Abstract
Background
Postzygotic isolation in the form of reduced viability and/or fertility of hybrids may help maintain species boundaries in the face of interspecific gene flow. Past hybridization events between house sparrows (Passer domesticus) and Spanish sparrows (P. hispaniolensis) have given rise to a homoploid hybrid species, the Italian sparrow (P. italiae). Although genetic incompatibilities are known to isolate these three species, the biological consequences of these incompatibilities are still unknown in early generation hybrids.
Methods
We investigated whether F1 hybrids between house and Spanish sparrows experience reduced viability or fertility. More specifically, we generated hybrids through controlled crosses in aviaries, and compared ovaries of female hybrids with female of pure-species sparrows.
Results
We found that overall, hybrid ovaries were underdeveloped and that half of all female hybrids exhibited symptoms of ovarian hypofunction (ovarian atrophy and complete absence of developed follicles).
Conclusions
Fertility in hybrids is a common consequence or post-zygotic barriers between species. We discuss these results in light of previous findings on genetic incompatibilities between the parent species and the potential role of incompatibilities in hybrid speciation, a rare evolutionary process in birds.
Similar content being viewed by others
Background
As divergence in allopatry between two species increases, levels of postzygotic isolation are also likely to increase (Coyne and Orr 1997). Extrinsic sources of postzygotic isolation, when hybrids are ecologically or biologically unfit, are thought to play an important role in the early stages of population divergence, while intrinsic genetic incompatibilities causing hybrid sterility and/or inviability are thought to develop later (Presgraves 2010; Seehausen et al. 2014). However, little is known about postzygotic isolation between pairs of species for which past hybridization has generated a new species that is reproductively isolated from both parents (a process called hybrid speciation; Mallet 2007). Such information is critical if we want to understand which isolating mechanisms may constrain or facilitate hybrid speciation (Abbott et al. 2013; Schumer et al. 2014). It has been hypothesized that postzygotic barriers may hinder the hybrid speciation process as they will, by necessity, involve the purging of genetic incompatibilities in early generation hybrids (Nolte and Tautz 2010). However, if instead these incompatibilities can be sorted in such a way that one subset of the genes function as reproductive barriers against one of the parents and a different subset against the other parent (Trier et al. 2014), they may favor the emergence of a fully isolated new hybrid lineage (Hermansen et al. 2014; Trier et al. 2014).
The house sparrow (Passer domesticus) and Spanish sparrow (P. hispaniolensis) are two closely related, ecologically similar passerine species. However, differences in habitat and timing of breeding appear to represent pre-mating barriers to gene exchange (Summers-Smith 1988; Hanh Tu 2013). In contrast, there is currently no evidence for post-mating prezygotic isolation (Cramer et al. 2014). Past episodes of hybridization between house sparrows and Spanish sparrows have resulted in the formation of a homoploid hybrid species, the Italian sparrow (P. italiae) (Elgvin et al. 2011; Hermansen et al. 2011). Further, the two parent species are still sympatric in many parts of their range and occasionally hybridize (Summers-Smith 1988; Hermansen et al. 2014). There is also molecular evidence that these two species are isolated by Z-linked genetic incompatibilities (Hermansen et al. 2014). However, little is known about the biological consequences of these genetic incompatibilities and whether they are of an extrinsic and/or intrinsic nature.
Haldane’s rule predicts that the heterogametic sex should suffer more from genetic incompatibilities (Haldane 1922). In birds, the female is the heterogametic sex (ZW) and is hence predicted to suffer more from intrinsic incompatibilities than male hybrids. Here, we investigate whether hybrid females between house sparrows and Spanish sparrows exhibit reduced fertility (through ovarian hypofunction indicated by ovarian atrophy and/or complete lack of follicular development) by dissecting and measuring the ovaries from experimental interspecific crosses between these species.
Methods
We used captive populations of the two species to form experimental crosses in a common garden environment. We captured house sparrows in Oslo, Norway (59.934°N, 10.723°E), and Spanish sparrows in Badajoz, Spain (38.649°N, 7.215°W); most birds were captured in 2010, and breeding activity was monitored the following years. Sparrows were maintained in mixed-sex aviaries housing 15–18 pairs of birds. Although there is no known information about the pedigrees of these different aviaries for logistic reasons, inter and intraspecific crosses were controlled through the following design: two aviaries contained males and females from a single species, while two other aviaries contained males from one species and females from the other species. Hence, hybrids sired from both types of parental combinations, as well as pure house and Spanish sparrows, were generated in a controlled fashion.
