Abstract
Introduction
Multiple gallbladders represent a rare congenital disorder, and coexistence with carcinoma is extremely rare, leading to a high possibility of misdiagnosis and surgical complications. In this study, a case was reported and the literature was reviewed.
Case presentation
An 80-year-old woman was diagnosed with acute cholecystitis via ultrasonography and was successfully treated with antibiotics. After the patient’s biliary colic relapsed, she was referred to our hospital. Multiple imaging modalities revealed duplication of her gallbladder (H-type) and suggested coexistence with carcinoma. According to preoperative evaluations, we assumed the patient had stage IIIA disease, and cholecystectomy, cholangiography using a near-infrared ray vision system, and sectionectomy of segments 4a and 5 were performed. Contrary to the high standardized uptake values obtained by 18F-fluoro-2-deoxy-D-glucose positron emission tomography, gallbladder carcinoma was pathologically diagnosed as stage 0 mucosal cancer. Seven days after the operation, portal thrombosis of the posterior branch was revealed, and conservative therapy was indicated; satisfactory results were achieved. The patient was discharged 65 days after surgery. No recurrence was observed for 1 year after surgery.
Conclusions
An extremely rare case of malignancy in a duplicated gallbladder was reported, and the literature was reviewed. Accurate estimations are feasible for diagnoses of multiple gallbladders, where correct evaluations are vital, especially in malignant cases. Because of the possibility of malignancy, resected accessory gallbladders should be scrutinized pathologically.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Multiple gallbladders (MGs) represent a rare congenital disorder, and coexistence with carcinoma is even more rare, resulting in a high risk of misdiagnosis and surgical complications. Although cases of MGs have been reported in 1 per 4000 autopsies [1], they are rarely recognized clinically. Boyden’s classification is the most widely accepted classification. He classifies double gallbladders into two types. Based on their relationship to the cystic duct, he describes “vesica fellea divisa” (a bilobed gallbladder that has one cystic duct) and “vesica fellea duplex” (true gallbladder duplication). True duplication is further subclassified into the “Y-shaped type” (two cystic ducts uniting before entering the common bile duct) and the “H-shaped or ductular type” (two cystic ducts enter individually into the common bile duct) [1]. Studies on MGs were identified in PubMed by searching for the words “multiple gallbladders,” “duplicated gallbladder,” “duplication of gallbladder,” “duplicate gallbladder,” and “double gallbladder” corresponding to the definition of true duplication according to Boyden’s classification. Furthermore, only five cases of gallbladder malignancy with MGs, except for MGs in a broad sense, have been reported in the English literature [2,3,4,5,6]; little is known about MGs associated with carcinoma. We herein report a rare case of MGs associated with carcinoma.
Case presentation
An 80-year-old woman was admitted to her home doctor’s practice with abdominal pain and fever. She was diagnosed with acute cholecystitis via ultrasonography (US) and was successfully treated with antibiotics. When biliary colic relapsed, the patient was referred to our hospital. She had suffered from hypertension for 10 years. There were no special notes in her family history or from her relevant physical examination, nor were there any other significant clinical findings.
