Abstract
Background
Echis ocellatus envenoming is potentially toxic initiating clinical damages on male reproductive system. Kaempferol is a therapeutic agent with neutralizing potentials on snake venom toxins. This study investigated the antagonistic effect of kaempferol on E. ocellatus venom (EoV)-induced reproductive toxicities.
Methods
Fifty adult male rats were sorted at random into five groups of ten rats for this study. The control rats were allotted to group 1, while rats in groups 2–5 were injected with 0.22 mg/kg bw (LD50) of EoV intraperitoneally. Rats in group 2 were not treated while groups 3–5 rats were treated with serum antivenom (0.2 ml), and 4 and 8 mg/kg bw of kaempferol post envenoming, respectively.
Results
EoV actuated reproductive toxicity, significantly decreased sperm parameters, and enhanced inflammatory, oxidative stress, and apoptotic biomarkers in reproductive organs of untreated envenomed rats. However, treatment with kaempferol alleviated the venom-induced reproductive disorders with a dose dependent effect. Kaempferol significantly increased the testicular weight, organo-somatic index, sperm parameters, and normalized the levels of serum luteinizing hormone, testosterone, and follicle stimulating hormone. Kaempferol ameliorated testicular and epididymal oxidative stress as evidenced by significant decrease in malondialdehyde (MDA) levels, enhancement of reduced glutathione (GSH) levels, superoxide dismutase (SOD) and glutathione peroxidase (GPX) activities. The inflammatory biomarkers; nitric oxide (NO) levels and myeloperoxidase activity (MPO), and apoptotic biomarkers; caspase 3 and caspase 9 activities were substantially suppressed in the testis and epididymis of envenomed rats treated with kaempferol.
Conclusion
Results revealed kaempferol as a potential remedial agent against reproductive toxicity that could manifest post-viper envenoming.
Similar content being viewed by others
Introduction
The reproductive system is a sensitive structure with specific organ functions that can be altered when exposed to toxic compounds [1]. Exposure to toxicants affects reproductive cell functions which have caused significantly deterioration most especially in male fertility over the years, thus becoming a global public health concern. Infertility associated with reproductive toxicity is on the rise affecting 15% of couples of reproductive ages [2]. Several factors that include genetic, environmental, chemicals, natural toxins and modern lifestyle are known to cause detrimental effect on male reproductive health [3, 4].
Studies have documented that one of the relevant factors that could affect reproductive health is exposure to natural toxins produced by venomous animals [5]. Most victims of snake envenoming are the inhabitants of tropical rural communities of the world, who are mostly farmers, agricultural workers and nomads as they are frequently exposed to contact with snakes due to their daily activities. This occupational hazard poses significant pathophysiology to normal reproductive organs and affect sperm functional parameters post envenoming. Furthermore, studies have documented venom induced-male reproductive pathologies and disorders after venom injection in rats [5, 6].
Snakebite envenoming is a serious public medical burden resulting to permanent disabilities and mortality, most especially in deprived communities of Africa, Latin America and Asia [7]. In Nigeria, incidences of snakebite envenoming are on the rise particularly in the northern region and has become a relevant occupational and public health hazard [8]. Echis ocellatus (African saw-scaled viper) is a specie of medical relevance, and its envenoming has been documented to result to deaths and permanent disabilities in Nigeria [9, 10].
E. ocellatus venom (EoV) contains diverse toxins exhibiting several clinical manifestation haemorrhages, uncoagulable blood, shock, and necrosis in viper envenomed victims [9]. E. ocellatus venom have been implicated in several damages to organs systems [11, 12]. Also, the venom has been documented to induced hormonal disorder, oxidative stress, inflammation and histomorphology in reproductive organs resulting into sperm deficits and abnormalities [6]. Induction of oxidative stress and inflammation in reproductive organs may represent a common mechanism in endocrine disruptor-mediated dysfunction, especially on testicular cell which can impair reproductive capacities by altering sperm counts motility, viability, and their cell membrane integrity [13]. Therefore, therapeutic strategies aimed at preventing reproductive disorders might be a reasonable choice against EoV-induced reproductive toxicity.
Treatment of snakebite envenoming involves the use of conventional antiserum which is very scarce, costly in most tropical rural communities of Africa [14]. In addition, antivenom is impotent in ameliorating local tissue injury combined with incapability to overturn pathophysiology caused to organ systems after envenoming [14]. Consequently, most victims of snakebite envenoming rely on phytotherapy as alternative. Natural bioactive phytochemicals have been known to exhibits great pharmacological capability and acknowledged in the treatment of snakebite envenoming [12].
Kaempferol is a flavonoid compound with 4 hydroxy-groups at −3, −40, −5 and −7 positions in their structure [15] and abundantly present in several edible plants [16]. Kaempferol is a plant-derived bioactive phytochemical that has been established as potent antivenom agent against snakebite toxicities [17]. Studies have documented the anticarcinogenic, antioxidant, cardioprotective, neuroprotective, antidiabetic, and antimicrobial properties of kaempferol and its glycosides [15]. The amelioration of E. ocellatus venom induced reproductive organs pathophysiology either by synthetic or natural active products from plant remain unreported. Thus, this present investigation explored the therapeutic potential of kaempferol on reproductive organs toxicities induced by E. ocellatus venom in male rats.
Materials and methods
Procurement of venom and antiserum
A lyophilized venom sample obtained from an adult male E. ocellatus was procured from the serpentarium of the Department of Zoology, University of Ibadan, Nigeria and preserved at 4oC in the laboratory. The EchiTAb-Plus ICP polyvalent serum antivenom was the reference drug for this research.
