Abstract
Background
In this study, the effect of heavy metals accumulation influence was evaluated on adult crayfish Procambarus clarkii (Decapoda, Astacidea) collected from three different Governmental locations (Kafr El-Shaikh, El-Menofya, and El-Gharbiya) of the Egyptian Delta. The activity of super oxidase dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX) of gills, hepatopancreas, and muscle tissue were measured. SDS Polyacrylamide gel electrophoresis (SDS-PAGE) and West blotting technique were performed to detect MT Protein expression.
Results
The results revealed that Kafr El-Shaikh reflected the highest Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) activity levels (97.2 u/100 mg, 28.5 u/100 mg, and 8.3 nmol mg (-1) protein min (-1) respectively. Superior protein polymorphism % (30%) remarked collected Freshwater crayfish P. clarkii from Kafr El-Shaikh location. Varied protein polymorphism % was shown between collected crayfish from El-Menofya, and El-Gharbiya locations (5.5 and 6.2 respectively) Increasing Metallothioneins intensity (15.4%) for collected Freshwater crayfish Procambarus clarkii from Kafr El-Shaikh Location.
Conclusion
Heavy metal stress influences antioxidant status and also induces increasing Metallothioneins intensity, especially samples that were collected from the Kafr El-Shaikh area.
Similar content being viewed by others
Background
The deterioration of the Nile River's water quality is a major issue in Egypt. Until it reaches the Delta, the water quality released from Aswan High Dam (AHD) is relatively clean and displays no degradation [1]. Egypt's main issue is the rise in pollution brought on by the Nile's low water level, particularly after the Ethiopia Dam is completed. Heavy metals are the most prevalent pollutant in the Nile River, with contamination originating from a variety of anthropogenic sources including industrial, agricultural, and home effluents. Heavy metal poisoning of surface water is a serious ecological risk due to its potential toxicity for both humans and the environment. They can bioaccumulate in the food chain since they are not biodegradable [2, 3].
The ability of some animals to accumulate relatively high levels of some contaminants, even from greatly diluted solutions, even without visible impacts, forms the basis for the biomonitoring of pollutants employing accumulator species. When their levels in the natural environment are below the detection limits of the generally employed methods, this methodology enables the measurement of trace element concentrations. Additionally, although the pollutant concentrations in the water only reflect the situation at the time of sampling, the pollutant concentrations in the organism are a product of both past and present pollution levels in the environment in which the organism lives [4].
Crayfish as well as other decapods are known to be vulnerable to pollution in freshwater ecosystems. These organisms are particularly responsive to changes in aquatic environments because of their sensitivity to changes in water quality [5, 6]. Both in the aquatic environment and in the laboratory, crayfish have been utilized as bioindicators. They show a strong preference for collecting contaminants in their tissues [7, 8] and respond to a variety of chemicals [5, 9]. As a result, they have the potential to be used as bioindicators in actual monitoring in industrial applications.
Organisms have developed a multitude of defense systems in response to exposure to free radicals from a variety of sources [10]. Preventive processes, healing mechanisms, physical defenses, and antioxidant defenses are all part of the defense against free radical-induced oxidative stress. Superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase are examples of enzymes that provide antioxidant protection (GPx). Vitamin C, tocopherol, glutathione (GSH), carotenoids, flavonoids, and other antioxidants are examples of non-enzymatic antioxidants. The antioxidant system in the body regulates the production and clearance of reactive oxygen species (ROS) [11,12,13,14].
Superoxide dismutases (SODs) are enzymes produced by organisms that can survive in the presence of oxygen. They help superoxide be transformed into oxygen and hydrogen peroxide through catalysis. SOD enzymes regulate a variety of reactive oxygen species (ROS) and reactive nitrogen species levels through their activity, which limits the potential toxicity of these molecules while also regulating a wide range of cellular processes that are governed by their signaling functions [15]. Furthermore, CAT can efficiently catalyze the breakdown of hydrogen peroxide (H2O2) to maintain the proper ratio between de novo H2O2 synthesis and rapid clearance, which is crucial for innate immunity. Furthermore, as H2O2 is involved in the sensing and signaling of various biological events, the up- or down-regulation of catalase can affect a variety of biological processes, such as growth, differentiation, emigration, and apoptotic [16]. In addition, glutathione S-transferases (GSTs) can reduce lipid hydroperoxides through their Se-independent glutathione-peroxidase activity [17].
Metallothioneins (MTs) are non-enzymatic, low-molecular-weight proteins rich in cysteine, free of aromatic amino acids, and heat stable. Metal stress in invertebrates could be detected using MTs as a biomarker. On the basis of molecular weights, UV absorption spectra, isoelectric points, and amino acid contents, crustacean MTs were shown to be very comparable to vertebrate MTs [18]. The biological activities of MTs are as follows: first, these proteins provide a non-toxic zinc and copper reserve for the creation of metalloenzymes, allowing several cellular processes to maintain homeostasis. Second, MTs can inhibit non-essential metals (Cd, Hg, or Ag) from binding within cells, hence limiting their harmful potential [19].
