Abstract
Many individuals have different blood pressure (BP) values in the office setting compared to that outside the office setting. Therefore, confirming hypertension based on office BP (OBP) measurement alone can lead to misdiagnosis and mistreatment. The limitations of OBP measurement have led to the complementary use of out-of-office BP measurements, including 24-hour ambulatory blood pressure monitoring (ABPM) and home blood pressure monitoring (HBPM). This review aims to describe when and how ABPM or HBPM can be used to accurately diagnose and treat hypertension. Both methods should be performed using validated automated oscillometric devices. To minimize user errors, ABPM should be performed using standard techniques, whereas HBPM requires patient education regarding proper BP measurements. ABPM provides short-term comprehensive information on BP, including daytime, nighttime, morning, and 24-h BP. Therefore, ABPM is recommended for the initial diagnosis of hypertension, assessment of BP phenotypes and circadian patterns, and detection of nocturnal hypertension, Furthermore, ABPM plays a critical role in confirming true resistant hypertension thereby excluding pseudo-resistant hypertension. However, it is not suitable for long-term follow-up of patients with hypertension. In contrast, HBPM involves multiple BP readings taken at specific times during the day and evening over a long period. Therefore, HBPM is recommended for diagnosing hypertension and assessing BP phenotypes. However, this method has limitations in measuring nocturnal BP and circadian BP patterns. HBPM is preferred over ABPM for the long-term follow-up of patients with hypertension. This approach improves patient adherence to treatment and ultimately enhances the rate of control of hypertension. Additionally, both methods play an important role in diagnosing and treating white coat hypertension during pregnancy. Consequently, out-of-office BP measurement is essential to prevent the misdiagnosis and mistreatment of hypertension. However, these two methods offer different information regarding the BP status of an individual, and they indeed show discrepancies in the diagnosis of hypertensive phenotypes. Therefore, it is crucial to understand the advantages and limitations of both ABPM and HBPM to ensure their appropriate use in clinical practice.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Hypertension is the primary cause of cardiovascular disease in Korea, [1] and accurate diagnosis and treatment of hypertension are critical for reducing cardiovascular morbidity and mortality. However, blood pressure (BP) fluctuates inherently, and many individuals have different BP readings in office settings compared to those outside the office. Thus, confirming hypertension based on office BP (OBP) measurement alone may lead to misdiagnosis and mistreatment. These limitations of OBP measurements have led to the complementary use of out-of-office BP measurements [2,3,4]. The major advantage of this method is its ability to provide multiple BP readings, away from medical settings, during routine daily activities. In addition, it is a stronger predictor of cardiovascular disease risk than OBP [5,6,7,8,9,10]. Out-of-office BP measurement comprises two techniques: 24-h ambulatory blood pressure monitoring (ABPM) and home blood pressure monitoring (HBPM). Among these, ABPM assesses both daytime and nighttime BP readings over 24 h, providing short-term comprehensive information on BP. However, it is inappropriate for long-term follow-up of patients with hypertension [11, 12]. Conversely, HBPM provides multiple BP readings at specific times during the day and night over a prolonged period, leading to improved patient adherence and physician inertia, and ultimately increased BP control rate [13, 14]. However, it is not suitable for detecting nocturnal BP values and all users require education on proper BP measurement [12, 15, 16]. Thus, these two methods offer somewhat different information on the BP status of an individual for the diagnosis and treatment of hypertension.
Therefore, we aimed to describe when and how ABPM or HBPM should be used for the proper diagnosis and treatment of hypertension in clinical practice.
Ambulatory blood pressure monitoring
Advantages and limitations
ABPM provides comprehensive information on BP, including daytime, nighttime, morning, and 24-h BP, thereby enabling the assessment of circadian BP patterns, BP phenotypes, and short-term BP variability.
Therefore, ABPM is preferred over HBPM for the initial diagnosis of hypertension; however, it is not suitable for long-term follow-up [4]. Yacong et al. reported that ABPM had excellent reproducibility at the population level, whereas the reproducibility of intra-individual BP values and dipping status from a 24-h ABPM was limited [17]. Poor intra-individual reproducibility may be related to differences in daily activities, decreased accuracy at higher BP, quality of sleep, and probably reduced accuracy of the device under real ambulant conditions [18]. Moreover, conventional ABPM is not widely feasible in primary clinics, is not acceptable for frequent use, and may cause problems such as sleep disturbance, anxiety, and skin irritation during monitoring owing to cuff inflation [19]. To overcome these shortcomings, cuffless BP monitoring using a wearable device has been attempted for continuous BP estimation [20]. However, currently, cuffless devices are not recommended for use in the diagnosis or management of hypertension in clinical practice [4]. HBPM may be considered for patients unwilling to undergo ABPM or for those who previously experienced considerable discomfort during ABPM [4].
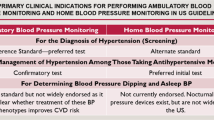
Device selection and procedures for ABPM (Fig. 1)
Clinical use of ABPM and HBPM. This figure shows the standard protocols and clinical indications for ABPM (left) and HBPM (right). The correct posture for ABPM is illustrated on the right. A small digital blood pressure monitor was attached to a belt around the patient’s waist and connected to a cuff around the upper arm. The cuff is wrapped around the non-dominant upper arm (usually left) at the heart level. It is to remain still with the arm relaxed during each measurement. The standard posture of HBPM is illustrated on the left: sitting in a chair with the back straight and supported, legs uncrossed, feet kept on the floor, bare arm rested on the table, cuff to fit arm circumference, and cuff placed mid-arm at heart level. The indications for ABPM and HBPM are listed in the center. The degree of recommendation is indicated by the degree of the plus mark: +++, strong recommendation; ++, moderate recommendation; +, weak recommendation; -, no recommendation, owing to the limited role of HBPM. ABPM, ambulatory blood pressure monitoring; HBPM, home blood pressure monitoring; WCH, white coat hypertension; WUCH, white coat uncontrolled hypertension; MH, masked hypertension; MUCH, masked uncontrolled hypertension; BP, blood pressure; RH, resistant hypertension; 24 h, 24 h; min, minutes
It is important to use a cuff-based, validated, upper arm oscillometric device according to established protocol. Separate validation is required in specific populations, such as children and pregnant women, for conventional 24-h ABPM [21]. Up-to-date lists of validated devices are available on both global and regional websites (www.stridebp.org, www.medaval.ie, www.dableducational.org, and www.validatebp.org) [22]. ABPM is programmed to take BP readings every 15–30 min during the day and every 30–60 min at night. It was performed on the non-dominant arm with an appropriate cuff according to the device instructions [11, 12]. The recently published 2023 ESH (European Society of Hypertension) Guidelines advise measuring blood pressure every 20 min throughout the day and night to minimize the risk of missing measurements and ensure the reliability of ABPM [4]. It is necessary to provide instructions for ABPM to patients, to preferably perform with usual daily activities and nighttime sleep, to remain still with the arm relaxed during each measurement, and to maintain a diary. ABPM data were considered satisfactory with 24-h recording with at least 70% of expected measurements, at least 20 valid awake measurements, and seven valid asleep measurements [11, 12].
