Abstract
The nuclear factor-kappa B (NF-κB) transcription factor plays an essential role in the host immune response to different pathogens and genotoxic stimuli. In addition and either dependently or independently, it also promotes malignant transformation. The first step in its activation is conversion of extracellular stimuli to a cascade of reactions mediated by a variety of membrane receptors. The activated receptor transmits the signal through downstream proteins to activate different kinases. Subsequently, the inhibitory proteins that sequester NF-κB in the cytoplasm are phosphorylated, ubiquitinated, and degraded in the proteasome. Free NF-κB then enters the nucleus to initiate its transcriptional program. An important signaling “code” in the multiple-step NF-κB activating cascade consists of different ubiquitin (Ub) chains that are assembled on the different effector proteins through different lysine residues in the Ub molecule. Together with this unique set of proteins in the cascade, Ub chains form the platform for the binding of downstream interacting partners. One unknown link in the activation of NF-κB was the ubiquitin ligase that catalyzes generation of the active p50 subunit from its p105 inactive precursor. We found that KPC1 ubiquitinates p105 and catalyzes its processing to p50 under both basal and signal-induced conditions. A variety of biological functions carried out by NF-κB, depends on the dimeric composition of the transcription factor, and can determine the tumor suppression/promotion fate of the cell. Overexpression of KPC1 probably shifts the balance between NF-κB subunits from the “canonical” p50·p65 heterodimer to a p50 homodimer which results in a strong tumor suppressive phenotype.
Similar content being viewed by others
Review
A leading role in regulating essential cellular processes such as proliferation, differentiation, cell survival, and immune response is carried out by the family of NF-κB transcription factors. The involvement of malfunction within the NF-κB pathway in the development of chronic inflammation and subsequent malignant transformation has recently become a scientific certainty (Ben-Neriah and Karin, 2011; DiDonato et al., 2012).
The NF-κB pathway depends in many of its steps on ubiquitination. Ubiquitin (Ub) is a small, 76 amino acid protein that can modify a variety of proteins. The reaction is catalyzed by three types of enzymes that act in concert: E1, a Ub activating enzyme; E2, a Ub conjugating enzyme (or Ub carrier protein); and E3, a substrate-specific Ub ligase. As a result, the C-terminal Gly of Ub is typically conjugated covalently to the ε-amino group of an internal lysine residue(s) in the target substrate (Glickman and Ciechanover, 2002). In many cases, the substrate is modified by a chain that is composed of Ubs bound to each other through internal lysine residues (K6, K11, K27, K29, K33, K48, and K63) in the Ub molecule. Sometimes the Ub molecules are linked “head-to-tail” with the C-terminus of Ub conjugated via peptide bond to the α-amino group of the first methionine of the previous Ub, resulting in generation of linear chains. Different linkages of the Ub chain have distinct structure and therefore modulate, via different interactions a variety of cellular processes such as proteasomal degradation, signal transduction, or specific cellular localization (Komander and Rape, 2012).
The activation of NF-κB starts in many cases with the recognition and binding of different pro-inflammatory cytokines by specific cell membrane receptors. Tumor necrosis factor alpha (TNFα), lipopolysaccharide (LPS), interleukin-1 beta (IL-1β), and cluster of differentiation 40 (CD40L) are examples of proteins that can activate NF-κB (Rahman and McFadden, 2011). In general, there are two types of NF-κB activation—“canonical” and “alternative.” They differ in the cascade of adaptor proteins downstream of the receptor, in subsequent activation of different kinases, and in generation of transcription factors that possess different subunits. Despite the difference, all the pathways have a similar scheme: ligand, specific receptor, and cascade of adaptor proteins (modified by Ub chains that serve a non-proteolytic function). These proteins “translate” the activation of the receptor to activation of a distinct kinase, modification of the inhibitory protein by Ub chains that leads to its proteasomal destruction with subsequent formation of active NF-κB that is translocated into the nucleus.
In this review, we shall focus on one particular example of TNFα-induced NF-κB activation pathway (Fig. 1).