The current study aimed to evaluate the effect of heavy metals on some tissues of crayfish P. clarkia (as a bioindicator), from three separate government locations (Kafr El-Shaikh, El-Menofya, and El-Gharbiya) of the Egyptian Delta, focusing on the immune and histological response of collected samples.
Results
Oxidative stress markers including Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) are estimated for collected Freshwater crayfish Procambarus clarkii from three locations as shown in Fig. 5 (Kafr El-Shaikh, El-Menofya, and El-Gharbiya) comparing with the reference sample. As shown in Table 1 and Fig. 1, among three locations, Kafr El-Shaikh reflected the highest Significant levels (P < 0.001) of Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) activity (97.2 u/100 mg, 28.5 u/100 mg and 8.3 nmol/mg protein min (−1) respectively. On the contrary, the lowest Oxidative stress markers values were remarked taken crap from El-Gharbiya (85.5 u/100 mg, 24 u/100 mg, and 7.4 nmol /mg protein min (−1) respectively, compared to the control and other locations On the other hand, the lowest level of enzymes were recorded in El-Gharbiya, compared to other locations, but still significsntly higher than the control group readings.
Different organs (muscle, hepatopancreas, and gills) of collected Freshwater crayfish P. clarkii from three locations reflected varied responses against heavy metals stresses. Generally, gills recorded the highest readings among all organs, and showed the highest significant level (P < 0.001) of Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) content in all of the studied areas, compared with control gills as shown in Table 2 and Fig. 2.
Lowest enzyme readings were observed in muscles samples from all studied areas, compared to other organs. Also, lowest muscle enzymes were found in Gharbiya, but on the other hand, still significantly higher than the control readings.
Also, it was found that all organs from Kafr El-Shaikh samples’ showed the highest readings compared to organs from other locations and control group, which emphasize results mentioned in the previous table.
Quantification and fragmentation of total soluble proteins for collected Freshwater crayfish P. clarkii from three locations (Kafr El-Shaikh, El-Menofya, and El-Gharbiya) were performed, and a positive correlation was detected between heavy metal stresses and superior protein content (Table 3 and Fig. 3).
Collected Freshwater crayfish P. clarkii from Kafr El-Shaikh,which was remarked with the highest heavy metals content, showed highly distinguishable protein content (51.63 mg/ml) compared with the control sample (40.56 mg/ml), and compared to other locations. Moderate and low protein content for collected samples from El-Gharbiya and El-Menofya,(48.81 and 44.64 and mg/ml) indicated our hypothesis.
Protein electrophoretic patterns for collected Freshwater crayfish P. clarkii from the three locations were studied to evaluate the heavy metal effect. As shown in Fig. 4 and Table 4, collected samples from Kafr El-Shaikh location reflected superior protein band number (20 bands). On the other hand, the lowest protein bands were observed in depurated Freshwater crayfish P. clarkii (12 bands).
As a direct influence of heavy metals stress, a positive relationship was detected between protein polymorphism % and degree of exposure to heavy metals (Table 4 and Fig. 5). Superior protein polymorphism % (30%) remarked from Kafr El-Shaikh’s samples. Also, varied protein polymorphism % between collected samples from El-Menofya, and El-Gharbiya locations (5.5% and 6.2% respectively) was shown. Furthermore, control samples reflected 100% of genetic similarity.
Metallothioneins, which consider the key role of protein to resist pollutant stresses, are significantly increased in all studied areas, compared to the control samples after exposure to heavy metals stresses (as showed Fig. 6 and Table 5). Increasing Metallothioneins intensity (15.4%) for collected Freshwater crayfish Procambarus clarkii from Kafr El-Shaikh location which remarked with high heavy metals concentrations area indicate its vital role for defense mechanism against heavy metals stresses, as the highest level of metalothioneins was observed in Kafr El-Shaikh (6.14), while the lowest was in El-Menofya (3.19), compared to the control group (1015).
The histological observations on hepatopancreas dissected from different locations and control groups’ samples showed that the group of depurated specimens (control group) had normal hepatopancreas with hepatocytes and sinusoids which were arranged in lobular shape with an asterisk-like appearance lumen. Hepatocytes were polyhedral cells having central spherical nuclei; each lobule had an interlobular connective tissue (Fig. 7A). The effect of heavy metals in freshwater crayfish caused diffuse damage of the hepatopancreas which was characterized by necrosis involving the lobular shape with irregular lumen. In both El-Gharbiya and El-Menofya collected samples, the hepatopancreas displayed necrosis; vaculation and epithelial cells were eroded with vaculation and hemolytic infiltration was observed (Fig. 7B, C) respectively. However, hepatopancreas of collected samples collected from Kafr El-Shaikh showed the same symptoms with rupture epithelial necrosis with a proliferation of nucleolysis and lumen deformation (Fig. 7D).
