Abstract
Introduction
Obesity-associated metabolic disorders are often associated with reproductive disorders affecting the spermatogenesis program and infertility. Recently, dietary calcium has shown a protective effect against obesity and its associated metabolic disorders. Calcium is a common nutrient in our daily diet with a diverse physiological role, but its role against reproductive function during obesity remains unknown.
Purpose
The present study aimed to investigate the dietary effect of calcium against male reproductive function in high-fat diet (HFD) rats.
Methods
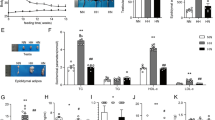
Male rats were randomly divided into four groups: the control group, the HFD obese group, the low calcium (0.25% Ca) HFD group, and the high calcium (1.0%Ca) HFD group. The rats were fed with the low or high-calcium diet for 12 weeks after inducing obesity. At the end of the study, several reproductive markers including sperm count, testicular steroidogenic enzymatic activity, testosterone, antioxidant, inflammatory and apoptotic markers were studied.
Results
High calcium (1.0% Ca) group was found to improve the reproductive parameters like sperm count, sperm motility, testosterone level, and steroidogenic enzymes indicating improvement in spermatogenesis which is affected by diet-induced obesity. These improvements in reproductive parameters are improved by restoring the physiological balance between antioxidant and oxidative stress levels and reducing the inflammatory and apoptosis markers in the high calcium group.
Conclusion
High calcium diet during obese conditions seemed to improve the male reproductive parameters and spermatogenesis program thereby preventing the risk of obesity-associated male infertility.






Similar content being viewed by others

Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Panth N, Gavarkovs A, Tamez M, Mattei J. The influence of diet on fertility and the implications for public health nutrition in the United States. Front Public Health. 2018;6:211. https://doi.org/10.3389/fpubh.2018.00211.
Martins AD, Majzoub A, Agawal A. Metabolic syndrome and male fertility. World J Mens Health. 2019;37:113–27. https://doi.org/10.5534/wjmh.180055.
Lotti F, Corona G, Degli Innocenti S, Filimberti E, Scognamiglio V, Vignozzi L, Forti G, Maggi M. Seminal, ultrasound and psychobiological parameters correlate with metabolic syndrome in male members of infertile couples. Andrology. 2013;1:229–39. https://doi.org/10.1111/j.2047-2927.2012.00031.x.
Leisegang K, Udodong A, Bouic PJ, Henkel RR. Effect of the metabolic syndrome on male reproductive function: a case controlled pilot study. Andrologia. 2014;46:167–76.
Zhang F, Ye J, Zhu X, Wang L, Gao P, Shu G, Jiang Q, Wang S. Anti-obesity effects of dietary calcium: the evidence and possible mechanisms. Int J Mol Sci. 2019;20:3072. https://doi.org/10.3390/ijms20123072.
Alomaim H, Griffin P, Swist E, Plouffe LJ, Vandeloo M, Demonty I, Kumar A, Bertinato J. Dietary calcium affects body composition and lipid metabolism in rats. PLoS One. 2019;14:e0210760. https://doi.org/10.1371/journal.pone.0210760.
Das S, Choudhuri D. Dietary calcium regulates the insulin sensitivity by altering the adipokine secretion in high fat diet induced obese rats. Life Sci. 2020;250:117560. https://doi.org/10.1016/j.lfs.2020.117560.
Das S, Choudhuri D. Calcium supplementation shows a hepatoprotective effect against high-fat diet by regulating oxidative-induced inflammatory response and lipogenesis activity in male rats. J Tradit Complement Med. 2019;10:511–9. https://doi.org/10.1016/j.jtcme.2019.06.002.
Zhang F, Su H, Song M, Zheng J, Liu F, Yuan C, Fu Q, Chen S, Zhu X, Wang L, Gao P, Shu G, Jiang Q, Wang S. Calcium supplementation alleviates high-fat diet-induced estrous cycle irregularity and subfertility associated with concomitantly enhanced thermogenesis of brown adipose tissue and browning of white adipose tissue. J Agric Food Chem. 2019;67:7073–81. https://doi.org/10.1021/acs.jafc.9b02663.