Home blood pressure monitoring
Advantages and limitations
HBPM assesses multiple BP readings outside the office setting in the usual environment of each individual. In addition, it is easy and relatively inexpensive to use. Thus, it can be used over a prolonged period, and its data are more reproducible than those obtained from office BP measurements [12, 15, 16]. Consequently, it provides insight into BP phenotypes such as white coat hypertension (WCH) and masked hypertension (MH), and long-term variability in BP [12, 15, 16]. Furthermore, it allows long-term follow-up of treated hypertension. However, HBPM is generally not well suited for assessing nocturnal hypertension and nocturnal BP dipping status, and may induce anxiety in some patients [12, 15, 16]. Additionally, since it is performed by the patients themselves without medical supervision, it is crucial to educate the patients regarding the standardized protocol to obtain accurate BP readings [3, 12, 15, 16].
Device selection and procedures for HBPM (Fig. 1)
To reduce device errors, validated upper arm devices are generally recommended for HBPM because BP values are computed by predetermined algorithms that are known only to the manufacturer [3, 12].. Picone et al. reported that a few devices for HBPM globally have evidence of validation for accuracy [23]. Moreover, devices may be inaccurate in arrhythmias, pregnancy, and pediatrics, [16] but recently, devices that can detect arrhythmias, such as atrial fibrillation, have become available on the market [24]. The standard protocol for HBPM is as follows: rest for 5 min before measurement, avoid smoking, alcohol, or caffeine for 30 min, and refrain from talking during and between measurements. For measurement, select a validated automated upper arm device, use the proper cuff size according to the device instructions, place the cuff at heart level, and take two measurements with 1-min interval in the morning and in the evening. The measurement should be performed for 7 days (at least 5 days) for the initial diagnosis, and duplicate measurements should be taken once or twice per week or per month for long-term follow-up. After measurement, all the readings should be accurately recorded in a BP log [3, 4, 12, 16]. The most common user errors for HBPM may include using the wrong-sized cuff, incorrect patient positioning, incorrect cuff placement, and using non-validated devices [25]. Therefore, ensuring that HBPM is measured correctly before using it in general practice is crucial.
Clinical implications for using ABPM and HBPM (Fig. 1)
Diagnosis of hypertension
Hodgkinson et al. reported that an OBP > 140/90 mmHg showed a mean sensitivity and specificity of 74.6% for diagnosing hypertension compared to ABPM at 135/85 mmHg. In contrast, HBPM > 135/85 mmHg exhibited a mean sensitivity of 85.7% and specificity of 62.4% [26]. These findings suggest that neither OBP nor HBPM have sufficient sensitivity or specificity to be recommended as a single diagnostic test for hypertension [26]. Therefore, when OBP is elevated, guidelines recommend repeated measurements of OBP at subsequent office visits or out-of-office BP measurements using either ABPM or HBPM to confirm hypertension [2,3,4]. Ideally, both ABPM and HBPM should be employed to confirm hypertension as they provide somewhat different and complementary information regarding the BP status.
Diagnosis and treatment of WCH and WUCH
Based on the results of the OBP and out-of-office measurements, the BP status was categorized into four phenotypes in untreated and treated patients [3, 4] (Fig. 2). The Korean Ambulatory BP Monitoring (Kor-ABP) Registry study showed that the discordancy rate between ambulatory BP and OBP in untreated and treated patients was 32.5% and 26.5%, respectively; WCH (14.9%) and MH (17.6%) in untreated patients; white coat uncontrolled hypertension (WUCH, 13.5%) and masked uncontrolled hypertension (MUCH, 13.0%) in treated patients. Therefore, approximately one-third of the patients with hypertension would have been misdiagnosed if they had considered OBP alone [27]. Kang et al. reported that HBPM has high specificity (86–94%) but low sensitivity (47–74%) compared to ABPM in the diagnosis of WCH and MH, regardless of treatment status [28]. This suggests that HBPM may complement ABPM in the diagnosis of WCH and WUCH. The ESH recommends that ABPM and/or HBPM be performed when WCH is suspected in patients with grade 1 hypertension [4]. Misdiagnosis can lead to over or undertreatment in real-world practice. Therefore, when BP is discordant between OBP and out-of-office BP, antihypertensive medications should be considered based on out-of-office BP. Compared to normotension, WCH was not an innocent condition [29,30,31,32,33]. Mancia et al. reported that WCH alone without hypertension-mediated organ damage (HMOD) is accompanied by a marked increase in new hypertension, new HMOD, and long-term risk of mortality compared to normotension [33]. Therefore, although patients with WCH should not initiate antihypertensive medication at this time, they should be recommended periodic BP monitoring with repeated ABPM or long-term HBPM and follow-up assessment of cardiovascular risk factors and HMOD [3, 4]. Lifestyle interventions are recommended for patients with WCH to reduce cardiovascular risk. Moreover, whether drug treatment should be used remains unresolved; however, it can be considered in patients with WCH, HMOD, and a high cardiovascular risk [4]. In contrast, among treated patients, those with WUCH did not have an increased cardiovascular risk compared to those with controlled OBP and controlled out-of-office BP [3, 4, 31]. Therefore, if OBP is elevated, ABPM or HBPM may be considered to rule out WUCH to avoid mistreating it as uncontrolled hypertension before up-titrating the antihypertensive medication. Additionally, repeat out-of-office BP measurements are recommended to confirm WCH and to detect the transition to sustained hypertension in individuals with WCH [3, 4].
