Abstract
Purpose
To investigate the effects of juçara pulp supplementation on energy homeostasis and metabolic complications in diet-induced obese mice.
Methods
Mice received a control (C) or a high-fat diet (H) for 16 weeks and for half the diet was supplemented with 0.5% freeze-dried juçara powder (CJ and HJ groups, respectively). Food intake, body weight, energy expenditure, glucose tolerance, insulin sensitivity, liver steatosis, and uncoupling protein 1 (UCP1) expression in brown adipocytes were analyzed.
Results
Juçara pulp supplementation reduced body weight gain in HJ compared to H group but increased body weight in CJ compared to C group. Energy intake was higher in the CJ group than in C, H, and HJ, but similar between H and HJ groups. Despite similar energy intake, juçara pulp supplementation increased energy expenditure in HJ compared to H group. However, UCP-1 expression was not affected by juçara. Also, H mice exhibited a severe diet-induced glucose intolerance and insulin resistance, all of which were significantly improved in HJ mice. In addition, H group had higher fat content in hepatocytes than HJ, whereas no difference was observed between C and CJ.
Conclusions
Juçara pulp has a great potential to reduce the metabolic complications associated with obesity.
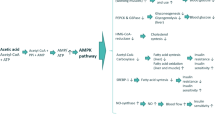
Graphical abstract

Juçara pulp supplementation in high-fat diet fed mice improved insulin resistance and glucose intolerance besides protecting mice from liver steatosis and obesity.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AIN:
-
American Institute of Nutrition
- ANOVA:
-
analysis of variance
- AUC:
-
area under the curve
- BSA:
-
bovine serum albumin
- C:
-
control diet group
- CAT:
-
catalase
- CEDEME:
-
Center for Development of Animal Models for Medicine and Biology
- CEUA:
-
Institutional Ethics Committee on Animal Use
- CJ:
-
control diet group supplemented with juçara 0.5% freeze-dried powder
- H:
-
high-fat diet group
- HE:
-
hematoxylin and eosin
- HJ:
-
high-fat diet group supplemented with juçara 0.5% freeze-dried powder
- IL-6:
-
interleukin 6
- IPEMA:
-
Institute of Permaculture and Ecovillages of the Atlantic
- ipGTT:
-
intraperitoneal glucose tolerance test
- ipITT:
-
intraperitoneal insulin tolerance test
- MCP-1:
-
monocyte chemoattractant protein-1
- NAFLD:
-
non-alcoholic fatty liver disease
- PUFAs:
-
polyunsaturated fatty acids
- SE:
-
standard error of the mean
- TNF-α:
-
tumor necrosis factor alpha
- UCP-1:
-
uncoupling protein 1
- WHO:
-
World Health Organization
References
WHO. Obesity and overweight. Fact Sheets. 2020 [Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight].
Collaborators GO. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017;377(1):13–27.
Schetz M, De Jong A, Deane AM, Druml W, Hemelaar P, Pelosi P, et al. Obesity in the critically ill: a narrative review. Intensive Care Med. 2019;45(6):757–769.
Ley SH, Hamdy O, Mohan V, Hu FB. Prevention and management of type 2 diabetes: dietary components and nutritional strategies. Lancet. 2014;383(9933):1999–2007.
Alissa EM, Ferns GA. Dietary fruits and vegetables and cardiovascular diseases risk. Crit Rev Food Sci Nutr. 2017;57(9):1950–62.
Jeznach-Steinhagen A, Ostrowska J, Czerwonogrodzka-Senczyna A, Boniecka I, Shahnazaryan U, Kuryłowicz A. Dietary and pharmacological treatment of nonalcoholic fatty liver disease. Medicina. 2019;55(5):166.
Scalbert A, Johnson IT, Saltmarsh M. Polyphenols: antioxidants and beyond. Am J Clin Nutr. 2005;81(1):215S–7S.