The birds were fed ad libitum with seeds and dried insects, and were supplemented with mealworms during the breeding season. We provided nest boxes and nesting material and allowed the birds to breed and pair without interference. A number of birds in all four aviaries bred successfully, and eggs were laid between April and September each year. During the breeding season (May) of 2014, on the same day, we sacrificed 6 hybrid females (5 fathered by a house sparrow male and Spanish sparrow female and 1 fathered by a Spanish sparrow male and house sparrow female) and 12 pure species females (8 house and 4 Spanish sparrows) and inspected their reproductive organs. All birds were mature adults (i.e. at least 2 or more years of age, either wild caught or aviary-born) and had been maintained in captivity under identical conditions for at least two years prior to the current experiment.
Prior to dissection, we measured tarsus length and beak height with a caliper to the nearest 0.1 mm, wing length with a ruler to the nearest 0.5 mm, and body mass with a Pesola balance to the nearest 0.1 g. Ovaries from a total of 18 adult females were scored as either normally developed or atrophied (the main symptom of ovarian hypofunction) and stored in RNA-later buffer for future transcriptomic work. We measured length and width for each ovary with a digital caliper to the nearest 0.1 mm and calculated their volume based on the assumption that ovaries are ellipsoids with equal height and width. We also inspected each ovary for the presence of prehierarchal follicles and counted the number of pre-ovulatory follicles if any were present. We took photographs of one normally developed ovary from a pure house sparrow female and one atrophied ovary from an F1-hybrid female (cross between a house sparrow sire and a Spanish sparrow dam). We performed an ANOVA to test for significant differences in ovary volume between hybrid and pure-species females and also between species. We also investigated whether the ovary width or length of some hybrid females (i.e. those that exhibited atrophied ovaries) were significant statistical outliers compared to the distribution of pure-species ovary widths or lengths using the Z-score method. A Fisher’s exact test was performed to determine whether the proportion of individuals with ovarian hypofunction was elevated in hybrids relative to pure individuals. Furthermore, to investigate whether potential differences in follicle development between normal and potentially atrophied ovaries were not only physical but also functional, we used an ANOVA to test for species and hybrid differences in the number of pre-ovulatory follicles. We then conducted a one sample t test to determine whether the average number of pre-ovulatory follicles found in normal ovaries, irrespective of species, was significantly different from 0 (the number found in all atrophied ovaries).
Results
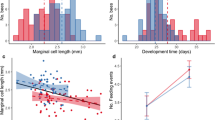
All individuals in the study had morphological features within their respective species ranges: F1 hybrids did not differ from either parent species in tarsus or wing length, beak height or body mass (Table 1). However, on average, hybrid females had significantly smaller ovaries than pure species females (F 1,17 = 5.64; p = 0.030; pure species volume: mean: 1202.7 mm3, SD: 472.8; hybrid volume: mean: 531.0 mm3, SD:729.8), while there was no significant difference in ovarian volume between house and Spanish sparrow females (F 1,11 = 1.85; p = 0.20) (Fig. 1a, b). Furthermore, three hybrid females (all fathered by a house sparrow male and Spanish sparrow female) were significant outliers (Fig. 1a; p = 0.05) in terms of ovary width and length, with surprisingly narrow and short ovaries, suggesting severe atrophy, which was not the case for any pure species females. This meant that 50 % of the hybrid females (n = 6) had atrophied ovaries, which were characterized by a drastic reduction in size (Fig. 1a, d) compared to pure house sparrow (n = 8) and Spanish sparrow females (n = 4). This difference represents a significantly higher incidence of ovarian atrophy in hybrids (p = 0.025; Fig. 1d). Moreover, prehierarchal follicles were detected in all ovaries scored as normal, whereas none could be detected in ovaries scored as atrophied (Table 2), which suggests that atrophied ovaries differ both physically and functionally from normal ovaries. Additionally, in a high proportion of individuals with a normal ovary (pure species: 7/12; hybrids: 3/3), pre-ovulatory follicles could be observed (although in the three hybrid individuals with normal ovaries only one pre-ovulatory follicle could be observed), unlike in individuals with an atrophied ovary (0/3) (Table 2). The number of pre-ovulatory follicles in pure species females was also not significantly different from hybrids with normal ovaries (ANOVA: F 1,14 = 7.86; p = 0.112) but the average number of pre-ovulatory follicles found in normal ovary individuals (mean: 1.47, standard deviation: 1.35) was significantly different from 0 (one-sample t test: t = 4.19; p = 0.001) and hence from what was found in hybrid individuals with atrophied ovaries.