Procurement of kaempferol
Kaempferol (200 mg, MF: C15H10O6, MW: 286.24 g/mol) was purchased from a standard chemical company, Sigma-Aldrich®, USA, St Louis, Missouri, USA for this research.
Ethics consideration
The use of animals for this study was approved by University of Ibadan-Animal Care and Use Research Ethics Committee (UI-ACUREC/19/0030). All experimental protocols were in compliance with the current rules and guidelines established for the care and use of laboratory animals [18]. Furthermore, the experiment was carried out in compliance with the revised ARRIVE guidelines 2.0.
Animals
Fifty male wistar rats with weight ranging between 125 and 155 g were procured from the Central Animal Facility of Osun State University, Osogbo, Nigeria. The animals were kept in transparent well-ventilated plastic cages and acclimatized for two weeks at the Department of Zoology Laboratory, Osun State University under the ambient standard conditions (25 ± 2 °C and relative humidity of 50 ± 15%). The rats were allowed free access to standard rat feed and clean water under 12 h daylight and darkness cycle.
Animal groupings
The animals were sorted unbiased into group of five (n = 10). Animals in group 1 served as the control and injected with saline while groups 2–5 were envenomed with 0.22 mg/kg−1 of EoV. Group 2 animals were left untreated while groups 3–5 animals were treated with 0.2 ml of polyvalent antivenom, 4 and 8 mg/kg of kaempferol respectively.
Envenoming and treatment protocols
The animals were injected with 0.2 ml of Lethal Dose (LD) concentration of EoV (LD50, 0.22 mg/kg−1 dissolved in 10 ml of saline) as previously determined by Adeyi et al. [12]. The treatment of the animals commenced 30 min post envenoming. Group 3 was treated intraperitoneally with 0.2 mg of polyvalent antivenom. Kaempferol at 4 and 8 mg/kg was dissolved separately in 1 ml of saline and injected intraperitoneally into rats in group 4 and 5 respectively, and this dosage were administered once daily to each respective treatment group for seven days in a row. However, the animals were euthanized 50 days post envenoming to complete the required duration of spermatogenesis in rats [19]. All experimental rats were observed for any clinical symptoms of envenoming and death.
Body weight changes
All the experimental animals were measured pre-venom injection as initial weight and at termination to determine the body weight changes before been euthanized. The changes in body weight in each group were determined using the formula:
Blood and organ collection
Retro-orbital sinus punctuation method was used to collect blood samples from each experimental animals into plain bottles for reproductive hormonal assays. The animals were then euthanized using cervical dislocation in accordance to Rowett [20] guides. The testes and cauda epididymis were surgically removed and weighed. The right epididymis was used for analysis of sperm parameters while the left epididymis and testes were used for biochemical assays. The relative organ weight was calculated using the formula:
Assessments of semen functional characteristics
Sperm volume assay
The epididymis was prepared to evaluate the sperm parameters and the sperm volume was determined by immersing the epididymis in 5 mL normal saline in a measuring cylinder and the volume displaced was taken as the volume of the epididymis [21].
Sperm motility assay
The right cauda epididymis was placed individually in a petri dish and minced in normal saline (1 ml) to form the sperm suspension. 10 µl of the suspension was dropped on a microscopic slide and observed for motility under the light microscope at a magnification of x400. Sperm motility was assessed by classifying 200 spermatozoa into two categories, motile and immotile spermatozoa. Three sperm classes were categorized as motile spermatozoa: rapid progressive, slow progressive and non-progressive spermatozoa [22].
Sperm counts assay
For the sperm count, a further 1:10 serial dilution of the sperm suspension was prepared out of which 10 µl of the dilution was counted under a light microscope at a 400× magnification with the aid of an improved Neubauer hemocytometer as described in the WHO [22]. Sperm count values were multiplied by the dilution factor and recorded as millions per milliliter (106/ml).
Sperm morphological abnormalities
Exactly 450 µL of the sperm suspension was mixed with 50 µL of 1% aqueous eosin Y for 45 min. The stained sperm suspension was used to make a thin smear on a pre-cleaned grease-free microscopic slide. Prepared slides were allowed to air dry and abnormalities were observed in 250 spermatozoa with four replicates in each rat at a magnification of ×1000 [23].
Assessment of serum reproductive hormones
Sex hormones levels in sera were analyzed using the Enzyme-Linked Immunosorbent Assay (ELISA) kits (CUSABIO, Houston, TX, 77054, USA) to determine the concentrations of Luteinizing Hormone (Cat no: CSB-E12654r), Follicle Stimulating Hormone (Cat no: CSB-E06869r) and testosterone (Cat no: CSB-E05100r). The assays were done according to manufactures’ instructions.
Assessments of testicular and epididymal biochemical analysis
Oxidative stress biomarkers and antioxidant enzymes
The levels of reduced glutathione (GSH) were carried out following the previous protocol [24]. Glutathione peroxidase (GPX) and Superoxide Dismutase (SOD) activities were measured using the previous methods of Marklund and Marklund, [25] and Rotruck et al. [26], respectively. The levels of lipid peroxidation end product; malondialdehyde (MDA), was determined as described by Ohkawa et al. [27].
Inflammatory biomarkers
The testicular and epididymal nitric oxide (NO) level was measured as described by Green et al. [28], while myeloperoxidase (MPO) activity was assessed by procedures previously described by Granell et al. [29].