Photomicrograph of the cross-section of P. clarkii hepatopancreas stained with hematoxylin and eosin stain (H&E) showing: A- normal tissues in the control group. Tubules(T); lumen(l); Intertubular tissue(IT); and central, spherical nuclei(N). B- affected hepatopancreas tubules with heavy metals of collected P. clarkii from El-Gharbia showing irregular lumen (l); the basal lamina was rippled and connective tissue (CT); within the inter-tubular space. C- P. clarkii hepatopancreas from El-Monofia showed, that epithelial cells were eroded with vaculation and hemolytic infiltration was observed (Arrow). D- collected P. clarkii from Kafr- El- Sheikh showing atrophied epithelium(A); necrosis (n); lumen erosion and hyperplasia vaculation (V)
Discussions
Our findings in this manuscript was a continuation on results obtained in a previous research paper, in which heavy metals were measured in Procambarus clarkii tissues in the same studied areas [20]. Our results from this study directly indicated that heavy metals stressed on different oxidative stress markers including Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) for collected Freshwater crayfish P. clarkii from three locations (Kafr El-Shaikh, El-Menofya, and El- Gharbiya). Changing oxidative stress markers of Freshwater crayfish Procambarus clarkii after exposure to heavy metals agreed with Zhang et al. [21] findings. They indicated significant changes in MDA content and antioxidant enzyme activity after Cd exposed P. clarkii to 2.0, 5.0, and 10.0 mg/L Cd for 24, 48, and 72 h. Also, our results came along with Kim et al. [22], who noticed an increase in SOD and GST activity after Daphnia magna exposure to Cd and Pb. In addition, Mi Kim et al. [23] results showed a significant increase in SOD and GST after exposure of copepod Tigriopus japonicus to Cd, Cu, and Zn.
In this study, different organs (Muscle, hepatopancreas, and gills) of collected Freshwater crayfish P. clarkii form three locations reflected varied responses against heavy metals stresses. Generally, gills showed the highest Superoxide dismutase (SOD), Catalase, and Glutathione S-transferase (GST) content compared with control gills, while hepatopancreas was the second highest organ in oxidative enzymes level. These results came side with Stara et al. [24] through recorded dramatic changes for antioxidant enzymes SOD and CAT, for hepatopancreas and muscle, of exposed Red Swamp Crayfish (Procambarus clarkii) to triazine herbicide. Also, our results came along with results of Wang et al. [25], who recorded a significant increase in SOD and GST activity after exposure of freshwater crab Sinopotamon henanense’s gills to Cd. Furthermore, an increase in SOD level in both gills and hepatopancreas of Unio macus was noticed after exposure to Cu [26]. The studies indicated that gill and hepatopancreas tissues are the most sensitive tissues exposed to toxicants. Since gills have a large surface in direct contact with water, they are considered a sensitive organ affected by water quality [27, 28].
It should be noticed that in our previous study [20], results showed that Kafr El-Shaikh governorate recorded the highest accumulation of all measured heavy metals (Cd, Cu, Fe, Pb, and Zn) in 2010, and also was the highest in accumulation of Cu and Pb in samples’ tissues, compared to other studied locations. These results definitely affected the oxidative enzymes measured in this study, as considering our heavy metals scanning findings, tissues of collected crayfish form distinguished Kafr El-Shiakh location were with the highest heavy metals content containing superior oxidative enzymes levels compared with collected tissues from El-Menofya and El-Gharbiya.
These findings agreed with Farombi et al. [29] findings that noticed a significant elevation in SOD activity after exposure to Cu in catfish Clarias garepinus. Also, Paris-Palacios et al. [30] recorded a significant increase in GST in the liver of Rutilus rutilus after Cu exposure.
As a co-factor for a number of enzymes, Cu+2 participates in oxidative stress and mitochondrial morphology with a narrow optimal range between essential and toxic concentrations. Numerous studies in a variety of species have shown that Cu+2 exposure resulted in apoptotic and autophagic cell death because of elevated ROS levels (Wang et al. [31]).
Monitoring and tracking protein profile and content for exposed Freshwater crayfish P. clarkii to increasing heavy metal stress levels revealed remarked variation in total protein content, band number, and polymorphism %.
Our results for a positive correlation between protein fractions and content of P. clarkii and exposure to increasing heavy metals were proved by Mohamed et al. [32]. They indicated that the total protein profile of Artemia salina tissues increased after treatment with Cd and Fe.
Cellular defense against metal toxicity is linked to the sequestration function of MT in the presence of toxic metals [33]. Heavy metals like zinc (Zn), copper (Cu), and cadmium (Cd) cause the promotion of MT in aquatic species when used in experiments [34]. These results have made MT a potentially useful biomarker for metal exposure. However, MT induction in aquatic species brought on by ambient exposure to heavy metals is more difficult to detect in field investigations, because environmental variables are less well characterized than in laboratory research [35]. Choosing a suitable organism to serve as a bioindicator is crucial [36].
Our results came along with Matin et al. [37], who found significant correlations between heavy metal levels and MT in hermit crab Clibanarius signatus. Similar results were showed by Hertika et al. [38], who stated that levels of MT among Crassostrea iredalei and Crassostrea glomerata, but always higher in gills than stomach. More support was added to our findings for increasing metallothioneins intensity after exposure to heavy metals by Viarengo and Nott [39], and Roesijadi [40]. They cleared that metallothioneins protein over expression in highly polluted sites may be related to its function for resistant pollutants via two main mechanisms. Firstly, preventing toxic potentials of non-essential metals through restriction of their binding to cells. Additionally, by providing a non-toxic zinc and copper pool for metalloenzyme production, the homeostasis of numerous cellular activities is enabled.
