EF, Hoffman DJ (1984) Avian models for toxicity testing. J Am Coll Toxicol 3(6):357–376. https://doi.org/10.3109/10915818409104398
Abass MA, Selim SA, Selim AO, El-Shal AS, Gouda ZA (2017) Effect of orally administered zinc oxide nanoparticles on albino rat thymus and spleen. IUBMB Life 69(7):528–539. https://doi.org/10.1002/iub.1638
Umrani R, Paknikar KM (2014) Zinc oxide nanoparticles show antidiabetic activity in streptozotocin-induced types 1 and 2 diabetic rats. Nanomed 9(1):89–104. https://doi.org/10.2217/nnm.12.205
Saddick S, Afifi M, Abu Zinada OA (2017) Effect of zinc nanoparticles on oxidative stress-related genes and antioxidant enzymes activity in the brain of Oreochromis niloticus and Tilapia zillii. Saudi J Biol Sci 24(7):1672–1678. https://doi.org/10.1016/j.sjbs.2015.10.021
Lightfoot TL, Yeager JM (2008) Pet bird toxicity and related environmental concerns. Vet Clin Exot Anim 11:229–259. https://doi.org/10.1016/j.cvex.2008.01.006
Puschner B, St. Judy L, Galey FD (1999) Differences normal and toxic zinc concentrations in serum/plasma and liver of psittacines with respect to genus. J Vet Diagn Invest 11(6):522–527. https://doi.org/10.1177/104063879901100606
Kelly SA, Havrilla CM, Brady TC, Abramo KH, Levin ED (1998) Oxidative stress in toxicology: established mammalian and emerging piscine model systems. Environ Health Perspect 106(7):375–384. https://doi.org/10.1289/ehp.98106375
Long TC, Saleh N, Tilton RD, Lowry GV, Veronesi B (2006) Titanium dioxide (P25) produces reactive oxygen species in immortalized brain microglia (BV2): implications for nanoparticle neurotoxicity. Environ Sci Technol 40(14):4346–4352. https://doi.org/10.1021/es060589n
Van Gestel CAM, Van Brummelen TC (1996) Incorporation of the biomarker concept in ecotoxicology calls for a redefinition of terms. Ecotoxicol 5:217–225. https://doi.org/10.1007/BF00118992
Hagger JA, Galloway TS, Langston WJ, Jones MB (2009) Application of biomarkers to assess the condition of European Marine Sites. Environ Pollut 157(7):2003–2010. https://doi.org/10.1016/j.envpol.2009.02.038
Halliwell B, Gutteridge JMC (1984) Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 219(1):1–14. https://doi.org/10.1042/bj2190001
Arabi M, Mahmoodian F (2023) Comparative toxicity of fresh and expired butachlor to earthworms Eisenia fetida in natural soil: biomarker responses. Environ Chem Ecotoxicol 5:108–119. https://doi.org/10.1016/j.enceco.2023.03.002
Ferreira M, Moradas-Ferreira P, Reis-Henriques MA (2005) Oxidative stress biomarkers in two resident species, mullet (Mugil cephalus) and flounder (Platichthys flesus), from a polluted site in River Douro Estuary. Portugal Aquat Toxicol 71(1):39–48. https://doi.org/10.1016/j.aquatox.2004.10.009
Ismaeil SAA, Emam H (2023) Morphological, histological and histochemical comparative investigations of the liver in young homing pigeon (Columba Livia Domestica) and cattle egret (Bubulcus Ibis). Adv Anim Vet Sci 10(3):607–620. https://doi.org/10.17582/journal.aavs/2022/10.3.607.620
Ahmadi F, Ebrahimnezhad Y, Sis NM, Ghalehkandi JG (2013) The effects of zinc oxide nanoparticles on performance, digestive organs and serum lipid concentrations in broiler chickens during starter period. Int J Biosci 3(7):23–29. https://doi.org/10.12692/ijb/3.7.23-29
Prasad K, Lauridsen C, Engberg RM, Vadalasetty R, Kutwin MP et al (2018) Influence of silver nanoparticles on growth and health of broiler chickens after infection with Campylobacter jejuni. BMC Vet Res 14(1):1. https://doi.org/10.1186/s12917-017-1323-x
