Abstract
Background
Polycystic ovary syndrome (PCOS) is a complex endocrine condition prevalent among a significant number of women during their reproductive years. Remarkably, 90–95% of women seeking infertility solutions due to anovulation are diagnosed with PCOS. Luteal phase support (LPS) is a crucial aspect of assisted reproductive technologies (ART). This systematic review aimed to evaluate the effectiveness of LPS in women with PCOS undergoing ART, with a focus on pregnancy rates as the primary endpoint.
Materials and methods
A systematic search was conducted on EMBASE, PubMed, and Scopus databases without language restrictions. We searched for studies up to August 1, 2023. The search strategy used terms related to PCOS and LPS. Clinical trials and cohort studies involving infertile women with PCOS undergoing ART were included. The Risk of Bias 2 (ROB2) and the Newcastle-Ottawa Scale (NOS) tool were used to assess the risk of bias.
Results
The review included five studies comprising a total of 818 patients. The studies used various ovulation induction medications, such as letrozole, clomiphene citrate, and human menopausal gonadotropin, in combination with different forms of progesterone for LPS (oral, intramuscular, and intravaginal). The overall results demonstrated inconsistent efficacy of LPS, with some studies showing significant improvements in pregnancy rates with LPS, while others showed no statistically significant difference.
Conclusion
The systematic review suggests that LPS may improve pregnancy rates in women with PCOS undergoing ART. However, the effectiveness appears to be influenced by the choice of ovulation induction agent and the route of progesterone administration. Personalized treatment approaches considering patient response and emerging evidence are essential.
Similar content being viewed by others
Background
Polycystic ovary syndrome (PCOS) is a complex endocrine condition prevalent among a significant number of women during their reproductive years. Globally, it is believed that about 10% of women grapple with PCOS challenges before menopause [1]. When it comes to anovulatory infertility, PCOS is the leading cause. Remarkably, 90–95% of women seeking infertility solutions due to anovulation are diagnosed with PCOS. Many display hormonal imbalances characterized by elevated luteinizing hormone levels, reduced follicle-stimulating hormone (FSH) levels, and increased amounts of insulin and androgens [2]. The primary treatment for this condition focuses on lifestyle modifications. If ineffective, the next step involves pharmacological ovulation induction using agents like letrozole or clomiphene citrate. In cases of pharmacological failure, assisted reproductive technologies (ART) or laparoscopic ovarian drilling are considered [3]. Assisted reproductive technologies mainly include artificial intrauterine insemination (IUI), in vitro fertilization (IVF), in vitro maturation (IVM), and intracytoplasmic sperm injection (ICSI) [4].
In the realm of reproductive physiology, the luteinizing hormone (LH) plays a pivotal role in upholding the function of the corpus luteum. It stimulates the release of progesterone and growth factors that are instrumental in processes like embryo implantation and placenta development [5]. Luteal phase support (LPS) is a crucial aspect of ART. It entails administering medications like progesterone, progestins, hCG, or GnRH agonists to bolster implantation success and early embryonic growth, enhancing the corpus luteum’s function [6, 7]. In ART, the woman’s pituitary gland is suppressed for controlled ovarian stimulation, yielding more mature eggs for external fertilization. However, intense stimulation can result in a luteal phase defect due to inadequate progesterone production by the corpus luteum, compromising implantation chances [7]. Without LPS post-IVF, the luteal phase often shortens, leading to early bleeding [8]. Thus, LPS is vital for luteal stability and early pregnancy support. A systematic review and meta-analysis affirm LPS’s positive impact on IVF outcomes [7, 9]. Luteal phase supplementation has yet to be well studied in women with PCOS undergoing ART. A recent systematic review examining the role of progesterone as LPS in ovarian stimulation and intrauterine insemination cycles indicated a potential advantage of progesterone in boosting live birth and clinical pregnancy rates compared to a placebo. However, the strength of this evidence is deemed between low and moderate, underscoring a pressing need for more rigorous trials to corroborate these findings [10]. Given the current absence of pooled evidence and the evolving landscape of reproductive health, a thorough systematic review on this subject is both timely and necessary. This review is poised to enrich the literature by providing more clarity and potentially influencing best practices for clinicians treating PCOS patients with ART. Our primary goal for this systematic review is to rigorously evaluate the effectiveness of luteal phase support in women with PCOS who are undergoing ART.