Kishimoto Y, Tani M, Kondo K. Pleiotropic preventive effects of dietary polyphenols in cardiovascular diseases. Eur J Clin Nutr. 2013;67(5):532–5.
Vauzour D. Dietary polyphenols as modulators of brain functions: biological actions and molecular mechanisms underpinning their beneficial effects. Oxidative Med Cell Longev. 2012;2012:1–16.
Kong J-M, Chia L-S, Goh N-K, Chia T-F, Brouillard R. Analysis and biological activities of anthocyanins. Phytochemistry. 2003;64(5):923–33.
Vendrame S, Del Bo C, Ciappellano S, Riso P, Klimis-Zacas D. Berry fruit consumption and metabolic syndrome. Antioxidants. 2016;5(4):34.
Tsuda T, Horio F, Uchida K, Aoki H, Osawa T. Dietary cyanidin 3-O-β-D-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J Nutr. 2003;133(7):2125–30.
Lee Y-M, Yoon Y, Yoon H, Park H-M, Song S, Yeum K-J. Dietary anthocyanins against obesity and inflammation. Nutrients. 2017;9(10):1089.
Ayoub HM, McDonald MR, Sullivan JA, Tsao R, Platt M, Simpson J, et al. The effect of anthocyanin-rich purple vegetable diets on metabolic syndrome in obese zucker rats. J Med Food. 2017;20(12):1240–9.
Jia Y, Wu C, Kim Y-S, Yang SO, Kim Y, Kim J-S, et al. A dietary anthocyanin cyanidin-3-O-glucoside binds to PPARs to regulate glucose metabolism and insulin sensitivity in mice. Commun Biol. 2020;3(1):1–10.
Peirce V, Vidal-Puig A. Regulation of glucose homoeostasis by brown adipose tissue. Lancet Diabetes Endocrinol. 2013;1(4):353–60.
Vieira GS, Marques AS, Machado MT, Silva VM, Hubinger MD. Determination of anthocyanins and non-anthocyanin polyphenols by ultra performance liquid chromatography/electrospray ionization mass spectrometry (UPLC/ESI–MS) in jussara (Euterpe edulis) extracts. J Food Sci Technol. 2017;54(7):2135–44.
Silva NAD, Rodrigues E, Mercadante AZ, de Rosso VV. Phenolic compounds and carotenoids from four fruits native from the Brazilian Atlantic forest. J Agric Food Chem. 2014;62(22):5072–84.
Oyama L, Silva F, Carnier J, De Miranda D, Santamarina A, Ribeiro E, et al. Juçara pulp supplementation improves glucose tolerance in mice. Diabetol Metab Syndr. 2016;8(1):8.
Santamarina AB, Jamar G, Mennitti LV, De Rosso VV, Cesar HC, Oyama LM, et al. The Use of Juçara (Euterpe edulis Mart.) supplementation for suppression of NF-κB pathway in the hypothalamus after high-fat diet in Wistar rats. Molecules. 2018;23(7):1814.
Santamarina AB, Jamar G, Mennitti LV, Ribeiro DA, Cardoso CM, de Rosso VV, et al. Polyphenols-rich fruit (Euterpe edulis Mart.) prevents peripheral inflammatory pathway activation by the short-term high-fat diet. Molecules. 2019;24(9):1655.
Santamarina AB, Jamar G, Mennitti LV, de Cássia Cesar H, Vasconcelos JR, Oyama LM, et al. Obesity-related inflammatory modulation by juçara berry (Euterpe edulis Mart.) supplementation in Brazilian adults: a double-blind randomized controlled trial. Eur J Nutr. 2020;59(4):1693–1705.
Reeves PG, Nielsen FH, Fahey GC Jr. AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993;123(11):1939–51.
Ribeiro JC, Antunes LMG, Aissa AF, Darin JDAC, De Rosso VV, Mercadante AZ, et al. Evaluation of the genotoxic and antigenotoxic effects after acute and subacute treatments with açai pulp (Euterpe oleracea Mart.) on mice using the erythrocytes micronucleus test and the comet assay. Mutat Res Genet Toxicol Environ Mutagen. 2010;695(1-2):22–8.
