Abstract
Peritoneal dialysis (PD) stands as an important modality among kidney replacement therapies for end-stage kidney disease, offering patients remarkable flexibility and autonomy. Despite its widespread use, challenges such as glucose-related complications, peritoneal membrane fibrosis, declining renal function, and cardiovascular risks persist, necessitating innovative therapeutic approaches. Sodium–glucose cotransporter 2 (SGLT2) inhibitors, originally developed for treating type 2 diabetes mellitus, have recently shown promise as add-on therapy for patients with diabetic and non-diabetic chronic kidney disease (CKD), even in advanced stages. This review describes the potential role of SGLT2 inhibitors as a breakthrough therapeutic option in PD, emphasizing their ability to address unmet clinical needs and improve patient outcomes. The multiple effects of SGLT2 inhibitors in CKD, including metabolic modulation, antihypertensive, diuretic, anemia-reducing, antioxidant, and antiinflammatory properties, are reviewed in the context of PD challenges. Additionally, the potentially protective influence of SGLT2 inhibitors on the integrity of the peritoneal membrane and the transport of solutes and water in the peritoneum are emphasized. Despite these encouraging results, the paper highlights the potential risks associated with SGLT2 inhibitors in PD and emphasizes the need for cautious and thorough investigation of dosing, long-term safety considerations, and patient-specific factors through comprehensive clinical trials. Looking forward, the review argues for well-designed studies to evaluate the expanded safety profile of SGLT2 inhibitors in PD, with particular attention paid to peritoneal membrane integrity and overall patient outcomes.
Similar content being viewed by others

Background
Sodium–glucose cotransporter 2 (SGLT2) inhibitors have emerged as potential game changers in the management of chronic kidney disease (CKD), showcasing not only remarkable glycemic control but also profound nonglycemic effects that intricately improve glomerular hemodynamics, modulate volume status, and influence local and systemic factors in CKD and cardiovascular disease (CVD) pathogenesis [1,2,3]. Robust evidence from pivotal trials, such as SCORED, CREDENCE, and DAPA-CKD, demonstrates that SGLT2 inhibitors may prevent the development of CKD, delay the worsening of CKD, and reduce the risk of adverse cardiovascular events in patients with moderate-to-severe CKD, including those without diabetes [4,5,6]. These groundbreaking findings have prompted the expansion of approved clinical indications for SGLT2 inhibitors, now encompassing patients with CKD, with or without diabetes, as well as those contending with heart failure, with or without reduced ejection fraction [1, 2, 7, 8]. However, it is worth noting that the trials’ lowest estimated glomerular filtration rate (eGFR) inclusion criterion was 20 mL/min/1.73 m2 [9], leaving an unanswered query about the feasibility and safety of SGLT2 inhibitor prescription in patients with end-stage kidney disease (ESKD).
The existing evidence for the potential benefits of SGLT2 inhibitors in patients with ESKD is limited, primarily originating from post hoc analyses of randomized clinical trials (RCTs), preclinical studies, or case reports. These investigations lend credence to the notion that, even with minimal diuresis and anticipated minimal delivery to the transporter in dialysis patients, SGLT2 inhibitors may exert direct and indirect effects, offering potential protection against cardiovascular events and mortality [10,11,12]. For example, the post hoc analysis of the DAPA-CKD study revealed that 14.5% of patients, irrespective of diabetes status, exhibited CKD stage 4 (eGFR < 30 mL/min/1.73 m2) [10]. Dapagliflozin demonstrated both safety and a significant reduction in major kidney and cardiovascular events, while mitigating progressive eGFR reduction [10]. Similar promising outcomes emerged from a post hoc analysis of CREDENCE data, indicating a potential slowing of CKD progression, even in cases of advanced diabetic nephropathy [11].
Despite these promising findings, a critical gap persists in our understanding of the feasibility and safety of prescribing SGLT2 inhibitors to patients with ESKD, including those undergoing peritoneal dialysis (PD). The nature of PD and its associated complications suggest several potential positive effects of SGLT2 inhibition, expounded upon in this review. This review navigates through the multifaceted benefits that SGLT2 inhibitors might confer in patients undergoing PD. Preservation of residual kidney function (RKF) and prevention of volume depletion, pivotal factors influencing patient outcomes in PD, are scrutinized in light of the renoprotective effects attributed to SGLT2 inhibitors. Improved control of glycemia and lipid levels, antiinflammatory effects, and potential cardiovascular advantages are among the discussed potential benefits. Concurrently, the impact of these agents on the peritoneal membrane, a critical component in the success of PD, is examined to determine whether SGLT2 inhibitors hold promise in maintaining peritoneal membrane integrity. However, every therapeutic intervention introduces a set of considerations, and SGLT2 inhibitors are no exception. This review aims to dissect the intricate interplay between the potential benefits and risks associated with the use of SGLT2 inhibitors in PD.
PD realities: a brief overview of the main benefits and challenges
PD emerges as a valuable modality for managing kidney failure, offering numerous advantages, such as home treatment convenience, enhanced quality of life, cost effectiveness in most countries, technical simplicity, a slower decline in residual kidney function (RKF) compared with hemodialysis (HD), and improved survival, especially in the initial years of therapy initiation [13, 14]. However, these benefits are accompanied by a distinctive set of challenges, including issues common to the broader dialysis population due to kidney failure and specific intricacies unique to PD [13, 14]. Key challenges shared with the HD population encompass a decline in RKF, overhydration, hypertension, metabolic disturbances, dyslipidemia, electrolyte imbalances, anemia, oxidative stress, and chronic inflammation [13, 14]. These challenges significantly contribute to heightened morbidity and mortality among patients undergoing PD. For instance, patients on PD commonly experience a gradual decline in RKF, influenced by factors inherent to kidney failure and the dialysis process [15]. While kidney replacement therapy typically begins at an eGFR of 5–10 mL/min/1.73 m2, most patients initiating dialysis possess some level of RKF [15, 16]. However, over time, there has been a gradual decline in RKF, contributing to increased morbidity and mortality among dialysis patients [15,16,17]. Preserving RKF in patients treated with PD has demonstrated significant associations with reduced incidences of infections, depression, improved nutrition, better volume and blood pressure control, as well as diminished requirements for erythropoietin, and decreased chronic inflammation [16,17,18]. This, in turn, holds the potential to lead to lower rates of PD technique failure and improved cardiovascular outcomes [16, 17, 19]. Overhydration [20, 21], anemia [22], dyslipidemia [23, 24], and chronic inflammation [25] have also been associated with PD technique failure and an increased risk of all-cause and cardiovascular morbidity and mortality among PD patients.
PD-specific challenges encompass concerns related to glucose absorption from PD solutions, PD-associated infections and the longevity of the peritoneal membrane [25,26,27]. Long-term exposure to glucose-based PD solutions and the accumulation of advanced glycation end-products in the peritoneal membrane, due to prolonged exposure to glucose-based PD solutions, have been proven to be associated with peritoneal membrane damage and fibrosis, leading to the loss of peritoneal ultrafiltration and technique failure [26, 28, 29]. Moreover, chronic exposure to hyperglycemic PD solutions induces oxidative stress in peritoneal mesothelial cells (PMCs), causing them to generate a considerable amount of reactive oxygen species (ROS), which initiate downstream inflammatory signaling pathways, increase the expression of fibrogenic factors and enhance the process of epithelial-to-mesenchymal transition (EMT) [26, 29, 30]. Consequently, this cascade of events leads to the development and accelerated progression of peritoneal fibrosis [26, 28, 30]. Therefore, minimizing the glucose load and optimizing the biocompatibility of PD solutions are essential for preserving the longevity of the peritoneal membrane and, consequently, PD technique survival.
Another major problem in PD is PD-related infections, which affect clinical outcomes, such as peritoneal ultrafiltration, technique failure, and cardiac and overall patient mortality [25, 31, 32]. Infection-related PD technique failure has been associated with an increased risk of mortality, with 30% of patients dying within the first 2 years of PD technique failure [32]. The number of peritonitis episodes has been shown to correlate with a decrease in RKF [33] and even a single episode of peritonitis leads to an increase in small solute transport and a decrease in the ultrafiltration rate [34]. These challenges underscore the intricate balance required in managing patients undergoing PD to optimize the benefits of the modality while effectively addressing the unique hurdles it presents. However, the current strategies to preserve RKF, reduce high glucose-induced transformation of the peritoneal membrane, and consequently prolong PD treatment and patient survival are presently limited [13, 16, 35]. These strategies include practices such as restricting the use of hyperglycemic PD solutions, preventing peritonitis, avoiding nephrotoxins, utilizing biocompatible PD fluids, and incorporating renin–angiotensin–aldosterone system (RAAS) inhibitors [16, 26, 35]. Within this complex landscape, emerging evidence suggests that SGLT2 inhibitors might offer potential advantages in managing some of the challenges associated with PD.
Navigating SGLT2 inhibitors in CKD: insights for PD perspectives
SGLT2 inhibitors, a relatively recent class of pharmacological agents initially designed for treating type 2 diabetes mellitus (T2DM), primarily target the SGLT2 protein responsible for glucose reabsorption in the kidney’s proximal tubules [36,37,38]. The SGLT2 protein functions by facilitating the reabsorption of glucose from the urine back into the bloodstream, and its mechanism involves the coupled movement of sodium ions (Na+) and glucose [37, 39]. In the kidneys, glucose reabsorption is intertwined with the movement of Na+ , where the Na+/K+-ATPase at the basolateral membrane maintains a lower Na+ concentration within tubular epithelial cells compared with the glomerular filtrate [39, 40]. Na+ passively moves into the cell along the electrochemical gradient, and glucose enters the cell alongside Na+ in a 1:1 ratio through the SGLT, following the Na+ concentration gradient. Subsequently, it exits the cell passively through basolateral glucose transporter 2 (GLUT2) [39]. SGLT2, primarily expressed in the kidney and located in the brush border membrane of the S1 segment of renal tubules, is a low-affinity, high-capacity transporter responsible for approximately 90% of glucose reabsorption [40,41,42]. Inhibiting SGLT2 thus reduces the reabsorption of Na+ and glucose in the proximal tubule, thereby decreasing blood glucose levels [2, 43].
Studies have demonstrated that SGLT2 inhibitors can lower hemoglobin A1c (HbA1c) by 0.6–1% and preserve kidney function in T2DM patients [2, 43]. The resulting glucosuria can exceed 100 g/d in T2DM patients and ranges from 50 to 60 g/d in those without diabetes [44, 45]. While glucosuric efficacy diminishes with a decline in estimated glomerular filtration rate (eGFR) and becomes minimal when eGFR falls below 30 ml/min per 1.73 m2 [2, 46], SGLT2 inhibitors offer various benefits beyond glycemic control, underscoring their relevance in the comprehensive management of patients with CKD [2, 7, 8]. The existing knowledge of the advantages of SGLT2 inhibitors in the broader CKD population, as outlined below, may offer valuable perspectives for the management of patients undergoing PD (Fig. 1).