As mentioned before, Cu and Pb accumulation was the highest heavy metals in Kafr El-Shaikh governorate, compared to other locations. It was found that Cu is important for MT biosynthesis due to significant correlation more than Ni, Cr, Cd and Hg. A possible justification for the findings is related to that Cu is an essential trace metals and MT play a major role in its hemostasis in crabs [36]. Also, Cu was reported to promote MT synthesis in different tissues, such as digestive gland and gills of mussels [41]. In addition, the analysis of the relationship between MT values and heavy metals level in a very recent study, at 2023, showed the highly significant effect of lead concentration on MT concentrations on Sulcospira testudinaria [42].
Remarked variation for protein content of P. clarkii could be explained by heavy metal exposure in the light of Filipovic and Raspor’s [43] results. For lipoprotein cell repair, they described protein variation as a result of tissue organization, in particular cellular components of cytoplasmic cell membranes and organelles following exposure to pollutants.
Expressing new protein bands with low molecular weight (8 to 4.5 KDa) after exposure of P. clarkii to high heavy metal stresses in Kafr El-Sheikh location could be explained via Seebaugh and Wallace [44]. They fragmented metal-binding proteins (MTs) which stimulate Ubiquitin (Ub) /proteasome-dependent proteolytic systems that break down proteins for decreasing muscle mass which enables claw in land crab, Gecarcinus lateralis, and American lobster to the adapted withdrawal of the appendage at ecdysis. Moreover, Mohamed et al. [32] add more support for expressing new low molecular weight protein bands after exposure to heavy metals. They cleared that ubiquitin considers a low molecular weight protein (8.5 kDa) that is only expressed in Artemia sp. under stress, like inflammation or heavy metals exposure.
In comparison to the control, our findings revealed that protein polymorphism was present in every area of study. Kafr El-Shaikh governorate has the most polymorphism found. These findings corroborated those of David et al. [45], who discovered polymorphism of the MT gene as a result of the stress of summer mortality. Additionally, the Pacific oyster Crassostrea gigas was shown to have an MT polymorphism, which may be connected to its higher resistance to heavy metals [46].
Hepatopancreas is considered the primary organ for the generation of enzymes for metabolism, the contamination of heavy metals in the surrounding medium had a marked effect on the hepatopancreas leading to its damage [47]. The findings of this study demonstrated that the hepatopancreas of crayfish suffered substantial structural damage as a result of accumulating distribution levels. Non-essential elements have been shown in other investigations [26, 48] to accumulate in crayfish tissues as environmental concentration and exposure time increase.
Conclusion
The results showed a clear effect of heavy metals accumulation on the Crayfish P. clarkii collected from three locations (Kafr El-Shaikh, El-Menofya, and El- Gharbiya), of the level of oxidative stress enzymes, and it also showed a remarked variation in total protein content, band number, and polymorphism %, in addition to a damaging effect on hepatopancreas compared to the depurated samples as control. The samples that were collected from the Kafr El-Shaikh area are considered the most affected by pollution, as the results showed the increasing Metallothioneins intensity (15.4%) which remarked with high heavy metals concentrations area.
These results indicated the vital role of MT as biomarker monitoring toxins that aquatic animals could be vulnerable to. Also, this work may highlights that accumulation of heavy metals could cause a genetic adaptation in some immune-related proteins as Metalothioneins, as a way of adaptation towards these contaminants.
Material and methods
Materials and methods
Sample collection
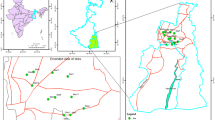
Freshwater crayfish Procambarus clarkii samples were gathered in the spring from three Egyptian Delta’s governments: El-Gharbiya (30.824722°N30.815278°E), El-Menofya (30.52°N30.99°E), and Kafr El-Shaikh (31°06′42″N 30°56′45″E), as shown in Fig. 8 cited by Negm et al. [49].
Fifty samples from each area were collected, cleaned, and brought to a lab at the University of Tanta in plastic bags in an icebox.A group of Samples from each was directly dissected to obtain the Gills, abdominal muscles, and hepatopancreas then preserved (-20 ◦C) for enzymes and protein extraction. Another group of 40 samples was kept in the lab for three weeks (21 days) in glass tanks (n = 10/tank). The water was switched out every two days, and each tank was then filled with dechlorinated, well-aerated stored freshwater to a specific capacity. In the climate-controlled laboratory, an air conditioner was installed and set at 25 ◦C. The P. clarkii specimens underwent depuration for 21 days (Control group) before being dissected.
Oxidative enzymes
Superoxide dismutase (SOD), Catalase (CAT), and Glutathione S-transferase (GST) estimation for Muscle, hepatopancreas, and Gills from Kafr El-Shaikh, El-Menofya, and El-Gharbiya locations, Superoxide Dismutase Activity Assay Kit (Colorimetric) (ab65354, Abcam), Catalase Activity Assay Kit (Colorimetric) (ab83464, Abcam) and GST Activity Assay Kit (Colorimetric) (ab65326) were used according to manufacturer protein.