Chandra A, Sengupta P, Goswami H, Sarkar M. Excessive dietary calcium in the disruption of structural and functional status of adult male reproductive system in rat with possible mechanism. Mol Cell Biochem. 2012;364:181–91. https://doi.org/10.1007/s11010-011-1217-3.
Beigi Harchegani A, Irandoost A, Mirnamniha M, Rahmani H, Tahmasbpour E, Shahriary A. Possible mechanisms for the effects of calcium deficiency on male infertility. Int. J Fertil Steril. 2019;12(267-272) https://doi.org/10.22074/IJFS.2019.5420.
Reeves PG, Nielsen FH, Fahey GC Jr. AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993;123:1939–51. https://doi.org/10.1093/jn/123.11.1939.
Noeman SA, Hamooda HE, Baalash AA. Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet induced obesity in rats. Diabetol Metab Syndr. 2011;3(1):17. https://doi.org/10.1186/1758-5996-3-17.
Wang EH, Yu ZL, Bu YJ, Xu PW, Xi JY, Liang HY. Grape seed proanthocyanidin extract alleviates high-fat diet induced testicular toxicity in rats. RSC Adv. 2019;9:11842–50. https://doi.org/10.1039/C9RA01017C.
Terpsidis KI, Papazahariadou MG, Taitzoglou IA, Papaioannou NG, Georgiadis MP, Theodoridis IT. Toxoplasma gondii: reproductive parameters in experimentally infected male rats. Exp Parasitol. 2009;121:238–41. https://doi.org/10.1016/j.exppara.2008.11.006.
Rezaei-Agdam H, Moshari S, Nahari E, Minas A, Daliri Z, Hallaj M, Razi M. Zeta and hyaluronic acid assessments, novel sperm selection procedures, in animal model for male infertility. Andrologia. 2019;51(11):e13447. https://doi.org/10.1111/and.13447.
Talalay P. Hydroxysteroid dehydrogenases. Methods Enzymol. 1962;5:512–32.
Arabak J, Adams JA, Williams-Ashman HG, Talalay P. Purification of 17 beta-hydroxysteroid dehydrogenase of human placenta and studies on its transhydrogenase function. J Biol Chem. 1962;237:345–57.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265–75.
Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–6.
Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974;47:469–74.
Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–69.
Erejuwa OO, Sulaiman SA, Wahab MS, Salam SK, Salleh MS, Gurtu S. Comparison of antioxidant effects of honey, glibenclamide, metformin, and their combinations in the kidneys of streptozotocin-induced diabetic rats. Int J Mol Sci. 2011;21(12):829–43. https://doi.org/10.3390/ijms12010829.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–8.
Reznick AZ, Packer L. Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol. 1994;233(3):57–63.
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem. 1982;126:131–8.
Minas A, Talebi H, Taravat Ray M, Yari Eisalou M, Alves MG, Razi M. Insulin treatment to type 1 male diabetic rats protects fertility by avoiding testicular apoptosis and cell cycle arrest. Gene. 2021;799:145847. https://doi.org/10.1016/j.gene.2021.145847.
Maadi MA, Minas A, Sepehri Vafa R, Tabatabaei-Naeini A, Bertolla RP. Apoptotic balance during testicular detorsion after one hour induced torsion in rats. Andrologia. 2022;54:e14349. https://doi.org/10.1111/and.14349.
Cui X, Long C, Zhu J, Tian J. Protective effects of fluvastatin on reproductive function in obese male rats induced by high-fat diet through enhanced signaling of mTOR. Cell PhysiolBiochem. 2017;41:598–608. https://doi.org/10.1159/000457881.
Han C, Liu C, Geng J, Tang Y, Li Y, Wang Y, Xie Z. Black and green tea supplements ameliorate male infertility in a murine model of obesity. J Med Food. 2020;23:1303–11. https://doi.org/10.1089/jmf.2020.4784.
Mu Y, Yan WJ, Yin TL, Zhang Y, Li J, Yang J. Diet-induced obesity impairs spermatogenesis: a potential role for autophagy. Sci Rep. 2017;9(7):43475. https://doi.org/10.1038/srep43475.
Leisegang K, Sengupta P, Agarwal A, Henkel R. Obesity and male infertility: mechanisms and management. Andrologia. 2021;53:e13617. https://doi.org/10.1111/and.13617.