SGLT2 inhibitors and hemodynamic regulation
The tubuloglomerular feedback (TGF) signaling pathway is crucial in regulating GFR by influencing preglomerular vascular tone [47]. When preglomerular vasodilatation occurs, it enhances glomerular filtration, leading to increased sodium delivery to the macula densa within the juxtaglomerular apparatus [48, 49]. However, chronic hyperglycemia disrupts this process as the obligatory reabsorption of sodium and glucose in the proximal tubule by SGLT2 results in reduced sodium delivery to the distal tubule and macula densa [40, 49]. Treatment with SGLT2 inhibitors addresses this issue by attenuating glucose and sodium reabsorption in the proximal tubule, thereby maintaining sodium delivery to the macula densa [47, 50]. Consequently, this prompts vasoconstriction in the preglomerular afferent arterioles, alleviating renal hyperfiltration and reducing glomerular pressure [51]. In a study by Thomson et al., diabetic rats given SGLT2 inhibitors showed a reduction of approximately 30% in proximal tubular sodium reabsorption, a decrease in glomerular capillary hydrostatic pressure by 5–8 mmHg, and a decrease in measured GFR (mGFR) by around 25% [50]. This study underscored the significance of TGF in responding to SGLT2 inhibition and suggested that postglomerular vasorelaxation might contribute to this response [50]. Clinical studies mirror these results, demonstrating a transient decline in mGFR by 2–5 mL/min/1.73 m2 upon SGLT2 inhibitor initiation, aligning with the decrease in glomerular pressure in patients with type 1 diabetes mellitus (T1DM) exhibiting renal hyperfiltration (GFR ≥ 135 mL/min/1.73 m2) [47]. Patients with T2DM exhibited comparable reductions in glomerular pressure [52, 53], and similar kidney-protective mechanisms, including natriuresis-induced GFR dips, have been demonstrated in non-diabetic patients with CKD [54]. Therefore, SGLT2 inhibitors, through TGF, cause increased sodium passage along the nephron, sensed by macula cells to constrict afferent glomerular arterioles, thereby protecting glomeruli by reducing intraglomerular pressure.
Nevertheless, the influence of SGLT2 inhibitors transcends the dynamics within the glomerulus to systemic hemodynamics, resulting in a decrease in systolic blood pressure by approximately 3–6 mmHg and diastolic blood pressure by around 1–2 mm Hg [46, 55]. This hypotensive impact is orchestrated through mechanisms such as natriuresis and associated plasma volume contraction [56], mitigation of arterial stiffness [57], and enhancement of endothelial function [58]. Importantly, this reduction in blood pressure remains consistent regardless of the individual’s hypertension status [59] and is observed in patients with a low eGFR [45].
SGLT inhibitors, albuminuria, and kidney protection
Clinical trials have consistently indicated that the use of an SGLT2 inhibitor can provide kidney protection through a decreased rate of decline in eGFR and reduced onset or progression of albuminuria [6, 9, 60, 61]. The pioneering CREDENCE trial was the first to explore the influence of SGLT2 inhibitors on kidney-related outcomes [6]. Enrolling 4401 participants diagnosed with T2DM and albuminuric CKD, the trial included patients with an eGFR ranging from 30 to 90 mL/min/1.73 m2 and a urinary albumin-to-creatinine ratio (UACR) between 300 and 5000 mg/g [6]. The participants were concurrently on maximally tolerated background therapy with renin–angiotensin system (RAS) inhibitors. The trial was prematurely halted following a planned interim analysis due to meeting preestablished efficacy criteria. Importantly, the impact of SGLT2 inhibitors on kidney-related outcomes was consistent across diverse levels of renal function and albuminuria. The EMPA-KIDNEY trial expanded the evidence supporting kidney protection to the lowest eGFR range by including participants with or without T2DM and having an eGFR of 20–45 mL/min/1.73 m2, regardless of albuminuria [9]. Alternatively, participants with an eGFR of 45–90 mL/min/1.73 m2 and UACR ≥ 200 mg/g, who were already on maximally tolerated RAS inhibitors were also enrolled. With 6609 participants, this trial stands as the largest investigation of SGLT2 inhibitors in chronic kidney disease (CKD) patients. With a mean eGFR of 37.5 mL/min/1.73 m2 and a median UACR of 412 mg/g, the trial provided compelling evidence of efficacy, demonstrating a lower risk of CKD progression or death from cardiovascular causes compared to the placebo [9].
A systematic review and meta-analysis of randomized controlled trials revealed a statistically significant reduction in albuminuria with SGLT2 inhibitors compared to placebo or active control, irrespective of the presence or absence of renal dysfunction [62]. In a meta-analysis using data from eligible cohorts in the Chronic Kidney Disease Prognosis Consortium, Coresh et al. demonstrated that SGLT2 inhibitors lowered albuminuria in patients with diabetic chronic kidney disease by at least 30% over a 2-year period [61].
The mechanisms underlying the beneficial effects of SGLT2 inhibitors on albuminuria and kidney function in patients with CKD are not fully understood, but the described mechanisms, such as a reduction in glomerular hypertension and an improvement in tubular function, have been proposed [60, 63]. Consequently, initiating SGLT2 inhibition earlier in the progression of albuminuria yielded more favorable outcomes in terms of kidney function preservation, as evidenced by the aforementioned studies. This implies that protective effects on the kidneys can be realized irrespective of the extent of albuminuria. The presence of normo-albuminuria should not be a justification for delaying treatment in conditions prone to developing albuminuria, nor should it deter the initiation of treatment in conditions frequently associated with non-albuminuric CKD, such as tubulointerstitial kidney disease [64].
The effects of SGLT2 inhibitors on kidneys in patients with kidney failure are still uncertain. Nevertheless, a retrospective study involving seven diabetic patients undergoing incremental HD revealed the safety and effectiveness of SGLT2 inhibitors in preserving RKF and improving cardiovascular health [12]. This study indicates that incorporating SGLT2 inhibitors into HD protocols may have the potential to alleviate interdialytic weight gain and sustain RKF [12].
SGLT2 inhibitors and metabolic effects
The mitigation of chronic hyperglycemia and the alleviation of glucose toxicity achieved through SGLT2 inhibitors contribute to the enhancement of both insulin secretion by beta cells and insulin sensitivity in peripheral tissues [65, 66].
Additionally, SGLT2 inhibitors can induce weight loss, a phenomenon well documented in various studies involving patients with T2DM. For approved SGLT2 inhibitors, an average weight loss of approximately 1.5–2 kg (placebo adjusted) is observed, and when combined with other drugs that possess anorectic effects or reduce hepatic glucose output, the weight loss can escalate to 3–5 kg [67, 68].
Moreover, SGLT2 inhibitors exhibit noteworthy effects on lipid metabolism, operating at various cellular levels [40, 69]. By diminishing lipid accumulation in visceral and subcutaneous fat, these inhibitors not only contribute to weight reduction but also bring about alterations in body composition. They modulate key molecules involved in lipid synthesis and oxidation, resulting in a decrease in circulating triglycerides and an increase in high-density lipoprotein cholesterol [69].
A comprehensive meta-analysis of 60 randomized trials revealed that SGLT2 inhibition led to an increase in total, LDL, and HDL cholesterol while decreasing triglycerides, with slight variations in effect sizes based on drug type, dosage, and ethnicity [70]. Consistent findings were reported in another systematic review and meta-analysis encompassing 48 randomized controlled trials, demonstrating that SGLT2 inhibitors significantly increased total cholesterol, LDL cholesterol, non-HDL cholesterol, and HDL cholesterol, while simultaneously decreasing triglycerides [71].
SGLT2 inhibitors and diuretic effect
Although SGLT2 inhibitors are known to induce osmotic diuresis and some natriuresis, their specific diuretic effects in patients with CKD are still being elucidated. Clinical studies have shown that SGLT2 increases urine volume in patients with T2DM during the first few days of treatment [72,73,74]. In the RECEDE-CHF trial, which investigated the renal and cardiovascular effects of SGLT2 inhibition in combination with loop diuretics in patients with T2DM and chronic heart failure, 26.1% of the recruited patients had CKD stage 3a (eGFR, 45–60 mL/min/1.73 m2) [75]. The study observed a mean increase in 24-h urine volume of 535 ml 3 days after SGLT2 inhibitor initiation and 545 ml by 6 weeks in these patients, indicating a diuretic effect of SGLT2 inhibitors in the presence of CKD and heart failure [75].
However, a comprehensive review of the diuretic effects of SGLT2 inhibitors highlighted the challenges in precisely quantifying the diuretic effect and the divergent results from studies using indirect estimations [76]. The review emphasized that SGLT2 inhibitors present some favorable properties compared with classical diuretics, such as reduced sympathetic activity, preserved potassium balance, lower risk of acute kidney injury, and decreased serum uric acid levels [76]. Another review discussed the diuretic effects of SGLT2 inhibitors and their influence on the renin–angiotensin system (RAS) [77]. It was noted that the diuretic effects of SGLT2 inhibitors, including diuresis, natriuresis, and associated body fluid loss, may activate the RAS, but these effects appear to be transient based on available data in patients with T2DM [77].
SGLT2 inhibitors and serum electrolyte levels
SGLT2 inhibitors can cause a reduction in serum sodium levels due to their enhanced natriuresis effect [39]. This effect is related to the inhibition of SGLT2 receptors, which promote glycosuria, natriuresis, and osmotic diuresis, leading to changes in electrolyte levels [39, 78]. The reduction in serum sodium levels is usually mild and transient, and it is not a common side effect of SGLT2 inhibitors [39, 79]. Apart from sodium, SGLT2 inhibitors may impact other electrolytes in patients with T2DM, including small increases in serum concentrations of magnesium, phosphate and calcium [78, 80, 81]. In a recent meta-analysis involving 28,269 patients with T2DM and six SGLT2 inhibitors, an elevation in serum magnesium by 0.07 mmol/L (95% CI, 0.06–0.08 mmol/L) and serum phosphate by 0.03 mmol/L (95% CI, 0.02–0.04 mmol/L) compared with placebo was demonstrated [81].
The elevation in serum magnesium levels observed with the use of SGLT2 inhibitors can be attributed to various mechanisms. These encompass the osmotic diuretic effect resulting from increased glucose excretion in the urine, causing heightened excretion of fluid and electrolytes, including magnesium, in the renal tubules [79]. Additionally, SGLT2 inhibitors have demonstrated the ability to decrease the fractional excretion of magnesium, signifying enhanced tubular reabsorption [79, 82]. They induce magnesium reabsorption and contribute to extracellular fluid volume depletion by activating the renin–angiotensin–aldosterone system [82]. Furthermore, these inhibitors promote the upregulation of magnesium transporters in the kidney or gut, leading to increased magnesium reabsorption and diminished excretion [82].
The elevation in serum phosphate concentration associated with the use of SGLT2 inhibitors may result from increased renal tubular phosphate reabsorption, hormonal changes affecting phosphate regulation, such as parathyroid hormone (PTH) and fibroblast growth factor 23 (FGF23), and direct pharmacological effects [79,80,81].
The existing data on increased serum calcium levels induced by SGLT2 inhibitors primarily stem from case report studies [78, 83]. However, a placebo-controlled randomized double-blind study involving 42 patients with T2DM found no significant effects on serum calcium [80]. The precise mechanisms underlying this phenomenon are not fully understood, but it is speculated to be associated with the osmotic diuresis induced by SGLT2 inhibitors, potentially causing plasma volume contraction and dehydration [78]. Additionally, SGLT2 inhibitors might enhance intestinal and urinary calcium absorption by inhibiting SGLT1, leading to carbohydrate malabsorption and decreased pH, which could contribute to increased calcium absorption [83].