Protein extraction and purification methods
In this investigation, SDS Polyacrylamide gel electrophoresis (SDS-PAGE) with 12% T was applied as a biochemical marker to evaluate heavy metals toxicity for control and collected samples from Kafr El-Shaikh, El-Menofya, and El- Gharbiya locations according to the methods of Lammli [50].
TriFast (Peqlab, VWR company) (Isolation of RNA, DNA and Protein simultaneously).
Homogenization
In 1 ml TriFast, 50–100 mg of tissue or pellet was added. For efficient lysis use a glass- Teflon or power homogenizer. The sample volume should not exceed 10% of the volume of TriFast used for the homogenization.
Phase separation
RNA is forced exclusively into the aqueous phase whereas DNA and the proteins partition into the interphase and lower phenol phase. The volume of the RNA-containing phase is about 60% of the volume of the TriFast used for homogenization.
RNA precipitation
Precipitate the RNA with 0.5 ml of isopropanol per 1 ml of TriFast used for the initial homogenization. Keep samples on ice for 5–15 min and centrifuge for 10 min at 4 °C at 12,000 × g max. The RNA pellet should form a gel like precipitate on the bottom and on the side of the tube.
DNA precipitation
DNA is sedimented by centrifugation at 2,000 × g at 4 °C for 15 min. (Careful removal of the aqueous phase is critical for the quality of the isolated DNA).
Protein precipitation
Precipitate the proteins from the ethanol/phenol supernatant with 1.5 ml Isopropanol. Keep the samples in washing solution for 20 min at room temperature before centrifuging at 7,500 × g for 5 min at 4 °C. Next vortex the protein pellet once with 2 ml of 100% ethanol, store for 20 min at room temperature and centrifuge at 7,500 × g for 5 min at 4 °C.
Protein solubilization
Remove any ethanol and dry the Protein pellet briefly for 5–10 min under vacuum and dissolve it in 1% SDS by pipetting it up and down. Incubation of the samples at higher temperatures (50–100 °C) may be necessary to yield complete solubilization.
Protein fraction quantification
Based on the interaction of the protein with Coomassie Brilliant Bleu G250 (CBBG-250) under acidic conditions, the protein fraction concentration was calculated using Bradford's [51] technique. According to Bamdad et al. [52], 12% slab gel was used.
After gel polymerization, 30 g of proteins were added to each sample, and electrophoresis was run at 75 V through a stacking gel for roughly 2 h.
Metallothionein (MT) Protein expression
GE Healthcare's HybondTM nylon membrane was used to electrophorese proteins on SDS-PAGE. The membrane was then treated overnight at 4°C with an antibody solution comprising the primary antibody Anti-Anti Metallothionein (Abcam, ab192385). Also, Anti- β-actin (Abcam, ab8226) was used as a housekeeping gene [53,54,55].
-
Membrane was washed at room temperature for 30–60 min with 5 or more changes of Blotting Buffer.
-
Membrane was incubated for 1 h at room temperature in an Antibody Solution containing appropriate dilution of HRP-conjugated secondary antibody (Antibody concentration. 0.1–0.5 µg/mL. Adjust antibody concentration from 0.05 to 2.0 µg/mL to obtain desired signal strength and low background.
-
Membrane was washed for 30–60 min with 5 or more changes of Blotting Buffer.
Light microscopic preparations
Dissected Hepatopancreas of the collected and control (14 days depurated samples) Samples were preserved in 10% formalin and dehydrated via an ascending series of ethanol, embedded in paraffin wax, sectioned at 4–6 μm thick, and stained with hematoxylin and eosin. An Olympus (E330-ADU1.2X) microscope with a digital camera was used to take photomicrographs Image-Pro Express software (Media Cybernetics).
Data analysis
Statistical analysis was performed by SPSS version 9.0 software. The enzymes differences between the control and the locations groups were analyzed by one-way analysis of variance (ANOVA). A value of P < 0.05 was accepted as statically significant. All data are expressed as the mean ± standard deviation (SD). Gel documentation system (Geldoc-it, UVP, England), was applied for data analysis using Totallab analysis software, ww.totallab.com, (Ver.1.0.1).