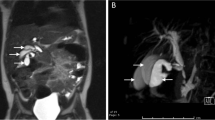
First, images obtained by US showed gallbladder duplication and an elevated lesion with a diameter of 1 cm in the proximal portion, which was identified as an accessory gallbladder (Fig. 1). An abdominal computed tomography (CT) scan with enhancement revealed a low-density mass with early enhancement in the proximal gallbladder. Direct liver invasion was less conspicuous on CT imaging (Fig. 2). Magnetic resonance cholangiopancreatography (MRCP) revealed that both cystic ducts had diverged from the common bile duct individually, so an H-type duplicated gallbladder was considered according to Boyden’s classification (Fig. 3). Endoscopic ultrasonography (EUS) indicated a hypoechoic lesion with a ragged edge, and an increased central area signified penetration into the hepatic parenchyma through the gallbladder bed (Fig. 4). The maximum standardized uptake value (SUV) from 18F-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) was revealed as 5.97 at the accessory gallbladder. No other metastases were identified in any of the images (Fig. 5). The patient’s laboratory tests revealed a normal complete blood count and mild liver dysfunction with an indocyanine green plasma disappearance rate of 15% at 15 min. The serum carcinoembryonic antigen (CEA) level, carbohydrate antigen 19-9 (CA19-9) level, and serum SPan-1 antigen level were 2.2 ng/mL (normal range, < 5 ng/mL), 35.6 U/mL (normal range, < 37 U/mL), and 21 U/mL (normal range, < 30 U/mL), respectively. Consequently, the patient was diagnosed with advanced gallbladder carcinoma of the accessory gallbladder (H-type), stage IIIA (T3, N0, M0) according to the 8th edition of the UICC TNM classification of malignant tumors [7]. The total liver volume (TLV) was estimated to be 832 mL by three-dimensional volumetric simulation software (Synapse Vincent®; Fujifilm, Tokyo, Japan), and the volume for the limited resection of segments 4a and 5 was estimated to be 93 mL, which was equivalent to 11.2% of the TLV. A double gallbladder was clearly identified macroscopically after laparotomy (Fig. 6). Intraoperative cholangiography was performed in the primary cystic duct with a near-infrared ray vision system (Photo Dynamic Eye®: PDE, HAMAMATSU Photonics, Shizuoka, Japan), which revealed a pair of cystic ducts that had been diagnosed preoperatively (Fig. 7). Intraoperative frozen-section examination of the accessory cystic duct was submitted. Although radical dissection of the lymph nodes in the hepatoduodenal ligament, including the no. 13 lymph nodes on the posterior surface of the pancreatic head, was performed during the examination while preserving the biliary tract using a near-infrared ray vision system, no enlarged lymph nodes were found. As several studies have addressed, a near-infrared ray vision system can be useful for visualizing the extrahepatic biliary anatomy during hepatobiliary surgery. The pathological report from the frozen specimen proved negative for carcinoma at the surgical margin, so sectionectomy of S4a and 5 with MGs was performed. The operation time was 325 min, and the intraoperative blood loss was 550 mL. No blood transfusion was needed. Examination of the resected specimen showed that the gallbladder wall was thin, and the demarcation between the wall and the mass was clear. The mass was pedunculated with a fine stalk, and there was space between the mass and the gallbladder wall. Pathological examination of the surgical specimen revealed biliary type adenocarcinoma (WHO); papillary adenocarcinoma; papillary expanding type (9 × 7 mm); pTis (M); int, ly0; v0; ne0; pEM0; pCM0; pPV0; pA0; pR0; and pN0 (0/7). The disease progressed through stages Tis, N0, and M0 and ultimately to stage 0 (according to the 8th edition of the UICC TNM classification of malignant tumors [7]) (Fig. 8). Seven days after the operation, portal thrombosis of the posterior branch was revealed, and conservative therapy was indicated; satisfactory results were achieved. The patient was discharged 65 days after surgery. No recurrence was observed for 1 year after surgery.
Abdominal enhanced CT imaging. Carcinoma in the proximal gallbladder, which was identified as the accessory gallbladder, shown by enhanced CT images. a Axial view. The low-density mass with a diameter of 1 cm was slightly enhanced (see the arrows). Direct liver invasion was less conspicuous on CT imaging. b Coronal view, and c sagittal view. Two separate cavities were clearly demonstrated
Intraoperative cholangiography. a Normal cholangiogram via iodine contrast agent. b, c Biliary images via a near-infrared ray vision system (Photo Dynamic Eye®: PDE). An intraoperative cholangiogram through the primary cystic duct showed the correct ductal anatomy. A pair of cystic ducts (indicated by arrows) were demonstrated
Macroscopic findings and histological findings of resected specimen. a Resected fresh specimen, proximal gallbladder identified as an accessory gallbladder containing carcinoma (single arrow), and distal gallbladder identified as a primary gallbladder (double arrows). b Formalin-fixed specimen, carcinoma in the proximal gallbladder identified as an accessory gallbladder (arrow). c, d Hematoxylin and eosin (H&E) staining at original magnification (× 20 and × 100). Pedunculated and non-invasive mucosal carcinoma had a fine stalk, and there was space between the mass and the gallbladder wall
Discussion
Cases of MGs are reported in 1 per 4000 autopsies [1], and anatomical recognition of the type of MGs is essential. Multiple imaging modalities, such as US, CT, MRCP, and endoscopic retrograde cholangiopancreatography (ERCP), can correctly diagnose MGs. Darnis et al. reviewed data from 181 patients extracted from 153 studies published between 1990 and 2017 [8]. They found that MGs were diagnosed prior to surgery in 76% of patients. Ozaki et al. claimed that the definitive diagnosis of a duplicated gallbladder could only be made intraoperatively [9]. Anatomical recognition resulted in the correct surgical procedure without complications such as bile duct injury or leakage.