Classification of BP phenotypes and their cardiovascular risk in untreated and treated patients with hypertension. According to the results of OBP and out-of-office measurements, an individual’s BP status is categorized into four phenotypes in untreated and treated patients; normotension (both OBP and out-of-office BP not elevated), WCH (elevated OBP but not out-of-office BP, MH (elevated out-of-office BP but not OBP), sustained hypertension (elevated OBP and out-of-office BP) in untreated patients; controlled hypertension (controlled OBP and out-of-office BP), WUCH (elevated OBP but not out-of-office BP), MUCH (elevated out-of-office BP but not OBP), and uncontrolled hypertension (elevated OBP and out-of-office BP) in treated patients with hypertension. Black and dark gray colors indicate increased risk of cardiovascular disease; light gray show intermediate risk between normotension and sustained hypertension; white color indicates no increased cardiovascular risk. BP, blood pressure; OBP, office BP; WCH, white coat hypertension; WUCH, white coat uncontrolled hypertension; MH, masked hypertension; MUCH, masked uncontrolled hypertension; 24-h, 24-hour
Diagnosis and treatment of MH and MUCH
MH is associated with a high risk of progression to sustained hypertension, metabolic abnormalities, HMOD, and adverse clinical outcomes [29, 34,35,36,37,38]. The prevalence of MH also varies depending on the time of BP monitoring. However, who should be screened and how the screening should be performed for MH has not yet been established. Booth et al. reported that performing ABPM in all adults in the U.S. with non-elevated OBP who were not taking antihypertensive medications is impractical because of the low probability of detecting MH [39]. An OBP in the prehypertensive range (120–139/80–89 mmHg) is associated with a higher prevalence of MH. However, Viera et al. demonstrated that an OBP cutoff of 120/82 mmHg would result in a high false-positive rate of approximately 40% [40]. Thus, OBP alone may not be sufficient to detect MH. Several factors, including young age, male sex, cigarette smoking, alcohol drinking, anxiety, job stress, higher levels of physical activity, diabetes, chronic kidney disease, obstructive sleep apnea, exaggerated BP response to exercise, and orthostatic posture, might be predictors of MH [3, 4, 41]. Thus, individuals with normal or high-normal OBP accompanying multiple risk factors for MH or HMOD may be considered for screening for MH [3, 4, 35]. A previous systemic review and meta-analysis reported that the reproducibility of MH was better with ABPM than with HBPM (Kappa reliability test: 0.41 in ABPM and 0.26 in HBPM) [42]. Kang et al. demonstrated that HBPM has high specificity (86–94%) but low sensitivity (47.74%) in the diagnosis of MH [28]. Thus, HBPM may serve as a complementary method to, but not a replacement for, ABPM. Cohen et al. demonstrated that the Kappa statistics between the first and second ABPM for the detection of MH were 0.50 for masked awake, 0.57 for masked 24-h, 0.57 for masked asleep, and 0.58 for any MH. Thus, the authors suggest that clinicians should consider the moderate reproducibility of MH when interpreting the results of a single ABPM [43]. Accordingly, the diagnosis of MH requires confirmation with at least a second set of office and out-of-office BP measurements. No randomized clinical trials have evaluated the efficacy of drug treatments for MH, and the effects of drug therapy are therefore unknown. However, considering MH as a poor prognostic factor, the ESH guidelines recommend stringent lifestyle modifications and close monitoring of patients with confirmed MH [4]. Antihypertensive drug therapy may be considered for patients with MH who have a higher cardiovascular risk and HMOD [4]. In treated patients, MUCH is associated with a worse metabolic profile, HMOD, and adverse clinical outcomes [37, 38, 44]. However, an optimal strategy for screening and treatment of MUCH has not been established. In the Korean-ABP registry, high-normal office BP, underuse of antihypertensive drugs, dyslipidemia, prior stroke, and left ventricular hypertrophy were suggested as predictors of MUCH [45]. ESH guidelines recommend that up-titration of medication should be advised in patients with MUCH above the recommended target BP values if it is well tolerated [4]. However, there is no established evidence to evaluate the efficacy of drug treatment and the optimal BP target for MUCH [4]. Therefore, physicians should consider excluding MH or MUCH with at least repeated ABPM or HBPM in patients with multiple risk factors for MH, MUCH, or HMOD despite normal or high-normal office BP to prevent the undertreatment of hypertension.
Detection of circadian BP patterns, nocturnal hypertension, and morning hypertension
The quantification of circadian BP changes is a unique advantage of ABPM, leading to the identification of dipping patterns. However, this is the main limitation of HBPM. Nondipping and reverse dipping were associated with an increased risk of cardiovascular events [46, 47]. Nocturnal hypertension is defined as an average BP ≥ 120/70 mmHg recorded during the night hours with ABPM [4]. HBPM may have limitations in measuring nocturnal BP, although some home devices have shown a level of accuracy comparable to that of ABPM [48]. However, further research on the appropriate measurement protocol and clinical significance of HBPM for detecting nocturnal BP is needed. Nocturnal hypertension is closely related to cardiovascular events and HMOD in the general population and patients with hypertension [49,50,51]. However, nocturnal hypertension is poorly reproducible in both untreated and treated patients. In addition, nighttime dipping and nondipping phenotypes can shift from one phenotype to another during treatment [52]. Therefore, these findings suggest that assessing nocturnal hypertension with ABPM is useful for predicting increased cardiovascular risk, but quantification of nighttime phenotype should be based on repeated ABPM rather than only a single recording. Morning hypertension was defined as elevated ABPM or HBPM in the morning (≥ 135/85 mm Hg) and a normal clinical BP (< 140/90 mmHg) [53]. Guo et al. reported that HBPM might be preferred over ABPM for detecting morning hypertension because of its better reproducibility and stronger correlation with vascular indices [54]. MH and MUCH comprise morning, daytime, and nocturnal hypertension [55]. Therefore, physicians should consider excluding MH or MUCH due to uncontrolled nocturnal and morning hypertension if they pose an increased risk of cardiovascular disease, even if their daytime BP is well controlled [55].
