Abstract
Background
Antimicrobial resistance (AMR) is accelerated by widespread and inappropriate use of antimicrobials. Many countries, including those in low- and middle- income contexts, have started implementing interventions to tackle AMR. However, for many interventions there is little or no economic evidence with respect to their cost-effectiveness. To help better understand the scale of this evidence gap, we conducted a systematic literature review to provide a comprehensive summary on the value for money of different interventions affecting AMR.
Methods
A systematic literature review was conducted of economic evaluations on interventions addressing AMR. a narrative synthesis of findings was produced. Systematic searches for relevant studies were performed across relevant databases and grey literature sources such as unpublished studies, reports, and other relevant documents. All identified economic evaluation studies were included provided that they reported an economic outcome and stated that the analysed intervention aimed to affect AMR or antimicrobial use in the abstract. Studies that reported clinical endpoints alone were excluded. Selection for final inclusion and data extraction was performed by two independent reviewers. A quality assessment of the evidence used in the included studies was also conducted.
Results
28,597 articles were screened and 35 articles were identified that satisfied the inclusion criteria. The review attempted to answer the following questions: (1) What interventions to address AMR have been the subject of an economic evaluation? (2) In what types of setting (e.g. high-income, low-income, regions etc.) have these economic evaluations been focused? (3) Which interventions have been estimated to be cost-effective, and has this result been replicated in other settings/contexts? (4) What economic evaluation methods or techniques have been used to evaluate these interventions? (5) What kind and quality of data has been used in conducting economic evaluations for these interventions?
Discussion
The review is one of the first of its kind, and the most recent, to systematically review the literature on the cost-effectiveness of AMR interventions. This review addresses an important evidence gap in the economics of AMR and can assist AMR researchers’ understanding of the state of the economic evaluation literature, and therefore inform future research.
Systematic review registration PROSPERO (CRD42020190310).
Similar content being viewed by others
Introduction
The World Health Organization (WHO) listed antimicrobial resistance (AMR) as one of the top ten threats to global health in 2019, with estimates that AMR could cause 10 million deaths per year by 2050 [1, 2]. AMR occurs when pathogens (bacteria, viruses, fungi and parasites) develop a resistance or tolerance to the medicines that are used to combat these microorganisms, resulting in treatments becoming less effective or ineffective [3]. The COVID-19 pandemic illustrated how quickly the spread of new pathogens with no effective treatment can take place, and how devastating the effects can be. AMR is already thought to be responsible for millions of deaths each year [4], and although AMR is a naturally occurring phenomenon, the rate at which it occurs is impacted by exposure pathogens to antimicrobial agents and their selective pressure [5]. There are numerous documented cases of pathogens developing AMR, with some of the pathogens responsible for the most deaths globally including methicillin-resistant Staphylococcus aureus (MRSA), drug-resistant tuberculosis and drug-resistant Escherichia coli [4, 6].
AMR has increased in low-, middle- and high-income countries around the world in recent years and this pattern is expected to continue [7,8,9]. Klein et al. [10] conducted a trend analysis on antibiotic consumption between 2005 and 2015 in 76 countries. The results indicate that between this time period, antibiotic consumption rose globally by 65% (measured by defined daily doses [DDD], a standard drug intake metric), primarily driven by increases in consumption in low-and middle-income countries (LMICs); estimates suggest a 77% increase in antibiotic consumption rate per 1,000 inhabitants in these regions. In many cases, antibiotics are overused or misused (e.g. the use of antibiotics for common viral infections like the flu in humans and as growth promoters in farm animals) [11,12,13]. Though antibiotic use is rising in LMICs, lack of access to appropriate and effective antimicrobials is a continuing problem in these settings, due to issues with affordability, supply chains and substandard and falsified medicines, amongst others [14, 15].
AMR has been recognised as a One Health issue by key international organisations [16], interventions to reduce AMR should consider its multisectoral nature, recognising the links between antimicrobial use in humans, animals and the environment [17]. The WHO’s 2015 Global Action Plan on AMR identified several key methods for reducing AMR as a threat, examples of which included: optimisation of the use of antimicrobials in both human and animal health; reducing infections, through effective sanitation, hygiene and other infection prevention measures; sustainable investment in the development of new antimicrobials, diagnostic tools and other interventions [18].