Comparison of the prevalence of ovarian hypofunction between females of both study species (Spanish sparrows, P. hispaniolensis, and house sparrows, P. domesticus) and their hybrids. a Ovary width and length for both hybrid and pure-species females. Black ellipse encircles individual datapoints which are significant outliers in terms of width and length. b Boxplots showing median, quartiles and 5- and 95-percentiles for ovary volume across hybrid and pure-species individuals. Single data-points indicate extreme observations. c Photograph of a typical (normal) ovary and an atrophied ovary. d Proportion of females exhibiting ovarian atrophy in each category (n is the sample size for each type of female)
Discussion
Our finding of underdeveloped or atrophied ovaries, combined with the complete absence of follicular development in otherwise phenotypically normal and viable adult hybrid females, is symptomatic of ovarian hypofunction, and suggests that these individuals were unable to produce ova (Oguntunji and Alabi 2010). Anecdotally, and consistent with Haldane’s rule (Haldane 1922), two hybrid males produced during the experiment exhibited normal sperm function (i.e. motility) relative to individuals from both pure species (Cramer et al. 2015). Strong postzygotic barriers between the house sparrow and Spanish sparrow may seem surprising given that historical hybridization between the two species led to the formation of the hybrid Italian sparrow (Hermansen et al. 2011). Although as many as 9 % of bird species are known to hybridize (Grant and Grant 1992; Mc Carthy 2006), the Italian sparrow is the only bird species that has been shown to be of hybrid origin and to have developed reproductive barriers against both parent species (Hermansen et al. 2011; Trier et al. 2014; but see Brelsford et al. (2011) for another case of an avian taxon of hybrid origin). Thus, there appear to be strong constraints on hybrid speciation in birds. Postzygotic isolation mechanisms may represent such an obstacle (Nolte and Tautz 2010), but these appear to evolve rather slowly in birds (Grant et al. 1996; Price and Bouvier 2002). Recently, Trier et al. (2014) found that genetic incompatibilities isolate the Italian sparrow from its parent species. Furthermore, these genetic incompatibilities represent a subset of those found to isolate house and Spanish sparrows where they occur in sympatry (Hermansen et al. 2014). Identifying the physiological effects of these incompatibilities and determining if these are extrinsic (leading to maladaptation in hybrids) or intrinsic (leading to inviable or infertile hybrids) increases our understanding of mechanisms acting during hybrid speciation.
Here, we experimentally confirm that intrinsic genetic incompatibilities isolate the house and Spanish sparrows, and that these manifest as an increase in the incidence of ovarian hypofunction in hybrid individuals. Interestingly, previous genetic studies have shown that genetic incompatibilities between these two species are predominantly mito-nuclear and sex-linked (Trier et al. 2014). Sex-linked incompatibilities have also been found to lead to hybrid sterility in many other systems (Payseur et al. 2004; Qvarnström and Bailey 2008). Trier et al. (2014) identified six Z-linked candidate incompatibility loci between the hybrid Italian sparrow and either of the parent species. Evidence suggests that these loci have been sorted during the hybrid speciation process in such a way that one subset of the genes function as reproductive barriers against the Spanish sparrow, and a different subset against the house sparrow (Hermansen et al. 2014; Trier et al. 2014). One of these six loci associated with reproductive isolation between the Italian and Spanish sparrow (Trier et al. 2014) as well as between the house and Spanish sparrow (Hermansen et al. 2014) is situated in the coding region of the gene GTF2H2, a transcription factor highly expressed in oocytes (Kakourou et al. 2013). This gene has been shown to be involved in premature ovarian hypofunction (Aboura et al. 2009) and polycystic ovary syndromes (Haouzi et al. 2012) in other vertebrates. If GTF2H2 is indeed contributing to ovarian hypofunction in sparrow hybrids, it may have been instrumental in this hybrid speciation event. In the future, mapping the occurrence of ovarian atrophy to candidate genes such as GTF2H2 in sparrow hybrids would yield the ultimate link needed to establish the importance of parental incompatibility sorting during hybrid speciation. Hence, more experimental work should be conducted on the biological consequences of postzygotic barriers to unravel the genetic predispositions to hybrid speciation.
