Abstract
Background
Anthocyanins (AC) showed positive effects on improving the intestinal health and alleviating intestinal pathogen infections, therefore, an experiment was conducted to explore the protective effects of supplemented AC on Salmonella-infected chickens.
Methods
A total of 240 hatchling chickens were randomly allocated to 4 treatments, each with 6 replicates. Birds were fed a basal diet supplemented with 0 (CON, and ST), 100 (ACL) and 400 (ACH) mg/kg of AC for d 60, and orally challenged with PBS (CON) or 109 CFU/bird (ST, ACL, ACH) Salmonella Typhimurium at d 14 and 16.
Results
(1) Compared with birds in ST, AC supplementation increased the body weight (BW) at d 18 and the average daily gain (ADG) from d 1 to 18 of the Salmonella-infected chickens (P < 0.05); (2) AC decreased the number of Salmonella cells in the liver and spleen, the contents of NO in plasma and inflammatory cytokines in ileal mucosa of Salmonella-infected chickens (P < 0.05); (3) Salmonella infection decreased the ileal villi height, villi height to crypt depth (V/C), and the expression of zonulaoccludins-1 (ZO-1), claudin-1, occludin, and mucin 2 (MUC2) in ileal mucosa. AC supplementation relieved these adverse effects, and decreased ileal crypt depth (P < 0.05); (4) In cecal microbiota of Salmonella-infected chickens, AC increased (P < 0.05) the alpha-diversity (Chao1, Pd, Shannon and Sobs indexes) and the relative abundance of Firmicutes, and decreased (P < 0.05) the relative abundance of Proteobacteria and Bacteroidota and the enrichment of drug antimicrobial resistance, infectious bacterial disease, and immune disease pathways.
Conclusions
Dietary AC protected chicken against Salmonella infection via inhibiting the Salmonella colonization in liver and spleen, suppressing secretion of inflammatory cytokines, up-regulating the expression of ileal barrier-related genes, and ameliorating the composition and function of cecal microbes. Under conditions here used, 100 mg/kg bilberry anthocyanin was recommended.
Similar content being viewed by others
Background
Salmonella Typhimurium has strong pathogenicity and causes human and animal infections worldwide. Salmonella infection damages the intestinal mucosal barrier of chicken, causing systemic inflammation and immune system disorders, leading to slow growth, diarrhea and even death [1, 2], thus causes serious economic loss in the animal industries. Additionally, infected chickens transmit Salmonella to humans through contaminated meat, eggs, or other products, causing public health and safety problems [3]. In production, repeated usage of antibiotics has been reported to cause chronic toxicity in animals, destroying the normal gut microbes, and triggering intestinal inflammation [4]. Many countries and regions have banned or restricted the addition of antibiotics in feed. Thus, under “antibiotic-free” production systems the development of a novel nutritional strategy provides a safer way to prevent Salmonella infection in chickens.
Anthocyanins (AC) are widely found in natural berries, fruits, and vegetables, also show positive effects on animal health mainly by anti-inflammatory [5], antioxidant [6], and anti-microbial [7] activities. These properties seem to indicate AC as an effective dietary supplement for alleviating intestinal pathogen infections and improving the intestinal health. Previous studies showed that AC in vitro inhibited the activity of pathogenic bacteria, including Salmonella Typhimurium, Escherichia coli and Staphylococcus aureus [8, 9]. Furthermore, AC improved intestinal health partly through the reduction of barrier permeability and secretion of inflammatory cytokines in mice [10, 11], and by activating the antioxidant adaptive response of intestinal epithelial cells to prevent intestinal inflammation [12, 13]. Our previous study found that supplementation with bilberry AC promoted immune status and suppressed oxidative stress of yellow-feathered chickens [14]. However, alleviation of Salmonella infection in yellow-feathered chickens by dint of the protective effect of AC has not been studied yet.
Therefore, the purpose of this study was to explore the effects of supplementation with bilberry AC on the growth performance, intestinal mucosal barrier and composition of cecal microbes in chickens challenged with Salmonella, in order to provide an explanation for a potential mechanism, thus to promote the application of AC in feed industry and broiler farming.
Material and methods
Animals, diets, and experimental design
Two hundred and forty 1-day-old Lingnan yellow-feathered chickens (male, slow-growing) were randomly allocated into 4 groups with 6 replicates and 10 birds per replicate. The basal diets were formulated in accordance with the recommendation of Nutrient Requirements of Yellow Chickens (Ministry of Agriculture, PRC, 2020) (Table 1) [15].
As shown in Fig. 1, birds in the control group (CON) and Salmonella infected group (ST) were fed the basal diets, and other birds in low dose of AC group (ACL) and high dose of AC group (ACH) were fed the basal diets supplemented with 100 or 400 mg/kg bilberry AC (Tianjin Jianfeng Natural Product R&D Co., Ltd., Tianjin, China; the purity > 36%), respectively. At d 14 and 16, birds in ST, ACL and ACH were orally challenged with 1.0 × 109 colony forming units (CFUs) of Salmonella Typhimurium in phosphate buffer saline (PBS), and birds in the CON were given the same volume of sterile PBS.
Chickens were allowed ad libitum access to fresh water and mashed diets. The temperature of the room was maintained at approximately 34 °C during d 1 to 3, then decreased gradually by 3 °C each week to reach a constant temperature of 26 °C. Artificial light was provided continuously during d 1 to 3, then decreased gradually by 2 h each day to reach a constant illumination time of 16 h. Feed consumption was recorded every day and body weight (BW) was recorded on d 1, 18, 28 and 60. The average daily gain (ADG), average feed intake (ADFI), and feed to gain ratio (F/G) were calculated.
Sample collection
At d 18, 2 birds closest to average BW of the replicate were deprived of feed for 12 h. Blood samples (5 mL) were taken from the wing vein and drawn into vacutainers with anticoagulant (BD™ Vacutainer, Franklin Lakes, NJ, USA), then the birds were euthanized. Plasma was obtained after centrifugation (3000 × g, 10 min) at room temperature. The liver and spleen were collected aseptically after the connective tissues were cleaned out. Ileal segments (around 1 cm long) were quickly fixed in 4% solution of paraformaldehyde. Furthermore, the samples of mucosa were gently scraped from the middle ileum, and cecal contents were collected. All samples were stored immediately at − 80 °C until analysis.