Raymond and Thrift reported the first case in 1956 [2], and only five cases of gallbladder malignancy associated with MGs, except for MGs in a broad sense, have been reported in the English literature. These five reports and our case are summarized in Table 1. Roeder et al. reported triplication of the gallbladder with papillary adenocarcinoma in 1971 [3]. They removed the two gallbladders identified intraoperatively, and the third gallbladder was detected on the fourteenth postoperative day via T tube cholangiography. Kin et al. reported a case of gallbladder duplication with advanced adenocarcinoma in 1996 [4]. They noted that magnetic resonance imaging (MRI) was the most useful modality in revealing the cystic formation of the accessory gallbladder with packed stones in addition to the main gallbladder. In addition, celiac angiography demonstrated the unusual form of the intrahepatic artery that enveloped the cystic tumor, and they noted that it might be a diagnostic characteristic of accessory gallbladders. Kawanishi et al. reported carcinoma visualized with abnormal accumulation by FDG-PET [5]. Well-differentiated tubular adenocarcinoma with infiltration into the submucosal layer was observed in the resected accessory gallbladder. Recently, Chen et al. reported a case of adenosquamous carcinoma in 2018 [6]. In their case, cholangiocarcinoma coexisted at the end of the common bile duct. Safioleas et al. stated that the incidence of gallstones in MG was similar to that in single gallbladders [10]. Additionally, Darnis et al. and Hurst et al. stated that there was no evidence of an increased risk for malignancy in MGs [8, 11]. However, as Kin et al. pointed out [4], attention must be paid to the fact that carcinoma in those 5 cases and in our case arose only from the accessory gallbladder. Although a higher frequency of malignancy in MGs that in single gallbladders has not been confirmed, resected accessory gallbladders should be scrutinized pathologically.
Recently, diagnostic precision has been improved by EUS, which has been suggested to be accurate for the clinical staging of gallbladder carcinoma [12,13,14]. However, we incorrectly interpreted our EUS findings to be reflective of advanced carcinoma.
The SUV max evaluated by FDG-PET was 5.97 in our case, which was supposed to be indicative of malignancy; the patient’s past history of cholecystitis was taken into account because an experimental animal model indicated that the SUV max would be normalized within 1 week of the onset of acute inflammation [15]. However, in our case, the serum C-reactive protein level was almost in the normal range on the day of the FDG-PET examination. In the case of Kawanishi et al, the SUV max evaluated by FDG-PET was 4.8, despite the presence of submucosal carcinoma [5]. Although we recognized the importance of FDG-PET, we could not reconcile the differences in these results. It is reasonable to speculate that remnants of inflammation or tumor volume but not a high level of malignancy affect the SUV max as evaluated by FDG-PET.
In the extremely rare case of malignancy, correct cancer staging is critical. This case presented as stage 0 pathologically but was overdiagnosed as clinical stage IIIA based on an incorrect interpretation of the EUS findings and an equivocal SUV max that was probably affected by previous inflammation. In cases of MGs with malignancy, attention should be paid to accurate staging in cholecystitis patients. However, few reports are available on this issue, so further studies are needed to assess the coexistence of gallbladder duplication and carcinoma.
Conclusions
We reported an extremely rare case of malignancy in a duplicated gallbladder. Accurate estimation is feasible for the diagnosis of MGs. In cases of malignancy, especially in cases with inflammation, correct cancer staging is vital. Because of the possibility of malignancy, resected accessory gallbladders should be scrutinized pathologically.
Availability of data and materials
All data generated or analyzed during this study are included in this article.