The multi-step activation pathway of the NF-κB transcription factor. For details, see the main text. Yellow circles denote Ub48-linked chains. Orange circles denote K64-based chains. Blue circles denote linear Ub chains. The yellow arrow denotes ubiquitination. The green arrow denotes de-ubiquitination. The red arrow denotes phosphorylation
The ligand protein TNFα is one of the most important inflammatory cytokines. It leads to the activation of NF-κB through the canonical pathway. A trimer of the extracellular transmembrane tumor necrosis factor receptor 1 (TNFR1) specifically recognizes a trimeric TNFα (Idriss and Naismith, 2000). In its bound state, the receptor interacts with the TNF receptor-associated protein with death domain (TRADD) (Hsu et al., 1995). The complex includes also the receptor-interacting protein-1 (RIP1) kinase, that is essential for TNF-dependent NF-κB activation independently of its kinase function (Kelliher et al., 1998). Following TNFα signaling, RIP1 is ubiquitinated by several ubiquitin ligases. TNF receptor-associated factor (TRAF2) (Lee et al., 2004) and cellular inhibitor of apoptosis-1 and apoptosis-2 (cIAP1 and cIAP2) (Bertrand et al., 2008; Park et al., 2004) modify RIP1 by K63-linked ubiquitin chains. The ubiquitination that occurs on K377 of RIP1 (Ea et al., 2006) serves as a platform for the binding of TAK1-binding protein (TAB)-TAK1-complex (Kanayama et al., 2004), as well as NF-κB essential modulator NEMO (Ea et al., 2006; Wu et al., 2006). This event promotes activation of IκB kinase (IKKα/β) with subsequent phosphorylation, ubiquitination, and degradation of IκB inhibitory proteins. Prior to their degradation, the IκB proteins retain NF-κB dimers in the cytoplasm.
The ubiquitin ligase cIAP1 along with the ubiquitin conjugation enzyme UbcH5 catalyzes yet another conjugation reaction—generation of K11-linked ubiquitin chains on RIP1. The resulting ubiquitin chain(s) have a structure that could be recognized by NEMO (Dynek et al., 2010).
To regulate the process, the dual function tumor necrosis factor alpha-induced protein 3 (A20) (that possesses both a deubiquitinating (DUB) and a K48 ubiquitin ligase activities) modifies RIP1 by removing the K63 chain and generating a K48-based chain, leading to destruction of the protein, thus attenuating the TNFα signal (Wertz et al., 2004).
Finally, linear ubiquitin chains were also described as an important regulatory signal in the inflammatory response and NF-κB activation. The linear Ub chain assembly complex (LUBAC) is a unique ligase that is able to synthetize linear Ub chains (Kirisako et al., 2006). LUBAC is made of the heme-oxidized IRP2 ligase-1 (HOIL-1L), HOIL-1L-interacting protein (HOIP), and SHANK-associated RH domain interacting protein (SHARPIN). HOIP is a catalytic subunit that through its Ub-associated (UbA) domain binds to Ub-like (UbL) domains of HOIL-1L and SHARPIN (Yagi et al., 2012). Linear Ub chains can serve as the platform for recruitment of NEMO through UBAN (Ub binding in ABIN and NEMO) domain (Rahighi et al., 2009). Alternatively, NEMO itself can be modified by linear ubiquitin chains on the specific Lys residues in the CC2-LZ domain (Tokunaga et al., 2009). In both cases, the result is TNFα-induced activation of the IKK complex (Iwai et al., 2014).
Active NF-κB transcription factor is a family of dimers, the members of which are made of different proteins in different combinations, including p50 (derived from processing of p105—NF-κB1) (Fan and Maniatis, 1991; Palombella et al., 1994), p52 (derived from processing of p100—NF-κB2) (Betts and Nabel, 1996), p65 (RelA), RelB, and c-Rel. All of them have a REL homology domain (RHD) that binds DNA. The NF-κB subunits form homo- or heterodimers that in resting cells stay in the cytoplasm bound to the ankyrin repeats of IκB inhibitory proteins (IκB, Bcl3, p100, and p105). Following different extracellular stimuli, the IκB proteins are degraded by 26S proteasome, and free and active NF-κB transcription factors are translocated to the nucleus. Depending on the dimers formed, distinct sets of genes are transcribed which results in differed outcomes.
NF-κB is excessively activated in many tumors, stimulating transcription of anti-apoptotic genes and cell cycle promoters. Nevertheless, the commonly held view that NF-κB is a pro-tumorigenic stimulator relates to the canonical tumorigenic p50·p65 heterodimer rather than to the less common p50·p50 homodimer. p50·p50 was described as possessing tumor suppressive activities (Perkins, 2012; Pikarsky and Ben-Neriah, 2006). Furthermore, p50·p50 dimers together with the co-repressor histone deacetylase 1 (HDAC1) downregulate the transcription of S100A8/9, CXCL1, and CXCL2 chemokines of neutrophils found in tumor microenvironment. This repression prevents development of neutrophil-driven hepatocellular carcinoma (Wilson et al., 2015). In addition, increased frequency of alkylator-induced mutations was identified in NF-κB1 (−/−) cells and mice. Moreover, after tumor induction by alkylating agents, NF-κB1 deficient mice developed more severe lymphomas compared to their NF-κB1 (+/+) counterparts (Voce et al., 2015). In a similar example, NF-κB2-derived dimers (p52·p52) contribute to the repression of Bcl2, XIAP, Bcl-XL, Cyclin D1, and c-Myc, leading to cell cycle arrest and cell death after cell damage (Barre et al., 2010; Barre and Perkins, 2007).