Determination of Salmonella colonization
The colonization of Salmonella in liver and spleen were determined as previously described [16]. Samples were homogenized in sterile PBS containing 0.1% Triton X-100, then diluted to 10%, 1%, and 0.1%. Each sub-dilution (100 μL) was evenly spread on Salmonella-Shigella (SS) agar plates and cultured in a constant temperature incubator (37 °C) until bacteria were grown.
Morphological analysis of ileum
Ileal segments were fixed with polyformaldehyde solution (4%) for more than 48 h, then dehydrated and processed into paraffin sections. Dewaxed sections were rehydrated, mounted, stained with hematoxylin-eosin (H&E), and then observed under the PANNORAMIC SCAN II (3DHISTECH, Budapest, Hungary). Villus height and crypt depth were measured on 5 fields for each section, and the ratio of villus height to crypt depth (V/C) was calculated.
Determination of nitric oxide in plasma and inflammatory cytokines in ileal mucosa
Nitric oxide (NO) concentration in plasma was determined with a commercial kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China); the contents of interleukin 1β (IL-1β), IL-6, IL-8, tumor necrosis factor-α (TNF-α), interferon-β (IFN-β), and IFN-γ in ileal mucosa were determined with chicken ELISA kits (Jiangsu Meimian Industrial Co., Ltd., Zhangjiagang, China) according to the manufacturer’s instructions, respectively.
Quantitative real-time PCR (qRT-PCR) analysis
The mRNA expression of zonula occludens 1 (ZO-1), occludin, claudin-1 and mucin 2 (MUC2) in ileal mucosa were determined. As previously described [17], total RNA was extracted from powdered frozen samples and reverse transcribed using RNAiso Plus and PrimeScript™II 1st Strand cDNA Synthesis Kits (Takara, Tokyo, Japan), respectively. Real-time PCR was performed on the CFX96 RT-PCR Detection System (Bio-Rad, CA, USA). Primers used are shown in Table 2. β-actin was used as a housekeeping gene, and 2–△△Ct method was used to quantify relative mRNA expression levels of genes.
Microbial DNA extraction and 16S rRNA sequencing
Microbial DNA was extracted from cecal contents (FastDNA® Spin Kit for Soil, MP Biomedical, Santa Ana, CA, USA) according to standard procedures. The DNA extract was checked and determined with NanoDrop 2000 UV-vis spectrophotometer (Thermo Scientific, Wilmington, DE, USA). The hypervariable region V3 to V4 of the bacterial 16S rRNA gene was amplified by using the 338F/806R primer pairs (338F: 5′-ACTCCTACGGGAGGCAGCAG-3′; 806R: 5′-GGACTACHVGGGTWTCTAAT-3′) and the sequencing was performed (Illumina NovaSeq PE250, San Diego, CA, USA). Raw sequences were quality-filtered and clustered into operational taxonomic units (OTUs) at 97% similarity by UPARSE (http://www.drive5.com/uparse) version 11.0.667, moreover, the chimeric sequences were identified and removed. Each OTU representative sequences were analyzed by RDP Classifier (https://sourceforge.net/projects/rdp-classifier) version 2.10, based on the release 138 the SILVA database (https://www.arb-silva.de).
Statistical analysis
Statistics of growth performance, bacteria load, histological measurements, biochemical variables, and gene expression levels were examined by one-way analysis of variance (ANOVA) in SPSS 20.0 for Windows (SPSS, Chicago, IL, USA). When treatment effects were significant (P < 0.05). Duncan’s multiple range tests were used to compare the individual means. Tabulated results were shown as means with SEM derived from the ANOVA error mean square.
For data of cecal microbiota, Venn diagram, community barplot, and community heatmap were created with R software (http://www.R-project.org) version 3.3.1 statistics and mapping. Alpha-diversity (Chao1, Pd, Shannon, and Sobs indexes) were calculated using Mothur (https://www.mothur.org) version 1.30.2, and Welch’s t-test was used to compare post-hoc means. Beta-diversity was evaluated by principal co-ordinates analysis (PCoA) to show the similarity and differences of community structures among different samples, and ANOSIM was used to test the significance of separation via R software (http://www.R-project.org) version 3.3.1. The non-parametric Kruskal-Wallis sum-rank test was used to detect the differences of abundance in species among different groups and obtain significantly different species. Wilcoxon Rank-sum test was used to test the difference consistency of the different species in the previous step. Linear discriminant analysis (LDA) of effect size (LEfSe) was used to estimate the impact of these different species on the difference between groups. Using Phylogenetic Investigation of Communities by Reconstruction of Unobserved State (PICRUSt2) (https://github.com/picrust/picrust2) version 2.2.0, the corresponded the Kyoto Encyclopedia of Genes and Genomes (KEGG) Ortholog (KO) information through the OTUs greengene ID to obtain and the relative abundance of KO was calculated. The abundance of pathway levels 2 functional categories was calculated based on the KEGG database (http://www.genome.jp/kegg) and corresponding OTUs abundance. Relative abundance of microbial composition and function were analyzed and visualized using Statistical Analysis of Metagenomic Profiles (STAMP) (https://beikolab.cs.dal.ca/software/STAMP) version 2.1.3 with 95% confidence interval. Welch’s t-test was used to compare post-hoc means.
Results
Growth performance
As shown in Table 3, Salmonella infection decreased (P < 0.05) the BW of birds at d 18, 28 and 60. From d 1 to 18, d 1 to 28 and d 1 to 60, the ADG of birds in the infected birds (ST) were all decreased (P < 0.05), while the F/G increased (P < 0.05), i.e., less efficient compared with both variables in CON. In addition, the ADG from d 1 to 18 was increased and F/G was decreased (P < 0.05) in the ACL and ACH compared with those in the challenged but non-supplemented birds (ST). There was no significant (P > 0.05) difference in average daily feed intake among treatments.
Bacterial load in the liver and spleen
As shown in Table 4, Salmonella infection increased (P < 0.05) the number of Salmonella cells in the liver and spleen of chickens. Compared with the ST-infected birds, supplementation with both 100 mg/kg and 400 mg/kg of AC decreased about 10-fold (P < 0.05) the number of Salmonella cells in the liver and spleen of birds.