However, the most frequent clinical challenge in the care of patients with CKD poses hyperkalemia, and the concern has been raised about the potential risk of hyperkalemia with the use of SGLT2 inhibitors in this population [84, 85]. Nevertheless, existing evidence suggests that SGLT2 inhibitors may influence potassium handling by accelerating the rate of sodium and water delivery to the distal nephron, a pivotal regulator of kidney-mediated potassium handling [84, 86]. This mechanism could result in an augmentation of urinary potassium excretion, potentially impacting serum potassium levels [84,85,86]. Studies indicate that the use of SGLT2 inhibitors is safe in both diabetic and non-diabetic CKD patients, without an increased risk of hyperkalemia [84, 85]. Illustratively, a recent meta-analysis encompassing 49,875 patients with T2DM and elevated cardiovascular risk or CKD revealed a significant reduction in the risk of severe hyperkalemia (≥ 6.0 mmol/L) with SGLT2 inhibitors, without a concurrent increase in the risk of hypokalemia [86].
SGLT2 inhibitors and anemia
SGLT2 inhibitors have also garnered attention for their potential impact on anemia in patients with CKD. Current evidence suggests that they may exhibit erythropoietic effects, contributing to elevated hemoglobin levels and potentially alleviating anemia in both diabetic and non-diabetic CKD patients [87, 88]. In a recent study, Osonoi et al. reported that dapagliflozin improves erythropoiesis and iron metabolism in patients with T2DM and anemia, suggesting that it has an erythropoietic effect and can help improve anemia in CKD [88]. A recent post hoc analysis of the DELIGHT trial revealed that patients with T2DM and albuminuria, with a mean eGFR level of 48.78 mL/min/1.73 m2, treated with dapagliflozin, experienced an increase in hemoglobin by 5.7 g/L (95% CI 4.0, 7.3) and a reduction in ferritin by 18.6% compared with the placebo group [89]. Another post hoc analysis of DAPA-CKD demonstrated similar results, indicating that dapagliflozin was associated with the prevention or correction of anemia in patients with CKD, both with and without T2DM. [87]. A comprehensive examination of both the mechanisms and clinical outcomes of the erythropoietic effects of SGLT2 inhibitors and prolyl hydroxylase inhibitors in CKD and anemia revealed that SGLT2 inhibitors lead to an increase in hemoglobin levels of approximately 0.6–0.7 g/dL [90]. Remarkably, the magnitude of this effect is comparable with that achieved with low-to-medium doses of prolyl hydroxylase inhibitors, and it remains evident even in patients with advanced CKD [90].
The proposed mechanism for the erythropoietic effects of SGLT2 inhibitors revolves around the idea that the heightened workload on renal tubule, caused by increased sodium delivery to the distal part after SGLT2 inhibition could induce renal medullary hypoxia [91]. This, in turn, is hypothesized to trigger the synthesis of erythropoietin, leading to an augmentation in hemoglobin levels [92]. However, despite this hypothesis, the magnetic resonance imaging study has not provided conclusive evidence supporting the changes in renal cortical or medullary tissue oxygenation in healthy volunteers undergoing SGLT2 inhibitor treatment [93]. This suggests that the observed elevation in erythropoietin levels is likely influenced by the modulation of hypoxia-inducible factors, rather than direct evidence of renal hypoxia [92].
Antioxidant and antiinflammatory feathers of SGLT2 inhibitors
Research findings have consistently demonstrated the antioxidant and antiinflammatory effects of SGLT2 inhibitors [94,95,96,97]. They function as indirect antioxidants by mitigating the oxidative stress induced by elevated glucose levels [95, 97]. Additionally, SGLT2 inhibitors exhibit the ability to reduce the generation of free radicals, inhibit pro-oxidants, such as NADPH oxidase 4 and thiobarbituric acid-reactive substances, and enhance the expression of antioxidants, such as superoxide dismutases and glutathione peroxidases [94, 98]. Furthermore, studies have demonstrated a reduction in a wide array of pro-inflammatory cytokines, including interleukin 6 (IL-6), IL-1, tumor necrosis factor alpha (TNF-α), and others following SGLT2 inhibitor administration [95,96,97, 99]. In a meta-analysis involving 6261 patients with T2DM, treatment with SGLT2 inhibitors significantly lowered levels of serum ferritin, C-reactive protein (CRP), and leptin compared to placebo or standard diabetes therapies [96]. While the precise mechanisms underlying the antioxidant and antiinflammatory effects of SGLT2 inhibitors remain unclear, studies have revealed the intricate molecular pathways involved in these processes [94, 97, 99]. It has been suggested that SGLT2 inhibitors activate adenosine monophosphate-activated protein kinase, contributing to antiinflammatory effects and the mitigation of oxidative stress [100]. Additionally, they influence the nod-like receptor (NLR) family pyrin domain containing 3 (NLRP3) inflammasome, contribute to the modulation of ROS, and impact cellular calcium homeostasis, leading to an antiinflammatory and antioxidative cellular environment [101, 102]. Furthermore, SGLT2 inhibitors have demonstrated antiinflammatory effects by inhibiting lipopolysaccharide (LPS)-induced Toll-like receptor 4 (TLR-4) overexpression and nuclear factor kappa B (NF-κB) activation in human endothelial cells and differentiated macrophages [103].
Gastrointestinal insights into SGLT2 inhibitor effects
One of the pivotal mechanisms through which SGLT2 inhibitors exert their effects in the gastrointestinal tract revolves around the impairment of small intestinal glucose absorption [104]. This process facilitates the redirection of glucose into the colon, culminating in an overall reduction in blood glucose levels, and is proposed to suppress protein fermentation and the generation of uremic toxins such as phenols and indoles [104,105,106]. Treatment with canagliflozin for 2 weeks significantly reduced the plasma levels of p-cresyl sulfate and indoxyl sulfate in mice with kidney failure (compared with the vehicle group a 75% and 26% reduction, respectively) [105]. The hypothesized role of SGLT2 inhibition in fostering renal clearance of protein-bound uremic toxins adds a layer of complexity to their gastrointestinal impact [104]. However, although the initial findings are promising, there remains an urgent need for further experimental and clinical research to comprehensively unravel the effects of SGLT2 inhibitors on the production, toxicity, and elimination of gut-derived uremic toxins.
Beyond glucose regulation, SGLT2 inhibitors exhibit a dual effect involving the heightened production of gastrointestinal hormones, including glucagon-like peptide 1 and peptide YY, which play a role in glucose regulation and satiety [107, 108]. Concurrently, SGLT2 inhibitors influence intestinal bacterial flora, fostering an environment characterized by a prevalence of balance-regulating bacteria and an increase in short-chain fatty acid (SCFA)-producing bacteria [105, 109, 110]. In a recent experimental study, Wu et al. demonstrated the protective effect of dapagliflozin on diabetic kidney disease in db/db mice, which appears to be associated with a dynamic improvement in gut microbiota over time [111]. This improvement may be related to the effects of dapagliflozin on the bile acid pool and its antioxidant activity [111]. In another diabetic nephropathy model, empagliflozin has also been shown to ameliorate T2DM-related diabetic nephropathy by altering the gut microbiota by reducing LPS-producing bacteria and increasing SCFA-producing bacteria in T2DM mice [112].
Cardiovascular protection by SGLT2 inhibitors
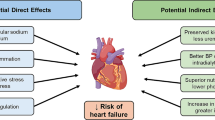
The multifaceted effects of SGLT2 inhibitors described above offer a unique opportunity to address both the local and systemic mechanisms involved in the pathogenesis of CVD. These effects result in a reduction of preload and afterload by optimizing body weight, inducing osmotic diuresis, regulating intravascular volume, influencing gut microbiota, enhancing endothelial function, and consequently mitigating vascular stiffness [1, 40, 92, 95, 112]. This collective action significantly contributes to improved cardiac function—an especially pivotal aspect for patients with CKD who are predisposed to cardiovascular complications [1, 2, 7]. Beyond hemodynamic improvements, SGLT2 inhibitors orchestrate a metabolic shift from free fatty acids to glucose oxidation [69, 71]. This metabolic redirection results in heightened cardiac ATP production, effectively preventing a decline in cardiac function [113].
Numerous RCTs focusing on primary cardiovascular outcomes, encompassing a substantial patient cohort with T2DM, provide compelling evidence of the significant cardiovascular benefits of SGLT2 inhibitors [114,115,116]. The outcomes reveal a noteworthy reduction in the risk of heart failure, frequency of hospitalizations due to heart failure, all-cause mortality, cardiovascular mortality and nonfatal myocardial infarction [113,114,115,116,117]. Although this robust evidence is less certain when extrapolated to non-diabetic CKD [118, 119], a recent meta-analysis conducted by Chen et al., incorporating findings from 13 RCTs, concludes that SGLT2 inhibitors confer cardiovascular protection in CKD patients. The analysis highlights a substantial reduction in the risk of cardiovascular disease, hospitalization for heart failure, and all-cause mortality, all achieved without an increased risk of serious adverse events and urinary tract infections [119]. Another meta-analysis involving a total of 27,823 patients with stage 3–4 CKD found that SGLT2 inhibitors significantly decreased the risk of primary cardiovascular outcomes, and these benefits were consistent across different levels of kidney function, even in stage 4 CKD [120]. The study highlighted the potential of SGLT2 inhibitors to reduce the risk of adverse cardiovascular events in patients with poor kidney function, which is particularly crucial given the increased risk of death and medical burden in this population [120]. As ongoing research continues to unravel the intricate mechanisms at play, the integration of SGLT2 inhibitors into the therapeutic landscape holds significant promise for enhancing cardiovascular outcomes in CKD.
PD-specific effects of SGLT2 inhibitors
Several recent studies have shed light on the specific effects of SGLT2 inhibitors in the realm of PD. Despite their somewhat limited scope, these studies indicate a potential paradigm shift that could reshape our understanding of the beneficial effects of SGLT2 inhibitors in addressing the distinctive challenges intrinsic to PD (Table 1).
One of the primary focuses of these studies has been on the role of SGLT2 inhibitors in managing glucose absorption from PD solutions and, consequently, their impact on peritoneal solute and water transport. The basis for these experiments stems from the finding that human PMCs express GLUT1, GLUT3, SGLT1, and SGLT2 receptors [121,122,123]. Therefore, SGLT2 inhibitors may have the potential to influence peritoneal solute and water transport. Supporting this hypothesis, Zhou et al. demonstrated a reduction in glucose uptake and an increase in ultrafiltration through the rat peritoneum, as well as an inhibition of glucose uptake by human PMCs mediated by empagliflozin [124]. Consistent results were found by Balzer [123] and Shentu [125], indicating that SGLT2 inhibition in cellular and animal models of PD was associated with reduced peritoneal fibrosis, decreased microvessel density, lower peritoneal glucose absorption, and improved peritoneal ultrafiltration. In contrast, Martus et al. observed no significant effect of empagliflozin on changes in sodium or water transport across the peritoneal membrane [126], and an increase in glucose concentration in the dialysate after 60 min of PD dwell was observed only with phlorizin [127].
