Abstract
Background
Coconut water is a relished beverage traditionally used as a remedy for childhood diarrhoea and gastroenteritis. It can be given to an individual who has hangover or suspected to have ingested a toxic substance. In this study, we evaluated the protective effect of coconut water against carbon tetrachloride (CCl4)-induced toxicity in rats.
Results
Administration of coconut water decreased significantly albumin, aspartate transaminase (AST), alanine transaminase (ALT), urea, creatinine, bicarbonate (HCO3−), total cholesterol, triglycerides, low-density lipoprotein, very low-density lipoprotein and malondialdehyde (MDA) levels compared to the non-pretreated group. Furthermore, high-density lipoprotein, glutathione (GSH), superoxide dismutase (SOD) and catalase (CAT) increased significantly (p < 0.05) in coconut water-pretreated groups compared to the negative control. There were no observed pathological changes in the coconut water-pretreated groups, but slight enlargement of the central veins and tubules was evident when compared to the negative control.
Conclusions
Therefore, intake of coconut water may be protective against toxicity induced by CCl4 as its pretreatment elevated antioxidant parameters (GSH, SOD and CAT) and did not alter biochemical parameters in rats.
Similar content being viewed by others
Background
Coconut water is a relished beverage gotten from coconuts (Cocos nucifera L) (Gordon and Jackson 2017). Previously, coconut water was mostly acquired as a fresh beverage in regions where it was accessible, by cutting open a green nut fresh off the tree; today, it is a $300 + million business (Gordon and Jackson 2017). It is a perennial plant that bears fruit 12–13 times a year for up to 60–70 years, providing between 30 and 75 fruits per year. The fruit, in the shape of a nut, grows on trees of different heights depending on the type of coconut and is an important source of money for many farmers and countries in the far East in particular, where it is utilized in a number of ways and exported to other countries across the world (Gordon and Jackson 2017). The fruit is a significant source of oilseeds, particularly in many developing nations such as the Philippines, Sri Lanka, India, Malaysia and portions of West Africa, Latin America and the Caribbean (Gordon and Jackson 2017; Ghosh et al. 2014).
The chemical composition of coconut water has been widely studied (Yong et al. 2009; Awua et al. 2011; Prades et al. 2012). Compositionally, it is made of about 96% water and 6% solids. The major components of the solids in coconut water are soluble sugars, proteins and minerals with relatively high potassium content and low sodium content. Phytochemical compositions of coconut water have shown to contain phenolic compounds, flavonoids, tannins and phytates (Akpro et al. 2019). However, the content of coconut water is highly influenced by the type of cultivar of coconut and the stage of maturation of the nut (Gordon and Jackson 2017). The reports of Yong et al. (2009) revealed the presence of some water-soluble vitamins in coconut water such as thiamine, riboflavin, nicotinic acid, pantothenic acid, pyridoxine, biotin, folic acid and ascorbic acid. Salicylic acid, syringic acid, m-coumaric acid, p-hydroxybenzoic acid, p-coumaric acid, caffeic acid and catechin are among the phenolic constituents found in coconut water (Mahoyothee et al. 2016). Aside from amino acids and L-arginine, coconut water contains plant hormones such as auxin, 1, 3-diphenylurea and cytokinin. It also contains acid phosphatase, catalase, dehydrogenase, diastase, peroxidase, RNA polymerases and growth factors. More so, it contains some minerals including sodium, potassium, magnesium, iron, calcium, copper and phosphorous (Kaliyamoorthy et al. 2015).
Coconut water is unstable to air, and it is best consumed fresh in tropical climates. Fresh coconut water has traditionally been used for oral rehydration, treatment of childhood diarrhoea and gastroenteritis (Mandal et al. 2009). It provides several nutritional, physiological and medicinal benefits including antifungal, antioxidant, antibacterial, hypoglycaemic, anti-cancer, antitumor, anti-dermatophyte, antiviral, anti-parasitic and hepatoprotective properties (Loki and Rajamohan 2003; Mohamad et al. 2019; Elekwa et al. 2021). These biological activities have been attributed to their high concentration of bioactive chemicals. Researchers have also uncovered that virgin coconut oil has anti-inflammatory, analgesic, antipyretic, immune-stimulatory and anti-cancer (Intahpuak et al. 2010; Varma et al. 2019).