Valenti L, Riso P, Mazzocchi A, Porrini M, Fargion S, Agostoni C. Dietary anthocyanins as nutritional therapy for nonalcoholic fatty liver disease. Oxidative Med Cell Longev. 2013;2013:1–8.
Karlsen A, Retterstøl L, Laake P, Paur I, Kjølsrud-Bøhn S, Sandvik L, et al. Anthocyanins inhibit nuclear factor-κ B activation in monocytes and reduce plasma concentrations of pro-inflammatory mediators in healthy adults. J Nutr. 2007;137(8):1951–4.
Center for Drug Evaluation and Research. Guidance for Industry. US Department of Health and Human Services; Washington, DC, USA: 2005. Estimating the maximum safe starting dose in initial clinical trials for therapeutics in adult healthy volunteers; pp. 1–27.
Matthews JN, Altman DG, Campbell MJ, Royston P. Analysis of serial measurements in medical research. BMJ. 1990;300(6719):230–5.
Zhang XG, Xu P, Liu Q, Yu CH, Zhang Y, Chen SH, et al. Effect of tea polyphenol on cytokine gene expression in rats with alcoholic liver disease. Hepatobiliary Pancreat Dis Int. 2006;5(2):268–72.
Aguiar O, Gollücke AP, de Moraes BB, Pasquini G, Catharino RR, Riccio MF, et al. Grape juice concentrate prevents oxidative DNA damage in peripheral blood cells of rats subjected to a high-cholesterol diet. Br J Nutr. 2011;105(5):694–702.
Wu T, Gao Y, Guo X, Zhang M, Gong L. Blackberry and blueberry anthocyanin supplementation counteract high-fat-diet-induced obesity by alleviating oxidative stress and inflammation and accelerating energy expenditure. Oxidative Med Cell Longev. 2018;2018:1–9.
Bertoia ML, Rimm EB, Mukamal KJ, Hu FB, Willett WC, Cassidy A. Dietary flavonoid intake and weight maintenance: three prospective cohorts of 124 086 US men and women followed for up to 24 years. BMJ. 2016; 352:i17.
Yamazaki T, Ikaga R, Li D, Nakae S, Tanaka S. A novel method for measuring diet-induced thermogenesis in mice. MethodsX. 2019;6:1950–6.
Hoek-van den Hil EF, van Schothorst EM, van der Stelt I, Swarts HJ, van Vliet M, Amolo T, et al. Direct comparison of metabolic health effects of the flavonoids quercetin, hesperetin, epicatechin, apigenin and anthocyanins in high-fat-diet-fed mice. Genes Nutr. 2015;10(4):23.
Argentato PP, Morais CA, Santamarina AB, de Cássia César H, Estadella D, de Rosso VV, et al. Jussara (Euterpe edulis Mart.) supplementation during pregnancy and lactation modulates UCP-1 and inflammation biomarkers induced by trans-fatty acids in the brown adipose tissue of offspring. Clin Nutr Exp. 2017;12:50–65.
Ikeda K, Yamada T. UCP1 dependent and independent thermogenesis in brown and beige adipocytes. Front Endocrinol. 2020;11:498.
Zhou F, Guo J, Han X, Gao Y, Chen Q, Huang W, et al. Cranberry polyphenolic extract exhibits an antiobesity effect on high-fat diet–fed mice through increased thermogenesis. J Nutr. 2020;150(8):2131–8.
Bjursell M, Gerdin AK, Lelliott CJ, Egecioglu E, Elmgren A, Törnell J, et al. Acutely reduced locomotor activity is a major contributor to Western diet-induced obesity in mice. Am J Physiol Endocrinol Metab. 2008;294(2):E251–60.