In addition to influencing peritoneal solute and water transport, the mentioned studies have shown that intraperitoneal dapagliflozin alleviates peritoneal fibrosis in a mouse model subjected to chronic exposure to high-glucose dialysate [123]. The treatment with empagliflozin has led to a significant reduction in inflammatory cytokine levels, including TNF-α, IL-1β, IL-6, and proteins associated with transforming growth factor β (TGF-β)/Smad signaling, such as TGF-β1 and phosphorylated Smad (p-Smad3) in both a mouse PD model and human PMCs [125]. This effect may be attributed to the regulation of SGLT1 and SGLT2 in the peritoneal membrane, as well as the suppression of glucose uptake and the inhibition of hypoxia-inducible factor 1 α (HIF-1α) [128]. Canagliflozin significantly decreased HIF-1α abundance, inhibited TGF-β/p-Smad3 signaling, and decreased the expression of fibrotic proteins (fibronectin, COL1A2, and α-SMA) in human PMCs and rats exposed to chronic high glucose [128]. Balzer et al. have demonstrated that dapagliflozin reduced monocyte chemoattractant protein-1 (MCP-1) specifically in the presence of a high-glucose environment within mesothelial cells [123]. Furthermore, SGLT2 inhibition significantly decreased both MCP-1 and TNF-α in the presence of an inflammatory stimulus such as LPS, suggesting the potential anti-inflammatory effects of SGLT2 inhibitors [123].
Moreover, Shi et al. demonstrated the ability to mitigate inflammatory signaling pathways activated by ROS, which could be a key mechanism contributing to the reduction of peritoneal inflammation in patients undergoing PD [98]. High glucose can induce oxidative stress in PMCs and stimulate EMT, leading to peritoneal fibrosis [26]. Empagliflozin has been found to inhibit high glucose-induced EMT and reduce oxidative stress via the Nrf2/HO-1 signaling pathway in human and rat PMCs, potentially protecting the structural and functional integrity of the peritoneum [98]. The antifibrogenic, antiinflammatory, and antioxidant properties exhibited by SGLT2 inhibitors may have profound implications for maintaining the integrity of the peritoneal membrane and, consequently, enhancing the longevity of the PD technique.
Considering the limited number and experimental nature of the aforementioned studies, it is important to highlight two pioneering clinical studies. One involved 50 patients undergoing PD [129], while the other provided details on four clinical cases [130]. Notably, both studies utilized dapagliflozin off-label, and neither study reported any adverse effects. Alhwiesh et al. demonstrated that a dose of 10 mg/day dapagliflozin significantly reduced insulin requirements, HbA1c, body weight, systolic blood pressure, blood glucose, and serum uric acid levels [129]. Additionally, it increased urine and peritoneal ultrafiltration volumes in patients with insulin-dependent T2DM treated with automated PD [129]. However, the authors did not observe any changes in the peritoneal transport status in these patients. Lai et al. administered a daily dose of 5 mg dapagliflozin to four overhydrated patients undergoing PD for a duration of 14–28 days, continuing until a euvolemic status was achieved [130]. In line with a report by Alhwiesh et al., all the patients demonstrated a significant increase in peritoneal ultrafiltration rate [130].
Although these preliminary experimental and clinical results are promising, the inclusion of SGLT2 inhibitors in PD protocols must be approached with caution. Robust clinical trials are required to confirm the benefits and risks of SGLT2 inhibitors in patients undergoing PD.
Safety concerns in SGLT2 inhibitor utilization for PD
Despite the consistently favorable safety profile of SGLT2 inhibitors in CKD [4, 35, 131] and the absence of prior clinical trials in PD, it is prudent to acknowledge that the reported side effects in broader patient populations may have relevance to PD patients. Adverse events such as hypotension, hypoglycemia, infections, limb amputations, bone fractures, and euglycemic ketoacidosis (EKA) have been documented in the product information provided by regulatory agencies and reported in multiple systematic reviews and meta-analyses [132,133,134]. These reported adverse events vary among different SGLT2 inhibitors and depend on factors such as sex, the presence of diabetes, and concurrent glucose-lowering therapy [133, 134]. Although most of these events (EKA, amputations, and Fournier gangrene) are rare, they can lead to serious and dangerous complications [132,133,134]. In the context of PD, three of the mentioned challenges in SGLT2 inhibitor utilization may pose specific risks and should be detailed.
EKA is a recognized complication of therapy with SGLT2 inhibitors and can occur in patients with normal or only slightly elevated plasma glucose concentrations, presenting a particular problem for individuals with preexisting CKD [135, 136]. In patients undergoing PD, continuous exposure of the peritoneal membrane to glucose-based PD solutions, along with the glycosuric effect of SGLT2 inhibitors, may create an environment conducive to metabolic imbalances, contributing to the risk of EKA [137]. Additionally, the higher proportion of diabetic patients undergoing PD adds another layer of complexity. Diabetic patients already face an increased risk of metabolic disturbances, and the addition of SGLT2 inhibitors may further accentuate this risk [138]. The theoretical synergistic effect of altered glucose metabolism in diabetes, coupled with the unique challenges of PD, may elevate susceptibility to EKA. However, on the flip side, as the glycosuric impact of SGLT2 inhibitors diminishes beyond stage 3 CKD, concerns regarding EKA are unlikely to be applicable to the PD population. Nevertheless, due to the absence of prior clinical trials investigating SGLT2 inhibitors in patients undergoing PD, it remains unclear whether their use would increase the risk of EKA, for example, in patients with preserved RKF.
The second concern involves the potential risk of PD-associated infections. SGLT2 inhibitors are recognized for elevating the likelihood of genital and urinary tract infections [132, 133], with genital mycotic infections and urinary tract infections being the most frequently reported adverse effects [139, 140]. The incidence of genital yeast infections associated with SGLT2 inhibitors is reported to be approximately three times higher compared with other treatments [141]. The adjusted incidence is reported as 135.5 per 1000 person-years for SGLT2 inhibitors, contrasting with 48.5 per 1000 person-years for alternative treatments, resulting in an excess risk of 87.6 per 1000 person-years [141]. Importantly, the risk of genital infections remains consistent across various SGLT2 inhibitors and persists throughout the duration of therapy [141]. In addition, female sex, diabetes duration of more than 10 years, and a previous genital infection are risk factors that contribute to an increased risk of infection [140, 142]. Considering the above, several factors might contribute to an elevated risk of PD-related infections in patients using SGLT2 inhibitors. In PD, where the peritoneal membrane is consistently exposed to glucose-based solutions, the utilization of SGLT2 inhibitors may lead to an increase in glucose levels in the peritoneal cavity, particularly within the initial 60 min [127]. This heightened concentration of glucose could provide a favorable environment for the growth of microorganisms, potentially amplifying the risk of peritoneal infections. Furthermore, PD patients encounter a direct entry point for microorganisms into the peritoneal cavity through the PD catheter. The presence of a higher glucose peritoneal environment and the catheter may potentially augment vulnerability to infections. Despite the minimal glycosuric effect of SGLT2 inhibitors in patients with kidney failure, PD patients are already confronted with an increased risk of infection. The susceptibility to genital and urinary tract infections may further escalate the overall infection risk, including the potentially life-threatening complication of fungal PD-related peritonitis. It is essential to emphasize that these potential risks are theoretical, and there is a lack of empirical evidence supporting an augmented risk of PD-associated infections specifically related to SGLT2 inhibitors.
Another significant issue is the possible impact of SGLT2 inhibitors on CKD-associated mineral and bone metabolism (MBD) in patients undergoing PD. Bone fractures are a recognized adverse effect of SGLT2, especially in patients aged over 65 with both diabetes and CKD [133, 134, 139, 143]. Several hypothesized mechanisms suggest that SGLT2 inhibitors can directly influence calcium–phosphate balance and indirectly increase bone turnover by promoting weight loss [144]. The elevation in serum phosphate levels, a known class effect of SGLT2 inhibitors, is likely facilitated by increased phosphate transport in the proximal tubule, leading to enhanced phosphate reabsorption and urinary calcium excretion due to sodium loss [144, 145]. This increase in phosphate levels is thought to stimulate FGF23, resulting in phosphaturia and the inhibition of 1.25-dihydroxyvitamin D to maintain the phosphate balance. In a randomized crossover study involving healthy volunteers using canagliflozin, the administration of 300 mg/day triggered the FGF23/1,25-dihydroxyvitamin D/PTH axis, leading to increased serum phosphate levels, plasma FGF23, and PTH, coupled with a decrease in 1,25-dihydroxyvitamin D [144]. Although there is controversy surrounding the evidence that SGLT2 inhibitors cause bone fractures [145, 146], the complex interplay of these factors may have implications for mineral and bone health in patients undergoing PD who already have MBD-CKD.
Lastly, some other potentially less serious problems would require continuous monitoring, such as blood pressure, fluid and electrolyte balance, blood glucose control, and drug interactions. SGLT2 inhibitors induce osmotic diuresis, which leads to increased urinary glucose excretion and fluid loss [39]. This diuretic effect may affect fluid status, potentially causing dehydration or affecting ultrafiltration requirements during PD. In addition, the prescription of SGLT2 inhibitors in PD patients may require adjustment of antihypertensive and/or diabetic treatment due to their antihypertensive effect and potential impact on peritoneal membrane function and fluid balance [2, 147]. As PD patients often take multiple medications, it is important to consider potential interactions between SGLT2 inhibitors and other prescribed medications [148]. Regular and careful monitoring is essential for optimal management of these various factors in PD patients treated with SGLT2 inhibitors.
Conclusions and future perspectives
This review presents a compelling case for the potential integration of SGLT2 inhibitors into the management of patients undergoing PD, extending beyond their established benefits in patients with T2DM, non-diabetic CKD, and cardiovascular health. The multifaceted effects of SGLT2 inhibitors, including metabolic modulation, hypotensive, diuretic, antianemic, antioxidant, and antiinflammatory properties, along with their protective impact on the peritoneal membrane and, consequently, solute and water transport, suggest a promising avenue for addressing challenges unique to PD. The observed renal and cardiovascular benefits in patients with T2DM and CKD underscore the potential of SGLT2 inhibitors to preserve RKF and mitigate cardiovascular complications in this patient population. Moreover, two preliminary clinical studies reported positive outcomes with the use of SGLT2 inhibitors, showing increased peritoneal ultrafiltration and other benefits similar to the general CKD population.
However, caution is warranted given the need for rigorous investigation into dosing, long-term safety, and patient-specific factors through comprehensive clinical trials. Currently, five registered clinical trials focused on PD are underway, aiming to address peritoneal glucose uptake, mechanisms and safety of SGLT2 inhibition, and the impact on ultrafiltration among PD patients: (1) “Reduction of Peritoneal Glucose Uptake With the Use of SGLT2 in Humans Undergoing Peritoneal Dialysis Treatment” (NCT05250752), (2) “Mechanisms and Safety of SGLT2 Inhibition in Peritoneal Dialysis” (NCT05715814), (3) “The Effect of Dapagliflozin on Ultrafiltration Among Peritoneal Dialysis Patients” (NCT04923295), (4) “Effect of Empagliflozin on Peritoneal and Kidney Function in End Stage Renal Disease (EMPA-PD)” (NCT05671991), and (5) “The effects of empagliflozin on ultrafiltration in patients with peritoneal dialysis” (jRCTs051230081). These trials are anticipated to provide comprehensive insights into the integration of SGLT2 inhibitors into PD protocols.
To address the potential risks associated with SGLT2 inhibitors in PD, such as EKA, infections, and effects on MBD, focused attention is essential. Future research should elucidate the underlying mechanisms and guide risk mitigation strategies tailored to patients undergoing PD, acknowledging the heterogeneity of this population. Individualized approaches to prescribing SGLT2 inhibitors, considering factors such as age, diabetes status, and PD-associated risks, should be explored. Continuous monitoring of vital factors, including blood pressure, fluid and electrolyte balance, blood glucose control, and potential drug interactions, is paramount. The development of comprehensive management protocols will ensure the optimal and safe utilization of SGLT2 inhibitors in PD. Longitudinal studies assessing the extended safety profile of SGLT2 inhibitors in PD, particularly their impact on peritoneal membrane integrity and patient outcomes, will be instrumental in establishing the long-term viability of these agents.