Assessment of pro-apoptotic biomarkers expression
Caspase 3 and caspase 9 activities
Caspase activities in the testis and epididymis were measured using specific enzyme-linked immunosorbent assay (ELISA) kits (CUSABIO, Houston, TX, 77054, USA) for caspase-3 (Cat no: CSB-E08857r) and caspase-9 (Cat no: CSB-E08863r). The procedure was done according to the manufacturer’s instructions.
Data analysis
Date obtained from this study were expressed as mean ± Standard Error. Significant differences between the control and envenomed groups were tested using one-way Analysis of Variance and Duncan multiple range test of the treatment groups. Analyzed values were considered significant at P < 0.05. Statistical Package for Social Sciences (SPSS, version 25) software produced by IBM Corp. Ltd. was used for data analysis.
Results
Clinical signs of toxicity, body and organ weight changes, and organo-somatic index
The envenomed animals showed symptoms of toxicity post-envenoming such as incoordination, restlessness, low food consumption, and local bleeding on site of venom injection. Mortality was observed on day 1 post envenoming with 50% recorded in group 2, while 40% deaths was each recorded in groups 3 and 4 (Table 1). However, mortality was not recorded in group 1 (control and group 5 (envenomed and treated with 8 mg/kg of kaempferol) all through the experimental period. The EoV caused substantial decrease (p < 0.05) in body weight of envenomed groups most especially in group 2 (envenomed untreated rats) compared to the control. However, there was substantial increase in the mean body weights of envenomed groups treated with varying doses of kaempferol (Table 2). Also, the organ and relative organ weights of envenomed untreated animals substantially decreased (p < 0.05) in contrast with the control and envenomed treated groups (Table 2). Kaempferol caused significant improvement in the weight of testis and organo-somatic index in the envenomed treated groups (Table 2).
Effect of kaempferol on sperm functional parameters
The percentage of motile sperm and fast progressive of envenomed untreated animals substantially decreased (p < 0.05) in contrast with the control and envenomed groups administered with antiserum and kaempferol (Table 3). However, group treated with varying doses of kaempferol recorded significant percentage in motile sperm and fast progressive which was dose dependent. EoV caused detrimental effect on sperm cells as evident in significant (p < 0.05) decrease in sperm volume and counts in envenomed untreated rats. However, kaempferol countered the effect of the venom and significantly improved the sperm parameters in envenomed treated groups (Table 3).
Effect of kaempferol on sperm cell abnormalities
EoV caused significant damaging effect on the spermatozoa of envenomed untreated rats as evident with higher structural deformities of sperm cell without hook, folded sperm, banana shape and tailless sperm (Table 4). Treatment with varied concentrations of kaempferol substantially decreased (p < 0.05) the sperm cells abnormalities dose dependently, thus decreasing the percentage of sperm abnormalities in the envenomed animals administered with the kaempferol in contrast with antivenom treated and envenomed untreated rats.
Effect of kaempferol on levels of serum reproductive hormones
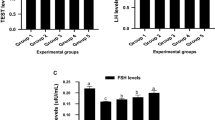
There was significant elevation of follicle stimulating hormone (Fig. 1A), testosterone (Fig. 1B), and luteinizing hormones (Fig. 1C) in sera of envenomed untreated animals in contrast with the control. However, serum hormonal levels significantly decrease (p < 0.05) in the envenomed groups treated with antivenom and varying doses of kaempferol. In comparison, kaempferol at 8 mg/kg best normalized the sera levels of the reproductive hormones in the envenomed treated groups.
Effect of kaempferol on reproductive hormones concentrations in the blood of envenomed treated rats. Data are represented as mean ± SE (n ≥ 5). Bar with different lower-case letter represent significant difference among the groups at p < 0.05 using DMRT. FSH Follicle Stimulating Hormone, TEST Testosterone Hormone, LH Luteinizing Hormone. Group 1: Control (Injected with saline only), Group 2: Venom control (Injected with EoV and not treated), Group 3: Injected with EoV and treated with antivenom (0.2 ml), Group 4: Injected with EoV and treated with kaempferol (4 mg/kg−1), Group 5: Injected with EoV and treated with kaempferol (8 mg/kg−1)
Effect of kaempferol on oxidative stress biomarkers and antioxidant enzymes
Malondialdehyde (MDA) levels
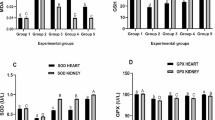
The testis and epididymal MDA levels substantially increased (p < 0.05) in envenomed untreated rats in contrast with the control and envenomed groups treated with antivenom and kaempferol (Fig. 2A). Treatment with varying doses of kaempferol suppressed the MDA levels in reproductive organs of envenomed treated groups, with more effectiveness noticed in group treated 8 mg/kg of kaempferol.
Reduced glutathione (GSH) levels
The EoV caused substantial (p < 0.05) decline in GSH levels in group envenomed and not treated post envenoming. However, kaempferol dose dependently improved the GSH levels in testis and epididymis of the envenomed treated groups (Fig. 2B).
Superoxide dismutase (SOD) activity
The activity of SOD was intense in epididymis and testis of envenomed untreated animals in comparison with the control and envenomed treated groups (Fig. 2C). Treatment with kaempferol caused significant decline in the SOD activity of reproductive organs of envenomed treated rats.
Glutathione peroxidase (GPX) activity
The EoV repressed the epididymal and testicular GPX activity in contrast with the control and group envenomed and treated (Fig. 2D). However, treatment with varying doses of kaempferol reinforced the GPX activity in the envenomed treated groups relative to the antivenom treated groups.