The optimal treatment for nocturnal and morning hypertension has not yet been established; however, several therapeutic strategies such as bedtime dosing and salt restriction have been proposed [56]. In certain instances of uncontrolled nocturnal and morning hypertension, particularly among individuals with conditions such as diabetes mellitus, chronic kidney disease, and obstructive sleep apnea, bedtime dosing has been shown to be effective in lowering evening and early morning BP [56]. However, the Treatment in Morning versus Evening (TIME) study failed to demonstrate the benefit of bedtime dosing in reducing cardiovascular outcomes in patients with hypertension [57]. In addition, evening dosing is associated with poor drug compliance. Therefore, ABPM may be the preferred method, whereas HBPM may have a limited role in evaluating circadian BP patterns and nocturnal hypertension. Morning BP can be assessed using both ABPM and HBPM [53]. Currently, it is unclear whether routine bedtime dosing is beneficial for reducing cardiovascular outcomes [56].
Confirmation of true RH
Resistant hypertension (RH) is defined as hypertension uncontrollable by ≥ 3 antihypertensive drugs, and it is associated with higher mortality and morbidity than non-RH [58, 59]. Therefore, it is important to accurately identify RH for proper management. In the Spanish ABPM Registry, among 8,295 patients diagnosed with RH based on OBP, 37.5% had normal ambulatory BP (WUCH) [60]. Thus, WUCH has been misdiagnosed as having RH, which is known as pseudo-RH. Therefore, excluding WUCH by using ABPM is a critical step in confirming the true RH. However, HBPM may have limitations in measuring nocturnal BP, although nondipping and nocturnal hypertension are commonly observed in RH. Therefore, HBPM may play a complementary role, but cannot replace ABPM in the diagnosis of RH. However, HBPM is useful in improving the long-term management of RH [59, 61]. It may provide information on the response to antihypertensive medication and improve drug adherence, ultimately leading to an improved hypertension control rate and decreased medical costs.
Detection of WCH with proper devices in pregnant women
The number of cases of pregnancy-induced hypertension is increasing in Korea [62]. During pregnancy, BP directly affects the mother’s health and fetal development. Thus, accurate BP measurement during pregnancy is crucial for the diagnosis and management of hypertension. Pregnancy induces significant hemodynamic changes including increased blood volume, stroke volume, and heart rate [63]. These alterations lead to an increased cardiac output and decreased peripheral vascular resistance. In a systematic review, Bello et al. found that the majority of validation studies evaluating BP measurement devices during pregnancy had violations [64]. Therefore, guidelines recommend the use of specially validated BP devices for OBP and out-of-office BP measurements during pregnancy [4]. Currently, OBP remains the primary method for diagnosing and treating hypertension in pregnant women. WCH is a common phenomenon in pregnant women; therefore, proper use of out-of-office BP monitoring is necessary to rule out WCH [65]. Moreover, women with WCH before 20 weeks are associated with worse perinatal and maternal outcomes than normotensive women but have better outcomes than those with gestational hypertension and chronic hypertension [66]. Therefore, if OBP is elevated in untreated pregnant women, it may be useful to confirm the diagnosis of hypertension using ABPM or HBPM to avoid unnecessary treatment [67, 68]. Consequently, women with WCH require continued HBPM throughout pregnancy to assess the risk of developing preeclampsia, having a small-for-gestational-age baby, and experiencing preterm birth. In treated pregnant women, WUCH may be interpreted as uncontrolled hypertension or a hypertensive emergency, potentially leading to overtreatment or unnecessary interruption of pregnancy [68]. Therefore, ABPM or HBPM is required for the proper management of all pregnant women, regardless of gestational age.
Assessing the efficacy of antihypertensive treatment and long-term control of hypertension
In a systemic review and meta-analysis, Agarwal et al. demonstrated that HBPM-based therapy resulted in reduction of BP (mean systolic/diastolic BP changes, -2.63 mmHg/-1.68 mmHg), and these reductions were greater when telemonitoring was used. Further, it led to more reductions in antihypertensive medication use and was associated with less therapeutic inertia [13]. Tucker et al. reported that BP-lowering effect with HBPM was significantly intensified with co-interventions such as patients’ education, feedback, and counselling (-1.0 mmHg with HBPM alone to -6.1 mmHg with HBPM combined with intervention) [69]. The randomized trial on the efficacy of self-monitored blood pressure, with or without telemonitoring, for the titration of antihypertensive medication (TASMINH4) demonstrated that HBPM-based titration of antihypertensive medications in patients with poorly controlled BP led to a significantly greater reduction in BP than that of OBP-based titration, regardless of telemonitoring [70]. Ultimately, this approach increased the rate of BP control [13] and drug adherence, [71] and reduced medical costs [72]. Therefore, HBPM is preferred over ABPM for long-term management of hypertension. However, Staessen et al. reported that adjusting antihypertensive treatment based on ABPM or HBPM instead of OBP led to less intensive drug treatment [73]. Consequently, further studies are needed to determine whether ABPM- or HBPM-based approaches lead to a reduction in cardiovascular outcomes compared to the OBP-based approach.