Yao Y, Zang Y, Qu J, Tang M, Zhang T (2019) The toxicity of metallic nanoparticles on liver: the subcellular damages, mechanisms, and outcomes. Int J Nanomed 14:8787–8804. https://doi.org/10.2147/IJN.S212907
Kelly L, Alworth L (2013) Techniques for collecting blood from the domestic chicken. Lab Anim 42:359–361. https://doi.org/10.1038/laban.394
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Luck H (1965) Catalase. In: Bergmeyer HU (ed) Method of enzymatic analysis, Academic Press, New York and London, 885–894 https://doi.org/10.1016/B978-0-12-395630-9.50158-4
Bradford MM (1976) A rapid sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239(1):70–76. https://doi.org/10.1006/abio.1996.0292
Dayem AA, Hossain MK, Lee SB, Kim K, Saha SK, Yang GM, Choi HY, Cho SG (2017) The role of Reactive Oxygen Species (ROS) in the biological activities of metallic nanoparticles. Int J Mol Sci 18:120. https://doi.org/10.3390/ijms18010120
Tong Liang SX, Wong LS, Lim YM, Leed PF, Djearamane S (2020) Effects of zinc oxide nanoparticles on Streptococcus pyogenes. S Afr J Chem Eng 34:63–71. https://doi.org/10.1016/j.sajce.2020.05.009
Hwang C, Choi MH, Kim HE, Jeong SH, Park JU (2022) Reactive oxygen species-generating hydrogel platform for enhanced antibacterial therapy. NPG Asia Mater 14:72. https://doi.org/10.1038/s41427-022-00420-5
Xia T, Kovochich M, Liong M, Mädler L, Gilbert B, Shi H, Yeh JI, Zink JI, Nel AE (2008) Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2(10):2121–2134. https://doi.org/10.1021/nn800511k
Yang X, Shao H, Liu W, Gu W, Shu X, Mo Y, Chen X, Zhang Q, Jiang M (2015) Endoplasmic reticulum stress and oxidative stress are involved in ZnO nanoparticle-induced hepatotoxicity. Toxicol Lett 234(1):40–49. https://doi.org/10.1016/j.toxlet.2015.02.004
Winston GW, Di Giulio RT (1991) Prooxidant and antioxidant mechanisms in aquatic organisms. Aquat Toxicol 19(2):137–161. https://doi.org/10.1016/0166-445X(91)90033-6
El-Bahr SM, Shousha S, Albokhadaim I, Shehab A, Khattab W et al (2020) Impact of dietary zinc oxide nanoparticles on selected serum biomarkers, lipid peroxidation and tissue gene expression of antioxidant enzymes and cytokines in Japanese quail. BMC Vet Res 16:349. https://doi.org/10.1186/s12917-020-02482-5
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9):405–410. https://doi.org/10.1016/s1360-1385(02)02312-9
Zhao CY, Tan SX, Xiao XY, Qiu XS, Pan JQ et al (2014) Effects of dietary zinc oxide nanoparticles on growth performance and antioxidative status in broilers. Biol Trace Elem Res 160(3):361–367. https://doi.org/10.1007/s12011-014-0052-2
Adeniyi AF, Adeleye J, Adeniyi CY (2010) Diabetes, sexual dysfunction and therapeutic exercise: a 20 year review. Curr Diabetes Rev 6(4):201–206. https://doi.org/10.2174/157339910791658907
Kori-Siakpere O, Ubogu EO (2008) Sub-lethal hematological effects of zinc on the freshwater fish, Heteroclarias sp. (Osteichthyes: Clariidae). Afr J Biotechnol 7(12):2068–2073. https://doi.org/10.5897/AJB07.706
Fazilati M (2013) Investigation toxicity properties of zinc oxide nanoparticles on liver enzymes in male rat. Eur J Exp Biol 3(1):97–103
Hosseini SM, Amani R, Moshrefi AH, Razavimehr SV, Aghajanikhah MH et al (2020) Chronic zinc oxide nanoparticles exposure produces hepatic and pancreatic impairment in female rats. Iran J Toxicol 14(3):145–154. https://doi.org/10.32598/ijt.14.3.626.1