Pollutants and chemical xenobiotics emitted by industrial waste, such as CCl4, can harm the liver and kidney by producing reactive oxygen species (ROS) (Dutta et al. 2018). ROS promotes oxidative stress, which has been linked to hepatorenal damages (Liu et al. 2011). ROS has also shown to induce cellular damage by lipid peroxidation and covalent binding, and an increase in ROS decreases oxidative stress defence enzymes. Its mode of action is evident in the liver cytochrome P450 enzyme system's reductive dehalogenation to create trichloromethyl free radical, which rapidly interacts with molecular oxygen to produce trichloromethylperoxyl radicals (Li et al. 2004). Excessive formation of CCl4 free radicals can result in membrane lipid oxidation, which can progress to hepatorenal injury (Li et al. 2004). In this study, we investigated the protective effects of coconut water against CCl4-induced toxicity in rats.
Methods
Collection and preparation of coconut water
Healthy tender coconuts were gathered from Ndi Ekpere Compound, Amaiyi Igbere in Bende Local Government Area of Abia State, Nigeria. The coconut was de-husked, and the epicarp was gently opened to avoid losing or contaminating the coconut water. Every day, new coconut water was collected and filtered in a sterile condition using a ready-to-use Whatman filter No 1.
Animal use and care
Healthy rats (male and female) (8 weeks old; weight 151.78 ± 19.45 g, n = 60) bred locally at the Department of Biochemistry, Faculty of Biological Sciences, Abia State University, Uturu, were used for the study. The rats were housed in a metal cage for at least a week before the commencement of the study to allow for acclimatization under natural atmospheric conditions. They were fed with standard rat chow (Vital Feed) and water ad libitum. The laboratory procedures and tests were carried out with the permission of the Abia State University Ethical Committee (ABSU/REC/BMR/0026) and in accordance with World Health Organization recommendations for ‘good laboratory practice'.
Experimental procedure
Healthy Wistar rats (male, n = 30 and female, n = 30) were randomly divided into five groups. To prevent mating, the male and female rats were placed in separate cages. Groups 3–5 were treated with different doses of coconut water for 14 days. After the 14th day pretreatment with coconut water, groups 2–5 were given 1 mL/kg of CCl4 (Merck, UK) diluted in olive oil at a 1:1 ratio by intraperitoneal (i.p.) route. The procedure is shown as:
-
Group 1: normal control- (distilled water; 1 mL/kg; p.o.)
-
Groups 2: negative control- CCl4 only (1 mL/kg; i.p.)
-
Group 3: Coconut water (2 mL/kg; p.o.) + CCl4 (1 mL/kg; i.p.)
-
Group 4: Coconut water (4 mL/kg; p.o.) + CCl4 (1 mL/kg; i.p.)
-
Group 5: Coconut water (6 mL/kg; p.o.) + CCl4 (1 mL/kg; i.p.)
The rats were sacrificed after 24 h of CCl4 treatment. The rats were weighed and humanely sacrificed by cervical dislocation. The blood samples were collected through cardiac puncture and distributed into dry plain bottles for biochemical tests and ethylenediaminetetraacetic acid (EDTA) bottles for haematological investigations. The liver and kidney were harvested and used to assess their histopathology.
Haematological and biochemical assays
Red blood cell (RBC), white blood cell (WBC), platelets, RBC indices and WBC indices were measured in accordance with the procedure previously described by Bain et al. (2017). Randox diagnostic kits (Randox Laboratory Ltd., Co., Antrim, UK) were used to evaluate spectrophotometrically the biomarkers of kidney, liver and lipids. Urea, creatinine and electrolytes (Na+, K+, HCO3− and Cl−) were the renal biomarkers studied. The hepatic biomarkers measured were alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase (ALP), bilirubin, total protein and albumin. Furthermore, lipids calculated included triglycerides (TAG), high-density lipoprotein (HDL), total cholesterol (TC) and low-density lipoprotein (LDL).
Antioxidant markers
Other researchers' methodologies were used to determine oxidative stress indicators. Techniques for measuring superoxide dismutase (SOD) were used as reported by Sun and Zigma (1978). Catalase was estimated as revealed by Aebi (1984). Malondialdehyde and glutathione were evaluated using the methods described by Buege and Aust (1978) and Sedlak and Lindsay (1968), respectively.