Decision making during discrepancy between ABPM and HBPM
Ntineri et al. reported a substantial disagreement between APBM and HBPM in the diagnosis of hypertensive phenotypes (20.1%) [74]. The significant determinants of this disagreement included age, sex, study center, body mass index, cardiovascular disease history, office hypertension, antihypertensive medication, and alcohol consumption [74]. In contrast, Kim et al. failed to demonstrate any demographic or clinical features that could predict disagreements [75]. Furthermore, they suggested that individuals with a diagnostic disagreement may have an intermediate cardiovascular risk between those with sustained normotension and those with hypertension. Consequently, this diagnostic disagreement could pose challenges in clinical decision-making. However, there is no consensus regarding the therapeutic strategies when diagnostic discrepancy occurs between ABPM and HBPM. Considering the advantages and limitations of both the methods, it is crucial to take the following factors into account when discrepancies occur in clinical situations. First, physicians should evaluate HBPM techniques, including the use of non-validated device, inadequate resting time before measurements, incorrect patient positioning, improper cuff size, improper cuff positioning (not mid-arm at heart level), and alcohol consumption before measurements. In such cases, physicians should re-educate individuals on proper HBPM techniques and reassess their home BP data. Secondly, it is essential to assess proper cuff positioning and ensure that patients remain still with a relaxed arm during each ABPM measurement. Repeated ABPM should be considered in these cases. Third, physicians should assess patient discomfort such as sleep disturbances and anxiety during ABPM. In such cases, ABPM data may be inaccurate, and repeated long-term HBPM may be used instead of repeated ABPM for the management of hypertension. Fourth, physicians should recognize the inherent limitations of HBPM, including its inability to detect nocturnal hypertension. Thus, if MH or MUCH is suspected due to nocturnal hypertension, ABPM is preferred over HBPM for diagnosis and decision-making regarding hypertension. Therefore, when discrepancies occur between ABPM and HBPM, physicians should evaluate and re-educate individuals on BP measurement techniques. Furthermore, long-term follow-up with HBPM or repeated ABPM should be considered.
Corresponding BP
There are well-known discrepancies between OBP and out-of-office BP owing to differences in measurement techniques, time window, white coat effect, and environmental factors [76]. Therefore, correctly categorizing individual BP status requires measuring both OBP and out-of-office BP. The prevalence of BP phenotypes can vary with different OBP and out-of-office BP thresholds [77]. Consequently, determining the threshold of out-of-office BP corresponding to OBP values is crucial for the accurate diagnoses and management of hypertension. Corresponding BP levels refer to out-of-office BP levels at which the risk of cardiovascular outcomes is similar to the risk associated with corresponding OBP [76]. Cohort-based or randomized controlled trial-based studies have suggested outcome-driven corresponding ambulatory and home BP values [76]. Generally, the difference between OBP and out-of-office BP thresholds is 5 mmHg. However, different guidelines and outcome-based studies have suggested different BP levels [76, 78,79,80]. The Korean Society of Hypertension (KSH) recommends the same corresponding BP values for diagnosing and treating hypertension [3] (Table 1). The corresponding value of OBP 140/90 mmHg is 135/85 mmHg in both daytime ambulatory BP and home BP. However, for an OBP ≤ 130 mmHg, the corresponding out-of-office BP is comparable to OBP because the white coat effect is likely to decrease at this level (Table 1).
Conclusion
This review describes device selection, implementation, and clinical implication of ABPM and HBPM for accurate diagnosis and treatment of hypertension in clinical practice. To prevent device errors, validated automated oscillometric devices should be used. ABPM is performed using standard techniques and patient education is essential for proper HBPM. ABPM offers short-term comprehensive information on BP values, making it the preferred method for initial diagnosis of hypertension and for understanding BP phenotypes and circadian BP patterns. However, HBPM is easy to use and provides multiple BP readings over a long period of time, making it the preferred choice for long-term monitoring to assess the effectiveness of antihypertensive treatment. If office BP is elevated, ABPM or HBPM should be considered before initiating or up-titrating antihypertensive medications, to rule out WCH or WUCH. Additionally, even with normal or high-normal office BP, ABPM or HBPM may be considered to exclude MH or MUCH in patients with HMOD. In cases of RH, ABPM is a critical step to confirm true RH, while HBPM is useful for long-term RH management. Thus, HBPM complements the diagnosis of MH/MUCH or true RH, although it has limitations in measuring nocturnal BP. WCH in pregnant women is associated with worse perinatal and maternal outcomes than in normotensive women. Thus, if OBP is elevated in pregnant women, ABPM or HBPM is required to confirm WCH or WUCH, regardless of gestational age. Therefore, continuous administration of HBPM throughout pregnancy is advisable. When the substantial discrepancies occur between ABPM and HBPM in clinical practice, physicians should evaluate and re-educate individuals on BP measurement techniques. Furthermore, long-term follow-up with HBPM or repeated ABPM should be considered. The corresponding value of OBP 140/90 mmHg is 135/85 mmHg in both daytime ambulatory BP and home BP. However, for an OBP ≤ 130 mmHg, the corresponding out-of-office BP is comparable to OBP because the white coat effect is likely to decrease at this level.
In conclusion, out-of-office BP monitoring, including ABPM and HBPM, is essential to prevent the misdiagnosis and mistreatment of hypertension. However, it is crucial to understand that these two methods provide somewhat different information about an individual’s BP status, making it necessary to appreciate the advantages and limitations of ABPM and HBPM for the accurate diagnosis and treatment of hypertension.
Data availability
Not applicable.
Abbreviations
- BP:
-
blood pressure
- OBP:
-
office blood pressure
- ABPM:
-
ambulatory blood pressure monitoring
- HBPM:
-
home blood pressure monitoring
- ESH:
-
European Society of Hypertension
- WCH:
-
white coat hypertension
- MH:
-
masked hypertension
- Kor-ABP registry:
-
Korea Ambulatory BP registry
- WUCH:
-
white coat uncontrolled hypertension
- MUCH:
-
masked uncontrolled hypertension
- HMOD:
-
hypertension-mediated organ damage
- TIME:
-
Treatment in Morning versus Evening
- RH:
-
resistant hypertension
- TASMIN4:
-
Efficacy of self-monitored blood pressure, with or without telemonitoring for the titration of antihypertensive medication
- KSH:
-
Korean Society of Hypertension
References
Kim HC, Lee H, Lee H-H, Lee G, Kim E, Song M, et al. Korea hypertension fact sheet 2022: analysis of nationwide population-based data with a special focus on hypertension in the elderly. Clin Hypertens. 2023;29(1):22.
Whelton PK, Carey RM, Aronow WS, Casey DE, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice guidelines. J Am Coll Cardiol. 2018;71(19):e127–e248.
Kim H-L, Lee EM, Ahn SY, Kim K-i, Kim HC, Kim JH, et al. The 2022 focused update of the 2018 Korean Hypertension Society Guidelines for the management of hypertension. Clin Hypertens. 2023;29(1):11.