Availability of supporting data
The data set supporting the results of this article will be available in the DRYAD repository [unique persistent identifier and hyperlink to dataset(s) in http://format], upon acceptance.
References
Abbott R, Albach D, Ansell S, Arntzen JW, Baird SJE, Bierne N, Boughman J, Brelsford A, Buerkle CA, Buggs CA, Butlin RK, Dieckmann U, Eroukhmanoff F, Grill A, Cahan SH, Hermansen JS, Hewitt G, Hudson AG, Jiggins C, Jones J, Keller B, Marczewski T, Mallet J, Martinez-Rodriguez P, Most M, Mullen S, Nichols R, Nolte AW, Parisod C, Pfennig K, Rice AM, Ritchie MG, Seifert B, Smadja CM, Stelkens R, Szymura JM, Vainola R, Wolf JBW, Zinner D. Hybridization and speciation. J Evol Biol. 2013;26:229–46.
Aboura A, Dupas C, Tachdjian G, Portnoï MF, Bourcigaux N, Dewailly D, Frydman R, Fauser B, Ronci-Chaix N, Donadille B, Bouchard P, Christin-Maitre S. Array comparative genomic hybridization profiling analysis reveals deoxyribonucleic acid copy number variations associated with premature ovarian failure. J Clin Endocrinol Metab. 2009;94:4540–6.
Brelsford A, Mila B, Irwin DE. Hybrid origin of Audubon’s warbler. Mol Ecol. 2011;20:2380–9.
Coyne JA, Orr HA. “Patterns of speciation in Drosophila” revisited. Evolution. 1997;51:295–303.
Cramer ERA, Laskemoen T, Eroukhmanoff F, Haas F, Hermansen JS, Lifjeld JT, Rowe M, Sætre GP, Johnsen A. Testing a post-copulatory pre-zygotic reproductive barrier in a passerine species pair. Behav Ecol Sociobiol. 2014;7:1133–44.
Cramer ERA, Laskemoen T, Stensrud E, Rowe M, Haas F, Lifjeld JT, Saetre GP, Johnsen A. Morphology-function relationships and repeatability in the sperm of Passer sparrows. J Morphol. 2015;276:370–7.
Elgvin TO, Hermansen JS, Fijarczyk A, Bonnet T, Borge T, Sæther SA, Voje KL, Sætre GP. Hybrid speciation in sparrows II: a role for sex chromosomes? Mol Ecol. 2011;20:3823–37.
Grant PR, Grant BR. Hybridization of bird species. Science. 1992;256:193–7.
Grant PR, Grant BR, Deutsch JC. Speciation and hybridization in Island birds. Phil Trans R Soc B. 1996;351:765–72.
Haldane JBS. Sex-ratio and unisexual sterility in hybrid animals. J Genet. 1922;12:101–9.
Hanh Tu M. Fine-and broad-scale premating isolation between two closely related species, the house sparrow (Passer domesticus) and the Spanish sparrow (P. hispaniolensis). M.Sc. Thesis. University of Oslo, Norway. 2013.
Haouzi D, Assou S, Monzo C, Vincens C, Dechaud H, Hamamah S. Altered gene expression profile in cumulus cells of mature MII oocytes from patients with polycystic ovary syndrome. Hum Reprod. 2012;27:3523–30.
Hermansen JS, Sæther SA, Elgvin TO, Borge T, Hjelle E, Sætre GP. Hybrid speciation in sparrows I: phenotypic intermediacy, genetic admixture and barriers to gene flow. Mol Ecol. 2011;20:3812–22.