Health technology assessment (HTA) is a multidisciplinary process that uses explicit methods to determine the value of health technologies or interventions at different points throughout the life cycle of the technology or intervention [19]. HTA often involves a health economic evaluation component which can produce comparative estimates of the cost-effectiveness of health interventions. In other words, economic evaluations can aid policymakers to understand whether certain interventions offer better value-for-money compared to other options. The Global Action Plan on AMR emphasised the need to promote more economic evidence-based use of interventions in development of a financial case for investment in AMR diagnostics and treatments [18]. However, some AMR interventions or initiatives have little or no evidence concerning their relative costs and benefits; such as educational and awareness raising interventions, or animal and environmental interventions. This is an issue of significant importance, particularly in more resource-constrained settings that need to use their budgets efficiently and where the burdens of AMR are rapidly rising [20].
The most recent systematic review on the cost-effectiveness of measures to contain the occurrence of AMR, looking at any type of intervention, dates back to 2002 [21], the study demonstrated that there was limited economic evidence available for AMR interventions. Since this review, research in the area has grown, although systematic reviews of evidence have focussed solely on the impact of antimicrobial stewardship programmes (ASPs) in hospitals, or the economic burden of AMR [22, 23].
Objectives
A broad systematic review was conducted with the aim to summarise and detail data from economic evaluations regarding the value-for-money of interventions impacting AMR as a step towards optimising resource use. Specifically, this review attempted to answer the following questions:
-
Objective 1 What interventions to address AMR have been the subject of an economic evaluation?
-
Objective 2 In what types of setting (e.g. high-income, low-income, regions etc.) have these economic evaluations been focused?
-
Objective 3 Which interventions have been estimated to be cost-effective, and has this result been replicated in other settings/contexts?
-
Objective 4 What economic evaluation methods or techniques have been used to evaluate these interventions?
-
Objective 5 What kind of data has been used in conducting economic evaluations for these interventions? What is the quality of this data?
Materials and methods
Search strategy and study selection
Search strategies were designed for accessing MEDLINE (Ovid), EMBASE (embase.com), Cochrane Library, Web of Science, Tufts Cost Effectiveness Analysis (CEA) Registry and Global Health (GH) CEA Registry, Centre for Reviews and Dissemination’s National Health Service Economic Evaluation Database (NHS-EED). Grey literature searches were also conducted, including using international conference databases for the following conferences: Health Technology Assessment International (HTAi), International Society of Pharmacoeconomics and Outcomes Research (ISPOR), International Health Economics Association (iHEA). Websites and databases from HTA or regulatory agencies such as the National Institute for Health and Care Excellence (NICE) or the Canadian Agency for Drugs and Technologies in Health (CADTH) were not searched. Hand searches of the bibliographies of any included studies were performed to identify any overlooked articles of relevance.
Both trial-based and model-based economic evaluations published in the English language and from the year 2000 to 2021 were included, to capture studies that wouldn’t have previously been identified in the review by Wilton et al. [21]. Any category of full economic evaluation was included, which did not include budget impact analyses. Studies on interventions to reduce AMR which report only clinical endpoints and do not investigate any economic outcomes were excluded from the review. Reviews, editorials, commentaries, and methodological articles were excluded. A detailed description of the search strategy can be found in the associated publication for the systematic review protocol [24].
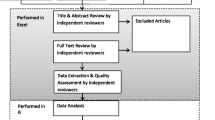
Data were exported to Covidence based on the inclusion and exclusion criteria of this review. The eligibility of the studies for review was assessed subjectively, and uncertainties were resolved in discussion amongst the reviewers. The inclusion of relevant studies was conducted with a two-step process: (1) Two reviewers independently screened titles and abstracts of all articles initially retrieved. (2) Full text screening of selected systematic reviews was conducted by two independent reviewers.
Population, interventions and outcomes
The populations considered included humans, animals, and the environment, consistent with the One Health approach of addressing AMR [11]. To be included in the review, the abstract of the article must have stated that a considered intervention in the evaluation would have an impact on AMR in some way, for example, through controlling the spread of resistant microbials, eradicating resistant microbials, or reducing antimicrobial use. There was no limitation on the type of intervention that could be included in the review.