Mancia G, Kreutz R, Brunström M, Burnier M, Grassi G, Januszewicz A, et al. 2023 ESH guidelines for the management of arterial hypertension the Task Force for the management of arterial hypertension of the European Society of Hypertension: endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA). J Hyperten. 2023;41(12):1874–2071.
Staessen JA, Thijs L, Fagard R, O’Brien ET, Clement D, de Leeuw PW, et al. Predicting cardiovascular risk using conventional vs ambulatory blood pressure in older patients with systolic hypertension. JAMA. 1999;282(6):539–46.
Dolan E, Stanton A, Thijs L, Hinedi K, Atkins N, McClory S, et al. Superiority of ambulatory over clinic blood pressure measurement in predicting mortality: the Dublin outcome study. Hypertension. 2005;46(1):156–61.
Redon J, Campos C, Narciso ML, Rodicio JL, Pascual JM, Ruilope LM. Prognostic value of ambulatory blood pressure monitoring in refractory hypertension: a prospective study. Hypertension. 1998;31(2):712–8.
Conen D, Bamberg F. Noninvasive 24-h ambulatory blood pressure and cardiovascular disease: a systematic review and meta-analysis. J Hypertens. 2008;26(7):1290–9.
Ohkubo T, Imai Y, Tsuji I, Nagai K, Kato J, Kikuchi N, et al. Home blood pressure measurement has a stronger predictive power for mortality than does screening blood pressure measurement: a population-based observation in Ohasama, Japan. J Hypertens. 1998;16(7):971–5.
Niiranen TJ, Hänninen M-R, Johansson J, Reunanen A, Jula AM. Home-measured blood pressure is a stronger predictor of cardiovascular risk than office blood pressure: the Finn-Home study. Hypertension. 2010;55(6):1346–51.
Kario K, Shin J, Chen CH, Buranakitjaroen P, Chia YC, Divinagracia R, et al. Expert panel consensus recommendations for ambulatory blood pressure monitoring in Asia: the HOPE Asia Network. J Clin Hypertens. 2019;21(9):1250–83.
Stergiou GS, Palatini P, Parati G, O’Brien E, Januszewicz A, Lurbe E, et al. 2021 European Society of Hypertension practice guidelines for office and out-of-office blood pressure measurement. J Hypertens. 2021;39(7):1293–302.
Agarwal R, Bills JE, Hecht TJ, Light RP. Role of home blood pressure monitoring in overcoming therapeutic inertia and improving hypertension control: a systematic review and meta-analysis. Hypertension. 2011;57(1):29–38.
Jo SH, Kim SA, Park KH, Kim HS, Han SJ, Park WJ. Self-blood pressure monitoring is associated with improved awareness, adherence, and attainment of target blood pressure goals: prospective observational study of 7751 patients. J Clin Hypertens. 2019;21(9):1298–304.
Park S, Buranakitjaroen P, Chen C-H, Chia Y-C, Divinagracia R, Hoshide S, et al. Expert panel consensus recommendations for home blood pressure monitoring in Asia: the Hope Asia Network. J Hum Hyperten. 2018;32(4):249–58.
Ihm S-H, Park J-H, Kim JY, Kim J-H, Kim K-I, Lee EM, et al. Home blood pressure monitoring: a position statement from the Korean Society of Hypertension Home blood pressure forum. Clin Hypertens. 2022;28(1):38.
Bo Y, Kwok K-O, Chung VC-H, Yu C-P, Tsoi KK-F, Wong SY-S, et al. Short-term reproducibility of ambulatory blood pressure measurements: a systematic review and meta-analysis of 35 observational studies. J Hypertens. 2020;38(11):2095–109.
Steen MSvd, Lenders JW, Graafsma SJ, Arend J, Thien T. Reproducibility of ambulatory blood pressure monitoring in daily practice. J Hum Hyperten. 1999;13(5):303–8.
Viera AJ, Lingley K, Hinderliter AL. Tolerability of the Oscar 2 ambulatory blood pressure monitor among research participants: a cross-sectional repeated measures study. BMC Med Res Methodol. 2011;11(1):1–7.
Joung J, Jung C-W, Lee H-C, Chae M-J, Kim H-S, Park J, et al. Continuous cuffless blood pressure monitoring using photoplethysmography-based PPG2BP-net for high intrasubject blood pressure variations. Sci Rep. 2023;13(1):8605.
Stergiou GS, Alpert B, Mieke S, Asmar R, Atkins N, Eckert S, et al. A universal standard for the validation of blood pressure measuring devices: Association for the Advancement of Medical Instrumentation/European Society of Hypertension/International Organization for Standardization (AAMI/ESH/ISO) collaboration Statement. Hypertension. 2018;71(3):368–74.
Picone DS, Padwal R, Campbell NR, Boutouyrie P, Brady TM, Olsen MH, et al. How to check whether a blood pressure monitor has been properly validated for accuracy. J Clin Hypertens. 2020;22(12):2167–74.
Picone DS, Campbell NR, Schutte AE, Olsen MH, Ordunez P, Whelton PK, et al. Validation status of blood pressure measuring devices sold globally. JAMA. 2022;327(7):680–1.
Balanis T, Sanner B. Detection of atrial fibrillation using a home blood pressure monitor. Vasc Health Risk Manag. 2021:407–14.
Nessler K, Krztoń-Królewiecka A, Suska A, Mann MR, Nessler MB, Windak A. The quality of patients’ self-blood pressure measurements: a cross-sectional study. BMC Cardiovasc Disord. 2021;21:1–8.
Hodgkinson J, Mant J, Martin U, Guo B, Hobbs F, Deeks J, et al. Relative effectiveness of clinic and home blood pressure monitoring compared with ambulatory blood pressure monitoring in diagnosis of hypertension: systematic review. BMJ. 2011;342:d3621.
Shin J, Park SH, Kim JH, Ihm SH, Kim K-i, Kim WS, et al. Discordance between ambulatory versus clinic blood pressure according to global cardiovascular risk group. Korean J Intern Med. 2015;30(5):610–9.
Kang Y-Y, Li Y, Huang Q-F, Song J, Shan X-L, Dou Y, et al. Accuracy of home versus ambulatory blood pressure monitoring in the diagnosis of white-coat and masked hypertension. J Hypertens. 2015;33(8):1580–7.