Materials and methods
We undertook a systematic review encompassing clinical trials and cohort studies focused on infertile women diagnosed with PCOS who had undergone assisted reproductive technology. Our aim was to collate and present evidence regarding the efficacy of luteal phase support for these women post-ART. Our search criteria were expansive, free from time or linguistic confines. Primary endpoints were centred on pregnancy rates. This research has been approved by The Health Research Ethics Committee Faculty of Medicine Universitas Indonesia.
We initiated a systematic search spanning three primary databases: EMBASE, PubMed, and Scopus, without language exclusions. Furthermore, a “snowball” technique was employed to unearth more studies by scrutinizing the references of pertinent papers and analysing studies that cited these primary sources. The search strategy encompassed terms like “Polycystic ovarian syndrome”, “Polycystic ovary syndrome”, PCOS in conjunction with “Luteal phase”, “Luteal support”, and “luteal phase support” (Table 1).
We searched for studies up to August 1, 2023. Dual authors took on the task of poring over the complete texts and gleaning pertinent details, including study demographics, quality evaluations, and core findings. Information was drawn solely from studies that aligned with our preset criteria. If studies were replicated, we prioritized the latest comprehensive version. When data extraction disagreements arose, they were amicably settled through dialogue or by seeking the insights of seasoned authors. Two independent evaluators gauged the integrity of the methods employed in the selected studies. The Risk of Bias 2 (ROB2) tool was deployed for RCTs, whereas cohort studies were appraised using the Newcastle–Ottawa Scale (NOS) tool. This systematic review was registered to the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) with protocol number INPLASY202440019.
Results
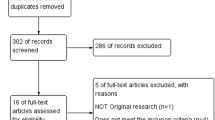
Our study included five studies, including two RCTs [11, 12], two retrospective cohorts [13, 14], and one prospective cohort [15], comprising 818 patients, to examine the effectiveness of luteal phase support in women with PCOS who are undergoing ART. Figure 1 depicts the search flow diagram illustrating the selection procedure. Table 2 provides detailed characteristics of the included investigations.
In the randomized controlled trial conducted by Alizzi in 2018 [11], a total of 149 women with PCOS undergoing ovulation induction were included and divided into two groups: one group received letrozole alone, and the other received a combination of letrozole and gonadotropin. Among these participants, 75 received oral progesterone in the form of dydrogesterone 10 mg twice daily for 14 days. The pregnancy rates were then analyzed, and the results showed that the pregnancy rate was 30.5% for the group receiving letrozole alone, 44% for the group receiving letrozole with LPS, 28% for the group receiving letrozole with gonadotropin-releasing hormone (GnRH), and 44% for the group receiving both letrozole and GnRH with LPS. However, the differences in pregnancy rates among these groups were not statistically significant (p = 0.347). Upon adjusting for potential confounding factors such as parity and the number of cycles, the researchers found that the group receiving letrozole with gonadotropin and LPS had a significantly higher likelihood of a successful pregnancy test. On the other hand, the group receiving letrozole with LPS showed a non-significant correlation with a positive pregnancy test when compared to the group receiving letrozole alone without LPS. Further examination of the data revealed odds ratios (OR) for the various treatment groups: 2.89 (0.901–9.259, p = 0.074) for letrozole with LPS, 2.06 (0.458–9.272, p = 0.346) for letrozole and GnRH alone, and 5.26 (1.323–20.884, p = 0.018) for letrozole and GnRH with LPS.