Ileal mucosal morphology
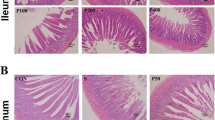
As shown in Table 5, compared with the non-infected controls, both the ileal villus height and V/C were decreased (P < 0.05) in the ST, and the ileal crypt depth was significantly increased. Compared with the infected birds (ST), supplementation with AC increased (P < 0.05) the ileal villus height, and 400 mg/kg of AC treatment reduced ileal crypt depth and increased V/C (P < 0.05). Representative images of H&E staining of ileal mucosa were shown in Fig. 2.
Biochemical variables
As shown in Fig. 3, the plasma NO content of chickens in the ST treatment was higher (P < 0.05) than that of the controls. Compared with the Salmonella-infected birds (ST), supplementation with both 100 mg/kg and 400 mg/kg of AC decreased (P < 0.05) plasma NO content, almost down to the level in control chickens.
As shown in Table 6, Salmonella infection significantly increased the ileal contents of inflammatory cytokines (IL-1β, IL-6, IL-8, TNF-α, IFN-β and IFN-γ) compared with the CON. These variables were all reduced (P < 0.05) in infected birds which supplemented with AC. In addition, the 100 mg/kg of AC treatment significantly increased ileal IL-1β and IL-6 contents in the chickens challenged with Salmonella, compared with the 400 mg/kg of AC treatment.
Gene expression in ileal mucosa
The relative expression of barrier-related genes in ileal mucosa was shown in Fig. 4. Salmonella infection down-regulated (P < 0.05) the expression of ZO-1, claudin-1 and occludin, but had no effect on MUC2 expression (P > 0.05). Compared with birds in ST, 100 mg/kg of AC up-regulated (P < 0.05) the expression of claudin-1 and MUC2, and 400 mg/kg of AC supplementation up-regulated (P < 0.05) the expression of ZO-1 and occludin.
Richness and diversity of cecal microbes
A total of 1,532,777 sequences (an average of 63,866 sequences per sample) were obtained, and the average length of the sequences was 413 bp. Overall, 697 OTUs were detected according to a nucleotide sequence identity of 97% between sequences.
Alpha-diversity of microbial communities were provided in Fig. 5A–D, Salmonella infection decreased (P < 0.05) Chao1, Pd, Shannon, and Sobs indexes compared with the CON. ACL and ACH treatments had higher (P < 0.05) Chao1, Pd, Shannon and Sobs indexes than the infected birds (ST). In addition, the PCoA chart (Fig. 5E) showed that the CON, ACL and ACH treatments, with some similarities between ACL and ACH, had some distinctive separation from the ST treatment.
Effects of bilberry anthocyanin on the average richness and diversity of the cecal bacteria community. A–D The effects of AC on the Alpha-diversity (Chao1, Pd, Shannon, and Sobs indexes). E Principal co-ordinates analysis (PCoA) of bacterial communities in CON, ST, ACL, and ACH. F Venn diagram of bacterial communities in CON, ST, ACL, and ACH. CON, control group; ST, Salmonella infected group; ACL, low dose of anthocyanin group; ACH, high dose of anthocyanin group. Date were shown as mean ± SEM (n = 6), *P < 0.05
The common and unique OTUs among the treatments at the genus level was shown in the Venn diagram (Fig. 5F). One hundred thirty-two OTUs were determined, and there were 100 common OTUs among the four groups. In addition, 3, 5, 5 and 2 OTUs were unique in the CON, ST, ACL and ACH, respectively.
Composition of cecal microbes
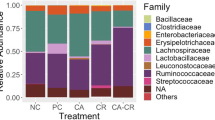
The relative abundance of bacteria at the phylum and genus level were presented in Figs. 6 and 7. Firmicutes, Bacteroidota and Proteobacteria were the top 3 phyla (> 1% at least in 1 of the 4 groups). From Fig. 7A, Salmonella infection decreased (P < 0.05) the relative abundance of Firmicutes, but increased (P < 0.05) the relative abundance of Proteobacteria compared with the CON. In comparison with the infected birds (ST), supplementation with AC (ACL and ACH) increased (P < 0.05) relative abundance of Firmicutes and decreased (P < 0.05) relative abundance of Proteobacteria, moreover, Bacteroidota was decreased (P < 0.05) in ACL and barely changed in ACH. As shown in Fig. 6B, Faecalibacterium, Bacteroides, Lactobacillus, Ruminococcus and Escherichia-Shigella were the major bacterial genera in cecal contents. Compared with the CON, Salmonella infection increased (P < 0.05) the relative abundances of Escherichia-Shigella and Enterococcus, but decreased (P < 0.05) the relative abundances of UCG-005, Shuttleworthia and Lactobacillus. Moreover, compared with birds in ST, in the ACL treatment the relative abundances of Bacteroides, Escherichia-Shigella and Fournierella were decreased (P < 0.05), along with increased (P < 0.05) Lactobacillus, Clostridia_UCG-014, Lachnospiraceae, Alistipes and Shuttleworthia (Fig. 7B); In the higher dose (ACH), the relative abundances of Bacteroides, Escherichia-Shigella and Enterococcus were decreased (P < 0.05), but those of Lactobacillus, Clostridia_UCG-014, Lachnospiraceae, Clostridia_vadinBB60_group, Shuttleworthia, Streptococcus and UCG-005 were increased (P < 0.05).
Relative abundance of bacterial composition in cecal contents at phylum (A) and genus (B) levels. The abscissa shows the name of the experimental group, the ordinate shows the proportion of the bacteria. Each color represents one bacterium, and the heigth of the band represents its percentage. CON, control group; ST, Salmonella infected group; ACL, low dose of anthocyanin group; ACH, high dose of anthocyanin group
The 30 most abundant genera were clustered and analyzed by heatmap and cluster tree (Fig. 8). The results showed that the dominant bacterial communities in ACL, ACH and CON treatment groups were clustered together, and those in infected birds were clearly segregated from the other groups.