Thaler JP, Yi CX, Schur EA, Guyenet SJ, Hwang BH, Dietrich MO, et al. Obesity is associated with hypothalamic injury in rodents and humans. J Clin Invest. 2012;122(1):153–62.
Winzell MS, Ahrén B. The high-fat diet-fed mouse: a model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes. 2004;53(Suppl 3):S215–9.
Nizamutdinova IT, Jin YC, Chung JI, Shin SC, Lee SJ, Seo HG, et al. The anti-diabetic effect of anthocyanins in streptozotocin-induced diabetic rats through glucose transporter 4 regulation and prevention of insulin resistance and pancreatic apoptosis. Mol Nutr Food Res. 2009;53(11):1419–29.
Takikawa M, Inoue S, Horio F, Tsuda T. Dietary anthocyanin-rich bilberry extract ameliorates hyperglycemia and insulin sensitivity via activation of AMP-activated protein kinase in diabetic mice. J Nutr. 2010;140(3):527–33.
Galisteo M, Duarte J, Zarzuelo A. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. J Nutr Biochem. 2008;19(2):71–84.
Almeida Morais C, Oyama LM, Oliveira JLD, Carvalho Garcia M, Rosso VVD, Sousa Mendes Amigo L, et al. Jussara (Euterpe edulis Mart.) supplementation during pregnancy and lactation modulates the gene and protein expression of inflammation biomarkers induced by trans-fatty acids in the colon of offspring. Mediat Inflamm. 2014;2014:987927.
Uysal KT, Wiesbrock SM, Marino MW, Hotamisligil GS. Protection from obesity-induced insulin resistance in mice lacking TNF-α function. Nature. 1997;389(6651):610–4.
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2017;114(12):1752–61.
Unger EK, Piper ML, Olofsson LE, Xu AW. Functional role of c-Jun-N-terminal kinase in feeding regulation. Endocrinology. 2010;151(2):671–82.
Jennings A, Welch AA, Spector T, Macgregor A, Cassidy A. Intakes of anthocyanins and flavones are associated with biomarkers of insulin resistance and inflammation in women. J Nutr. 2014;144(2):202–8.
Cassidy A, Rogers G, Peterson JJ, Dwyer JT, Lin H, Jacques PF. Higher dietary anthocyanin and flavonol intakes are associated with anti-inflammatory effects in a population of US adults. Am J Clin Nutr. 2015;102(1):172–81.
de Liz S, Cardoso AL, Copetti CLK, de Fragas Hinnig P, Vieira FGK, da Silva EL, et al. Açaí (Euterpe oleracea Mart.) and juçara (Euterpe edulis Mart.) juices improved HDL-c levels and antioxidant defense of healthy adults in a 4-week randomized cross-over study. Clin Nutr. 2020;39(12):3629–3636.
Alvarez-Suarez JM, Giampieri F, Tulipani S, Casoli T, Di Stefano G, González-Paramás AM, et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J Nutr Biochem. 2014;25(3):289–94.
Code availability
Not applicable.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)—Finance Code 001 and by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, grant 2011/05932-3 and 2017/26075-8).
Author information
Authors and Affiliations
Contributions
MB, CAMO and LPP were responsible for the design of the study; MB and CAMO conducted the study; MB, CAMO, TLM, FPC, IDB, DAR, DE, LLM and VVR helped with experimental procedures, data analysis and discussion.
Corresponding author
Ethics declarations
Ethics approval
All procedures were approved by the Institutional Ethics Committee on Animal Use (CEUA n.1750/11) from Federal University of Sao Paulo.
Consent to participate
Not applicable.
Consent for publication
All authors listed have contributed to the work, read, approved, and agreed to submit the manuscript.
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Barthichoto, M., Moretto, T.L., de Carvalho, F.P. et al. Juçara (Euterpe edulis Mart.) supplementation reduces body weight gain and protects mice from metabolic complications induced by high-fat diet. Nutrire 46, 6 (2021). https://doi.org/10.1186/s41110-021-00135-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-021-00135-7