Effects of kaempferol on oxidative stress biomarkers and antioxidant enzymes in the testis and epididymis of envenomed treated rats. Data are represented as mean ± SE (n ≥ 5). Bar with different lower-case letter represent significant difference in stress profile of testis among the groups at p < 0.05 using DMRT. Bar with different upper-case letter represent significant difference in stress profile of epididymis among the groups at p < 0.05 using DMRT. MDA Malondialdehyde, GSH Glutathione, SOD Superoxide dismutase, GPX Glutathione Peroxidase. Group 1: Control (Injected with saline only), Group 2: Venom control (Injected with EoV and not treated), Group 3: Injected with EoV and treated with antivenom (0.2 ml), Group 4: Injected with EoV and treated with kaempferol (4 mg/kg−1), Group 5: Injected with EoV and treated with kaempferol (8 mg/kg−1)
Effect of kaempferol on inflammatory biomarkers
Nitric oxide (NO) levels
The testicular and epididymal NO levels were very intense in group envenomed and not treated in contrast with the control and envenomed treated groups (Fig. 3A). However, treatment of envenomed rats with varying doses of kaempferol and antivenom substantially decreased (p < 0.05) the levels of NO, with intense effect in group treated with high dose of kaempferol.
Myeloperoxidase (MPO) activity
EoV enhanced the MPO activity in the epididymis and testis of envenomed untreated animals relative to the control and envenomed treated groups. However, kaempferol suppressed the MPO activities in a dose dependent effect and high dose of kaempferol (8 mg/kg) was best effective (Fig. 3B).
Effect of kaempferol on inflammatory biomarkers in the testis and epididymis of envenomed treated rats. Data are represented as mean ± SE (n ≥ 5). Bar with different lower-case letter represent significant difference in inflammatory markers of the testis among the groups at p < 0.05 using DMRT. Bar with different upper-case letter represent significant difference in inflammatory markers of epididymis among the groups at p < 0.05 using DMRT. NO Nitric Oxide, MPO Myeloperoxidase. Group 1: Control (Injected with saline only), Group 2: Venom control (Injected with EoV and not treated), Group 3: Injected with EoV and treated with antivenom (0.2 ml), Group 4: Injected with EoV and treated with kaempferol (4 mg/kg−1), Group 5: Injected with EoV and treated with kaempferol (8 mg/kg−1)
Effect of kaempferol on activity of pro-apoptosis biomarkers
Caspase 3 activity
Casp-3 activity in the testis and epididymis of the envenomed untreated group was more prominent in contrast with the control and envenomed treated rats (Fig. 4A). However, kaempferol and antivenom suppressed the activity casp-3 in groups envenomed and treated. The effect was more prominent in envenomed group administered with 8 mg/kg of kaempferol.
Caspase 9 activity
The EoV caused significant enhancement of casp-9 activity in the epididymis and testis of envenomed untreated rats relative to the control and groups treated after venom injection (Fig. 4B). Kaempferol showed substantial (p < 0.05) dose dependent reduction in casp-9 activity with best effect noticed in group treated with 8 mg/kg.
Effect of kaempferol on Caspase 3 and 9 activities in the testis and epididymis of envenomed treated rats. Data are represented as mean ± SE (n ≥ 5). Bar with different lower-case letter represent significant difference in apoptosis markers of the testis among the groups at p < 0.05 using DMRT. Bar with different upper-case letter represent significant difference in apoptosis markers of epididymis among the groups at p < 0.05 using DMRT. Casp-3 Caspase 3, Casp-9 Caspase 9. Group 1: Control (Injected with saline only), Group 2: Venom control (Injected with EoV and not treated), Group 3: Injected with EoV and treated with antivenom (0.2 ml), Group 4: Injected with EoV and treated with kaempferol (4 mg/kg−1), Group 5: Injected with EoV and treated with kaempferol (8 mg/kg−1)
Discussion
The damaging effect of chemicals and toxins on male reproductive functions is of great concern across the globe over the years. The harmful effects of these toxicants and natural toxins can influence the testicular and epididymal functions which could lead to infertility [6, 30]. Snake venom is composed of many toxins predominantly metalloproteinases in viper venoms, exhibiting both local and systemic effects following envenoming in humans [31].
In this current study, the overall assessment of health status of the envenomed animals indicates organ toxicity attributed to EoV toxins. These was evident as clinical manifestation of toxicity such as local bleeding, substantial decline in body and testicular weights including testiculo-somatic index were observed in the envenomed untreated rats which was in tandem with previous studies using viper venoms [6, 32]. The decrease in the testicular weight may be attributed to distortion in metabolic pathways leading to tissue breakdown [33] and/or reduction in number of germ cells, inhibition of spermatogenesis and steroidogenic enzyme activity [13]. However, these toxic effects were ameliorated by kaempferol with significant improvement in the body and organ weights including organo-somatic index of the envenomed treated rats suggesting kaempferol could interact with EoV toxins and prevent biological response on the overall organ system post-envenoming.
Hypothalamic-pituitary-testicular axis and gonadotropin-releasing hormone (GnRH) regulates spermatogenesis in the testes via the secretion of LH and FSH by the pituitary gland [33]. LH and FSH initiates the physiology of reproduction and co-ordinate the synthesis of testosterone to initiates spermatogenesis, sperm production and production of seminal fluid [34]. In this study, EoV elevated sera FSH, testosterone, and LH levels in the envenomed untreated rats suggesting an undesirable response from hypothalamus-pituitary-gonadal axis, which could impair the initiation and completion of spermatogenesis, and subsequently, testicular malfunction [33, 34]. Toxicants including natural toxins are known to influence endocrine parameters resulting in hormonal imbalances by acting on Leydig cells leading to reproductive hormones synthesis disorder [5, 35]. Finding from this study is evidence that the venom is capable of disrupting endocrine functions revealing a pattern of hormonal disorders similar to previous report in viper envenomed male rats [6]. The normalized serum sex hormones levels following treatment of envenomed rats with kaempferol suggests protective effect of kaempferol on venom-induced hormonal disruption to improve spermatogenesis.