Clustering heatmap of bacterial community composition data at the genus level. The color blocks in the Heatmap represent the relative abundance of a genus, and the values were taken as the average values of each group of samples. Gradient from red to blue indicates high to low relative abundance. CON, control group; ST, Salmonella infected group; ACL, low dose of anthocyanin group; ACH, high dose of anthocyanin group
As presented in the cladogram of the microbe structure axis (Fig. 9A), a significant change of microbes was found in CON, ST, ACL and ACH. The results of LEfSe confirmed the higher abundance of Bacteroidota and Proteobacteria in the ST, Firmicutes in the ACL, and Actinobacteriota in the ACH (Fig. 9B). In the ST, 3 genera were significantly more abundant, namely Bacteroides, Escherichia-Shigella, and Enterococcus (Fig. 9C). In addition, Clostridia, ASF356, and DTU089 were significantly more abundant in the CON (Fig. 9C). Similarly, there were significant enrichment of Clostridia_UCG-014, Clostridia, Anaeroplasma, Christensenellaceae_R-7_group, UCG-005, Shuttleworthia, Ruminococcus, Oscillospirales and NK4A213_group in the ACL, and a profusion of Lachnaspiraceae, Fimicutes, Marvinbryantia, Lactobacillales, CHKCI001 and Bifidobacterium in the ACH (Fig. 9C).
Different taxa microbe analysis in cecal contents based on LEfSe method. A Cladogram of the microbe structure axis. The circles from inside to out represent the classification level (phylum, class, order, family, and genus). The color of circles with letters mean that the bacteria was higher at CON, ST, ACL or ACH respectively, the diameter of each circle is proportional to the abundance of the group. LEfSe bar at phylum (B) and genus (C) level. The default parameters were LDA score > 2 and P < 0.05. Different-colored regions represent different constituents (blue: CON, red: ST, green: ACL, pink: ACH). CON, control group; ST, Salmonella infected group; ACL, low dose of anthocyanin group; ACH, high dose of anthocyanin group
Predicted function of cecal microbes
PICRUSt2 metagenome prediction (Fig. 10) showed that Salmonella infection enriched (P < 0.05) 4 pathways (drug antimicrobial resistance, specific cancer types, infectious bacterial disease, and development and regeneration), whereas it decreased (P < 0.05) transcription and endocrine system. Compared with the ST, dietary supplementation with AC (ACL and ACH) increased (P < 0.05) the abundances of 5 pathways, including transcription, ‘folding, sorting and degradation’, amino acid metabolism, lipid metabolism, cell motility and nervous system, while decreased (P < 0.05) the abundances of 9 pathways, including drug antimicrobial resistance, development and regeneration, specific cancer types, infectious bacterial disease, energy metabolism, aging, neurodegenerative disease, digestive system, and transport and catabolism.
Discussion
Growth performance
Salmonella infection causes diarrhea, damaging the immune system and composition of cecal microbes, resulting in seriously limited growth performance of chickens [18]. The present study confirmed Salmonella as a pathogen causing a decrease of growth performance in yellow-feathered chicken. Furthermore, AC alleviated weight loss and the increased F/G caused by Salmonella infection. However, there was no significant difference between 100 mg/kg and 400 mg/kg of AC in improving performance. A previous study showed that supplementation with cranberry AC did not improve growth performance of broilers [19], which was consistent with the research of bilberry AC on chickens [14]. These results indicated that AC did not directly promote growth of chickens and the main effect of AC on improving growth performance in the present study might be indirectly related to alleviating Salmonella infection, which also indicated the potentiality of AC as an antibiotic substitute for prevention of pathogens in poultry industry.
Bacterial load in liver and spleen
Liver and spleen are the main immune organs of birds impacted by Salmonella infection [20], and Salmonella transferred to liver and spleen after colonizing the intestine, resulting in persistent infection in chickens [21, 22]. The present study found that after Salmonella infection, the number of Salmonella cells in the liver and spleen increased, but less so in birds fed diets supplemented with AC. This is in line with the reported anti-bacterial activities of AC, such as preventing Salmonella enterititis and Escherichia coli, which might be related to the ability to target and destroy lipopolysaccharide (LPS) in the cell wall, reducing adhesion and increasing the outflow of ATP from the cytoplasm [10, 23]. On the other hand, Salmonella-induced damages the intestinal barrier resulted in transfer of bacteria from gut to the organs via the bloodstream. These results indicated that AC ameliorated the extent of injury of the intestinal barrier.
Intestinal barrier function
Intestinal barrier function, inflammatory cytokines levels, and composition of microbes together determine the intestinal health. It was worth noting that in some pathogen-infected models, antibiotics not only killed pathogenic bacteria but also affected the structure and diversity of normal microbes, thus had adverse influence on intestinal health [24]. Furthermore, previous studies in mice showed that antibiotic treatment resulted reduction in claudin 4 expression [25], destroyed the integrity of colon, triggered the transfer of pathogens to organs [26], and induced acute inflammation in the cecal mucosa [4]. Therefore, nutritional strategies of antibiotic substitute to improve intestinal health are needed.
The integrity of the intestinal barrier reflects conditions of the health and function of the intestine, and its damage is associated with decreased nutrient digestion and absorption, intestinal inflammation, as well as the transfer of pathogenic microbes and their metabolites such as endotoxin [27]. Salmonella infection causes the rupture and shedding of intestinal villi in chickens. Through the increasing of ileal villus height and V/C and the decreasing of ileal crypt depth, AC efficiently alleviated the ileal mucosal structure damages caused by Salmonella.
ZOs, claudins, occludin and MUC2 are the key components involved in intestinal barrier protection and immune response [28]. Previous studies showed that Salmonella infection induced intestinal barrier injury by suppressing gene expression of tight junction proteins (ZO-1, claudin-1 and occludin) and MUC2 [29, 30]. In the current experiment, dietary AC alleviated the adverse effects of chickens caused by Salmonella infection by up-regulating expression of ZO-1, claudin-1, occludin, and MUC2. Similarly, a previous study showed that substances containing AC reduced the impact of intestinal inflammatory damage by up-regulating the expression of ZO-1, claudin-1, occludin and MUC2 in high-fat diet-induced mice [31]. Based on the above results, AC clearly improved the integrity of the intestinal barrier, thereby alleviating intestinal damage in Salmonella infected chickens.
Intestinal inflammation
Inflammatory responses are self-regulated processes recognizing and eliminating the invading pathogens and restoring the normal physiological function of tissue. Recent studies found that Salmonella infection induced large amounts of NO in macrophages of chickens [32], which promoted inflammatory responses in tissues and activated host immune function [33]. Then, NO reacted with O2− to produce peroxynitrite, leading to lipid peroxidation of cell membranes and damage to DNA. A previous study found that AC reduced LPS-induced NO production in RAW264.7 cells [34], which was consistent with decreasing in plasma NO in current finding, indicating that AC alleviated Salmonella-induced immune responses.