Abbreviations
- MGs:
-
Multiple gallbladders
- US:
-
Ultrasonography
- CT:
-
Computed tomography scan
- MRCP:
-
Magnetic resonance cholangiopancreatography
- EUS:
-
Endoscopic ultrasonography
- SUV:
-
Standardized uptake value
- FDG-PET:
-
18F-fluoro-2-deoxy-D-glucose positron emission tomography
- CEA:
-
Carcinoembryonic antigen
- CA19-9:
-
Carbohydrate antigen 19-9
- TLV:
-
Total liver volume
- WHO:
-
World Health Organization
- ERCP:
-
Endoscopic retrograde cholangiopancreatography
- MRI:
-
Magnetic resonance imaging
- PTBD:
-
Percutaneous transhepatic biliary drainage
- ENBD:
-
Endoscopic nasobiliary drainage
References
Boyden EA. The accessory gallbladder-an embryological and comparative study of aberrant biliary vesicles occurring in man and domestic mammals. Am J Anast. 1926;38:177–231.
Raymond SW, Thrift CB. Carcinoma of a duplicated gallbladder. Illinois Med J. 1956;110:239–40.
Roeder WJ, Mersheimer WL, Kazarian KK. Triplication of the gallbladder with cholecystitis, cholelithiasis, and papillary adenocarcinoma. Am J Surg. 1971;121:746–8.
Kin Y, Yamaguchi A, Isogai M, Hori A. Duplication of the gallbladder with advanced adenocarcinoma: diagnostic value of MRI and angiography. Hepatogastroenterology. 1996;43:405–8.
Kuwanishi M, Kuwada Y, Mitsuoka Y, et al. A case of double gallbladder with adenocarcinoma arising from the left hepatic duct: a case report and review of the literature. Gastroenterol Res Pract. 2010;721946. https://doi.org/10.1155/2010/721946.
Chen W, Han R. Coexistence of double gallbladder with cholangiocarcinoma: a case report. Medicine (Baltimore). 2018; 97(25): doi:10.1097/MD.0000000000011015.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumors. 8th ed. Oxford: Wiley-Blackwell; 2017.
Darnis B, Mohkam K, Cauchy F, et al. A systematic review of the anatomical findings of multiple gallbladders. HPB (Oxford). 2018;20(11):985–91. https://doi.org/10.1016/j.hpb.2018.04.002.
Ozaki N, Hashimoto D, Ikuta Y, Chikamoto A, Takamori H, Baba H. Definitive diagnosis of a duplicate gallbladder can only be made intraoperatively: report of a case. Clin J Gastroenterol. 2014;7:338–41.
Safioleas MC, Papavassiliou VG, Moulakakis KG, Angouras DC, Skandalakis P. Congenital duplication of the gallbladder. Am Surg. 2006;72:217–20.
Hurst JM, Mayo RA. Unsuspected latent pairing of the cystic primordium. South Med J. 1980;73:950–1.
Sadamoto Y, Kubo H, Harada N, Tanaka M, Eguchi T, Nawata H. Preoperative diagnosis and staging of gallbladder carcinoma by EUS. Gastrointest Endosc. 2003;58:536–41.
Mitake M, Nakazawa S, Naitoh Y, Kimoto E, Tsukamoto Y, Asai T, Yamao K, Inui K. Endoscopic ultrasonography in diagnosis of the extent of gallbladder carcinoma. Gastrointest Endosc. 1990;36:562–6.
Mocellin S, Pasquali S. Diagnostic accuracy of endoscopic ultrasonography (EUS) for the preoperative locoregional staging of primary gastric cancer. Cochrane Database Syst. Rev. 2015;2:CD009944. https://doi.org/10.1002/14651856.CD009944.pub2 Review.
Türkan E, Mine Sencan E, Meral K, Gülgün O, Hatice D. 18F-FDG-PET/CT in initiation and progression of inflammation and infection. Mol Imaging Radionucl Ther. 2017 Jun;26(2):47–52. https://doi.org/10.4274/mirt.18291.
Acknowledgements
The authors thank all the people who contributed to this work.
Endnotes
Not applicable
Funding
The authors declare that they received no funding support for this study.
Author information
Authors and Affiliations
Contributions
TFur described and designed the article. KS, TA, and TFur performed the operation and followed up the patient; additionally, KS supervised the editing of the manuscript. TFuc, HF, and NO contributed to the preoperative check and diagnosis. NI and MN participated in critically revising the manuscript. TH performed the pathological examination and provided the slides. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The patient has provided informed consent, and the study design was approved by an ethics review board.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Furuhashi, T., Abe, T., Sakata, K. et al. Extremely rare case of H-type gallbladder duplication coexistent with carcinoma: a case report and review of the literature. surg case rep 6, 193 (2020). https://doi.org/10.1186/s40792-020-00953-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40792-020-00953-3