Sperm motility and count are vital functional parameters that facilitate fertilization [36]. Results herein showed that the venom was toxic on sperms with substantial decrease in sperm counts, volume, and motility including fast progressive cells in envenomed untreated rats. The observed decrease in the epididymal spermatozoa counts may suggest that the venom alters the processes of spermiogenesis, which substantiates the observed decrease in sperms quantity which was in accordance with previous studies [6, 13]. Also, the decrease in sperm mobility could be linked with reduction in steroid-regulated antioxidant defense in the epididymis [33]. In addition, EoV caused significant detrimental effects on sperm morphology with consequent increase in sperm structural defects which aligned with our previous reports in male rats envenomed with cobra [5] and viper venoms [6]. The reproductive deficits observed may be linked to hormonal imbalance, importantly significant elevation of serum sex hormones which were correlated to decrease in semen characteristics as reported in other study [37]. However, treatment with kaempferol mitigated the venom-induced sperm toxicity by normalizing and maintaining the sperm functional parameters suggesting the positive influence of the kaempferol on epididymal and testicular wellbeing.
Lipid peroxidation is an important biomarker for oxidative damage, where reactive oxygen species (ROS) actuate lipid peroxidation of cellular membrane enhancing MDA levels as a by-product [38]. Oxidative stress is another common toxicity pathway that is initiated by snake venom toxins through rapid production of ROS [39]. EoV induced oxidative stress with substantial elevation of MDA levels in reproductive organs of the envenomed untreated animals which corroborated our earlier study [6]. The resultant impact of oxidative stress in the reproductive organs could affects spermatogenesis, sperm cells and overall reproductive health.
The spermatozoa membranes are susceptible to attack by ROS and LPO due to their richness in polyunsaturated fatty acids. Consequently, peroxidation of sperm membrane lipids could alter the membrane fluidity and firmness resulting to sperm defective structure, immotile, non-viable, DNA damage, sperm destruction, organelle breakdown and cell death [38]. Furthermore, studies have posited that the underlying mechanism of reprotoxicity could be via induction of ROS in cells to cause alterations on intracellular antioxidants, thereby resulting in oxidative stress [13].
Antioxidants play a crucial role via continuous inactivation of ROS to maintain the cell functions and only a small quantity is required for this task [39]. Superoxide dismutase (SOD) is an antioxidant enzyme responsible for direct elimination of ROS through oxygen and hydrogen peroxide generation [39]. The elevated testicular and epididymal SOD activity in untreated envenomed rats could be a response of cells of the immune system to alleviate the venom-actuated oxidative stress, and to maintain oxi-redox balance abating lipid peroxidation [38]. Studies have reported elevated SOD levels in reproductive organs of rats after exposure to toxicants [40].
Glutathione (GSH) is a vital intracellular antioxidant that spontaneously neutralizes the effects of ROS [13]. In this present study, EoV depleted GSH content in the testis and epididymis which made spermatogenic cells more susceptible to attack by free radicals [41]. Glutathione peroxidase (GPx) is known to act against the free radicals in cells of the tissues [42]. However, EoV caused GPx reduction in reproductive organs of the envenomed untreated animals. Therefore, depreciation in GPx activity and GSH content indicates the venom caused the impairment of the enzymatic and non-enzymatic antioxidants functions due to failure of the primary antioxidant system as posited by Ayala et al. [42].
Elevation of myeloperoxidase (MPO) activity and nitric oxide (NO) levels is evidence of potential inflammation associated to reprotoxicity initiated majorly by ROS [33]. The venom induced inflammation as evidenced by enhancement of MPO activity, and NO levels in the epididymis and testes of envenomed untreated rats. Inflammation induced via elevation of NO levels has been implicated in endocrine toxicity [43] as reported in this study. However, treatment of envenomed rats with kaempferol downregulated the inflammatory markers thus, affirming the anti-inflammatory ability of kaempferol against venom induced inflammation [44, 45].
Apoptosis is considered a programmed cell death that is regulated by the caspase family of proteins [46]. Caspase 3 and caspase 9 are important biomarkers indicating apoptosis in organ tissues. Caspase-9 is a known cysteine-aspartic protease, an initiator of intrinsic apoptosis and regulator of physiological cell death including pathological tissue degeneration [47]. On the other hand, caspase 3 is the main effector caspase that is activated by oxidative stress or inflammation to execute apoptosis [48]. EoV induced apoptosis as evidence by enhancement of casp-3 and casp-9 activities in testis and epididymis of envenomed untreated rats which support previous reports on pro-apoptotic effect of EoV in vital organs of envenomed rats [49]. The pro-apoptotic effect of the venom could be associated directly or indirectly to upsurge in ROS generation and inflammation [50] leading to the disruption of spermatogenesis and sperm functional parameters in this study.