The activation of intestinal mucosal immune function is mainly manifested by the releasing of cytokines. IL-1β is released rapidly in response to bacterial and viral infections and helps stimulate early innate immune responses. IL-6 acts as both proinflammatory and anti-inflammatory, responding quickly to Salmonella invasion [35]. IL-8 induces changes in cell morphology and enhances the antibacterial effect of immune cells. IFN-β affects the proliferation of immune cells and regulates the immune response, providing resistance to Salmonella infection [36]. IFN-γ and TNF-α were essential for the clearance of Salmonella in vivo [37, 38].
A previous study showed that Salmonella infection increased contents of IL-1β, IL-8, IFN-β, and IFN-γ in plasma of chickens [39]. Although the immune response plays an important role in eliminating pathogens, excessive release of inflammatory cytokines causes the destruction of tissue structure [40]. AC showed anti-inflammatory activity in vitro and in vivo, such as reducing the excessive release of pro-inflammatory mediator NO and cytokines (IL-1β, IL-6, IL-8, TNF-α etc.) in RAW 264.7 macrophages, and its intake was a strategy to prevent and inhibit inflammation [41, 42]. Also, bilberry extract inhibited intestinal inflammation in mice, reduced intestinal bleeding and improved intestinal histological structure [43]. In the current experiment, AC at a level of 100 mg/kg in the diet decreased the ileal cytokines (IL-1β, IL-6, IL-8, TNF-α, IFN-β, and IFN-γ) production in chickens challenged with Salmonella.
These results indicated that AC played a significant anti-inflammatory role by inhibiting the release of proinflammatory mediator and cytokines to reduce the inflammatory damage in the ileum, thereby limiting the infection of Salmonella.
Diversity of intestinal microbiota
Salmonella infection inhibited the colonization of other normal microbiota in the ileum of chickens [44] and affected the composition and diversity of the ileal and cecal microbiota [32]. In the present research, Salmonella infection caused a decrease in cecal microbial diversity. Dietary AC increased alpha-diversity including Chao1, Pd, Shannon, and Sobs indexes, which might contribute to the richer and more stable intestinal microbes. Using β-diversity to indicate the degree of similarity between microbial communities, dietary supplementation with AC affected the structure of cecal microbes, making it different from that in Salmonella-infected chickens. The author inferred that the increase of diversity might be correlated with improving the integrity of intestinal barrier and the development of normal microbiota in Salmonella-infected chickens by dietary AC. Moreover, abundant hindgut microbes were beneficial to the ability to degrade AC that were not absorbed by the upper digestive tract, and the metabolites produced would be further utilized by intestinal epithelial cells [45].
Composition of intestinal microbiota
Firmicutes, Bacteroidetes and Proteobacteria were the major phyla of all detected in chickens [46]. Salmonella infection resulted in the increasing of pathogenic and facultative anaerobic bacteria in the cecal microbiota of chickens, which destabilized the microbiota [47]. Supplementation with AC were effective in regulating the composition of cecal microbes in mice [48, 49]. In the present experiment, Firmicutes was increased and Proteobacteria was decreased by supplementation with AC. Proteobacteria contains various of pathogens such as Salmonella, Escherichia coli, and Shigella. At the genus level, Escherichia-shigella in Proteobacteria and Enterococcus were dominant in infected chickens; these were potentially harmful bacteria, triggering inflammation through up-regulating Hsps and inflammation genes [50, 51]. In addition, Escherichia-Shigella was correlated negatively with growth in chickens [29], and it activated NLRP3 inflammasomes and induced inflammation in broilers, associating with up-regulating IL-6 expression [52, 53]. In the current experiment, AC supplementation decreased the abundance of these pathogens, thus alleviated of intestinal disease. Also, AC increased the abundance of beneficial bacteria, such as Bacteroidetes, Lactobacillus, Clostridium etc. Bacteroidetes was participated in the metabolism of feeding, maintained the stability of intestinal environment [54, 55], and inhibited the growth of pathogenic bacteria by producing antimicrobial peptides [56]. Lactobacillus inhibited the proliferation of other bacterial species by synthesizing biotin and producing lactic acid through fermentation, which thereby reducing intestinal pH and inhibiting the activity of pathogens [57, 58]. In addition, Bacteroides, Clostridium, Ruminococcus, Alistipes and Lactobacillus hydrolyzed starch and other macromolecules to form short-chain fatty acids through fermentation; these end-products showed anti-inflammatory activity when absorbed [46]. The increase of these beneficial bacteria thereby alleviating intestinal inflammation caused by Salmonella infection.
Functional prediction of intestinal microbiota
Using PICRUSt2 to predict the potential function of microbial communities, Salmonella infection significantly enriched diseased-related pathways, such as drug antimicrobial resistance and infectious bacterial disease. Virulence genes and antibiotic resistance enhanced virulence of bacteria [3] and brought benefits to Salmonella survival in adverse environments [59]. The reduction of those enriched pathways in dietary AC group indicated that AC might inhibit the intestinal colonization and transfer of Salmonella-infected chickens by reducing the expression of Salmonella virulence and drug resistance genes.
Moreover, genomic prediction showed that, dietary AC significantly increased the enrichment of amino acid metabolism, lipid metabolism, and decreased the enrichment of transport and catabolism, and energy metabolism in Salmonella-infected chickens. The author inferred that the changes in these pathways might relate to the reduction of weight loss in Salmonella-infected chickens by dietary AC. After Salmonella invading the intestine, the intestinal mucosal immune system of host was activated through recognition of pathogen-associated molecular patterns such as flagellin and LPS [60]. In the present experiment, 100 mg/kg of AC decreased the enrichment of immune disease in Salmonella-infected chickens, which might contain some connection between reduction in immune system activation and secretion of immune cytokines caused by infection. Overall, dietary AC offset the negative effects caused by Salmonella infection through comprehensive regulation on microbiota composition and function.