Together, while the initial evidence is promising, the integration of SGLT2 inhibitors into PD necessitates careful consideration, with future research aimed at refining protocols, addressing risks, and personalizing treatment strategies to ensure optimal patient outcomes.
Availability of data and materials
Not applicable.
Abbreviations
- APD:
-
Automated peritoneal dialysis
- CKD:
-
Chronic kidney disease
- EKA:
-
Euglycemic ketoacidosis
- EMT:
-
Epithelial to mesenchymal transformation
- eGFR:
-
Estimated glomerular filtration rate
- FGF23:
-
Fibroblast growth factor 23
- GLUT1/3:
-
Glucose transporter 1/3
- HbA1c:
-
Hemoglobin A1c
- HDL:
-
High-density lipoprotein
- HIF-1α:
-
Hypoxia-inducible factor 1-alpha
- IL:
-
Interleukin
- LPS:
-
Lipopolysaccharide
- LDL:
-
Low-density lipoprotein
- MCP-1:
-
Monocyte chemoattractant protein-1
- MBD:
-
Mineral and bone metabolism
- NLRP3:
-
Nod-like receptor pyrin domain containing 3
- PD:
-
Peritoneal dialysis
- PDE:
-
Peritoneal dialysis effluent
- PMCs:
-
Peritoneal mesothelial cells
- PTH:
-
Parathyroid hormone
- p-Smad3:
-
Phosphorylated Smad3
- RAS:
-
Renin-angiotensin system
- RCTs:
-
Randomized controlled trials
- ROS:
-
Reactive oxygen species
- SCFA:
-
Short-chain fatty acid
- SGLT2:
-
Sodium–glucose cotransporter 2
- T2DM:
-
Type 2 diabetes mellitus
- TLR-4:
-
Toll-like receptor 4
- TGF-β:
-
Transforming growth factor β
- TNF-α:
-
Tumor necrosis factor alpha
References
Kansara A, Mubeen F, Shakil J. SGLT2 inhibitors in patients with chronic kidney disease and heart disease: a literature review. Methodist Debakey Cardiovasc J. 2022;18(4):62–72. https://doi.org/10.14797/mdcvj.1120.
Yau K, Dharia A, Alrowiyti I, Cherney DZI. Prescribing SGLT2 inhibitors in patients with CKD: expanding indications and practical considerations. Kidney Int Rep. 2022;7(11):2546–7. https://doi.org/10.1016/j.ekir.2022.08.016.
Ellison DH. SGLT2 inhibitors, hemodynamics, and kidney protection. Am J Physiol Renal Physiol. 2021;321(1):F47–9. https://doi.org/10.1152/ajprenal.00092.2021.
Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, et al. DAPA-CKD Trial Committees and Investigators. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383(15):1436–46. https://doi.org/10.1056/NEJMoa2024816.
Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, McGuire DK, et al. SCORED investigators. Sotagliflozin in patients with diabetes and chronic kidney disease. N Engl J Med. 2021;384(2):129–39. https://doi.org/10.1056/NEJMoa2030186.
Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, et al. CREDENCE Trial Investigators. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295–2306. https://doi.org/10.1056/NEJMoa1811744.
Giorgino F, Vora J, Fenici P, Solini A. Renoprotection with SGLT2 inhibitors in type 2 diabetes over a spectrum of cardiovascular and renal risk. Cardiovasc Diabetol. 2020;19(1):196. https://doi.org/10.1186/s12933-020-01163-9.
Almaimani M, Sridhar VS, Cherney DZI. Sodium-glucose cotransporter 2 inhibition in non-diabetic kidney disease. Curr Opin Nephrol Hypertens. 2021;30(5):474–81. https://doi.org/10.1097/MNH.0000000000000724.
The EMPA-KIDNEY Collaborative Group; Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, Emberson JR, et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med. 2023;388(2):117–27. https://doi.org/10.1056/NEJMoa2204233.
Waijer SW, Vart P, Cherney DZI, Chertow GM, Jongs N, Langkilde AM, et al. Effect of dapagliflozin on kidney and cardiovascular outcomes by baseline KDIGO risk categories: a post hoc analysis of the DAPA-CKD trial. Diabetologia. 2022;65(7):1085–97. https://doi.org/10.1007/s00125-022-05694-6.
Bakris G, Oshima M, Mahaffey KW, Agarwal R, Cannon CP, Capuano G, et al. Effects of canagliflozin in patients with baseline eGFR <30 ml/min per 1.73 m2: subgroup analysis of the randomized CREDENCE Trial. Clin J Am Soc Nephrol. 2020;15(12):1705–14. https://doi.org/10.2215/CJN.10140620.
De La Flor JC, Villa D, Cruzado L, Apaza J, Valga F, Zamora R, et al. Efficacy and safety of the use of SGLT2 inhibitors in patients on incremental hemodialysis: maximizing residual renal function, is there a role for SGLT2 inhibitors? Biomedicines. 2023;11(7):1908. https://doi.org/10.3390/biomedicines11071908.
Bello AK, Okpechi IG, Osman MA, Cho Y, Cullis B, Htay H, et al. Epidemiology of peritoneal dialysis outcomes. Nat Rev Nephrol. 2022;18(12):779–93. https://doi.org/10.1038/s41581-022-00623-7.
François K, Bargman JM. Evaluating the benefits of home-based peritoneal dialysis. Int J Nephrol Renovasc Dis. 2014;7:447–55. https://doi.org/10.2147/IJNRD.S50527.
Ethier I, Cho Y, Hawley C, Pascoe EM, Viecelli AK, Campbell SB, et al. Rate of decline in residual kidney function pre and post peritoneal dialysis initiation: a post hoc analysis of the IDEAL study. PLoS ONE. 2020;15(11):e0242254. https://doi.org/10.1371/journal.pone.0242254.
Li T, Wilcox CS, Lipkowitz MS, Gordon-Cappitelli J, Dragoi S. Rationale and strategies for preserving residual kidney function in dialysis patients. Am J Nephrol. 2019;50(6):411–21. https://doi.org/10.1159/000503805.
Alrowiyti IM, Bargman J. A review of residual kidney function in peritoneal dialysis patients. Indian J Nephrol. 2023;33(4):239–46. https://doi.org/10.4103/ijn.ijn_242_23.
John MM, Gupta A, Sharma RK, Kaul A. Impact of residual renal function on clinical outcome and quality of life in patients on peritoneal dialysis. Saudi J Kidney Dis Transpl. 2017;28(1):30–5. https://doi.org/10.4103/1319-2442.198109.
Liu X, Dai C. Advances in understanding and management of residual renal function in patients with chronic kidney disease. Kidney Dis (Basel). 2017;2(4):187–96. https://doi.org/10.1159/000449029.
Shu Y, Liu J, Zeng X, Hong HG, Li Y, Zhong H, et al. The effect of overhydration on mortality and technique failure among peritoneal dialysis patients: a systematic review and meta-analysis. Blood Purif. 2018;46(4):350–8. https://doi.org/10.1159/000492148.
Jotterand Drepper V, Kihm LP, Kälble F, Diekmann C, Seckinger J, Sommerer C, et al. Overhydration is a strong predictor of mortality in peritoneal dialysis patients - independently of cardiac failure. PLoS ONE. 2016;11(7):e0158741. https://doi.org/10.1371/journal.pone.0158741.
Kuo KL, Liu JS, Lin MH, Hsu CC, Tarng DC, Taiwan Society of Nephrology Renal Registry Data System Research Group. Association of anemia and iron parameters with mortality among prevalent peritoneal dialysis patients in Taiwan: the AIM-PD study. Sci Rep. 2022;12(1):1269. https://doi.org/10.1038/s41598-022-05200-3.
Stepanova N, Burdeyna O. Association between Dyslipidemia and peritoneal dialysis technique survival. Open Access Maced J Med Sci. 2019;7(15):2467–73. https://doi.org/10.3889/oamjms.2019.664.
Stepanova N, Driianska V, Savchenko S. Dyslipidemia and intraperitoneal inflammation axis in peritoneal dialysis patients: a cross-sectional pilot study. Kidney Dis (Basel). 2020;6(1):35–42. https://doi.org/10.1159/000503632.
Ye H, Zhou Q, Fan L, Guo Q, Mao H, Huang F, et al. The impact of peritoneal dialysis-related peritonitis on mortality in peritoneal dialysis patients. BMC Nephrol. 2017;18(1):186. https://doi.org/10.1186/s12882-017-0588-4.
Krediet RT. Aging of the peritoneal dialysis membrane. Front Physiol. 2022;13:885802. https://doi.org/10.3389/fphys.2022.885802.
Wu HY, Hung KY, Huang TM, Hu FC, Peng YS, Huang JW, et al. Safety issues of long-term glucose load in patients on peritoneal dialysis–a 7-year cohort study. PLoS ONE. 2012;7(1):e30337. https://doi.org/10.1371/journal.pone.0030337.
Roumeliotis S, Dounousi E, Salmas M, Eleftheriadis T, Liakopoulos V. Unfavorable effects of peritoneal dialysis solutions on the peritoneal membrane: the role of oxidative stress. Biomolecules. 2020;10(5):768. https://doi.org/10.3390/biom10050768.
Stepanova N. The gut-peritoneum axis in peritoneal dialysis and peritoneal fibrosis. Kidney Med. 2023;5(6):100645. https://doi.org/10.1016/j.xkme.2023.100645.
Li J, Liu Y, Liu J. A review of research progress on mechanisms of peritoneal fibrosis related to peritoneal dialysis. Front Physiol. 2023;14:1220450. https://doi.org/10.3389/fphys.2023.1220450.
Jeon Y, Kim HD, Hong YA, Kim HW, Yang CW, Chang YK, et al. Clinical outcomes of infection-related hospitalization in incident peritoneal dialysis patients. Kidney Res Clin Pract. 2020;39(4):460–8. https://doi.org/10.23876/j.krcp.20.069.
Chen JHC, Johnson DW, Hawley C, Boudville N, Lim WH. Association between causes of peritoneal dialysis technique failure and all-cause mortality. Sci Rep. 2018;8(1):3980. https://doi.org/10.1038/s41598-018-22335-4.
Szeto CC, Kwan BC, Chow KM, Chung S, Yu V, Cheng PM, et al. Predictors of residual renal function decline in patients undergoing continuous ambulatory peritoneal dialysis. Perit Dial Int. 2015;35(2):180–8. https://doi.org/10.3747/pdi.2013.00075.
van Diepen AT, van Esch S, Struijk DG, Krediet RT. The first peritonitis episode alters the natural course of peritoneal membrane characteristics in peritoneal dialysis patients. Perit Dial Int. 2015;35(3):324–32. https://doi.org/10.3747/pdi.2014.00277.
Borkum M, Jamal A, Suneet Singh R, Levin A. The rationale for the need to study sodium-glucose co-transport 2 inhibitor usage in peritoneal dialysis patients. Perit Dial Int. 2023;43(2):139–44. https://doi.org/10.1177/08968608221096556.
Hsia DS, Grove O, Cefalu WT. An update on sodium-glucose co-transporter-2 inhibitors for the treatment of diabetes mellitus. Curr Opin Endocrinol Diabetes Obes. 2017;24(1):73–9. https://doi.org/10.1097/MED.0000000000000311.