Availability of data and materials
The datasets used and analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- Ag:
-
Chemical symbol for Silver
- AHD:
-
Aswan High Dam
- ANOVA:
-
One way analysis of variance
- BSA:
-
Bovine Serum Albumin
- CAT:
-
Catalyse
- Cd:
-
Chemical symbol for Cadmium
- Cr:
-
Chemical symbol for Chromium
- Cu:
-
Chemical symbol for Copper
- DNA:
-
Deoxyribonucleic acid
- Fe:
-
Chemical symbol for Iron
- GPX:
-
Glutathione peroxidase
- GSH:
-
Glutathione
- GSTs:
-
Glutathione S-Transferases
- H2O2 :
-
Hydrogen peroxide
- Hepato:
-
Hepatopancreas
- Hg:
-
Chemical symbol for Mercury
- IACUC:
-
Institutional Animal Care and Use Committee
- KDa:
-
KiloDalton
- MTs:
-
Metallothioneins
- n:
-
Number
- Ni:
-
Chemical symbol for Nickel
- OD:
-
Optical Denisty
- P. clarkii:
-
Procambarus clarkia
- Pb:
-
Chemical symbol for Lead
- REC:
-
Research ethical Committee
- RNA:
-
Ribonucleic acid
- ROS:
-
Reactive Oxygen Species
- SD:
-
Standard Deviation
- SDS:
-
Sodium dodecyl-sulfate polyacrylamide gel electrophoresis
- SOD:
-
Super Oxide Dismutase
- SPSS:
-
Statistical Package for the Social Sciences
- Zn:
-
Chemical symbol for Zinc
References
Masoud MS, Elewa AA, Abdel-Halim AM. Some environmental analysis on Nile water. J Saudi Chem Soc. 2002;6(3):377–98.
Ezzat SM, Mahdy HM, Abo-State MA, Abd El Shakour EH, El-Bahnasawy MA. Water quality assessment of River Nileat Rosetta Branch: impact of drains discharge. MEJSR. 2012;12(4):413–23.
Goher ME, Hassan AM, Abdel-Moniem IA, Fahmy AH, ElSayed SM. Evaluation of surface water quality and heavy metal indices of Ismailia Canal, Nile River. Egypt Egypt J Aquat Res. 2014;40:225–33.
Ravera RC, Beone GM, Dantas M, Lodigiani P. Trace element concentrations in freshwater mussels and macrophytes as related to those in their environment. J Limnol. 2003;62(1):61–70.
Soedarini B, Klaver L, Roessink I, Widianarko B, van Straalen NM, van Gestel CAM. Copper kinetics and internal distribution in the marbled crayfish (Procambarus sp.). Chemosphere. 2012;87:333–8.
Pennuto CM, Lane OP, Evers DC, Taylor RJ, Loukmas J. Mercury in the northern crayfish, Orconectes virilis (Hagen), in New England, USA. Ecotoxicology. 2005;14:149–62 ([CrossRef] [PubMed]).
Alcorlo P, Otero M, Crehuet M, Baltanas A, Montes C. The use of the red swamp crayfish (Procambarus clarkii, Girard) as indicator of the bioavailability of heavy metals in environmental monitoring in the River Guadiamar (SW, Spain). Sci Total Environ. 2006;366:380–90 ([CrossRef] [PubMed]).
Kuklina I, Sladkova S, Kouba A, Kholodkevich S, Kozak P, Investigation of chloramine-T impact on crayfish Astacusleptodactylus (Esch. cardiac activity. Environ Sci Pollut Res. 1823;2014(21):10262–9 ([CrossRef] [PubMed]).
Nathaniel TI, Huber R, Panksepp J. Repeated cocaine treatments induce distinct locomotor effects in Crayfish. Brain Res Bull. 2012;87:328–33 ([CrossRef] [PubMed]).
Cadenas E. Production of superoxide radicals and hydrogen peroxide in Mitochondria. Mol Aspects Med. 1997;25(1):17–26.
Xu DD, Liu XH, Cao JM, Du ZY, Huang YY, Zhao HX, Zhu X. Dietary glutathione as an antioxidant improves resistance to ammonia exposure in Litopenaeus vannamei. Aquac Res. 2012;43:311–6. https://doi.org/10.1111/j.1365-2109.2011.02820.x.
Chen M, Chen XQ, Tian LX, Liu YJ, Niu J. Enhanced intestinal health, immune responses and ammonia resistance in pacific white shrimp (Litopenaeus vannamei) fed dietary hydrolyzed yeast (Rhodotorula mucilaginosa) and Bacillus licheniformis. Aquaculture Reports. 2020;17:10385. https://doi.org/10.1016/j.aqrep.2020.100385.
Ebadi H, Zakeri M, Mousavi SM, Yavari V, Souri M. The interaction effects of dietary lipid, vitamin E and vitamin C on growth performance, feed utilization, muscle proximate composition and antioxidant enzyme activity of white leg shrimp (Litopenaeus vannamei). Aquaculture Res. 2020;1–13. https://doi.org/10.1111/are.1505.
Yu Y, Liu Y, Yin P, Zhou W, Tian L, Liu Y, Xu D, Niu J. Astaxanthin attenuates fish oil-related hepatotoxicity and oxidative insult in juvenile Pacific white shrimp (Litopenaeus vannamei). Mar Drugs. 2020;18(4):218. https://doi.org/10.3390/md18040218.
Wang Y, Branicky R, Noe A, Hekimi S. Superoxide dimutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 2013;217(6):1915–28.
Wang C, Yue X, Lu X, Liu B. The role of catalase in the immune response to oxidative stress and pathogen challenge in the clam Meretrix meretrix. Fish Shellfish Immunol. 2013;34:91–9.
Singhal SS, Singh SP, Singhal P, Horne D, Singhal J, Awasthi S. Antioxidant role of glutathione S-transferases: 4-Hydroxynonenal, a key molecule in stress-mediated signaling. Toxicol Appl Pharmacol. 2015;289(3):361–70.