Histopathological assessments
The liver and kidneys were fixed in 10% v/v formalin before being encased in paraffin wax and sectioned. Each specimen was cut at 5 µm and stained with haematoxylin and eosin dyes. Using X40 magnification light microscope, the stained portions were examined for histological changes and photomicrographs were taken.
Statistical analysis
The generated data were represented as mean ± standard deviation. To determine the difference between the groups, statistical analysis was performed with R™ statistical program (https://cran.r-project.org/src/base/R3/), version 3.0.3. A significant relationship was determined using the Tukey test post hoc at a 95% level of confidence.
Results
Body weight of 14 days treatment of coconut water on CCl4-induced toxicity
Figure 1 represents the protective effect of coconut water on body weight of CCl4-induced toxicity in both male and female rats. No significant changes (p > 0.05) in body weight were observed in both sexes in all the treatment groups.
Protective effect of coconut water on liver biomarkers of CCl4-induced toxicity
Intake of coconut water decreased the values of albumin, AST and ALT compared to the negative control. However, total protein and bilirubin levels had a dose-dependent significant difference (p < 0.05) between the coconut water-pretreated groups (4 and 6 mL/kg) and the negative (CCl4) control. ALP values did not show any significant difference in the male rats. In the female rats, there were significant differences (p < 0.05) in the values of albumin, AST, ALT, ALP and bilirubin among the coconut water-pretreated groups compared to the CCl4 group (Table 1).
Protective effect of coconut water on kidney biomarkers of CCl4-induced toxicity
Table 2 shows the data set obtained from the protective effect of coconut water on kidney function of CCl4-induced toxicity in both sexes. There were significant differences observed in the values of urea, creatinine, Na+, K+ and HCO3− in the coconut water-pretreated groups compared to the CCl4-treated group.
Protective effect of coconut water on haematological parameters of CCl4-induced toxicity
In the CCl4 group, the values of RBC, PCV, HB, platelet, MCV and neutrophils were decreased compared to the coconut water-treated groups. Furthermore, the values of WBC and lymphocytes were increased in the negative control compared to coconut water-treated groups (Table 3).
Protective effect of coconut water on lipid profiles of CCl4-induced toxicity
Table 4 shows the result obtained from the protective effect of coconut water on lipid profiles of carbon tetrachloride-induced toxicity in both male and female rats. Groups administered with coconut water had a significant decrease (p < 0.05) in the values of TC, triglycerides, LDL and VLDL. The values of the HDL decreased significantly in the CCl4 group compared to the coconut water-treated groups.
Protective effect of coconut water on antioxidant enzymes of CCl4-induced toxicity
The result presented in Table 5 is the protective effect of coconut water on antioxidant enzymes of CCl4-induced toxicity in both male and female rats. Administration of coconut water caused a significant elevation in the values of GSH, SOD and CAT. The values of MDA decreased in the treatment group compared to its untreated counterparts in both male and female rats.
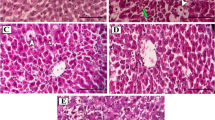
Protective effect of coconut water on histology section of liver and kidney
The liver and kidney histology sections of the male and female rats are presented in Figs. 2, 3, 4 and 5. The photomicrograph of the normal control showed a normal liver architectural design, but the treated rats showed a slight enlarged central vein compared to the negative control. More so, the hepatocytes appeared a bit distorted and many microcysts within the stroma were found (A-J). In the kidney histology (K-T), no pathological change was observed in the normal control unlike the CCl4-treated groups. While the negative control had a slight dilated and shrunken glomerulus, a slight enlargement of the tubules was observed in the coconut water-treated group, although there were no pathological changes observed amongst the groups (Figs. 4, 5).