Fonseca-Correa JI, Correa-Rotter R. Sodium-glucose cotransporter 2 inhibitors mechanisms of action: a review. Front Med (Lausanne). 2021;8: 777861. https://doi.org/10.3389/fmed.2021.777861.
Chao EC. SGLT-2 inhibitors: a new mechanism for glycemic control. Clin Diabetes. 2014;32(1):4–11. https://doi.org/10.2337/diaclin.32.1.4.
Tang J, Ye L, Yan Q, Zhang X, Wang L. Effects of sodium-glucose cotransporter 2 inhibitors on water and sodium metabolism. Front Pharmacol. 2022;13:800490. https://doi.org/10.3389/fphar.2022.800490.
Hou YC, Zheng CM, Yen TH, Lu KC. Molecular mechanisms of SGLT2 inhibitor on cardiorenal protection. Int J Mol Sci. 2020;21(21):7833. https://doi.org/10.3390/ijms21217833.
Vallon V, Platt KA, Cunard R, Schroth J, Whaley J, Thomson SC, et al. SGLT2 mediates glucose reabsorption in the early proximal tubule. J Am Soc Nephrol. 2011;22(1):104–12. https://doi.org/10.1681/ASN.2010030246.
Ghezzi C, Loo DDF, Wright EM. Physiology of renal glucose handling via SGLT1, SGLT2 and GLUT2. Diabetologia. 2018;61(10):2087–97. https://doi.org/10.1007/s00125-018-4656-5.
Bashier A, Khalifa AA, Rashid F, Abdelgadir EI, Al Qaysi AA, Ali R, et al. Efficacy and safety of SGLT2 inhibitors in reducing glycated hemoglobin and weight in emirati patients with type 2 diabetes. J Clin Med Res. 2017;9(6):499–507. https://doi.org/10.14740/jocmr2976w.
Abdul-Ghani MA, Norton L, DeFronzo RA. Renal sodium-glucose cotransporter inhibition in the management of type 2 diabetes mellitus. Am J Physiol Renal Physiol. 2015;309(11):F889-900. https://doi.org/10.1152/ajprenal.00267.2015.
Cherney DZI, Cooper ME, Tikkanen I, Pfarr E, Johansen OE, Woerle HJ, et al. Pooled analysis of Phase III trials indicate contrasting influences of renal function on blood pressure, body weight, and HbA1c reductions with empagliflozin. Kidney Int. 2018;93(1):231–44. https://doi.org/10.1016/j.kint.2017.06.017.
Thomas MC, Cherney DZI. The actions of SGLT2 inhibitors on metabolism, renal function and blood pressure. Diabetologia. 2018;61(10):2098–107. https://doi.org/10.1007/s00125-018-4669-0.
Cherney DZ, Perkins BA, Soleymanlou N, Maione M, Lai V, Lee A, et al. Renal hemodynamic effect of sodium-glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation. 2014;129(5):587–97. https://doi.org/10.1161/CIRCULATIONAHA.113.005081.
Peti-Peterdi J, Harris RC. Macula densa sensing and signaling mechanisms of renin release. J Am Soc Nephrol. 2010;21(7):1093–6. https://doi.org/10.1681/ASN.2009070759.
Wu J. Paradigm shift in hyperglycemic glomerular hyperfiltration: blunted tubuloglomerular feedback or preglomerular vasodilation? Hypertension. 2023;80(8):1611–3. https://doi.org/10.1161/HYPERTENSIONAHA.123.21505.
Thomson SC, Vallon V. Effects of SGLT2 inhibitor and dietary NaCl on glomerular hemodynamics assessed by micropuncture in diabetic rats. Am J Physiol Renal Physiol. 2021;320(5):F761–71. https://doi.org/10.1152/ajprenal.00552.2020.
Skrtić M, Yang GK, Perkins BA, Soleymanlou N, Lytvyn Y, von Eynatten M, et al. Characterisation of glomerular haemodynamic responses to SGLT2 inhibition in patients with type 1 diabetes and renal hyperfiltration. Diabetologia. 2014;57(12):2599–602. https://doi.org/10.1007/s00125-014-3396-4.
van Bommel EJM, Muskiet MHA, van Baar MJB, Tonneijck L, Smits MM, Emanuel AL, et al. The renal hemodynamic effects of the SGLT2 inhibitor dapagliflozin are caused by post-glomerular vasodilatation rather than pre-glomerular vasoconstriction in metformin-treated patients with type 2 diabetes in the randomized, double-blind RED trial. Kidney Int. 2020;97(1):202–12. https://doi.org/10.1016/j.kint.2019.09.013.
Ott C, Jung S, Korn M, Kannenkeril D, Bosch A, Kolwelter J, et al. Renal hemodynamic effects differ between antidiabetic combination strategies: randomized controlled clinical trial comparing empagliflozin/linagliptin with metformin/insulin glargine. Cardiovasc Diabetol. 2021;20(1):178. https://doi.org/10.1186/s12933-021-01358-8.
Cherney DZI, Dekkers CCJ, Barbour SJ, Cattran D, Abdul Gafor AH, et al; DIAMOND investigators. Effects of the SGLT2 inhibitor dapagliflozin on proteinuria in non-diabetic patients with chronic kidney disease (DIAMOND): a randomised, double-blind, crossover trial. Lancet Diabetes Endocrinol. 2020;8(7):582–93. https://doi.org/10.1016/S2213-8587(20)30162-5.
Zaccardi F, Webb DR, Htike ZZ, Youssef D, Khunti K, Davies MJ. Efficacy and safety of sodium-glucose co-transporter-2 inhibitors in type 2 diabetes mellitus: systematic review and network meta-analysis. Diabetes Obes Metab. 2016;18(8):783–94. https://doi.org/10.1111/dom.12670.
Lambers Heerspink HJ, de Zeeuw D, Wie L, Leslie B, List J. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes Metab. 2013;15(9):853–62. https://doi.org/10.1111/dom.12127.
Cherney DZ, Perkins BA, Soleymanlou N, Har R, Fagan N, Johansen OE, et al. The effect of empagliflozin on arterial stiffness and heart rate variability in subjects with uncomplicated type 1 diabetes mellitus. Cardiovasc Diabetol. 2014;13:28. https://doi.org/10.1186/1475-2840-13-28.
Sugiyama S, Jinnouchi H, Kurinami N, Hieshima K, Yoshida A, Jinnouchi K, et al. The SGLT2 inhibitor dapagliflozin significantly improves the peripheral microvascular endothelial function in patients with uncontrolled type 2 diabetes mellitus. Intern Med. 2018;57(15):2147–56. https://doi.org/10.2169/internalmedicine.0701-17.
Mazidi M, Rezaie P, Gao HK, Kengne AP. Effect of sodium-glucose cotransport-2 inhibitors on blood pressure in people with type 2 diabetes mellitus: a systematic review and meta-analysis of 43 randomized control trials with 22,528 patients. J Am Heart Assoc. 2017;6(6):e004007. https://doi.org/10.1161/JAHA.116.004007.
Bailey CJ, Day C, Bellary S. Renal protection with SGLT2 inhibitors: effects in acute and chronic kidney disease. Curr Diab Rep. 2022;22(1):39–52. https://doi.org/10.1007/s11892-021-01442-z.
Coresh J, Heerspink HJL, Sang Y, Matsushita K, Arnlov J, Astor BC, et al. Chronic kidney disease prognosis consortium and chronic kidney disease epidemiology collaboration. Change in albuminuria and subsequent risk of end-stage kidney disease: an individual participant-level consortium meta-analysis of observational studies. Lancet Diabetes Endocrinol. 2019;7(2):115–27. https://doi.org/10.1016/S2213-8587(18)30313-9.
Kelly MS, Lewis J, Huntsberry AM, Dea L, Portillo I. Efficacy and renal outcomes of SGLT2 inhibitors in patients with type 2 diabetes and chronic kidney disease. Postgrad Med. 2019;131(1):31–42. https://doi.org/10.1080/00325481.2019.1549459.
Sato S, Takayanagi K, Shimizu T, Kanozawa K, Iwashita T, Hasegawa H. Correlation between albuminuria and interstitial injury marker reductions associated with SGLT2 inhibitor treatment in diabetic patients with renal dysfunction. Eur J Med Res. 2022;27(1):140. https://doi.org/10.1186/s40001-022-00737-5.
Fernández-Fernandez B, Sarafidis P, Soler MJ, Ortiz A. EMPA-KIDNEY: expanding the range of kidney protection by SGLT2 inhibitors. Clin Kidney J. 2023;16(8):1187–98. https://doi.org/10.1093/ckj/sfad082.
Xu L, Ota T. Emerging roles of SGLT2 inhibitors in obesity and insulin resistance: focus on fat browning and macrophage polarization. Adipocyte. 2018;7(2):121–8. https://doi.org/10.1080/21623945.2017.1413516.
Waseda N, Satoh H, Yoshida C, Ikeda F, Kanazawa A, Watada H. Effects of SGLT2 inhibitors on insulin secretion and insulin resistance—results from a cross-sectional study. Diabetes 2018;67(Suppl 1):1187-P. https://doi.org/10.2337/db18-1187-P.
Zheng H, Liu M, Li S, Shi Q, Zhang S, Zhou Y, Su N. Sodium-glucose co-transporter-2 inhibitors in non-diabetic adults with overweight or obesity: a systematic review and meta-analysis. Front Endocrinol (Lausanne). 2021;12:706914. https://doi.org/10.3389/fendo.2021.706914.
Pereira MJ, Eriksson JW. Emerging role of SGLT-2 inhibitors for the treatment of obesity. Drugs. 2019;79(3):219–30. https://doi.org/10.1007/s40265-019-1057-0.
Szekeres Z, Toth K, Szabados E. The effects of SGLT2 inhibitors on lipid metabolism. Metabolites. 2021;11(2):87. https://doi.org/10.3390/metabo11020087.
Bechmann LE, Emanuelsson F, Nordestgaard BG, Benn M. SGLT2-inhibition increases total, LDL, and HDL cholesterol and lowers triglycerides: meta-analyses of 60 randomized trials, overall and by dose, ethnicity, and drug type. Atherosclerosis. 2023:117236. https://doi.org/10.1016/j.atherosclerosis.2023.117236.
Sánchez-García A, Simental-Mendía M, Millán-Alanís JM, Simental-Mendía LE. Effect of sodium-glucose co-transporter 2 inhibitors on lipid profile: a systematic review and meta-analysis of 48 randomized controlled trials. Pharmacol Res. 2020;160:105068. https://doi.org/10.1016/j.phrs.2020.105068.
Weir MR, Januszewicz A, Gilbert RE, Vijapurkar U, Kline I, Fung A, et al. Effect of canagliflozin on blood pressure and adverse events related to osmotic diuresis and reduced intravascular volume in patients with type 2 diabetes mellitus. J Clin Hypertens (Greenwich). 2014;16(12):875–82. https://doi.org/10.1111/jch.12425.
Yasui A, Lee G, Hirase T, Kaneko T, Kaspers S, von Eynatten M, et al. Empagliflozin induces transient diuresis without changing long-term overall fluid balance in Japanese patients with type 2 diabetes. Diabetes Ther. 2018;9(2):863–71. https://doi.org/10.1007/s13300-018-0385-5.
Sha S, Polidori D, Heise T, Natarajan J, Farrell K, Wang SS, et al. Effect of the sodium glucose co-transporter 2 inhibitor canagliflozin on plasma volume in patients with type 2 diabetes mellitus. Diabetes Obes Metab. 2014;16(11):1087–95. https://doi.org/10.1111/dom.12322.