The pharmacological attributes of kaempferol against reprotoxicity is demonstrated in this study through alleviation of venom-induced sperm functional deficits, the depreciated antioxidants were reinforced, and the elevated inflammatory markers in testis and epididymis were countered post-kaempferol intervention in envenomed treated rats which substantiated our previous reports [51]. Similarly, enhancement of apoptotic markers of casp-3 and casp-9 activities induced by EoV were suppressed after treatment with kaempferol, thus, suggesting anti-apoptotic effect of kaempferol via downregulation of the initiator casp-3. In our previous study, kaempferol ameliorated male reproductive toxicities induced by cobra venom [51], while quercetin a similar flavonoid like kaempferol have been reported to attenuates toxicants-induced reproductive toxicity in other studies [13, 52]. Studies have reported that the possible ameliorative mechanism of kaempferol on induced toxicity could be through the activation of hemeoxygenase-1 (HO-1), to suppress nitric oxide expression levels and via lipid peroxidation inhibition [53, 54]. Furthermore, kaempferol is known to possess potent antioxidant properties and can react with hydrogen peroxide (H2O2), hypochlorous acid (HOCl), superoxide, nitric oxide with excellent radical scavenging activity through reinforcement of the endogenous antioxidants [53].
Conclusion
Findings from this study highlighted E. ocellatus venom-induced reprotoxicity in envenomed male rats. However, kaempferol antagonized the venom induced-reprotoxicity through enhancement of the sperm parameters, regularization of sex hormone functions, reinforcement of antioxidant system, anti-inflammatory and anti-apoptotic properties in reproductive organs of envenomed treated rats. Thus, establishing the pharmacological potentials of kaempferol in mitigating clinical reprotoxicity that could manifest post E. ocellatus envenoming in snakebite victims.
Data availability
The data generated in the current study is available from the corresponding author on reasonable request.
References
Bouabdallah N, Mallem L, Abdennour C, Chouabbia A, Tektaka M. Toxic impacts of a mixture of three pesticides on the reproduction and oxidative stress in male rats. J Anim Behav Biometeorol. 2022;10:2204. https://doi.org/10.31893/jabb.22004.
Massányi P, Massányi M, Madeddu R, Stawarz R, Lukác N. Effects of Cadmium, lead, and Mercury on the structure and function of Reproductive Organ. Toxics. 2020;8:94. https://doi.org/10.3390/toxics804009.
Agarwal A, Mulgund A, Hamada A, Chyatte MR. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:37. https://doi.org/10.1186/s12958-015-0032-1.
Otasevic V, Stancic A, Korac A, Jankovic A, Korac B. Reactive oxygen, nitrogen, and sulfur species in human male fertility. A crossroad of cellular signaling and pathology. BioFactors. 2020;46(2):206–19. https://doi.org/10.1002/biof.1535.
Ajisebiola BS, Adeniji OB, James AS, Ajayi BO, Adeyi AO. Naja nigricollis venom altered reproductive and neurological functions via modulation of pro-inflammatory cytokines and oxidative damage in male rats. Metabol Open. 2022a;14:100188. https://doi.org/10.1016/j.metop.2022.100188.
Ajisebiola BS, Alamu PI, James AS, Adeyi AO. Echis ocellatus Venom-Induced Reproductive pathologies in Rat Model; roles of oxidative stress and pro-inflammatory cytokines. Toxins. 2022b;14:378. https://doi.org/10.3390/toxins14060378.
Harrison RA, Gutiérrez JM. Priority actions and progress to substantially and sustainably reduce the mortality, morbidity and socioeconomic burden of Tropical Snakebite. Toxins (Basel). 2016;8(12):351. https://doi.org/10.3390/toxins8120351.
Habib AG, Musa BM, Iliyasu G, Hamza M, Kuznik A, Chippaux JP. Challenges and prospects of Snake Antivenom Supply in Sub-saharan Africa. PLoS Negl Trop Dis. 2020;14(8):e0008374. https://doi.org/10.1371/journal.pntd.0008374.
Habib AG, Gebi UI, Onyemelukwe GC. Snake bite in Nigeria. Afri J Med Sci. 2001;30:171–8.
Habib AG. Public health aspects of snakebite care in West Africa: perspectives from Nigeria. J Venom Anim Toxins Incl Trop Dis. 2013;19(1):27. https://doi.org/10.1186/1678-9199-19-27.
Ajisebiola BS, Fawole AB, Adeyi OE, Adeyi AO. An in vivo assessment of inflammatory and oxidative stress responses in Echis ocellatus-venom induced cardiotoxicity. Med Omics. 2022;5–6:100017. https://doi.org/10.1016/j.meomic.2022.100017.
Adeyi AO, Adeyemi SO, Effiong E-OP, Ajisebiola BS, Adeyi OE, James AS. Moringa oleifera Extract extenuates Echis ocellatus Venom-Induced Toxicities, histopathological impairments and inflammation via enhancement of Nrf2 expression in rats. Pathophysiology. 2021;28:98–115. https://doi.org/10.3390/pathophysiology28010009.
Abd-Ellah MF, Aly HAA, Mokhlis HAM, Abdel-Aziz AH. Quercetin attenuates di-(2-ethylhexyl) phthalate-induced testicular toxicity in adult rats. Hum Exp Toxicol. 2016;35(3):232–43. https://doi.org/10.1177/0960327115580602.
WHO. Snakebite envenoming: a strategy for prevention and control. Geneva: World Health Organization. Licence 2019: CC BY-NC-SA 3.0 IGO. Accessed 29 July 2023.
Qattan MY, Khan MI, Alharbi SH, Verma AK, Al-Saeed FA, Abduallah AM, Al Areefy AA. Therapeutic importance of Kaempferol in the treatment of Cancer through the modulation of cell signalling pathways. Molecules. 2022;27:8864. https://doi.org/10.3390/molecules27248864.
Imran M, Salehi B, Sharifi-Rad J, Aslam Gondal T, Saeed F, Imran A, Shahbaz M, Tsouh Fokou PV, Umair Arshad M, Khan H, et al. Kaempferol: a key emphasis to its Anticancer potential. Molecules. 2019;24(12):2277. https://doi.org/10.3390/molecules24122277.