Olafson RW, Sim RG, Boto KG. Isolation and chemical characterization of the heavy metal-binding protein metallothionein from marine invertebrates. Comp Biochem Physiol. 1979;62(4):407–16.
Zaroogian G, Jackim E. In vivo metallothionein and glutathione status in an acute response to cadmium in Mercenaria mercenaria brown cells. Comparative Biochem Physiol C. 2000;127:251–61.
Younis ML, Mona MM. Rizk E-ST, and Atlam AI Monitoring changes in accumulation of heavy metals in the Egyptian Delta region in 2010 and 2021, using the crayfish Procambarus clarkii (Girard, 1852) (Decapoda, Astacidea) as bio-indicator. Crustaceana. 2022;95(7):763–78.
Zhang Y, Li Z, Kholodkevich S, Sharov A, Feng Y, Ren N, Sun K. Cadmium-induced oxidative stress, histopathology, and transcriptome changes in the hepatopancreas of freshwater crayfish (Procambarus clarkii). Sci Total Environ. 2019;20(666):944–55. https://doi.org/10.1016/j.scitotenv.02.159.
Kim H, Kim J, Kim P, Won E, Lee Y. Response of antioxidant enzymes to Cd and Pb exposure in water flea Daphnia magna: Differential metal and age specific patterns. Comparative Biochem Physiology. 2018;C209:28–36.
Kim B, Rhee J, Jeong C, Seo J, Park G, Lee Y, Lee J. Heavy metals induce oxidative stress and trigger oxidative stress-mediated heat shock protein (hsp) modulation innthe intertidal copepod Tigriopus japonicas. Comparative Biochemistry Physiol. 2014;c166:65–74.
Stará A, Kouba A, Velíšek J. Effect of Chronic Exposure to Prometryne on Oxidative Stress and Antioxidant Response in Red Swamp Crayfish (Procambarus clarkii)". BioMed Res Int. 2014;2014(Article ID 680131):6.
Wang J, Zhang P, Shen Q, Wang Q, Liu D, Li J, Wang L. The effects of cadmium exposure on the oxidative state and cell death in the gill of freshwater crab Sinopotamon henanense. PLoS ONE. 2013;8(5):e64020. https://doi.org/10.1371/journal.
Pacal E, Gumus BA, Gunal C, Erkmen B, Arslan P, Yildirim Z, Erkoc F. Oxidative stress response as biomarker of exposure of a freshwater invertebrate model organism (Unio mancus Lamark, 1819) to antifouling copper pyrithione. Pestic Phytomed (Belgrade). 2022;37(2):63–76.
Aarab N, Godal BF, Bechmann RK. Seasonal variation of histopathological and histochemical markers of PAH exposure in blue mussel (Mytilus edulis L.). Marine Environ Res. 2011;71(3):213–7. https://doi.org/10.1016/j.marenvres.2011.01.005.
Pinto J, Costa M, Leite C, Borges C, Coppola F, Henriques B, Freitas R. Ecotoxicological effects of lanthanum in Mytilus galloprovincialis: Biochemical and histopathological impacts. Aquat Toxicol. 2019;211:181–92. https://doi.org/10.1016/j.aquatox.2019.03.017.
Farombi EO, Adelowo OA, Ajimoko YR. Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in African cat fish (Clarias gariepinus) from Nigeria Ogun River. Int J Environ Res Public Health. 2007;4:158–65.
Paris-Palacios S, Biagianti-Risbourg S, Vernet G. Metallothionein analyzed in liver of Rutilus rutilus exposed to Cu with three methods: metal summation, SH determination and original spectrofluorimetric method. Comp Biochem Physiol. 2000;C 126:122–33.
Wang Y, Zhao H, Shao Y, Liu J, Li J, Luo L, Xing M. Copper (II) and/or arsenite-induced oxidative stress cascades apoptosis and autophagy in the skeletal muscles of chicken. Chemosphere. 2018;206:597–605. https://doi.org/10.1016/j.chemosphere.2018.05.013.
Mohamed AH, Sheir SK, Osman GY, Abd-El Azeem HH. Toxic effects of heavy metals pollution on biochemical activities of the adult brine shrimp. Artemia salina Can J Pure App Sci. 2014;8:3019–28.
Roesijadi G. Metallothioneins in metal regulation and toxicity in aquatic animals. Aquat Toxicol. 1992;22(2):81–113.
Linde AR, Garcia-Vazquez E. A simple assay to quantify metallothionein helps to learn about bioindicators and environmental health. Biochem Mol Biol Educ. 2006;34(5):360–3.
Linde AR, Sánchez-Galán S, Vallés-Mota P, García-Vázquez E. Metallothionein as bioindicator of freshwater metal pollution: European eel and brown trout. Ecotoxicol Environ Saf. 2001;49(1):60–3.
Chen Z, Eaton B, Davies J. The Appropriateness of Using Aquatic Snails as Bioindicators of Toxicity for Oil Sands Process-Affected Water. Pollutants. 2021;1(1):10–7.