In Aygün’s 2023 retrospective cohort study [13], 164 patients underwent IVF with frozen-thawed embryo transfer. Among them, 57 patients received vaginal progesterone gel, 30 received oral progesterone tablets, and 77 received intramuscular (IM) progesterone, all administered for 21 days. The pregnancy rates were 58.1% in the IM progesterone group, 46.2% in the oral progesterone group, and 20% in the vaginal progesterone group. A statistically significant difference was observed between the IM progesterone and vaginal progesterone groups. Furthermore, when comparing pregnancy outcomes between the use of vaginal progesterone gel and oral progesterone tablets, it was found that the pregnancy rate increased significantly for those receiving oral progesterone tablets (OR 3.66, 95% CI 1.61–8.31).
Foroozanfard conducted a randomized controlled trial in 2014 involving 198 PCOS patients who underwent ART with ovulation induction [12]. The patients were divided into two groups: group 1 received clomiphene citrate (CC) + human menopausal gonadotropin (HMG), while group 2 received letrozole + HMG. Luteal phase support was administered to 122 patients in the form of intravaginal progesterone, specifically Cyclogest, at a daily dosage of 400 mg for 14 days. The results revealed a marginally significant association, with an odds ratio of 0.741 (95% CI = 0.539–1.019, p = 0.06).
In a retrospective cohort study by Montville in 2010 involving 121 PCOS patients who underwent ovulation induction, group 1 received clomiphene citrate (CC) and group 2 received letrozole [14]. For the LPS, intravaginal micronized progesterone was used at a dosage of 200 mg twice daily for 14 days. The observed pregnancy rates were as follows: in the CC group, the pregnancy rate was 12.1% (11 out of 91) without LPS and 15.3% (19 out of 124) with LPS, showing no statistically significant difference. In contrast, the letrozole group had a pregnancy rate of 0% (0 out of 13) without LPS, while with LPS, the pregnancy rate increased to 21.1% (8 out of 38), which was statistically significant with p < 0.01.
In a prospective cohort study conducted by Rezk in 2019 [15], a total of 186 participants underwent ovulation induction using letrozole. Of these, 94 participants received intravaginal progesterone for LPS (dydrogesterone, 10 mg twice daily for 14 days). The pregnancy rate in the LPS group was 48.9%, while it was 23.9% in the group without LPS (p < 0.001).
Discussion
The efficacy of LPS in women with PCOS undergoing ART is an important concern in reproductive medicine. Given the prevalence of PCOS and its often-challenging interplay with fertility, optimizing treatment strategies can make a marked difference in patient outcomes and the success rates of ART procedures. Such advancements have the potential to reshape clinical practices, providing clearer guidance to practitioners and offering hope to countless women seeking to conceive. This systematic review aimed to assess the efficacy of LPS in women diagnosed with PCOS undergoing ART and offer a comprehensive and updated understanding of its role and impact.
The included studies exhibited diverse treatment strategies for LPS. Progesterone, administered via oral, intramuscular, or intravaginal routes, was the primary means of LPS. There were variations in the accompanying ovulation induction medications, with letrozole, clomiphene citrate, and human menopausal gonadotropin being the key agents. While some studies focused on comparing the efficacy of LPS with different ovulation induction regimens, others explored the differential success rates between various routes of progesterone administration.
The efficacy of LPS across the studies was inconsistent. In Alizzi’s study [11], while there were observable differences in pregnancy rates across the groups, only the combination of letrozole, gonadotropin, and LPS yielded statistically significant results. Aygün’s findings highlighted the superiority of intramuscular progesterone over vaginal progesterone gel, and a distinct advantage was seen for oral progesterone tablets over vaginal progesterone gel [13]. Foroozanfard’s study presented a marginally significant in favour of LPS [12], while Montville’s investigation showcased the pronounced positive impact of LPS on pregnancy rates for those treated with letrozole [14]. Finally, Rezk’s research indicated an apparent enhancement in pregnancy rates when LPS was incorporated into the letrozole regimen [15].
Despite the disparities in methodologies and outcomes, an overarching trend emerges: LPS tends to augment the chances of a successful pregnancy in women with PCOS undergoing ART. However, the extent of this benefit appears to be influenced by the choice of ovulation induction agent and the route of progesterone administration.