Conclusion
Salmonella Typhimurium infection of starter-phase chickens reduced weight gain, caused severe ileal inflammatory injury, and destroyed the optimal composition of cecal microbes. Dietary AC reduced Salmonella colonization in liver and spleen of challenged chickens, decreased secretion of inflammatory cytokines, up-regulated the expression of ileal genes encoding mucosal tight junction and mucin proteins, and modulated the composition of cecal microbes. In the current study, there was no significant difference between 100 and 400 mg/kg of AC in the parameters measured of Salmonella-infected chickens, therefore 100 mg/kg of AC was recommended to diets in broiler production to combat Salmonella infection.
Availability of data and materials
All data generated or analyzed during this study are available from the corresponding author on request.
Abbreviations
- AC:
-
Anthocyanins
- ACH:
-
High dose of anthocyanin group
- ACL:
-
Low dose of anthocyanin group
- ADG:
-
Average daily gain
- ANOVA:
-
Analysis of variance
- BW:
-
Body weight
- CFU:
-
Colony forming unit
- CON:
-
Control group
- F/G:
-
Feed to gain ratio
- H&E:
-
Hematoxylin-eosin
- IFN-β:
-
Interferon-β
- IFN-γ:
-
Interferon-γ
- IL-1β:
-
Interleukin 1β
- IL-6:
-
Interleukin 6
- IL-8:
-
Interleukin 8
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- KO:
-
KEGG Ortholog
- LDA:
-
Linear discriminant analysis
- LESfe:
-
Linear discriminant analysis of effect size
- LPS:
-
Lipopolysaccharide
- MUC2:
-
Mucin 2
- NO:
-
Nitric oxide
- OTUs:
-
Operational taxonomic units
- PBS:
-
Phosphate buffer saline
- PCoA:
-
Principal co-ordinates analysis
- PICRUSt2:
-
Phylogenetic Investigation of Communities by Reconstruction of Unobserved States
- qRT-PCR:
-
Quantitative real-time PCR
- SEM:
-
Standard error of the mean
- ST:
-
Salmonella infected group
- STAMP:
-
Statistical Analysis of Metagenomic Profiles
- TNF-α:
-
Tumor necrosis factor-α
- V/C:
-
The ratio of villus height to crypt depth
- ZO-1:
-
Zonula occluden 1
References
Dar MA, Urwat U, Ahmad SM, Ahmad R, Kashoo ZA, Dar TA, et al. Gene expression and antibody response in chicken against Salmonella Typhimurium challenge. Poultry Sci. 2019;98(5):2008–13.
Lamas A, Miranda JM, Regal P, Vázquez B, Franco CM, Cepeda A, et al. A comprehensive review of non-enterica subspecies of Salmonella enterica. Microbiol Res. 2018;206:60–73.
Hai D, Yin XP, Lu ZX, Lu FX, Zhao HZ, Bie XM. Occurrence, drug resistance, and virulence genes of Salmonella isolated from chicken and eggs. Food Control. 2020;113:107109.
Rivera-Chávez F, Zhang LF, Faber F, Lopez CA, Byndloss MX, Olsan EE, et al. Depletion of butyrate-producing Clostridia from the gut microbiota drives an aerobic luminal expansion of Salmonella. Cell Host Microbe. 2016;19(4):443–54.
Phan MAT, Bucknall M, Arcot J. Effect of different anthocyanidin glucosides on lutein uptake by Caco-2 cells, and their combined activities on anti-oxidation and anti-inflammation in vitro and ex vivo. Molecules. 2018;23(8):2035.
Kim I, Lee J. Variations in anthocyanin profiles and antioxidant activity of 12 genotypes of mulberry (Morus spp.) fruits and their changes during processing. Antioxidants. 2020;9(3):242.
Genskowsky E, Puente LA, Pérez-Álvarez JA, Fernández-López J, Muñoz LA, Viuda-Martos M. Determination of polyphenolic profile, antioxidant activity and antibacterial properties of maqui [Aristotelia chilensi s (Molina) Stuntz] a Chilean blackberry. J Sci Food Agr. 2016;96(12):4235–42.
Deng HT, Zhu JY, Tong YQ, Kong YW, Tan C, Wang MY, et al. Antibacterial characteristics and mechanisms of action of Aronia melanocarpa anthocyanins against Escherichia coli. LWT-Food Sci Techonol. 2021;150:112018.
Zhou TT, Wei CH, Lan WQ, Zhao Y, Pan YJ, Sun XH, et al. The effect of Chinese wild blueberry fractions on the growth and membrane integrity of various foodborne pathogens. J Food Sci. 2020;85(5):1513–22.
Gao J, Yu WC, Zhang CJ, Liu HW, Fan JG, Wei J. The protective effect and mechanism of Aornia melanocarpa Elliot anthocyanins on IBD model mice. Food Biosci. 2021;41:101075.
Chen T, Hu SH, Zhang HW, Guan QF, Yang YH, Wang XM. Anti-inflammatory effects of Dioscorea alata L. anthocyanins in a TNBS-induced colitis model. Food Funct. 2017;8(2):659–69.
Bashllari R, Molonia MS, Muscarà C, Speciale A, Wilde PJ, Saija A, et al. Cyanidin-3-O-glucoside protects intestinal epithelial cells from palmitate-induced lipotoxicity. Arch Physiol Biochem. 2020;6:1–8.
Ershad M, Shigenaga MK, Bandy B. Differential protection by anthocyanin-rich bilberry extract and resveratrol against lipid micelle-induced oxidative stress and monolayer permeability in Caco-2 intestinal epithelial cells. Food Funct. 2021;12(7):2950–61.
Wang YB, Ma XY, Ye JL, Zhang S, Chen ZL, Jiang SQ. Effects of dietary supplementation with bilberry extract on growth performance, immune function, antioxidant capacity, and meat quality of yellow-feathered chickens. Animals. 2021;11(7):1989.
Ministry of Agriculture of the People’s Republic of China (PRC). Nutrient requirements of yellow chicken. Beijing: China Agricultural Press; 2020.
Xu XG, Gong L, Wang BK, Wu YP, Wang Y, Mei XQ, et al. Glycyrrhizin attenuates Salmonella enterica Serovar Typhimurium infection: new insights into its protective mechanism. Front Immunol. 2018;9:2321.
Wang YB, Wang Y, Lin XJ, Gou ZY, Fan QL, Jiang SQ. Effects of Clostridium butyricum, sodium butyrate, and butyric acid glycerides on the reproductive performance, egg quality, intestinal health, and offspring performance of yellow-feathered breeder hens. Front Microbiol. 2021;12:657542.