Hermansen JS, Haas F, Bailey RJ, Nederbragt AJ, Trier CN, Reynolds AM, Sætre GP. Hybrid speciation by sorting of parental incompatibilities in Italian sparrows. Mol Ecol. 2014;23:5831–42.
Kakourou G, Jaroudi S, Tulay P, Heath C, Serhal P, Harper JC, SenGupta SB. Investigation of gene expression profiles before and after embryonic genome activation and assessment of functional pathways at the human metaphase II oocyte and blastocyst stage. Fertil Steril. 2013;99:803–14.
Mallet J. Hybrid speciation. Nature. 2007;446:279–83.
Mc Carthy EM. Handbook of avian hybrids of the world. New York: Oxford University Press; 2006.
Nolte AW, Tautz D. Understanding the onset of hybrid speciation. Trends Genet. 2010;26:54–8.
Oguntunji AO, Alabi OM. Influence of high environmental temperature on egg production and shell quality: a review. World Poult Sci J. 2010;66:739–50.
Payseur BA, Krenz JG, Nachman MW. Differential patterns of introgression across the X chromosome in a hybrid zone between two species of house mice. Evolution. 2004;58:2064–78.
Presgraves DC. The molecular evolutionary basis of species formation. Nat Rev Genet. 2010;11:175–80.
Price TD, Bouvier MM. The evolution of F1 postzygotic incompatibilities in birds. Evolution. 2002;56:2083–9.
Qvarnström A, Bailey RI. Speciation through evolution of sex-linked genes. Heredity. 2008;102:4–15.
Schumer M, Rosenthal GG, Andolfatto P. How common is homoploid hybrid speciation? Evolution. 2014;68:1553–60.
Seehausen O, Butlin RK, Keller I, Wagner CE, Boughman JW, Hohenlohe PA, Peichel CL, Saetre G-P, Bank C, Brännström A, Brelsford A, Clarkson CS, Eroukhmanoff F, Feder JL, Fischer MC, Foote AD, Franchini P, Jiggins CD, Jones FC, Lindholm AK, Lucek K, Maan ME, Marques DA, Martin SH, Matthews B, Meier JI, Möst M, Nachman MW, Nonaka E, Rennison DJ, Schwarzer J, Watson ET, Westram AM, Widmer A. Genomics and the origin of species. Nat Rev Genet. 2014;15:176–92.
Summers-Smith JD. The sparrows: a study of the genus Passer. 1st ed. Calton: T&AD Poyser; 1988.
Trier CN, Hermansen JS, Sætre GP, Bailey RI. Evidence for mito-nuclear and sex-linked reproductive barriers between the hybrid Italian sparrow and its parent species. PLoS Genet. 2014;10:e1004075.
Authors’ contributions
FE, FH, JSH, GPS carried out the breeding experiment. FE, MR, AR and ERAC carried out or assisted with the dissections, AR measured the ovaries, MR evaluated follicular activity. FE and ERAC conducted the statistical analyses. FE, MR, AJ and GPS participated in the design of the study and its coordination. FE wrote the manuscript and all authors helped to draft it. All authors read and approved the final manuscript.
Authors' information
This study is the result of a collaboration between two research groups: the Sparrow Group at the Department of Biosciences (University of Oslo) and the Sex and Evolution Research Group at the Natural History Museum (University of Oslo) and was initiated by GPS as part of large research project on hybridization in Passer species.
Acknowledgements
We thank the numerous assistants who helped in the field and during the experimental phase of this project. This study was funded by the Research Council of Norway (GPS, AJ), the Swedish Research Council (FH, FE, AR), the University of Oslo (JSH) and the Marie-Curie Foundation (FE). All authors declare that the present study complies with the current laws and ethical standards of animal research in Norway. Ethical permission was issued to F. Haas and M. Rowe (Norwegian Animal Research Authority—FOTS ID 2394 and Norwegian Animal Research Authority—FOTS ID 6323).
Competing interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Eroukhmanoff, F., Rowe, M., Cramer, E.R.A. et al. Experimental evidence for ovarian hypofunction in sparrow hybrids. Avian Res 7, 3 (2016). https://doi.org/10.1186/s40657-016-0038-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40657-016-0038-1