In the realm of ART, the role of LPS and its impact on pregnancy outcomes remains a topic of keen interest. Our systematic review specifically illuminated the influence of various progesterone formulations used as LPS in women with PCOS undergoing ART. Rezk’s study [15], in particular, indicated the positive influence of LPS on pregnancy rates. Conversely, several studies involving a broader range of patients, not restricted to those with PCOS, have explored the effectiveness of vaginal progesterone as LPS in individuals undergoing letrozole-induced ovulation. Dilday’s 2023 study [16], which delved into the efficacy of vaginal progesterone as LPS in patients with letrozole-induced ovulation, did not discern a statistically significant advantage in terms of pregnancy or live birth rates, a sentiment echoed by Alizzi’s RCT [11], which also did not observe a marked difference in outcomes with or without LPS in conjunction with letrozole.
The value of LPS is underscored by Casarramona’s 2022 meta-analysis [10], which noted that progesterone LPS post-ovulation-stimulated-intrauterine insemination led to higher clinical outcomes, especially when gonadotropins were employed. Such findings harmonize with several studies in our review where the positive impact of LPS was palpable, especially evident in the letrozole cohort in Montville’s study [14]. This consensus is further solidified by Wu’s 2021 meta-analysis [17], which touted the benefits of timely initiated LPS in ongoing pregnancy. Wu also expanded the discussion on the efficacy of various progesterone delivery mechanisms, dovetailing with our exploration of different progesterone formulations and administration pathways [17].
The narrative woven by these myriad studies highlights both consistencies and discrepancies. One clear consensus is the beneficial role of LPS, especially progesterone, across multiple reproductive contexts. Both our review and studies like Casarramona’s and Wu’s meta-analyses support this [10, 17]. However, nuances emerge when diving deeper. For instance, the impact of LPS on IUI cycles, as presented by Aytac et al., seems at odds with its more evident benefit in IVF and other ART contexts [18]. This highlights the potential for LPS’s efficacy to fluctuate based on the reproductive procedure in question. Another consistent thread, regardless of the varied outcomes, is the prevalent preference for progesterone as the primary agent for LPS, irrespective of its formulation or delivery method. However, when juxtaposing specific findings like those from Dilday et al. with our review, it is clear that the beneficial impact of LPS can vary depending on factors like ovulation induction agents [16].
Numerous studies have demonstrated a favourable trend in pregnancy rates when utilizing LPS, particularly in individuals undergoing letrozole medication. This observation suggests a concrete clinical benefit in including LPS in ART for patients with PCOS. In the context of clinical practice, there are numerous practical ideas that might be implemented. Considering the extensive range of progesterone formulations and administration techniques observed in the studies under review, it would be advantageous for doctors to prioritize approaches that offer both effectiveness and limited adverse effects. This is particularly relevant when developing treatment plans specifically customized for patients with PCOS. Moreover, although a uniform method of dosing might provide a fundamental framework, it is crucial to personalize the dosage according to the distinct reaction of each patient. A potential avenue for developing best practice recommendations in reproductive medicine might be achieved by adopting a more integrated approach, which is informed by collaborative and evidence-based deliberations among practitioners in the field.
Although our systematic review covers a wide range of topics, it is important to acknowledge its limits. Various study designs were analyzed, ranging from randomized controlled trials to observational cohort studies. The presence of diversity can give rise to outcome variations, which may facilitate the emergence of biases. Furthermore, an additional concern arises with the variation observed among patient populations and the various therapies employed. Although the primary focus revolved around PCOS, there were variations in the therapies, demographic characteristics, and specific phases of PCOS in the research, which could complicate the generalizability of the findings. Furthermore, it is imperative to exercise critical examination when considering inherent biases and the overall quality of evidence in particular research.