Du Plessis SS, Cabler S, McAlister DA, Sabanegh E, Agarwal A. The effect of obesity on sperm disorders and male infertility. Nat Rev Urol. 2010;7(3):153–61. https://doi.org/10.1038/nrurol.2010.6.
Leisegang K, Henkel R, Agarwal A. Obesity and metabolic syndrome associated with systemic inflammation and the impact on the male reproductive system. Am J Reprod Immunol. 2019;82:e13178. https://doi.org/10.1111/aji.13178.
Mu Y, Dai HG, Luo LB, Yang J. Irisin alleviates obesity-related spermatogenesis dysfunction via the regulation of the AMPKα signalling pathway. Reprod Biol Endocrinol. 2021;8(19):135. https://doi.org/10.1186/s12958-021-00821-1.
Bellastella G, Menafra D, Puliani G, Colao A, Savastano S. Obesity Programs of nutrition, Education, Research and Assessment (OPERA) Group. How much does obesity affect the male reproductive function? Int J Obes Suppl. 2019;9:50–64. https://doi.org/10.1038/s41367-019-0008-2.
Das S, Choudhuri D. Dietary calcium regulates the risk renal injury in high fat diet induced obese rats by regulating renal lipid metabolism, oxidative stress and inflammation. Arch Physiol Biochem. 2022;128:1039–49. https://doi.org/10.1080/13813455.2020.1746812.
Suleiman JB, Nna VU, Zakaria Z, Othman ZA, Bakar ABA, Mohamed M. Obesity-induced testicular oxidative stress, inflammation and apoptosis: protective and therapeutic effects of orlistat. Reprod Toxicol. 2020;95:113–22. https://doi.org/10.1016/j.reprotox.2020.05.009.
Lui WY, Lee WM, Cheng CY. TGF-betas: their role in testicular function and Sertoli cell tight junction dynamics. Int J Androl. 2003;26(3):147–60. https://doi.org/10.1046/j.1365-2605.2003.00410.x.
Al-Megrin WA, El-Khadragy MF, Hussein MH, Mahgoub S, Abdel-Mohsen DM, Taha H, Bakkar AAA, Abdel Moneim AE, Amin HK. Green Coffea arabica extract ameliorates testicular injury in high-fat diet/streptozotocin-induced diabetes in rats. J Diabetes Res. 2020;2020:6762709. https://doi.org/10.1021/acsomega.2c02796.
Jia YF, Feng Q, Ge ZY, Guo Y, Zhou F, Zhang KS, Wang XW, Lu WH, Liang XW, Gu YQ. Obesity impairs male fertility through long-term effects on spermatogenesis. BMC Urol. 2018;18(1):42. https://doi.org/10.1186/s12894-018-0360-5.
Acknowledgements
Financial support for the project by the Indian Council of Medical Research (ICMR) Govt. of India, New Delhi is deeply acknowledged. The Infrastructural facility provided by The State Biotech Hub, Tripura University is acknowledged.
Funding
This study was funded by the Indian Council of Medical Research (ICMR), Govt. of India, grant number (5/7/1213/2014-RCH and 5/7/1617/2018 RBMH & CH).
Author information
Authors and Affiliations
Contributions
Conception and design: Sandeep Das and Dipayan Choudhuri. Acquisition, analysis, or interpretation of data: Soma Choudhuri, Sandeep Das, Susmita Sarkar, Panisree Roy Chowdhury, and Dipayan Choudhuri. Drafting the manuscript: Soma Choudhuri and Sandeep Das. Supervision: Sandeep Das and Dipayan Choudhuri. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The entire study was in conducted healthy male Wistar albino rats purchased from a government-registered animal supplier (Reg. No. 1443/PO/b/11/CPCSEA) nominated by Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests Govt of India. The entire study protocol was approved and followed by the guidelines of the Institutional Animal Ethical Committee, Tripura University (Ref.No: TU/IAEC/2014/VIII/2-6 dated 12.09.2014).
Consent for publication
All authors have consented to the publication of the manuscript.
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Choudhuri, S., Sarkar, S., Chowdhury, P.R. et al. Dietary calcium improves the reproductive functions against high-fat diet (HFD)–induced testicular toxicity in male obese rats. Nutrire 48, 55 (2023). https://doi.org/10.1186/s41110-023-00243-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-023-00243-6