Mordi NA, Mordi IR, Singh JS, McCrimmon RJ, Struthers AD, Lang CC. Renal and cardiovascular effects of SGLT2 inhibition in combination with loop diuretics in patients with type 2 diabetes and chronic heart failure: the RECEDE-CHF trial. Circulation. 2020;142(18):1713–24. https://doi.org/10.1161/CIRCULATIONAHA.120.048739.
Delanaye P, Scheen AJ. The diuretic effects of SGLT2 inhibitors: a comprehensive review of their specificities and their role in renal protection. Diabetes Metab. 2021;47(6):101285. https://doi.org/10.1016/j.diabet.2021.101285.
Ansary TM, Nakano D, Nishiyama A. Diuretic effects of sodium glucose cotransporter 2 inhibitors and their influence on the renin-angiotensin system. Int J Mol Sci. 2019;20(3):629. https://doi.org/10.3390/ijms20030629.
El Masri D, Jamil Y, Eid FJ. Sodium-glucose co-transporter protein 2 inhibitors induced hypercalcemia: a case series and literature review. AACE Clin Case Rep. 2021;8(1):30–3. https://doi.org/10.1016/j.aace.2021.07.002.
Cianciolo G, De Pascalis A, Capelli I, Gasperoni L, Di Lullo L, Bellasi A, et al. Mineral and electrolyte disorders with SGLT2i therapy. JBMR Plus. 2019;3(11):e10242. https://doi.org/10.1002/jbm4.10242.
Rau M, Thiele K, Hartmann NK, Möllmann J, Wied S, Hohl M, et al. Effects of empagliflozin on markers of calcium and phosphate homeostasis in patients with type 2 diabetes—data from a randomized, placebo-controlled study. Bone Rep. 2022;16:101175. https://doi.org/10.1016/j.bonr.2022.101175.
Zhang J, Huan Y, Leibensperger M, Seo B, Song Y. Comparative effects of sodium-glucose cotransporter 2 inhibitors on serum electrolyte levels in patients with type 2 diabetes: a pairwise and network meta-analysis of randomized controlled trials. Kidney360. 2022;3(3):477–87. https://doi.org/10.34067/KID.0006672021.
Saha A, Omer A, Trivedi N. Improvement in serum magnesium levels with sodium-glucose cotransporter 2 inhibitors. JCEM Case Rep. 2022;1(1):luac018. https://doi.org/10.1210/jcemcr/luac018.
Kaur A, Winters SJ. Severe hypercalcemia and hypernatremia in a patient treated with canagliflozin. Endocrinol Diabetes Metab Case Rep. 2015;2015:150042. https://doi.org/10.1530/EDM-15-0042.
Albakr RB, Sridhar VS, Cherney DZI. Novel therapies in diabetic kidney disease and risk of hyperkalemia: a review of the evidence from clinical trials. Am J Kidney Dis. 2023;82(6):737–42. https://doi.org/10.1053/j.ajkd.2023.04.015.
Gabai P, Fouque D. SGLT2 inhibitors: new kids on the block to control hyperkalemia. Nephrol Dial Transpl. 2023;38(6):1345–8. https://doi.org/10.1093/ndt/gfad026.
Neuen BL, Oshima M, Agarwal R, Arnott C, Cherney DZ, Edwards R, et al. Sodium-glucose cotransporter 2 inhibitors and risk of hyperkalemia in people with type 2 diabetes: a meta-analysis of individual participant data from randomized. Controlled Trials Circulation. 2022;145(19):1460–70. https://doi.org/10.1161/CIRCULATIONAHA.121.057736.
Koshino A, Schechter M, Chertow GM, Vart P, Jongs N, Toto RD, et al. Dapagliflozin and anemia in patients with chronic kidney disease. NEJM Evid 2023;2(6). https://doi.org/10.1056/EVIDoa2300049.
Osonoi T, Shirabe S, Saito M, Hosoya M, Watahiki N, Douguchi S, et al. Dapagliflozin improves erythropoiesis and iron metabolism in type 2 diabetic patients with renal anemia. Diabetes Metab Syndr Obes. 2023;16:1799–808. https://doi.org/10.2147/DMSO.S411504.
Koshino A, Neuen BL, Jongs N, Pollock C, Greasley PJ, Andersson EM, et al. Effects of dapagliflozin and dapagliflozin-saxagliptin on erythropoiesis, iron and inflammation markers in patients with type 2 diabetes and chronic kidney disease: data from the DELIGHT trial. Cardiovasc Diabetol. 2023;22(1):330. https://doi.org/10.1186/s12933-023-02027-8.
Packer M. Mechanistic and clinical comparison of the erythropoietic effects of SGLT2 inhibitors and prolyl hydroxylase inhibitors in patients with chronic kidney disease and renal anemia. Am J Nephrol. 2023. https://doi.org/10.1159/000531084. Epub ahead of print.
Hare GMT, Zhang Y, Chin K, Thai K, Jacobs E, Cazorla-Bak MP, et al. Impact of sodium glucose linked cotransporter-2 inhibition on renal microvascular oxygen tension in a rodent model of diabetes mellitus. Physiol Rep. 2021;9(12):e14890. https://doi.org/10.14814/phy2.14890.
Packer M. Critical reanalysis of the mechanisms underlying the cardiorenal benefits of SGLT2 inhibitors and reaffirmation of the nutrient deprivation signaling/autophagy hypothesis. Circulation. 2022;146(18):1383–405. https://doi.org/10.1161/CIRCULATIONAHA.122.061732.
Zanchi A, Burnier M, Muller ME, Ghajarzadeh-Wurzner A, Maillard M, Loncle N, et al. Acute and chronic effects of SGLT2 inhibitor empagliflozin on renal oxygenation and blood pressure control in nondiabetic normotensive subjects: a randomized, placebo-controlled trial. J Am Heart Assoc. 2020;9(13):e016173. https://doi.org/10.1161/JAHA.119.016173.
Tsai KF, Chen YL, Chiou TT, Chu TH, Li LC, Ng HY, et al. Emergence of SGLT2 inhibitors as powerful antioxidants in human diseases. Antioxidants (Basel). 2021;10(8):1166. https://doi.org/10.3390/antiox10081166.
Scisciola L, Cataldo V, Taktaz F, Fontanella RA, Pesapane A, Ghosh P, et al. Anti-inflammatory role of SGLT2 inhibitors as part of their anti-atherosclerotic activity: data from basic science and clinical trials. Front Cardiovasc Med. 2022;9:1008922. https://doi.org/10.3389/fcvm.2022.1008922.
Wang D, Liu J, Zhong L, Li S, Zhou L, Zhang Q, et al. The effect of sodium-glucose cotransporter 2 inhibitors on biomarkers of inflammation: a systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2022;13:1045235. https://doi.org/10.3389/fphar.2022.1045235.
Schönberger E, Mihaljević V, Steiner K, Šarić S, Kurevija T, Majnarić LT, et al. Immunomodulatory effects of SGLT2 Inhibitors-targeting inflammation and oxidative stress in aging. Int J Environ Res Public Health. 2023;20(17):6671. https://doi.org/10.3390/ijerph20176671.
Shi P, Zhan Z, Ye X, Lu Y, Song K, Sheng F, et al. The antioxidative effects of empagliflozin on high glucose-induced epithelial-mesenchymal transition in peritoneal mesothelial cells via the Nrf2/HO-1 signaling. Ren Fail. 2022;44(1):1528–42. https://doi.org/10.1080/0886022X.2022.2118066.
Theofilis P, Sagris M, Oikonomou E, Antonopoulos AS, Siasos G, Tsioufis K, et al. The impact of SGLT2 inhibitors on inflammation: a systematic review and meta-analysis of studies in rodents. Int Immunopharmacol. 2022;111:109080. https://doi.org/10.1016/j.intimp.2022.109080.
Mazzieri A, Basta G, Calafiore R, Luca G. GLP-1 RAs and SGLT2i: two antidiabetic agents associated with immune and inflammation modulatory properties through the common AMPK pathway. Front Immunol. 2023;14:1163288. https://doi.org/10.3389/fimmu.2023.1163288.
Yang L, Zhang X, Wang Q. Effects and mechanisms of SGLT2 inhibitors on the NLRP3 inflammasome, with a focus on atherosclerosis. Front Endocrinol (Lausanne). 2022;13:992937. https://doi.org/10.3389/fendo.2022.992937.
Kim SR, Lee SG, Kim SH, Kim JH, Choi E, Cho W, et al. SGLT2 inhibition modulates NLRP3 inflammasome activity via ketones and insulin in diabetes with cardiovascular disease. Nat Commun. 2020;11(1):2127. https://doi.org/10.1038/s41467-020-15983-6.
Abdollahi E, Keyhanfar F, Delbandi AA, Falak R, Hajimiresmaiel SJ, Shafiei M. Dapagliflozin exerts anti-inflammatory effects via inhibition of LPS-induced TLR-4 overexpression and NF-κB activation in human endothelial cells and differentiated macrophages. Eur J Pharmacol. 2022;918: 174715. https://doi.org/10.1016/j.ejphar.2021.174715.
Evenepoel P, Meijers B, Masereeuw R, Lowenstein J. Effects of an SGLT inhibitor on the production, toxicity, and elimination of gut-derived uremic toxins: a call for additional evidence. Toxins (Basel). 2022;14(3):210. https://doi.org/10.3390/toxins14030210.
Mishima E, Fukuda S, Kanemitsu Y, Saigusa D, Mukawa C, Asaji K, et al. Canagliflozin reduces plasma uremic toxins and alters the intestinal microbiota composition in a chronic kidney disease mouse model. Am J Physiol Renal Physiol. 2018;315(4):F824–33. https://doi.org/10.1152/ajprenal.00314.2017.
Ho HJ, Kikuchi K, Oikawa D, Watanabe S, Kanemitsu Y, Saigusa D, et al. SGLT-1-specific inhibition ameliorates renal failure and alters the gut microbial community in mice with adenine-induced renal failure. Physiol Rep. 2021;9(24):e15092. https://doi.org/10.14814/phy2.15092.
Takebayashi K, Inukai T. Effect of sodium glucose cotransporter 2 inhibitors with low SGLT2/SGLT1 selectivity on circulating glucagon-like peptide 1 levels in type 2 diabetes mellitus. J Clin Med Res. 2017;9(9):745–53. https://doi.org/10.14740/jocmr3112w.
Zambrowicz B, Ding ZM, Ogbaa I, Frazier K, Banks P, Turnage A, et al. Effects of LX4211, a dual SGLT1/SGLT2 inhibitor, plus sitagliptin on postprandial active GLP-1 and glycemic control in type 2 diabetes. Clin Ther. 2013;35(3):273–85. https://doi.org/10.1016/j.clinthera.2013.01.010.
Kusunoki M, Hisano F, Matsuda SI, Kusunoki A, Wakazono N, Tsutsumi K, et al. Effects of SGLT2 inhibitors on the intestinal bacterial flora in Japanese patients with type 2 diabetes mellitus. Drug Res (Stuttg). 2023;73(7):412–6. https://doi.org/10.1055/a-2037-5250.
Hata S, Okamura T, Kobayashi A, Bamba R, Miyoshi T, Nakajima H, et al. Gut microbiota changes by an SGLT2 inhibitor, luseogliflozin, alters metabolites compared with those in a low carbohydrate diet in db/db mice. Nutrients. 2022;14(17):3531. https://doi.org/10.3390/nu14173531.