An imperative domain that necessitates further investigation in future research is the analysis of live birth rates. This result provides a clear and measurable indicator of the effectiveness of ART. Furthermore, investigating the many health complications that may arise during pregnancy can contribute to a more extensive comprehension of the benefits and potential drawbacks of LPS. There is a significant opportunity for implementing meticulously designed, multicenter, randomized controlled trials that effectively address the needs of various patient populations. This particular strategy could reduce biases and effectively generate more definitive findings. Future research may benefit from focusing on a singular ovulation induction agent or implementing parallel study arms to facilitate direct comparisons, as the diverse outcomes associated with different agents warrant more investigation. The foundation of achieving the best possible results is built upon two fundamental principles: individualized patient care and ongoing advancements in research.
Conclusion
The effectiveness of LPS depends on the selection of the ovulation induction drug and the route of progesterone delivery. While it may be reasonable to generally support using LPS in ART protocols for individuals with PCOS, it is essential to emphasize the importance of personalized treatment approaches that include patient response and emerging evidence. There is a need for future randomized controlled studies that are well-designed, particularly those that prioritize tangible outcomes such as live birth rates, in order to provide more conclusive clinical recommendations.
Registration
This systematic review was registered to the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) with protocol number INPLASY202440019.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ART:
-
Assisted reproductive technology
- CC:
-
Clomiphene citrate
- CI:
-
Confidence interval
- FSH:
-
Follicle-stimulating hormone
- GnRH:
-
Gonadotropin-releasing hormone
- hCG:
-
Human chorionic gonadotropin
- HMG:
-
Human menopausal gonadotropin
- ICSI:
-
Intracytoplasmic sperm injection
- IM:
-
Intramuscular
- IUI:
-
Intrauterine insemination
- IVF:
-
In vitro fertilization
- IVM:
-
In vitro maturation
- LH:
-
Luteinizing hormone
- LPS:
-
Luteal phase support
- NOS:
-
Newcastle-Ottawa Scale
- OR:
-
Odds ratio
- PCOS:
-
Polycystic ovary syndrome
- ROB2:
-
Risk of Bias 2
References
Sadeghi HM, Adeli I, Calina D, Docea AO, Mousavi T, Daniali M et al (2022) Polycystic ovary syndrome: a comprehensive review of pathogenesis, management, and drug repurposing. Int J Mol Sci 23(2):583. https://doi.org/10.3390/ijms23020583
Dennett CC, Simon J (2015) The role of polycystic ovary syndrome in reproductive and metabolic health: overview and approaches for treatment. Diabetes Spectr 28(2):116–120. https://doi.org/10.2337/diaspect.28.2.116
Cunha A, Póvoa AM (2021) Infertility management in women with polycystic ovary syndrome: a review. Porto Biomed J 6(1):e116. https://doi.org/10.1097/j.pbj.0000000000000116
Li M, Ruan X, Mueck AO (2022) Management strategy of infertility in polycystic ovary syndrome. Glob Health J 6(2):70–74. https://doi.org/10.1016/j.glohj.2022.03.002
Carosso AR, Canosa S, Gennarelli G, Sestero M, Evangelisti B, Charrier L et al (2021) Luteal support with very low daily dose of human chorionic gonadotropin after fresh embryo transfer as an alternative to cycle segmentation for high responders patients undergoing gonadotropin-releasing hormone agonist-triggered ivf. Pharmaceuticals 14(3):228. https://doi.org/10.3390/ph14030228
Farquhar C, Marjoribanks J (2018) Assisted reproductive technology: an overview of Cochrane Reviews. Cochrane Database Syst Rev 8(8):CD010537. https://doi.org/10.1002/14651858
van der Linden M, Buckingham K, Farquhar C, Kremer JA (2015) Metwally M (2015) Luteal phase support for assisted reproduction cycles. Cochrane Database Syst Rev 7:Cd009154. https://doi.org/10.1002/14651858