Protective effect of coconut water on histology section of liver against CCl4-induced toxicity in male rats (A–E). A Control, B CCl4 only, C 2 mL/kg, D 4 mL/kg, E 6 mL/kg. V: central vein, S: sinusoids, NS: unmarkable sinusoids, M: multicystic space. The liver sections were stained with haematoxylin and eosin staining (H&E) solution and observed under an Olympus light microscope fixed with camera, at a magnification (40×)
Protective effect of coconut water on histology section of liver against CCl4-induced toxicity in female rats (A–E). A Control, B CCl4 only, C 2 mL/kg, D 4 mL/kg, E 6 mL/kg. V: central vein, S: sinusoids, NS: unmarkable sinusoids, M: multicystic space, D: dilated cystic space, H: distorted hepatocytes. The liver sections were stained with haematoxylin and eosin staining (H&E) solution and observed under an Olympus light microscope fixed with camera, at a magnification (40×)
Protective effect of coconut water on histology section of kidney against CCl4-induced toxicity in male rats (A–E). A Control, B CCl4 only, C 2 mL/kg, D 4 mL/kg, E 6 mL/kg. G: glomeruli, T: tubules, D: glomerular degeneration, GS: shrunken glomerula, BCS: Bowman’s space, B: Bowman’s capsule, D: dilated tubules, GN: network of the glomerulus. The kidney sections were stained with haematoxylin and eosin staining (H&E) solution and observed under an Olympus light microscope fixed with camera, at a magnification (40×)
Protective effect of coconut water on histology section of kidney against CCl4-induced toxicity in female rats (A–E). A Control, B CCl4 only, C 2 mL/kg, D 4 mL/kg, E 6 mL/kg. G: glomeruli, T: tubules, D: glomerular degeneration, GS: shrunken glomerula, BCS: Bowman’s space, B: Bowman’s capsule, D: dilated tubules, GN: network of the glomerulus. The kidney sections were stained with haematoxylin and eosin staining (H&E) solution and observed under an Olympus light microscope fixed with camera, at a magnification (40×)
Discussion
In this study, we examined the protective effect of coconut water against CCl4-induced toxicity in rats. The findings of the study revealed no significant changes in body weight in the experimental rats. This might be attributable to the brief period of CCl4 delivery following coconut water pretreatment. In other reports, body weight measurement has been described as plausible evidence in toxicological research (Emmanuel et al. 2021).
Elevations of liver biomarkers; enzymes (AST, ALT and ALP), bilirubin, albumin and total proteins are strong indicators for liver insults (Orieke et al. 2019; Ugbogu et al. 2019; Emmanuel et al. 2021). When the liver is under severe attack, these enzymes are released into the blood and thus give insight on the hepatic status of the liver (Ugbogu et al. 2019). Dutta et al. (2018) reported that CCl4 is hepatotoxic by producing ROS. Increased ROS has been associated with decreased antioxidant levels (Emmanuel et al. 2021). In the present study, the negative control had elevated levels of liver enzymes and administration of coconut water decreased the values of albumin, AST and ALT. Our findings correlate with the results of other researchers (Anurag and Rajamohan 2003). Anurag and Rajamohan (2003) showed that coconut water counteracted an increased level of liver enzymes caused by isoproterenol exposure. The reported hepatoprotective activities of coconut water might be linked to its antioxidant activities (Tsai et al. 2009).
A functional renal system may be related to the kidney's filtration capacity, commonly known as the glomerular filtration rate (GFR). A reduction in GFR may produce an increase in urea and creatinine in the serum, indicating renal disease (Jose 2014). Additionally, in kidney test, a high serum urea level indicates renal failure, which usually progresses to severe kidney damage in long-term instances (Gowda et al. 2010). In our study, urea, creatinine and HCO3− significantly decreased in the coconut water-treated groups when compared to the negative control. Abnormal elevations of creatinine, urea and electrolytes levels are implicated in kidney insults (Ugbogu et al. 2019; Emmanuel et al. 2021).
Haematological parameters are those that relate to blood and blood-forming organs. The quantity and shape of RBC, WBC and platelets are important in disease diagnosis and monitoring (Owoeye et al. 2011). In this work, groups pretreated with coconut water prior to CCl4 induction had a significant haematoprotective effect in both male and female rats compared to the negative control. In the negative control, the levels of RBC, PCV and HB were decreased, and this may be associated with the toxic free radicals generated from CCl4 which consequently affected haematopoiesis in the experimental animals as revealed in other studies (Rahmouni et al. 2011). Therefore, haematological parameters are very sensitive and are crucial for predicting the toxicological and pathological effect of potentially toxic substances in both human and animals (Kong et al. 2016). Previous study on the effects of CCl4 on haematological parameters showed that acute CCl4 toxicity led to transient decrease in the HB concentration and reticulocyte count as well as PCV and RBC counts by extension which is in consonant with this present study (Saba et al. 2010; Emmanuel et al. 2021).