Wu J, Chen Y, Yang H, Gu L, Ni Z, Mou S, et al. Sodium glucose co-transporter 2 (SGLT2) inhibition via dapagliflozin improves diabetic kidney disease (DKD) over time associatied with increasing effect on the gut microbiota in db/db mice. Front Endocrinol (Lausanne). 2023;14:1026040. https://doi.org/10.3389/fendo.2023.1026040.
Deng L, Yang Y, Xu G. Empagliflozin ameliorates type 2 diabetes mellitus-related diabetic nephropathy via altering the gut microbiota. Biochim Biophys Acta Mol Cell Biol Lipids. 2022;1867(12):159234. https://doi.org/10.1016/j.bbalip.2022.159234.
Das US, Paul A, Banerjee S. SGLT2 inhibitors in heart failure with reduced ejection fraction. Egypt Heart J. 2021;73(1):93. https://doi.org/10.1186/s43044-021-00218-w.
Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al; DECLARE–TIMI 58 Investigators. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380(4):347–57. https://doi.org/10.1056/NEJMoa1812389.
Neal B, Perkovic V, Matthews DR. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377(21):2099. https://doi.org/10.1056/NEJMc1712572.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al; EMPA-REG OUTCOME investigators. empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–28. https://doi.org/10.1056/NEJMoa1504720.
Tuttle KR, Brosius FC 3rd, Cavender MA, Fioretto P, Fowler KJ, Heerspink HJL, et al. SGLT2 inhibition for CKD and cardiovascular disease in type 2 diabetes: report of a scientific workshop sponsored by the national kidney foundation. Diabetes. 2021;70(1):1–16. https://doi.org/10.2337/dbi20-0040.
Khiali S, Taban-Sadeghi M, Sarbakhsh P, Khezerlouy-Aghdam N, Rezagholizadeh A, Asham H, et al. SGLT2 inhibitors’ cardiovascular benefits in individuals without diabetes, heart failure, and/or chronic kidney disease: a systematic review. J Clin Pharmacol. 2023;63(12):1307–23. https://doi.org/10.1002/jcph.2311.
Chen X, Wang J, Lin Y, Yao K, Xie Y, Zhou T. Cardiovascular outcomes and safety of SGLT2 inhibitors in chronic kidney disease patients. Front Endocrinol (Lausanne). 2023;14:1236404. https://doi.org/10.3389/fendo.2023.1236404.
Li N, Zhou G, Zheng Y, Lv D, Zhu X, Wei P, et al. Effects of SGLT2 inhibitors on cardiovascular outcomes in patients with stage 3/4 CKD: a meta-analysis. PLoS ONE. 2022;17(1):e0261986. https://doi.org/10.1371/journal.pone.0261986.
Schricker S, Oberacker T, Fritz P, Ketteler M, Alscher MD, Schanz M. Peritoneal expression of SGLT-2, GLUT1, and GLUT3 in peritoneal dialysis patients. Kidney Blood Press Res. 2022;47(2):125–34. https://doi.org/10.1159/000520894.
Schröppel B, Fischereder M, Wiese P, Segerer S, Huber S, Kretzler M, et al. Expression of glucose transporters in human peritoneal mesothelial cells. Kidney Int. 1998;53(5):1278–87. https://doi.org/10.1046/j.1523-1755.1998.00899.x.
Balzer MS, Rong S, Nordlohne J, Zemtsovski JD, Schmidt S, Stapel B, et al. SGLT2 inhibition by intraperitoneal dapagliflozin mitigates peritoneal fibrosis and ultrafiltration failure in a mouse model of chronic peritoneal exposure to high-glucose dialysate. Biomolecules. 2020;10(11):1573. https://doi.org/10.3390/biom10111573.
Zhou Y, Fan J, Zheng C, Yin P, Wu H, Li X, et al. SGLT-2 inhibitors reduce glucose absorption from peritoneal dialysis solution by suppressing the activity of SGLT-2. Biomed Pharmacother. 2019;109:1327–38. https://doi.org/10.1016/j.biopha.2018.10.106.
Shentu Y, Li Y, Xie S, Jiang H, Sun S, Lin R, et al. Empagliflozin, a sodium glucose cotransporter-2 inhibitor, ameliorates peritoneal fibrosis via suppressing TGF-β/Smad signaling. Int Immunopharmacol. 2021;93:107374. https://doi.org/10.1016/j.intimp.2021.107374.
Martus G, Bergling K, de Arteaga J, Öberg CM. SGLT2 inhibition does not reduce glucose absorption during experimental peritoneal dialysis. Perit Dial Int. 2021;41(4):373–80. https://doi.org/10.1177/08968608211008095.
Martus G, Bergling K, Öberg CM. Dual SGLT1/SGLT2 inhibitor phlorizin reduces glucose transport in experimental peritoneal dialysis. Perit Dial Int. 2023;43(2):145–50. https://doi.org/10.1177/08968608221080170.
Wang J, Lv X, A-Ni-Wan AS, Tian SS, Wang JM, Liu HY, et al. Canagliflozin alleviates high glucose-induced peritoneal fibrosis via HIF-1α inhibition. Front Pharmacol. 2023;14:1152611. https://doi.org/10.3389/fphar.2023.1152611.
Alhwiesh AK, Abdul-Rahman IS, Nasreldin MA, Mohammed AM, Al-Oudah S, Al-Thwainy R, et al. The use of SGLT2 inhibitors in peritoneal dialysis patients: a shade of light on dapagliflozin. Arch Nephrol Urol. 2022;5(1):1–8. https://doi.org/10.26502/anu.2644-2833047.
Lai JW, Lin HJ, Chou CY. SGLT-2 inhibitors may increase ultrafiltration in incident peritoneal dialysis patients: a case report. BMC Nephrol. 2023;24(1):106. https://doi.org/10.1186/s12882-023-03164-8.
van der Aart-van der Beek AB, de Boer RA, Heerspink HJL. Kidney and heart failure outcomes associated with SGLT2 inhibitor use. Nat Rev Nephrol. 2022;18(5):294–306. https://doi.org/10.1038/s41581-022-00535-6.
Mascolo A, Di Napoli R, Balzano N, Cappetta D, Urbanek K, De Angelis A, et al. Safety profile of sodium glucose co-transporter 2 (SGLT2) inhibitors: A brief summary. Front Cardiovasc Med. 2022;9:1010693. https://doi.org/10.3389/fcvm.2022.1010693.
Zhou X, Ye X, Guo X, Liu D, Xu J, Hu F, et al. Safety of SGLT2 inhibitors: a pharmacovigilance study from 2013 to 2021 based on FAERS. Front Pharmacol. 2021;12:766125. https://doi.org/10.3389/fphar.2021.766125.
Pelletier R, Ng K, Alkabbani W, Labib Y, Mourad N, Gamble JM. Adverse events associated with sodium glucose co-transporter 2 inhibitors: an overview of quantitative systematic reviews. Ther Adv Drug Saf. 2021;12:2042098621989134. https://doi.org/10.1177/2042098621989134.
Palmer BF, Clegg DJ. Euglycemic ketoacidosis as a complication of SGLT2 inhibitor therapy. Clin J Am Soc Nephrol. 2021;16(8):1284–91. https://doi.org/10.2215/CJN.17621120.
Ata F, Yousaf Z, Khan AA, Razok A, Akram J, Ali EAH, et al. SGLT-2 inhibitors associated euglycemic and hyperglycemic DKA in a multicentric cohort. Sci Rep. 2021;11(1):10293. https://doi.org/10.1038/s41598-021-89752-w.
Sampani E, Sarafidis P, Dimitriadis C, Kasimatis E, Daikidou D, Bantis K, et al. Severe euglycemic diabetic ketoacidosis of multifactorial etiology in a type 2 diabetic patient treated with empagliflozin: case report and literature review. BMC Nephrol. 2020;21(1):276. https://doi.org/10.1186/s12882-020-01930-6.
Ogawa W, Sakaguchi K. Euglycemic diabetic ketoacidosis induced by SGLT2 inhibitors: possible mechanism and contributing factors. J Diabetes Investig. 2016;7(2):135–8. https://doi.org/10.1111/jdi.12401.
Choi H, Nguyen LA, Wan J, Milani H, McGill K, Park J. Adverse events of sodium-glucose cotransporter-2 inhibitors in chronic kidney disease: a retrospective chart review. Perm J. 2021;25:20.242. https://doi.org/10.7812/TPP/20.242.
Caro MKC, Cunanan EC, Kho SA. Incidence and factors associated with genitourinary infections among Type 2 diabetes patients on SGLT2 Inhibitors: a single retrospective cohort study. Diabetes Epidemiol Manag. 2022;7:100082. https://doi.org/10.1016/j.deman.2022.100082.
Dave CV, Schneeweiss S, Patorno E. Comparative risk of genital infections associated with sodium-glucose co-transporter-2 inhibitors. Diabetes Obes Metab. 2019;21(2):434–8. https://doi.org/10.1111/dom.13531.
McGovern AP, Hogg M, Shields BM, Sattar NA, Holman RR, Pearson ER, et al; MASTERMIND consortium. Risk factors for genital infections in people initiating SGLT2 inhibitors and their impact on discontinuation. BMJ Open Diabetes Res Care. 2020;8(1):e001238. https://doi.org/10.1136/bmjdrc-2020-001238.
Masajtis-Zagajewska A, Hołub T, Pęczek K, Makówka A, Nowicki M. Different effects of empagliflozin on markers of mineral-bone metabolism in diabetic and non-diabetic patients with stage 3 chronic kidney disease. Medicina. 2021;57(12):1352. https://doi.org/10.3390/medicina57121352.
Blau JE, Bauman V, Conway EM, Piaggi P, Walter MF, Wright EC, et al. Canagliflozin triggers the FGF23/1,25-dihydroxyvitamin D/PTH axis in healthy volunteers in a randomized crossover study. JCI Insight. 2018;3(8):e99123. https://doi.org/10.1172/jci.insight.99123.
Dobre M. Safety of SGLT2 Inhibitors in CKD: walking the fine line. Clin J Am Soc Nephrol. 2022;17(6):774–6. https://doi.org/10.2215/CJN.04900422.
Cowan A, Jeyakumar N, Kang Y, Dixon SN, Garg AX, Naylor K, et al. Fracture risk of sodium-glucose cotransporter-2 inhibitors in chronic kidney disease. Clin J Am Soc Nephrol. 2022;17(6):835–42. https://doi.org/10.2215/CJN.16171221.
Swanner AA, Hawley CE, Li K, Triantafylidis LK, Li J, Paik JM. Medication optimization for new initiators of empagliflozin for diabetic kidney disease. Clin Diabetes. 2022;40(2):158–67. https://doi.org/10.2337/cd21-0078.
Lalagkas PN, Poulentzas G, Kontogiorgis C, Douros A. Potential drug-drug interaction between sodium-glucose co-transporter 2 inhibitors and statins: pharmacological and clinical evidence. Expert Opin Drug Metab Toxicol. 2021;17(6):697–705. https://doi.org/10.1080/17425255.2021.1921735.
Acknowledgements
Not applicable.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
N.S. is the only contributor to the paper.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The author declares no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Stepanova, N. SGLT2 inhibitors in peritoneal dialysis: a promising frontier toward improved patient outcomes. Ren Replace Ther 10, 5 (2024). https://doi.org/10.1186/s41100-024-00523-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41100-024-00523-5