The types of outcome measures that this review recorded included any cost–benefit measurement such as incremental cost-effectiveness ratio (ICER), incremental cost per quality-adjusted life year (QALY), incremental cost per disability-adjusted life year (DALY), incremental cost–benefit ratio, net monetary benefit (NMB), incremental net benefit (INB), net health benefit (NHB), costs avoided, net costs, cost-consequence measures and budget impact. Our review also included outcomes that are specific to the AMR context, such as incremental cost per resistant infection avoided. Furthermore, this review analysed the types of settings (countries or regions, country income-levels, farms, pharmacies or hospitals, types of hospitals (primary, community or tertiary), that these analyses were focussed on.
Data analysis and synthesis
A standardised template was created to facilitate the extraction of data from the included articles. Two researchers assisted in extracting the data of included studies. One researcher extracted the data and another researcher was responsible for checking the accuracy of the extracted data. In the case of disagreements, a third researcher was consulted to resolve the conflict. A narrative synthesis of the economic evaluations was conducted to report the study findings [25]. The narrative synthesis focused on important contextual factors of the available literature, including the countries analysed, the target populations represented, types of interventions and technical aspects of the economic analysis including costs, effectiveness and the sources of such data, discounting and perspectives. The country-level data was aggregated regionally and into income brackets based on the 2021 revised categorisations of the World Bank [26]. In addition to these main themes, the researchers also recorded details on first and corresponding authorship and their affiliations, whether any conflicts of interest were declared and what they were as well as funding sources. The extraction tables were generated using Microsoft Excel and data visualisation was conducted using Datawrapper [27].
To understand which interventions were deemed cost-effective and to make international comparisons, all extracted incremental cost-effectiveness ratios (ICERs) that reported a cost per QALY ICERs were converted to international Dollars using purchasing price parity (PPP) indices, consistent with the approach reported by Velasco et al. 2012 [28, 29]. The ICER values were also inflated to the 2020 cost year, where necessary. To attempt to understand whether the transformed ICER values could be considered cost-effective in different countries, the ICERs were presented next to the cost-effectiveness threshold ranges for low/middle income and middle/high income countries, reported by Woods et al. [30]. The country income levels were classified according to the World Bank income classifications, and the values inflated from 2013 to 2020 values to $18–600 for low/middle income countries, and $2,576–10,081 for middle/high income countries.
Quality assessment
The quality of the data used in the included economic evaluation studies was assessed using an adapted framework for the hierarchy of evidence scoring system detailed in Cooper et al. [31]. Where multiple sources of evidence were used for a single dimension, the highest ranked source of evidence was recorded for consistency.
Results
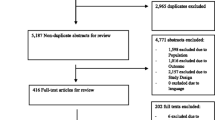
The database search yielded 40,169 articles, 11,572 of which were identified as duplicates by Covidence. Out of 28,597 titles and abstracts screened, 65 article full-texts were reviewed and considered for inclusion. Their reference lists were also hand searched for further relevant articles. Articles were most commonly excluded at the full-text stage due to being determined to have the wrong study design, no available full-text, and in one case there was an identified duplicate that had not already been identified by the Covidence software. This resulted in 35 articles that fulfilled the inclusion criteria. The PRISMA flowchart is illustrated in Fig. 1.
Summary of included studies
Table 1 below summarises the key features of evaluated interventions. All included studies were published between the years of 2001–2021. Health facilities were the most common study setting (n = 26), followed by community (n = 5), and other, which included prisons and multi-sectoral interventions (n = 3). The evaluated interventions included a range of intervention types, such as health care processes and guidelines (n = 20), pharmaceutical intervention (n = 5), antimicrobial stewardship (n = 4), awareness generation activities (n = 2), government policies and legislation (n = 2), medical technologies (n = 1), and combination of pharmaceutical intervention and health care processes and guidelines (n = 1).
Healthcare processes and guidelines include interventions included screening, surveillance and infection control mechanisms such as isolation, disinfectant application and the use of personal protective equipment. Stewardship programs broadly focused on the rational use of antimicrobials and were tailored to the specific contexts. For instance, Voermans et al. [59] describe an intervention of using procalcitonin algorithm to guide clinical decisions on drug use for lower-respiratory tract infection (LRTI) patients admitted in hospital. Awareness generation or educational interventions have been used in two studies in this review, a dialysis clinic and another was a training initiative for general practitioners in primary care when testing and communications when diagnosing patients presenting with respiratory symptoms [49]. The only medical technology included in this review was a study about a rapid point of care test for improved diagnosis and prescription of antibiotics for those with suspected LRTIs [47]. Of the two studies that analysed government policies or legislation, one analysed the role of macroeconomic approaches such as in tackling AMR while another examined the cost-effectiveness of a comprehensive action plan for multi-drug resistant TB in the WHO European region [41, 57].