The rats administered with coconut water prior CCl4 induction had decreased levels TC, triglycerides, LDL and VLDL, while HDL levels increased compared to the untreated group. This demonstrates that CCl4 could disrupt lipid metabolism. Atherosclerosis and coronary heart disease have been linked to higher levels of cholesterol, triglycerides, LDL and VLDL (Sandhya and Rajamohan 2008; Emmanuel et al. 2021). Our findings confirmed the previously reported evidence that coconut water lowered the increased levels of TC, triglycerides, LDL and VLDL in rats induced by a high cholesterol fat feed (Sandhya and Rajamohan 2006).
CCl4 has been found to be toxic by causing the production of free radicals capable of interacting with sulphur-containing proteins, which can impair antioxidant activity (Tsai et al. 2009). This study revealed that CCl4 has a negative effect on antioxidants, as seen by a decrease in SOD, GSH and CAT and an increase in MDA. Our findings are consistent with the findings of Rahmouni et al. (2011) that discovered that CCl4 may reduce erythrocyte GSH, SOD and CAT activities while increasing erythrocyte MDA levels.
In the histology of the liver and kidney, pathological changes were evident; however, there were slight enlargement of the central veins and tubules, respectively, compared to the negative control. Coconut water administration revealed a dose-dependent protection compared to the untreated group. Injuries in the liver included deformed hepatocytes, dilated sinusoids, microvascular steatosis and an enlarged central vein, while congested blood vessels and dilated tubules were detected in the kidney of the negative control. These findings support the assertion that CCl4 has haematotoxic, hepatotoxic and nephrotoxic potentials (Rahmouni et al. 2011; Elshater et al. 2013; Emmanuel et al. 2021).
Conclusions
Our findings show that coconut water protects rats from CCl4-induced toxicity. Its pretreatment for 14 days following CCl4 toxicity elevated GSH, SOD, CAT levels and decreased MDA levels. These data show that coconut water may protect against lipid peroxidation by scavenging the harmful radicals produced by CCl4 and thereby normalizing the quantity and distribution of lipids in rats’ systemic circulation. The observed increase in these antioxidants may be related to coconut water’s hepato/reno-protective potentials and reduced cellular oxidative damage. Notwithstanding the health benefits of coconut water, it can be noted that its contents may be influenced and varied by the type of cultivar, geographical location and the stage of maturation of the nut.
Availability of data and materials
All the data used for this manuscript are available on request from the corresponding author.
Abbreviations
- ALT:
-
Alanine transaminase
- AST:
-
Aspartate transaminase
- ALP:
-
Alanine phosphatase
- CAT:
-
Catalase
- CCl4 :
-
Carbon tetrachloride
- Cl− :
-
Chloride ion
- CVD:
-
Cardiovascular disease
- EDTA:
-
Ethylenediaminetetraacetic acid
- HB:
-
Haemoglobin
- HCO3 − :
-
Bicarbonate
- HDL:
-
High-density lipoprotein
- GSH:
-
Glutathione
- K+ :
-
Potassium ion
- LDL:
-
Low-density lipoprotein
- MCV:
-
Mean corpuscular volume
- MCH:
-
Mean corpuscular haemoglobin
- MCHC:
-
Mean corpuscular haemoglobin concentration
- MDA:
-
Malondialdehyde
- Na + :
-
Sodium ion
- PCV:
-
Packed cell volume
- RBC:
-
Red blood cell
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TC:
-
Total cholesterol
- VLDL:
-
Very low-density lipoprotein
- WBC:
-
White blood cell
References
Aebi H (1984) Catalase in vitro. In: Method in enzymology, vol 105. Academic Press, New York, pp 121–126 https://doi.org/10.1016/S0076-6879(84)05016-3