Albano C, Grimbizis G, Smitz J, Riethmüller-Winzen H, Reissmann T, Van Steirteghem A et al (1998) The luteal phase of nonsupplemented cycles after ovarian superovulation with human menopausal gonadotropin and the gonadotropin-releasing hormone antagonist Cetrorelix. Fertil Steril 70(2):357–359. https://doi.org/10.1016/s0015-0282(98)00135-6
Nosarka S, Kruger T, Siebert I, Grové D (2005) Luteal phase support in in vitro fertilization: meta-analysis of randomised trials. Gynecol Obstet Invest 60(2):67–74. https://doi.org/10.1159/000084546
Casarramona G, Lalmahomed T, Lemmen C, Eijkemans M, Broekmans F, Cantineau A et al (2022) The efficacy and safety of luteal phase support with progesterone following ovarian stimulation and intrauterine insemination: a systematic review and meta-analysis. Front Endocrinol (Lausanne) 13:960393. https://doi.org/10.3389/fendo.2022.960393
Alizzi FJ (2018) Pregnancy rate following luteal phase support in polycystic ovary women using letrozole with or without gonadotropin as ovulation induction. Asian J Pharm Clin Res 11(9):321–324. https://doi.org/10.22159/ajpcr.2018.v11i9.27047
Foroozanfard F, Saberi H, Moraveji SA, Bazarganipour F (2014) Pregnancy rate following luteal phase support in Iranian women with polycystic ovarian syndrome. Int J Fertil Steril 8(3):235–242
Aygün EG, Özbaşlı E, Köse MF (2023) The effect of different luteal phase support applications on clinical pregnancy outcomes in frozen-thawed embryo transfer. Biomed Res Int 2023:8157210. https://doi.org/10.1155/2023/8157210
Montville CP, Khabbaz M, Aubuchon M, Williams DB, Thomas MA (2010) Luteal support with intravaginal progesterone increases clinical pregnancy rates in women with polycystic ovary syndrome using letrozole for ovulation induction. Fertil Steril 94(2):678–683. https://doi.org/10.1016/j.fertnstert.2009.03.088
Rezk M, Hamza H, El-Shamy ES (2019) Luteal support with vaginal dydrogesterone increases pregnancy rate in patients with clomifene resistant polycystic ovary syndrome receiving letrozole for ovulation induction. Gynecol Endocrinol 35(3):217–219. https://doi.org/10.1080/09513590.2018.1512571
Dilday E, Gigg M, Hoyos L, Quinn M, Markovic D, Kroener L (2023) Luteal phase support with progesterone does not improve pregnancy rates in patients undergoing ovarian stimulation with letrozole. Reprod Biomed Online 46(1):123–128. https://doi.org/10.1016/j.rbmo.2022.09.012
Wu H, Zhang S, Lin X, Wang S, Zhou P (2021) Luteal phase support for in vitro fertilization/intracytoplasmic sperm injection fresh cycles: a systematic review and network meta-analysis. Reprod Biol Endocrinol 19(1):103. https://doi.org/10.1186/s12958-021-00782-5
Aytac PC, Bulgan Kilicdag E, Haydardedeoglu B, Simsek E, Cok T, Coban G (2016) Luteal phase support after mild ovulation induction with intrauterine insemination: an on-going debate. Gynecol Endocrinol 32(7):543–547. https://doi.org/10.3109/09513590.2016.1138460
Acknowledgements
The authors sincerely thank the Department of Obstetrics and Gynecology, Faculty of Medicine Universitas Indonesia, for supporting this research.
Funding
The authors receive no funding for this research.
Author information
Authors and Affiliations
Contributions
A.K.H.: conceptualization, methodology, validation, writing—review and editing, visualization, funding administration. S.M.R.P., C.T.P.R.: formal analysis, investigation, data curation, writing—original draft. N.U., H.D.M., P.N., A.R.P: visualization, writing—review and editing. B.W; Supervision, writing—review and editing. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Health Research Ethics Committee Faculty of Medicine, Universitas Indonesia, under No. KET-1294/UN2.F1/ETIK/PPM.00.02/2023.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Harzif, A.K., Pratamasari, S.M.R., Rumapea, C.T.P. et al. The efficacy of luteal phase support in women with polycystic ovary syndrome following assisted reproductive technology: a systematic review. Middle East Fertil Soc J 29, 40 (2024). https://doi.org/10.1186/s43043-024-00187-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43043-024-00187-5