Matin MT, Mashinchain A, Sinaei M, Jamili S. Metallothionein as biomarker of heavy metal (Cd, Cu, Zn, Pb, Hg, Ni, Cr) pollution in hermit crab (Clibanarius signatus). EnvironmentAsia. 2019;12(2):164–71.
Hertika AMS, Kusriani K, Indrayani E, Nurdiani R, Putra R. Relationship between levels of the heavy metals lead, cadmium and mercury, and glomerata. F1000Research. 2018;7:1239.
Viarengo A, Nott JA. Mechanisms of heavy metal cation homeostasis in marine invertebrates. Comparative Biochem Physiol C. 1993;104(3):355–72.
Roesijadi G. Metallothionein and its role in toxicmetal regulation. Comparative Biochem Physiol C. 1996;113(2):117–23.
Geret F, Cosson RP. Induction of specific isoforms of metallothionein in mussel tissues after exposure to cadmium or mercury. Arch Environ Contam Toxicol. 2002;42(1):36–42.
Hertika AMS, Arfiati D, Lusiana ED, Putra R. Performance of metallothionein biomarker from Sulcospira testudinaria to assess heavy metal pollution in the Brantas Riverwatershed Indonesia. J Ecol Eng. 2023;24(3):276–86.
Filipovic V, Raspor B. Metallothionein and metal levels in cytosol of liver, kidney, and brain in relation to growth parameters of Mullus surmuletus and Liza aurata. From the eastern Adriatic Sea. Water Res. 2003;37(13):3253–62. https://doi.org/10.1016/S0043-1354(03)00162-3.
Seebaugh D, William W. Importance of metal-binding proteins in the partitioning of Cd and Zn as trophically available metal (TAM) in the brine shrimp Artemia franciscana. Marine Ecol Progress Series. 2004;272:215–30. https://doi.org/10.3354/meps272215.
David E, Tanguy A, Moraga D. Characterization and genetic polymorphism of metallothionein gene CgMT4 in experimental families of Pacific oyster Crassostrea gigas displaying summer mortality. Biomarkers. 2012;17(1):85–95.
Tanguy A, Boutet I, Bonhomme F, Boudry P, Moraga D. Polymorphism of metallothionein genes in the Pacific oyster Crassostrea gigas as a biomarker of response to metal exposure. 2002.
Padmanaban AM, Mohan K. Tissue histopathology of freshwater crab Paratelphusa Hydrodromous under cadmium chloride toxicity. Int J Universal Pharm Bio Sci. 2013;2(4):109–17.
Suàrez-Serrano A, Alcaraz C, Ibanez C, Troajo R, Barata C. Procambarus clark as bioindicator of heavy metal pollution sources in the lower Ebro River and Delta. Ecotoxicol Environ Saf. 2010;73:280–6.
Negm AM, Sakr S, Abd-Elaty I, Abd-Elhamid HF. An Overview of Groundwater Resources in Nile Delta Aquifer. Groundw. Nile Delta. 2018;73:3–44. [Google Scholar] [CrossRef].
Lammli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680–5. https://doi.org/10.1038/227680a0. (PMID: 5432063).
Bradford MM. Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal Biochem. 1976;72:248–54. https://doi.org/10.1016/0003-2697(76)90527-3.
Bamdad F, Dokhani S, Keramat J. Functional Assessment and Subunit Constitution of Lentil (Lens culinaris) Proteins during Germination. Int J Agric Biol. 2009;11(6):690–4.
Malik VS, Lillehoj P. Antibody Techniques. San Diego - New York - Boston - London - Sydney - Tokyo - Toronto: Academic Press; 1994. p. 353.
Pound JD. Immunochemical Protocols. 2nd ed. Humana Press; 1998. p. 207–216.
Harlow Ed, Lane D. Using Antibodies, A Laboratory Manual. A Cold Spring Harbor Laboratory Press; 1999. p. 267–309.
Acknowledgements
Thanks to all authors Assistant Professor Mahy M. Mona, Dr. Mai L. Younis, and Dr. Aalaa I. Atlam for collecting samples, dissecting, methodology, and writing the original draft, reviewing, and editing.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This work was all self-funded by the authors.
Author information
Authors and Affiliations
Contributions
The authors confirm contribution to the paper as follows: study conception and design: MMM, MLY; data collection: MMM, AIA, MLY; analysis and interpretation of results: MMM, AIA; figures preparation: AIA; draft manuscript preparation: MMM, MLY. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental protocols were approved by the Research ethical committee (REC), and The Institutional Animal Care and Use Committee (IACUC), Faculty of Science, Tanta University, Tanta, Egypt. Reference Number: IACUC-SCI-TU-0371. All methods were performed in accordance with the relevant guidelines and regulations, as stated in the ethical approval.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Mona, M.M., Younis, M.L. & Atlam, A.I. Evaluation of freshwater heavy metals accumulation effect on oxidative stress, Metallothionein biosynthesis and histopathology of Procambarus clarkii (Girard,1985) collected from three locations in the Delta region, Egypt. BMC Zool 8, 21 (2023). https://doi.org/10.1186/s40850-023-00183-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40850-023-00183-8