Akpro LA, Gbogouri GA, Konan BR, Issali AE, Konan KJL, Brou KD, Nemlin GJ (2019) Phytochemical compounds, antioxidant activity and non-enzymatic browning of sugars extracted from the water of immature coconut (Cocos nucifera L.). Sci Afr 6:e00123. https://doi.org/10.1016/j.sciaf.2019.e00123
Anurag P, Rajamohan T (2003) Cardioprotective effect of tender coconut water in experimental myocardial infarction. Plant Foods Human Nutri 58(3):1–12
Awua AK, Doe ED, Agyare R (2011) Exploring the influence of sterilization and storage on some physicochemical properties of coconut (Cocos nucifera L.) water. BMC Res Notes 4(1):451
Bain BJ, Bates I, Laffan MA (2017) Dacie and Lewis practical haematology. Elsevier Health Sciences, New York
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Dutta S, Chakraborty AK, Dey P, Kar P, Guha P, Sen S, Kumar A, Sen A, Chaudhuri TK (2018) Amelioration of CCl4 induced liver injury in Swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill. PLoS ONE 13(4):e0196411
Elekwa I, Ude VC, Emmanuel O, Amachaghi VO, Ugbogu EA (2021) In vivo studies on the ameliorative effect of coconut water against carbon tetrachloride induced toxicity in rats. Biomarkers. https://doi.org/10.1080/1354750X.2021.1946848
Elshater AEA, Salman MMA, Mohamed SA (2013) The hepato-ameliorating effect of Solanum nigrum against CCl4 induced liver toxicity in albino rats. Egypt Acad J Biol Sci 5(1):59–66
Emmanuel O, Okezie UM, Iweala EJ, Ugbogu EA (2021) Pretreatment of red palm oil extracted from palm fruit (Elaeis guineensis) attenuates carbon tetrachloride induced toxicity in Wistar rats. Phytomed plus 1:100079. https://doi.org/10.1016/j.phyplu.2021.100079
Ghosh PK, Bhattacharjee P, Mitra S, Poddar-Sarkar M (2014) Physicochemical and phytochemical analyses of copra and oil of Cocos nucifera L. (West Coast Tall Variety). Inter J Food Sci. https://doi.org/10.1155/2014/310852
Gordon A, Jackson J (2017) Case study: application of appropriate technologies to improve the quality and safety of coconut water. Food Safety Qual Syst Dev Countries. https://doi.org/10.1016/B978-0-12-801226-0.00007-4
Gowda S, Desai PB, Kulkarni SS, Hull VV, Math AAK, Vernekar SN (2010) Markers of renal function tests. North Am J Med Sci 2:170–173
Intahphuak S, Khonsung P, Panthong A (2010) Anti-inflammatory, analgesic, and antipyretic activities of virgin coconut oil. Pharmaceut Biol 48(2):151–157
Jose HS (2014) Overview of urea and creatinine. Lab Med 45(1):e19–e20. https://doi.org/10.1309/lm920sbnzpjrjgut
Kaliyamoorthy J, Rajasekaran S, Nagarajan M, Vijayarengan P (2015) Bioactive enzyme activity and medicinal properties of tender coconut (Cocos nucifera L.). Int J Modern Biochem 4(1):10–14
Kong BH, Tan NH, Fung SY, Pailoor J (2016) Sub-acute toxicity study of tiger milk mushroom Lignosus tigris Chon S. Tan Cultivar E sclerotium in Sprague Dawley rats. Front Pharmacol 7:246
Li JX, Pang YZ, Tang CS, Li ZQ (2004) Protective effect of taurine on hypochlorous acid toxicity to nuclear nucleoside triphosphatase in isolated nuclei from rat liver. World J Gastroenterol 10:694–698
Liu Q, Kong B, Li G, Liu N, Xia X (2011) Hepatoprotective and antioxidant effects of porcine plasma protein hydrolysates on carbon tetrachloride-induced liver damage in rats. Food Chem Toxicol 49(6):1316–1321
Loki AL, Rajamohan T (2003) Hepatoprotective and antioxidant effect of tender coconut water on CCl4 induced liver injury in rats. Indian J Biochem Biophy 40:354–357
Mahayothee B, Koomyart I, Khuwijitjaru P, Siriwongwilaichat P, Nagle M, Müller J (2016) Phenolic compounds, antioxidant activity, and medium chain fatty acids profiles of coconut water and meat at different maturity stages. Int J Food Propert 19(9):2041–2051
Mandal SM, Dey S, Mandal M, Sarkar S, Maria-Neto S, Franco OL (2009) Identification and structural insights of three novel antimicrobial peptides isolated from green coconut water. Peptides 30:633–637