Ohkubo T, Kikuya M, Metoki H, Asayama K, Obara T, Hashimoto J, et al. Prognosis of masked hypertension and white-coat hypertension detected by 24-h ambulatory blood pressure monitoring: 10-year follow-up from the Ohasama study. J Am Coll Cardiol. 2005;46(3):508–15.
Mancia G, Bombelli M, Facchetti R, Madotto F, Quarti-Trevano F, Friz HP, et al. Long-term risk of sustained hypertension in white-coat or masked hypertension. Hypertension. 2009;54(2):226–32.
Cohen JB, Lotito MJ, Trivedi UK, Denker MG, Cohen DL, Townsend RR. Cardiovascular events and mortality in white coat hypertension: a systematic review and meta-analysis. Ann Intern Med. 2019;170(12):853–62.
Paiva AM, Gomes MI, Campana ÉM, Feitosa AD, Sposito AC, Mota-Gomes MA, et al. Impact of hypertension phenotypes on the office and 24-h pulse wave velocity and augmentation index in individuals with or without antihypertensive medication use. Hypertens Res. 2019;42(12):1989–95.
Mancia G, Facchetti R, Vanoli J, Dell’Oro R, Seravalle G, Grassi G. White-coat hypertension without organ damage: impact on long-term mortality, new hypertension, and new organ damage. Hypertension. 2022;79(5):1057–66.
Franklin SS, Thijs L, Li Y, Hansen TW, Boggia J, Liu Y, et al. Masked hypertension in diabetes mellitus: treatment implications for clinical practice. Hypertension. 2013;61(5):964–71.
Anstey DE, Pugliese D, Abdalla M, Bello NA, Givens R, Shimbo D. An update on masked hypertension. Curr Hypertens Rep. 2017;19:1–8.
Fujiwara T, Yano Y, Hoshide S, Kanegae H, Kario K. Association of cardiovascular outcomes with masked hypertension defined by home blood pressure monitoring in a Japanese general practice population. JAMA Cardiol. 2018;3(7):583–90.
Palla M, Saber H, Konda S, Briasoulis A. Masked hypertension and cardiovascular outcomes: an updated systematic review and meta-analysis. Integr Blood Press Control. 2018:11–24.
Zhang D-Y, Guo Q-H, An D-W, Li Y, Wang J-G. A comparative meta-analysis of prospective observational studies on masked hypertension and masked uncontrolled hypertension defined by ambulatory and home blood pressure. J Hypertens. 2019;37(9):1775–85.
Booth IIIJN, Muntner P, Diaz KM, Viera AJ, Bello NA, Schwartz JE, et al. Evaluation of criteria to detect masked hypertension. J Clin Hypertens. 2016;18(11):1086–94.
Viera AJ, Lin F-C, Tuttle LA, Shimbo D, Diaz KM, Olsson E, et al. Levels of office blood pressure and their operating characteristics for detecting masked hypertension based on ambulatory blood pressure monitoring. Am J Hypertens. 2015;28(1):42–9.
Franklin SS, O’Brien E, Thijs L, Asayama K, Staessen JA. Masked hypertension: a phenomenon of measurement. Hypertension. 2015;65(1):16–20.
Antza C, Farmakis I, Doundoulakis I, Akrivos E, Stalikas N, Zafeiropoulos S, et al. Reproducibility of masked hypertension and office-based hypertension: a systematic review and meta-analysis. J Hypertens. 2022;40(6):1053–9.
Cohen LP, Schwartz JE, Pugliese DN, Anstey DE, Christian JP, Jou S, et al. Short-term reproducibility of masked hypertension among adults without office hypertension. Hypertension. 2020;76(4):1169–75.
Fu X, Ren H, Xie J, Wang W, Li Y, Gao P, et al. Association of nighttime masked uncontrolled hypertension with left ventricular hypertrophy and kidney function among patients with chronic kidney disease not receiving dialysis. JAMA Netw Open. 2022;5(5):e2214460.
Kim H-J, Shin J-H, Lee Y, Kim JH, Hwang SH, Kim WS, et al. Clinical features and predictors of masked uncontrolled hypertension from the Korean ambulatory blood pressure monitoring Registry. Korean J Intern Med. 2021;36(5):1102–14.
Pierdomenico SD, Bucci A, Costantini F, Lapenna D, Cuccurullo F, Mezzetti A. Circadian blood pressure changes and myocardial ischemia in hypertensive patients with coronary artery disease. J Am Coll Cardiol. 1998;31(7):1627–34.
Ohkubo T, Hozawa A, Yamaguchi J, Kikuya M, Ohmori K, Michimata M, et al. Prognostic significance of the nocturnal decline in blood pressure in individuals with and without high 24-h blood pressure: the Ohasama study. J Hypertens. 2002;20(11):2183–9.
Kario K. Nocturnal hypertension: new technology and evidence. Hypertension. 2018;71(6):997–1009.
Fan H-Q, Li Y, Thijs L, Hansen TW, Boggia J, Kikuya M, et al. Prognostic value of isolated nocturnal hypertension on ambulatory measurement in 8711 individuals from 10 populations. J Hypertens. 2010;28(10):2036–45.
Salles GF, Reboldi G, Fagard RH, Cardoso CR, Pierdomenico SD, Verdecchia P, et al. Prognostic effect of the nocturnal blood pressure fall in hypertensive patients: the ambulatory blood pressure collaboration in patients with hypertension (ABC-H) meta-analysis. Hypertension. 2016;67(4):693–700.
Fujiwara T, Hoshide S, Kanegae H, Kario K. Cardiovascular event risks associated with masked nocturnal hypertension defined by home blood pressure monitoring in the J-HOP nocturnal blood pressure study. Hypertension. 2020;76(1):259–66.
Mancia G, Facchetti R, Bombelli M, Quarti-Trevano F, Cuspidi C, Grassi G. Short-and long-term reproducibility of nighttime blood pressure phenotypes and nocturnal blood pressure reduction. Hypertension. 2021;77(5):1745–55.