Alaeldein MA, Mashael RA, Manal MA, Abdulaziz A, Gamal MS, Ali R, et al. Comparative effects of Bacillus subtilis and Bacillus licheniformis on live performance, blood metabolites and intestinal features in broiler inoculated with Salmonella infection during the finisher phase. Microb Pathog. 2020;139:103870.
Leusink G, Rempel H, Skura B, Berkyto M, White W, Yang Y, et al. Growth performance, meat quality, and gut microflora of broiler chickens fed with cranberry extract. Poultry Sci. 2010;89(7):1514–23.
Zhang S, Shen YR, Wu S, Xiao YQ, He Q, Shi SR. The dietary combination of essential oils and organic acids reduces Salmonella enteritidis in challenged chicks. Poultry Sci. 2019;98(12):6349–55.
Zeng JX, Lei CW, Wang YL, Chen YP, Zhang XZ, Kang ZZ, et al. Distribution of Salmonella Enteritidis in internal organs and variation of cecum microbiota in chicken after oral challenge. Microb Pathog. 2018;122:174–9.
Wang G, Song QL, Huang S, Cai S, Wang YM, Yu HT, et al. Effect of antimicrobial peptide microcin J25 on growth performance, immune regulation, and intestinal microbiota in broiler chickens challenged with Escherichia coli and Salmonella. Animals. 2020;10(2):345.
Lacombe A, Wu VC, White J, Tadepalli S, Andre EE. The antimicrobial properties of the lowbush blueberry (Vaccinium angustifolium) fractional components against foodborne pathogens and the conservation of probiotic Lactobacillus rhamnosus. Food Microbiol. 2012;30(1):124–31.
Duan H, Yu L, Tian F, Zhai Q, Fan L, Chen W. Antibiotic-induced gut dysbiosis and barrier disruption and the potential protective strategies. Crit Rev Food Sci Nutr. 2022;62(6):1427–52.
Ran Y, Fukui H, Xu X, Wang X, Ebisutani N, Tanaka Y, et al. Alteration of colonic mucosal permeability during antibiotic-induced dysbiosis. Int J Mol Sci. 2020;21(17):6108.
Guo WN, Zhou X, Li XR, Zhu QF, Peng J, Zhu B, et al. Depletion of gut microbiota impairs gut barrier function and antiviral immune defense in the liver. Front Immunol. 2021;12:636803.
Hernández-Ramírez JO, Nava-Ramírez MJ, Merino-Guzmán R, Téllez-Isaías G, Vázquez-Durán A, Méndez-Albores A. The effect of moderate-dose aflatoxin B1 and Salmonella enteritidis infection on intestinal permeability in broiler chickens. Mycotoxin Res. 2020;36(1):31–9.
Murai A, Kitahara K, Terada H, Ueno A, Ohmori Y, Kobayashi M, et al. Ingestion of paddy rice increases intestinal mucin secretion and goblet cell number and prevents dextran sodium sulfate-induced intestinal barrier defect in chickens. Poultry Sci. 2018;97(10):3577–86.
Zhang BB, Li G, Shahid MS, Gan LP, Fan H, Lv ZP, et al. Dietary L-arginine supplementation ameliorates inflammatory response and alters gut microbiota composition in broiler chickens infected with Salmonella enterica serovar Typhimurium. Poultry Sci. 2020;99(4):1862–74.
Song J, Li QH, Everaert N, Liu RR, Zheng MQ, Zhao GP, et al. Effects of inulin supplementation on intestinal barrier function and immunity in specific pathogen-free chickens with Salmonella infection. J Anim Sci. 2020;98(1):skz396.
Tian B, Zhao JH, Zhang M, Chen ZF, Ma QY, Liu HC, et al. Lycium ruthenicum anthocyanins attenuate high-fat diet-induced colonic barrier dysfunction and inflammation in mice by modulating the gut microbiota. Mol Nutr Food Res. 2021;65(8):e2000745.
He Y, Yang YY, Dong YY, Yan CL, Zhang BK. The effects of flavomycin and colistin sulfate pre-treatment on ileal bacterial community composition, the response to Salmonella typhimurium and host gene expression in broiler chickens. Microorganisms. 2019;7(11):574.
Jiang PP, Yang WT, Jin YB, Huang HB, Shi CW, Jiang YL, et al. Lactobacillus reuteri protects mice against Salmonella typhimurium challenge by activating macrophages to produce nitric oxide. Microb Pathog. 2019;137:103754.
Hu C, Cai YZ, Li W, Corke H, Kitts DD. Anthocyanin characterization and bioactivity assessment of a dark blue grained wheat (Triticum aestivum L. cv. Hedong Wumai) extract. Food Chem. 2007;104(3):955–61.
Waititu SM, Yitbarek A, Matini E, Echeverry H, Kiarie E, Rodriguez-Lecompte JC, et al. Effect of supplementing direct-fed microbials on broiler performance, nutrient digestibilities, and immune responses. Poultry Sci. 2014;93(3):625–35.
Li XC, Zhang P, Jiang XS, Hu HR, Yang CW, Zhang ZR, et al. Differences in expression of genes in the MyD88 and TRIF signalling pathways and methylation of TLR4 and TRIF in Tibetan chickens and DaHeng S03 chickens infected with Salmonella enterica serovar enteritidis. Vet Immunol Immunopathol. 2017;189:28–35.
Wang BK, Ye XL, Zhou YH, Zhao PW, Mao YL. Glycyrrhizin attenuates Salmonella Typhimurium-induced tissue injury, inflammatory response, and intestinal dysbiosis in C57BL/6 mice. Front Vet Sci. 2021;8:648698.
Li NY, Ansari AR, Sun ZJ, Huang HB, Cui L, Hu YF, et al. Toll like receptor 4 signaling pathway participated in Salmonella lipopolysaccharide-induced spleen injury in young chicks. Microb Pathog. 2017;112:288–94.
Liu N, Lin L, Wang JQ, Zhang FK, Wang JP. Tetramethylpyrazine supplementation reduced Salmonella Typhimurium load and inflammatory response in broilers. Poultry Sci. 2019;98(8):3158–64.