Abu Zeid EH, Alam RTM, Ali SA, Hendawi MY (2019) Dose-related impacts of imidacloprid oral intoxication on brain and liver of rock pigeon (Columba livia domestica), residues analysis in different organs. Ecotoxicol Environ Saf 167:60–68. https://doi.org/10.1016/j.ecoenv.2018.09.121
Vandebriel RJ, De Jong WH (2012) A review of mammalian toxicity of ZnO nanoparticles. Nanotechnol Sci Appl 5:61–71. https://doi.org/10.2147/NSA.S23932
Pellegrini N, Vitaglione P, Granato D, Fogliano V (2020) Twenty-five years of total antioxidant capacity measurement of foods and biological fluids: merits and limitations. J Sci Food Agri 100(14):5064–5078. https://doi.org/10.1002/jsfa.9550
Ghiselli A, Serafini M, Natella F, Scaccini C (2000) Total antioxidant capacity as a tool to assess redox status: critical view and experimental data. Free Radic Biol Med 29(11):1106–1114. https://doi.org/10.1016/S0891-5849(00)00394-4
Haliwell B (1994) Free radicals, antioxidants and human diseases: curiosity, cause or consequence? Lancet 344(8924):721–724. https://doi.org/10.1016/S0140-6736(94)92211-X
Wang L, Zhang L, Niu Y, Sitia R, Wang CC (2014) Glutathione peroxidase 7 utilizes hydrogen peroxide generated by Ero1ato promote oxidative protein folding. Antioxid Redox Signal 20(4):545–556. https://doi.org/10.1089/ars.2013.5236
Muthukaruppan G, Janardhanan S, Vijayalakshmi G (2005) Sublethal toxicity of the herbicide butachlor on the earthworm Perionyx sansibaricus and its histological changes. J Soils Sediments 5(2):82–86. https://doi.org/10.1065/jss2004.09.111
Bbrown DM, Donaldson K, Borm PJ, Schins RP, Dehnhardt M et al (2004) Calcium and ROS-mediated activation of transcription factors and TNF-α cytokine gene expression in macrophages exposed to ultrafine particles. Am J Physiol-Lung Cell Mol Physiol 286:L344–L353. https://doi.org/10.1152/ajplung.00139.2003
Dashti HM, Mathew TC, Jadaon MM, Ashkanani E (1997) Zinc and liver cirrhosis: biochemical and histopathological assessment. Nutrition 13(3):206–212. https://doi.org/10.1016/S0899-9007(96)00403-0
Wang B, Feng WY, Wang TC, Jia G, Wang M, Shi JW, Zhang F, Zhao YL, Chai ZF (2006) Acute toxicity of nano- and micro-scale zinc powder in healthy adult mice. Toxicol Lett 161(2):115–123. https://doi.org/10.1016/j.toxlet.2005.08.007
Khanam S (2020) Toxicological effect of zinc on liver of broiler chicks. Egypt Liver J 10:21. https://doi.org/10.1186/s43066-020-00028-w
Acknowledgements
The authors kindly thank Shahrekord University, Shahrekord, Iran, for providing financial support to this study. We are also grateful to Dr. Mohammad Shadkhast for his valuable suggestions.
Funding
This study, as a MSc project, is financially supported by Shahrekord University, Iran (1389-91).
Author information
Authors and Affiliations
Contributions
MA designed and supervised the study and drafted the original manuscript. HRN did the investigation. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study protocol was approved by the Research Ethical Committee of Shahrekord University, Iran; SKU-FBS/1389-91. All animal experiments have complied with the ARRIVE guidelines 2.0 and carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986, and also with Directive 2010/63/EU revising Directive 86/609/EEC on the protection of animals used for scientific purposes was adopted on September 22, 2010.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arabi, M., Naseri, HR. Hepatic function and structure in feral pigeons (Columba livia domestica) exposed to zinc oxide nanoparticles. Egypt Liver Journal 13, 44 (2023). https://doi.org/10.1186/s43066-023-00278-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43066-023-00278-4