Wang JG, Kario K, Chen CH, Park JB, Hoshide S, Huo Y, et al. Management of morning hypertension: a consensus statement of an Asian expert panel. J Clin Hypertens. 2018;20(1):39–44.
Guo Q-H, Cheng Y-B, Zhang D-Y, Wang Y, Huang Q-F, Sheng C-S, et al. Comparison between home and ambulatory morning blood pressure and morning hypertension in their reproducibility and associations with vascular injury. Hypertension. 2019;74(1):137–44.
Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, et al. The Japanese Society of Hypertension guidelines for the management of hypertension (JSH 2019). Hypertens Res. 2019;42(9):1235–481.
Park S, Ihm S-H, Cho I-J, Kim D-H, Park JH, Chung W-B, et al. Statement on chronotherapy for the treatment of hypertension: consensus document from the Korean society of hypertension. Clin Hypertens. 2023;29(1):25.
Mackenzie IS, Rogers A, Poulter NR, Williams B, Brown MJ, Webb DJ, et al. Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial. Lancet. 2022;400(10361):1417–25.
Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72(5):e53–e90.
Park S, Shin J, Ihm SH, Kim K-i, Kim H-L, Kim HC, et al. Resistant hypertension: consensus document from the Korean society of hypertension. Clin Hypertens. 2023;29(1):30.
de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension. 2011;57(5):898–902.
Kim HM, Shin J. Role of home blood pressure monitoring in resistant hypertension. Clin Hypertens. 2023;29(1):2.
Kim HC, Lee H, Lee H-H, Eunsun S, Kim E, Han J, et al. Korea hypertension fact sheet 2021: analysis of nationwide population-based data with special focus on hypertension in women. Clin Hypertens. 2022;28:1.
Sanghavi M, Rutherford JD. Cardiovascular physiology of pregnancy. Circulation. 2014;130(12):1003–8.
Bello NA, Woolley JJ, Cleary KL, Falzon L, Alpert BS, Oparil S, et al. Accuracy of blood pressure measurement devices in pregnancy: a systematic review of validation studies. Hypertension. 2018;71(2):326–35.
Brown MA, Mangos G, Davis G, Homer C. The natural history of white coat hypertension during pregnancy. BJOG. 2005;112(5):601–6.
Johnson S, Liu B, Kalafat E, Thilaganathan B, Khalil A. Maternal and perinatal outcomes of white coat hypertension during pregnancy: a systematic review and meta-analysis. Hypertension. 2020;76(1):157–66.
Johnson S, Gordijn S, Damhuis S, Ganzevoort W, Brown M, von Dadelszen P, et al. Diagnosis and monitoring of White Coat hypertension in pregnancy: an ISSHP Consensus Delphi Procedure. Hypertension. 2022;79(5):993–1005.
Espeche WG, Salazar MR. Ambulatory blood pressure monitoring for diagnosis and management of hypertension in pregnant women. Diagnostics. 2023;13(8):1457.
Tucker KL, Sheppard JP, Stevens R, Bosworth HB, Bove A, Bray EP, et al. Self-monitoring of blood pressure in hypertension: a systematic review and individual patient data meta-analysis. PLoS Med. 2017;14(9):e1002389.
McManus RJ, Mant J, Franssen M, Nickless A, Schwartz C, Hodgkinson J, et al. Efficacy of self-monitored blood pressure, with or without telemonitoring, for titration of antihypertensive medication (TASMINH4): an unmasked randomised controlled trial. Lancet. 2018;391(10124):949–59.
Ogedegbe G, Schoenthaler A. A systematic review of the effects of home blood pressure monitoring on medication adherence. J Clin Hypertens. 2006;8(3):174–80.
Arrieta A, Woods JR, Qiao N, Jay SJ. Cost–benefit analysis of home blood pressure monitoring in hypertension diagnosis and treatment: an insurer perspective. Hypertension. 2014;64(4):891–6.
Staessen JA, Byttebier G, Buntinx F, Celis H, O’Brien ET, Fagard R. Antihypertensive treatment based on conventional or ambulatory blood pressure measurement: a randomized controlled trial. Ambul Blood Press Monit Treat Hypertens Investigators JAMA. 1997;278(13):1065–72.
Ntineri A, Niiranen TJ, McManus RJ, Lindroos A, Jula A, Schwartz C, et al. Ambulatory versus home blood pressure monitoring: frequency and determinants of blood pressure difference and diagnostic disagreement. J Hypertens. 2019;37(10):1974–81.
Kim CH, Kim JS, Rhee M-Y. Characteristics of individuals with disagreement between home and ambulatory blood pressure measurements for the diagnosis of hypertension. Healthcare. 2020;8(4):457.
Shin J, Wang JG, Chia YC, Kario K, Chen CH, Cheng HM et al. The HOPE Asia Network consensus on blood pressure measurements corresponding to office measurements: automated office, home, and a ambulatory blood pressures. J Clin Hypertens. 2023:1–11.
Kim B-K, Rhee M-Y. Impact of 2018 ESC/ESH and 2017 ACC/AHA hypertension guidelines: difference in prevalence of White-Coat and masked hypertension. Healthcare. 2020;8(2):122.
Ohkubo T, Imai Y, Tsuji I, Nagai K, Ito S, Satoh H, et al. Reference values for 24-hour ambulatory blood pressure monitoring based on a prognostic criterion: the Ohasama Study. Hypertension. 1998;32(2):255–9.
Kikuya M, Hansen TW, Thijs L, Björklund-Bodegård K, Kuznetsova T, Ohkubo T, et al. Diagnostic thresholds for ambulatory blood pressure monitoring based on 10-year cardiovascular risk. Circulation. 2007;115(16):2145–52.
Niiranen TJ, Asayama K, Thijs L, Johansson JK, Ohkubo T, Kikuya M, et al. Outcome-driven thresholds for home blood pressure measurement: international database of home blood pressure in relation to cardiovascular outcome. Hypertension. 2013;61(1):27–34.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
EML wrote the draft.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The author has no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Lee, E.M. When and how to use ambulatory blood pressure monitoring and home blood pressure monitoring for managing hypertension. Clin Hypertens 30, 10 (2024). https://doi.org/10.1186/s40885-024-00265-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40885-024-00265-w