Horiguchi H, Loftus TJ, Hawkins RB, Raymond SL, Stortz JA, Hollen MK, et al. Innate immunity in the persistent inflammation, immunosuppression, and catabolism syndrome and its implications for therapy. Front Immunol. 2018;9:595.
Karunarathne WAHM, Lee KT, Choi YH, Jin CY, Kim GY. Anthocyanins isolated from Hibiscus syriacus L. attenuate lipopolysaccharide-induced inflammation and endotoxic shock by inhibiting the TLR4/MD2-mediated NF-κB signaling pathway. Phytomedicine. 2020;76:153237.
Lee SG, Brownmiller CR, Lee SO, Kang HW. Anti-inflammatory and antioxidant effects of anthocyanins of Trifolium pratense (red clover) in lipopolysaccharide-stimulated RAW-267.4 macrophages. Nutrients. 2020;12(4):1089.
Martin DA, Bolling BW. A review of the efficacy of dietary polyphenols in experimental models of inflammatory bowel diseases. Food Funct. 2015;6(6):1773–86.
Pedroso AA, Lee MD, Maurer JJ. Strength lies in diversity: how community diversity limits Salmonella abundance in the chicken intestine. Front Microbiol. 2021;12:694215.
Krga I, Tamaian R, Mercier S, Celine B, Monfoulet LE, Glibetic M, et al. Anthocyanins and their gut metabolites attenuate monocyte adhesion and transendothelial migration through nutrigenomic mechanisms regulating endothelial cell permeability. Free Radical Bio Med. 2018;124:364–79.
Pandit RJ, Hinsu AT, Patel NV, Koringa PG, Jakhesara SJ, Thakkar JR, et al. Microbial diversity and community composition of caecal microbiota in commercial and indigenous Indian chickens determined using 16s rDNA amplicon sequencing. Microbiome. 2018;6(1):115.
Hayashi RM, Lourenço MC, Kraieski AL, Raquel BA, Gonzalez-Esquerra R, Leonardecz E, et al. Effect of feeding Bacillus subtilis spores to broilers challenged with Salmonella enterica serovar Heidelberg Brazilian strain UFPR1 on performance, immune response, and gut health. Front Vet Sci. 2018;5:13.
Sun MB, Li D, Hua M, Miao XY, Su Y, Chi YP, et al. Black bean husk and black rice anthocyanin extracts modulated gut microbiota and serum metabolites for improvement in type 2 diabetic rats. Food Funct. 2022;13(13):7377–91.
Song HZ, Shen XC, Deng R, Zhang Y, Zheng XD. Dietary anthocyanin-rich extract of açai protects from diet-induced obesity, liver steatosis, and insulin resistance with modulation of gut microbiota in mice. Nutrition. 2021;86:111176.
Yang YT, Li X, Cao ZH, Qiao YG, Lin QY, Liu JP, et al. Effects of different ambient temperatures on caecal microbial composition in broilers. Pol J Microbiol. 2021;70(1):33–43.
Kong C, Gao RY, Yan XB, Huang LS, Qin HL. Probiotics improve gut microbiota dysbiosis in obese mice fed a high-fat or high-sucrose diet. Nutrition. 2019;60:175–84.
Liu QX, Zhou Y, Li XM, Ma DD, Shuang X, Feng HF, et al. Ammonia induce lung tissue injury in broilers by activating NLRP3 inflammasome via Escherichia/Shigella. Poultry Sci. 2020;99(7):3402–10.
Oakley BB, Kogut MH. Spatial and temporal changes in the broiler chicken cecal and fecal microbiomes and correlations of bacterial taxa with cytokine gene expression. Front Vet Sci. 2016;3:11.
Jandhyala SM, Talukdar R, Subramanyam C, Vuyyuru H, Sasikala M, Nageshwar Reddy D. Role of the normal gut microbiota. World J Gastroenterol. 2015;21(29):8787–803.
Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr. 2018;57(1):1–24.
Collins FW, O’Connor PM, O’Sullivan O, Rea MC, Hill C, Ross RP. Formicin–a novel broad-spectrum two-component lantibiotic produced by Bacillus paralicheniformis APC 1576. Microbiology. 2016;162(9):1662–71.
Pan D, Yu ZY. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes. 2014;5(1):108–19.
Neal-McKinney JM, Lu XN, Duong T, Larson CL, Call DR, Shah DH, et al. Production of organic acids by probiotic Lactobacilli can be used to reduce pathogen load in poultry. PLoS One. 2018;7(9):e43928.
Lei CW, Zhang Y, Kang ZZ, Kong LH, Tang YZ, Zhang AY, et al. Vertical transmission of Salmonella enteritidis with heterogeneous antimicrobial resistance from breeding chickens to commercial chickens in China. Vet Microbiol. 2020;240:108538.
Park JH, Kim IH. Supplemental effect of probiotic Bacillus subtilis B2A on productivity, organ weight, intestinal Salmonella microflora, and breast meat quality of growing broiler chicks. Poultry Sci. 2014;93(8):2054–9.
Acknowledgements
We are thankful to W. Bruce Currie (Emeritus Professor) from Cornell University for his suggestions on presentation.
Funding
This work was financially supported by Natural Science Foundation from Guangdong Province (2021A1515010830, 2021A1515012412), National Key R&D Project (2018YFD0500600, 2021YFD300404), China Agriculture Research System of MOF and MARA (CARS-41), the Key Realm R&D Program of Guangdong Province (2020B0202090004), National Natural Science Foundation of China (31802104), the Science and Technology Program of Guangdong Academy of Agricultural Sciences (202106TD, R2019PY-QF008), P. R. China.
Author information
Authors and Affiliations
Contributions
Conceptualization, YBW and SQJ; Methodology, SZ, QLF and JLY; Formal analysis, XJL and ZYG; Writing, SZ; Supervision, YBW and SQJ; Funding acquisition, YBW and SQJ. The author(s) read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Experimental procedures followed to guidelines established by the Institutional Animal Care and Use Committee, Guangdong Academy of Agricultural Sciences in China (Number: GAASISA-2019-009).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, S., Wang, Y., Ye, J. et al. Dietary supplementation of bilberry anthocyanin on growth performance, intestinal mucosal barrier and cecal microbes of chickens challenged with Salmonella Typhimurium. J Animal Sci Biotechnol 14, 15 (2023). https://doi.org/10.1186/s40104-022-00799-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40104-022-00799-9