Mohamad NE, Yeap SK, Abu N, Lim KL, Zamberi NR, Nordin N, Sharifuddin SA, Alitheen LK, NB, (2019) In vitro and in vivo antitumour effects of coconut water vinegar on 4T1 breast cancer cells. Food Nutr Res 10:63
Orieke D, Ohaeri OC, Ijeh II, Ijioma SN, Achi NK (2019) Acute and sub-acute evaluations of methanolic leaf extract of Corchorus olitorius in experimental animal models. Asian J Res Anim Vet Sci 2(4):1–12
Owoeye O, Onwuka SK, Farombi EO (2011) Vernonia amygdalina leaf extract and alpha-tocopherol alleviated gamma radiation-induced haematological and biochemical changes in rats. Int J Biol Chem Sci 5(5):1978–1992
Prades A, Dornier M, Diop N, Pain JP (2012) Coconut water uses, composition and properties: a review. Fruits 67(2):87–107
Rahmouni F, Hamdaoui L, Badraoui R, Rebai T (2011) Protective effects of Teucrium polium aqueous extract and ascorbic acid on hematological and some biochemical parameters against carbon tetrachloride (CCl4) induced toxicity in rats. Biomed Pharmacother 91:43–48
Saba AB, Oyagbemi AA, Azeez OI (2010) Amelioration of carbon tetrachloride-induced hepatotoxicity and haemotoxicity by aqueous leaf extract of Cnidoscolus aconitifolius in rats. Nig J Physiol Sci 25:139–147
Sandhya VG, Rajamohan T (2006) Beneficial effects of coconut water feeding on lipid metabolism in cholesterol-fed rats. J Med Food 9(3):400–407
Sandhya VG, Rajamohan T (2008) Comparative evaluation of the hypolipidemic effects of coconut water and lovastatin in rats fed fat–cholesterol enriched diet. Food Chem Toxicol 46(12):3586–3592
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and non-protein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:1192–1205
Sun M, Zigma S (1978) An improved spectrophotometer assay of superoxide dismutase based on epinephrine antioxidation. Anal Biochem 90:81–89
Tsai CF, Hsu YW, Chen WK, Chang WH, Yen CC, Ho YC, Lu FJ (2009) Hepatoprotective effect of electrolyzed reduced water against carbon tetrachloride-induced liver damage in mice. Food Chem Toxicol 47(8):2031–2036
Ugbogu EA, Akubugwo EI, Ude VC, Emmanuel O, Okomba NO, Ibe C, Onyero O (2019) Safety evaluation of an aqueous extract of Termitomyces robustus (Agaricomycetes) in Wistar rats. Inter J Med Mushrooms 21(2):193–203
Varma SR, Sivaprakasam TO, Arumugam I, Dilip N, Raghuraman M, Pavan KB, Rafiq M, Paramesh R (2019) In vitro anti-inflammatory and skin protective properties of virgin coconut oil. J Trad Complement Med 9(1):5–14
Yong JW, Ge L, Ng YF, Tan SN (2009) The chemical composition and biological properties of coconut (Cocos nucifera L.) water. Molecules 14:5144–5164
Acknowledgements
Not applicable.
Funding
This study was sponsored by Tertiary Education Trust Fund Institutional Base Research Grant from Abia State University, Uturu, Nigeria.
Author information
Authors and Affiliations
Contributions
OE, IE and EAU conceptualized the work, sourced the literature and wrote the original draft. CP, VCU, OGE, SNI, VOA and CU wrote the original draft and read the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The laboratory procedures and tests were carried out with the permission of the Abia State University Ethical Committee (ABSU/REC/BMR/0026) and in accordance with World Health Organization recommendations for ‘good laboratory practice'.
Consent for publication
Not applicable.
Competing interests
The authors declare none whatsoever.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Emmanuel, O., Elekwa, I., Paul-Joseph, C. et al. Protective effects of coconut water against the intraperitoneal infused carbon tetrachloride-induced toxicity—evaluations of biochemical, haematological and histopathological profiles in rats. Bull Natl Res Cent 46, 206 (2022). https://doi.org/10.1186/s42269-022-00893-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42269-022-00893-y