Ajisebiola BS, Oladele JO, Adeyi AO. Kaempferol from Moringa oleifera demonstrated potent antivenom activities via inhibition of metalloproteinase and attenuation of Bitis arietans venom–induced toxicities. Toxicon. 2023;233:107242. https://doi.org/10.1016/j.toxicon.2023.107242.
National Research Council. Guide for the care and use of laboratory animals. Washington, DC, USA: National Academies; 2010.
Adler I. Comparison of the duration of spermatogenesis between male rodents and humans. Mutat Res. 1996;352:169–72. https://doi.org/10.1016/0027-5107(95)00223-5.
Rowett HGO. Dissecting guides of rats with notes on mouse. 111. London: Bulter and tanner LTD; 1997. pp. 5–23.
Freund M, Carol B. Factors affecting haemocytometer count of sperm concentration in human semen. J Reprod Fertil. 1964;8:149–55. https://doi.org/10.1530/jrf.0.0080149.
World Health Organisation. WHO laboratory manual for the examination and processing of human semen. fifth ed. Geneva: WHO (2010). http://www.who.int/iris/handle/10665/44261
Wyrobek AJ, Gordon LA, Burkhart JG, Francis MW, Kapp RW, Letz G, Malling HV, Topham JC, Whorton MD. An evaluation of the mouse sperm morphology test and other sperm tests in non-human mammals. A report of the United States Environmental Protection Agency Gene-Tox Program. Mutat Res Rev Genet Toxicol. 1983;115:1–72. https://doi.org/10.1016/0165-1110(83)90014-3.
Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959;82(1):70–7. https://doi.org/10.1016/0003-9861(59)90090-6.
Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974;47:469–74. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x.
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoektra EG. Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973;179:588–90. https://doi.org/10.1126/science.179.4073.588.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–8. https://doi.org/10.1016/0003-2697(79)90738-3.
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15 N] nitrate in biological fluids. Anal Biochem. 1982;126:131–8. https://doi.org/10.1016/0003-2697(82)90118-x.
Granell S, Gironella M, Bulbena O, Panes J, Mauri M, Sabater L, Aparisi L, Gelpi E, Closa D. Heparin mobilizes xanthine oxidase and induces lung inflammation in acute pancreatitis. Crit Care Med. 2003;31:525–30. https://doi.org/10.1097/01.CCM.0000049948.64660.06.
Selvaraju V, Baskaran S, Agarwal A, Henkel R. Environmental contaminants and male infertility: effects and mechanisms. Andrologia. 2021;53:13646. https://doi.org/10.1111/and.13646.
Williams HF, Mellows BA, Mitchell R, Sfyri P, Layfield HJ, Salamah M, Vaiyapuri R, Collins-Hooper H, Bicknell AB, Matsakas A, Patel K. Mechanisms underpinning the permanent muscle damage induced by snake venom metalloprotease. PLoS Negl Trop Dis. 2019;13(1):e0007041. https://doi.org/10.1371/journal.pntd.0007041.
Alberto-Silva C, De Araujo AC, Bonfim RS, Gilio JM. The peptide fraction of Bothrops jararaca snake venom induces toxicological effects on the male reproductive system after local envenomation in mice. Biocell. 2023;47(2):289–95.
Tijani AS, Farombi EO, Olori DO. Thymol co-administration abrogates hexachlorobenzene-induced reproductive toxicities in male rats. Hum Exp Toxicol. 2023;421–15. https://doi.org/10.1177/09603271221149201.
Fraczek M, Kurpisz M. Inflammatory mediators exert toxic effects of oxidative stress on human spermatozoa. J Androl. 2007;28(2):325–33.
Wang Y, Dong Y, Wu S, Zhu Q, Li X, Liu S, Huang T, Li H, Ge RS. Acephate interferes with androgen synthesis in rat immature leydig cells. Chemosphere 2020:245125597. https://doi.org/10.1016/j.chemosphere.2019.125597
Milardi D, Grande G, Sacchini D, Astorri AL, Pompa G, Giampietro A, De Marinis L, Pontecorvi A, Spagnolo AG, Marana R. Male fertility and reduction in semen parameters: a single tertiary-care center experience. Int J Endocrinol. 2012;649149. https://doi.org/10.1155/2012/649149.
Ollero M, Gil-Guzman E, Lopez MC, Sharma RK, Agarwal A, Larson K, Evenson D, Thomas AJ Jr., Alvarez JG. Characterization of subsets of human spermatozoa at different stages of maturation: implications in the diagnosis and treatment of male infertility. Hum Reprod. 2001;16(9):1912–21. https://doi.org/10.1093/humrep/16.9.1912.
Ritchie C, Ko EY. Oxidative stress in the pathophysiology of male infertility. Andrologia. 2020;23(e13581). https://doi.org/10.1111/and.13581.
Garcia MS, Cavalcante DND, Santiago MDA, de Medeiros PD, do Nascimento CC, Fonseca GFC, Sueur-Maluf LL, Perobelli. JE. Reproductive toxicity in male juvenile rats: antagonistic effects between isolated agrochemicals and in binary or ternary combinations. Ecotoxicol Environ Saf. 2021;209:111766. https://doi.org/10.1016/j.ecoenv.2020.111766.
Sardar A, David M, Jahan S, Afsar T, Ahmad A, Ullah A, Almajwal A, Shafique H, Razak S. Determination of biochemical and histopathological changes on testicular and epididymis tissues induced by exposure to insecticide Imidacloprid during postnatal development in rats. BMC Pharmacol Toxicol. 2023;2468. https://doi.org/10.1186/s40360-023-00709-3.