Conclusions
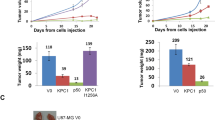
In our recent study (Kravtsova-Ivantsiv et al., 2015), we demonstrated that over expression of the NF-κB1 p50 subunit has a strong suppressive effect on glioblastoma and breast xenograft development. In the same study, we found one of the missing enzymes in the activation cascade of NF-κB, KIP1 ubiquitination-promoting complex (KPC). KPC, a heterodimer made of KPC1 (RNF123) and KPC2 (UBAC1), is the ligase that is responsible for the generation of p50 from p105 both under basal conditions and following stimulation. RNA seq analysis of the glioblastoma tumors that overexpress KPC1 (or p50) revealed significant expression of tumor suppressors. In addition, functional analysis has shown alteration of proteins in the tumor interaction with its microenvironment (e.g., cell adhesion, cell migration, and cell-cell signaling proteins), all in the direction of tumor suppression. Analysis of human head and neck and glioblastoma sections reveled decreased staining of KPC1 and nuclear p50 compared to normal tissue. We hypothesize that one of the critical steps in malignant transformation may be the downregulation of these proteins that in the normal tissue support the expression of “beneficial” proteins such as tumor suppressors.
Altogether, we can hypothesize that determination of the cell outcome can be due, at least partially, to the balance between NF-κB subunits, and shifting to p50·p50 homodimers is beneficial to cell homeostasis. Further experiments are needed to substantiate this assumption.
References
Barre B, Perkins ND. A cell cycle regulatory network controlling NF-kappaB subunit activity and function. EMBO J. 2007;26:4841–55.
Barre B, Coqueret O, Perkins ND. Regulation of activity and function of the p52 NF-kappaB subunit following DNA damage. Cell Cycle. 2010;9:4795–804.
Ben-Neriah Y, Karin M. Inflammation meets cancer, with NF-kappaB as the matchmaker. Nat Immunol. 2011;12:715–23.
Bertrand MJ, Milutinovic S, Dickson KM, Ho WC, Boudreault A, Durkin J, et al. cIAP1 and cIAP2 facilitate cancer cell survival by functioning as E3 ligases that promote RIP1 ubiquitination. Mol Cell. 2008;30:689–700.
Betts JC, Nabel GJ. Differential regulation of NF-kappaB2(p100) processing and control by amino-terminal sequences. Mol Cell Biol. 1996;16:6363–71.
DiDonato JA, Mercurio F, Karin M. NF-kappaB and the link between inflammation and cancer. Immunol Rev. 2012;246:379–400.
Dynek JN, Goncharov T, Dueber EC, Fedorova AV, Izrael-Tomasevic A, Phu L, et al. c-IAP1 and UbcH5 promote K11-linked polyubiquitination of RIP1 in TNF signalling. EMBO J. 2010;29:4198–209.
Ea CK, Deng L, Xia ZP, Pineda G, Chen ZJ. Activation of IKK by TNFalpha requires site-specific ubiquitination of RIP1 and polyubiquitin binding by NEMO. Mol Cell. 2006;22:245–57.
Fan CM, Maniatis T. Generation of p50 subunit of NF-kappa B by processing of p105 through an ATP-dependent pathway. Nature. 1991;354:395–8.
Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002;82:373–428.
Hsu H, Xiong J, Goeddel DV. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. Cell. 1995;81:495–504.
Idriss HT, Naismith JH. TNF alpha and the TNF receptor superfamily: structure-function relationship(s). Microsc Res Tech. 2000;50:184–95.
Iwai K, Fujita H, Sasaki Y. Linear ubiquitin chains: NF-kappaB signalling, cell death and beyond. Nat Rev Mol Cell Biol. 2014;15:503–8.
Kanayama A, Seth RB, Sun L, Ea CK, Hong M, Shaito A, et al. TAB2 and TAB3 activate the NF-kappaB pathway through binding to polyubiquitin chains. Mol Cell. 2004;15:535–48.
Kelliher MA, Grimm S, Ishida Y, Kuo F, Stanger BZ, Leder P. The death domain kinase RIP mediates the TNF-induced NF-kappaB signal. Immunity. 1998;8:297–303.