El-Desoky GE, Bashandy SA, Alhazza IM, Othman ZA, Aboul-Soud MA, Yusuf K. Improvement of mercuric chloride-induced testis injuries and sperm quality deteriorations by Spirulina platensis in rats. PLoS ONE. 2013;8:e59177. https://doi.org/10.1371/journal.pone.0059177.
Ayala A, Muñoz MF, Argüelles S. Lipid peroxidation: production, metabolism, and Signaling mechanisms of Malondialdehyde and 4-Hydroxy2-Nonenal. Oxid Med Cell Longev. 2014;360438. https://doi.org/10.1155/2014/360438.
Dobashi M, Fujisawa M, Yamazaki T, Okuda Y, Kanzaki M, Tatsumi N, Tsuji T, Okada H, Kamidono S. Inhibition of steroidogenesis in Leydig cells by exogenous nitric oxide occurs independently of steroidogenic acute regulatory protein (star) mRNA, Arch. Androl. 2001;47(3):203–9. https://doi.org/10.1080/014850101753145915.
Ajisebiola BS, Mustapha AK, Oyedara OO, Oladele JO, Adeyi AO. Kaempferol alleviates neurodegenerative disorders induced by Naja nigricollis venom via mechanisms of antioxidants, anti-inflammatory, dopaminergic and neuronal functions. Phytomed Plus. 2024;4:100584. https://doi.org/10.1016/j.phyplu.2024.100584.
Hofer S, Geisler S, Lisandrelli R, Ngoc HN, Ganzera M, Schennach H, Fuchs D, Fuchs JE, Gostner JM, Kurz K. Pharmacological targets of kaempferol within inflammatory pathways—a hint towards the central role of tryptophan metabolism. Antioxidants. 2020;9:180. https://doi.org/10.3390/antiox9020180.
Brentnall M, Rodriguez-Menocal L, De Guevara RL, Cepero E, Boise LH. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013;14:32. http://www.biomedcentral.com/1471-2121/14/32.
Avrutsky MI, Troy CM. Caspase-9: a Multimodal Therapeutic Target with Diverse Cellular expression in Human Disease. Front Pharmacol. 2021;12:701301. https://doi.org/10.3389/fphar.2021.701301.
Akhigbe R, Ajayi A. Testicular toxicity following chronic codeine administration is via oxidative DNA damage and upregulation of NO/TNF-α and caspase 3 activities. PLoS ONE. 2020;15(3):e0224052. https://doi.org/10.1371/journal.pone.0224052.
Ajisebiola BS, Durodola BP, Mustapha AK, Oladele JO, Adeyi OA. Cardio-nephrotoxicity mediated by Echis ocellatus venom and its amelioration through kaempferol’s suppressive effect on oxidative stress, inflammation, and apoptosis expression. Clin Phytosci. 2024;10:7. https://doi.org/10.1186/s40816-024-00370-1.
Habib R, Wahdan SA, Gad AM, Azab SS. Infliximab abrogates cadmium-induced testicular damage and spermiotoxicity via enhancement of steroidogenesis and suppression of inflammation and apoptosis mediators. Ecotoxicol Environ Saf. 2019;182:109398. https://doi.org/10.1016/j.ecoenv.2019.109398.
Adeyi AO, Ajisebiola BS, Sanni AA, Oladele JO, Mustapha AK, Oyedara OO, Fagbenro OS. Kaempferol mitigates reproductive dysfunctions induced by Naja nigricollis venom through antioxidant system and anti-inflammatory response in male rats. Sci Rep. 2024;14:3933. https://doi.org/10.1038/s41598-024-54523-w.
Sonmez M, Turk G, Ceribasi S, Ciftci M, Yuce A, Guvenc M, Ozer S, Cay M, Aksakal M. Quercetin attenuates carbon tetrachloride-induced testicular damage in rats. Andrologia. 2013;46(8):848–58. https://doi.org/10.1111/and.12159.
Devi KP, Malar DS, Nabavi FS, Sureda A, Xiao J, Nabavi SM, Dagliaf M. Kaempferol and inflammation: from chemistry to medicine. Pharmacol Res. 2015;99:1–10. https://doi.org/10.1016/j.phrs.2015.05.002.
Lin CW, Chen PN, Chen MK, Yang WE, Tang CH, Yang SF, Hsieh YS. Kaempferol reduces matrix metalloproteinase-2 expression by downregulating erk1/2 and the activator protein-1 signaling pathways in oral cancer cells. PLoS ONE. 2013;8(11):e80883. https://doi.org/10.1371/journal.pone.0080883.
Funding
The authors reported there is no funding associated with the work featured in this article.
Author information
Authors and Affiliations
Contributions
BSA and JOO designed the experiment; BSA, AAT, AOA and JOO guided the study, coordinated and conducted the laboratory experimental work. BSA, JOO and OSF interpreted the results. BSA and AKM analyzed the data. BSA, JOO, AKM, OSF and AOA wrote and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The use of experimental animals for this study strictly followed the guidelines of University of Ibadan-Animal Care and Use Research Ethics Committee (UI-ACUREC/19/0030).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ajisebiola, B.S., Toromade, A.A., Oladele, J.O. et al. Echis ocellatus venom-induced sperm functional deficits, pro-apoptotic and inflammatory activities in male reproductive organs in rats: antagonistic role of kaempferol. BMC Pharmacol Toxicol 25, 46 (2024). https://doi.org/10.1186/s40360-024-00776-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40360-024-00776-0