Kirisako T, Kamei K, Murata S, Kato M, Fukumoto H, Kanie M, et al. A ubiquitin ligase complex assembles linear polyubiquitin chains. EMBO J. 2006;25:4877–87.
Komander D, Rape M. The ubiquitin code. Annu Rev Biochem. 2012;81:203–29.
Kravtsova-Ivantsiv Y, Shomer I, Cohen-Kaplan V, Snijder B, Superti-Furga G, Gonen H, et al. KPC1-mediated ubiquitination and proteasomal processing of NF-kappaB1 p105 to p50 restricts tumor growth. Cell. 2015;161:333–47.
Lee TH, Shank J, Cusson N, Kelliher MA. The kinase activity of Rip1 is not required for tumor necrosis factor-alpha-induced IkappaB kinase or p38 MAP kinase activation or for the ubiquitination of Rip1 by Traf2. J Biol Chem. 2004;279:33185–91.
Palombella VJ, Rando OJ, Goldberg AL, Maniatis T. The ubiquitin-proteasome pathway is required for processing the NF-kappa B1 precursor protein and the activation of NF-kappa B. Cell. 1994;78:773–85.
Park SM, Yoon JB, Lee TH. Receptor interacting protein is ubiquitinated by cellular inhibitor of apoptosis proteins (c-IAP1 and c-IAP2) in vitro. FEBS Lett. 2004;566:151–6.
Perkins ND. The diverse and complex roles of NF-kappaB subunits in cancer. Nat Rev Cancer. 2012;12:121–32.
Pikarsky E, Ben-Neriah Y. NF-kappaB inhibition: a double-edged sword in cancer? Eur J Cancer. 2006;42:779–84.
Rahighi S, Ikeda F, Kawasaki M, Akutsu M, Suzuki N, Kato R, et al. Specific recognition of linear ubiquitin chains by NEMO is important for NF-kappaB activation. Cell. 2009;136:1098–109.
Rahman MM, McFadden G. Modulation of NF-kappaB signalling by microbial pathogens. Nat Rev Microbiol. 2011;9:291–306.
Tokunaga F, Sakata S, Saeki Y, Satomi Y, Kirisako T, Kamei K, et al. Involvement of linear polyubiquitylation of NEMO in NF-kappaB activation. Nat Cell Biol. 2009;11:123–32.
Voce DJ, Schmitt AM, Uppal A, McNerney ME, Bernal GM, Cahill KE, et al. Nfkb1 is a haploinsufficient DNA damage-specific tumor suppressor. Oncogene. 2015;34:2807–13.
Wertz IE, O’Rourke KM, Zhou H, Eby M, Aravind L, Seshagiri S, et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-kappaB signalling. Nature. 2004;430:694–9.
Wilson CL, Jurk D, Fullard N, Banks P, Page A, Luli S, et al. NFkappaB1 is a suppressor of neutrophil-driven hepatocellular carcinoma. Nat Commun. 2015;6:6818.
Wu CJ, Conze DB, Li T, Srinivasula SM, Ashwell JD. Sensing of Lys 63-linked polyubiquitination by NEMO is a key event in NF-kappaB activation [corrected]. Nat Cell Biol. 2006;8:398–406.
Yagi H, Ishimoto K, Hiromoto T, Fujita H, Mizushima T, Uekusa Y, et al. A non-canonical UBA-UBL interaction forms the linear-ubiquitin-chain assembly complex. EMBO Rep. 2012;13:462–8.
Acknowledgements
Research in the laboratory of A.C. is supported by grants from the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (AMRF), the Israel Science Foundation (ISF), the I-CORE Program of the Planning and Budgeting Committee and the ISF (Grant1775/12), the Deutsch-Israelische Projektkooperation (DIP), and the Program for Targeting Cancer by Modulating Protein Dynamics supported by Albert Sweet, Malibu, CA, USA. A.C. is an Israel Cancer Research Fund (ICRF) USA Professor. The laboratory of Y.T.K. is supported by the Basic Science Research Program of the National Research Foundation (NRF) funded by the Ministry of Science, ICT and Future Planning (MSIP) of Korea (NRF-2013R1A2A2A01014170). A.C. and Y.T.K. are supported by the Nobel Laureates Invitation Program of Seoul National University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
YK-I, YTK and AC drafted the manuscript. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kravtsova-Ivantsiv, Y., Kwon, Y.T. & Ciechanover, A. Role of the ubiquitin ligase KPC1 in NF-κB activation and tumor suppression. J Anal Sci Technol 7, 8 (2016). https://doi.org/10.1186/s40543-016-0087-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40543-016-0087-4