Twenty-seven out of 35 studies had received funding support and 26 were authored by academics. Two studies reported first authorship from private consultancies. Sixteen of the 35 included studies did not report conflicts of interest in these studies. Of the 19 that did, only approximately half (n = 9) stated they had no conflicts of interest for the study.
Objective 1: What interventions to address AMR have been the subject of an economic evaluation?
Given the One Health lens of the review, it was notable that 34 of the 35 focussed on interventions to address AMR exclusively among human populations. Only one study addressed livestock-associated methicillin-resistant Staphylococcus aureus (LA MRSA) [39], and was focussed on the transmission of LA-MRSA from animal to human populations. None of the reviewed studies considered AMR concerns in the environment in any capacity.
Twenty out of 35 studies compared types of health care processes and guidelines. These interventions focussed on screening, diagnostics, surveillance and isolation systems in primary clinics, hospitals and tertiary facilities. Only two were community-focussed interventions, that used modified guidelines to ensure greater adherence to prescribed antimicrobial therapy and also as a means to implement a more stringent system of prescription itself [45, 50]. Five studies evaluated different types of ASPs, such as stewardship teams to guide clinical decision-making compared to routine care, or the use of bio-markers for patients with lower-respiratory tract infections. All of the ASPs identified in the review were hospital-based interventions [33, 46, 55, 56, 59].
Five studies detailed pharmaceutical interventions, three in community settings and two in clinics, which analysed recommendations for drug prescription such as amoxicillin for acute LRTIs, preventative fluoroquine therapy for drug-resistant tuberculosis and pre-exposure prophylaxis (PrEP) for patients with HIV. In other cases, these interventions compared two alternate courses of drugs or scenarios where a second- or third-line of drugs may be more cost-effective [34, 37, 42, 48, 51].Three of these interventions were at the community-level and at primary care facilities, including preventive daily drugs against multi-drug resistant tuberculosis. One study in Hong Kong used a combination of interventions; an expanded screening program with pharmaceutical prescriptions for intensive care unit (ICU) patients [66].
Only two studies considered broader national plans as an intervention: One study considered interventions that were specifically related to government policies and legislations that could influence the production, manufacturing and sale of antimicrobials [57], whilst the other focussed on the impact on TB specifically [41]. Examples of these interventions include, regulations, taxes, tariffs and other macroeconomic instruments. Education and training interventions were also being used to address AMR with two studies of this kind included in the review [49, 58]. Only a single study in our review reported on a new, point of care testing method [47]; no other examples of technological solutions for AMR were identified.
Not all included studies focussed on individual pathogens, though the majority did (26/35). The most common pathogen of focus was MRSA, whilst others included minority multi-drug resistant tuberculosis, HIV, gonorrhoea, carbapenem-resistant Enterobacteriaceae. Of the nine studies that did not specify an individual pathogen, they either described the class or location of infections, such as acute respiratory or LRTIs, intrabdominal infections, bloodstream infections, sepsis, nosocomial infections and multi-drug resistant organisms [33, 42, 46, 47, 49, 55, 56, 58, 60].
Objective 2: In what types of setting (by income level and geography) have these economic evaluations been focused?
Twenty-nine studies were conducted in a single country or territory, whereas six studies were conducted in multiple countries or territories. Thirty of the 35 studies were conducted in high-income settings, the majority of which were concentrated in the USA and in Europe; other countries and territories included Hong Kong and Singapore. The upper middle-income settings that were analysed were China and South Africa and Russia, the latter was part of one study that analysed screening for multi-drug resistant tuberculosis for prisoners in the Former Soviet Union (FSU) bloc. The other two countries included in the FSU study were Latvia, a high-income country from Europe and another Tajikistan, a lower-middle income country. The only individual study which focussed on a lower-middle income country in this review was for Vietnam in East Asia. This review has no representation from the South Asian or Latin American and Caribbean region, as well as from low-income countries which includes much of the African continent (see Fig. 2).
Geographical study populations of included articles in the review by income level. Income and regional categorisations are based on World Bank guidelines. Countries or territories listed: Belgium, China, France, Germany, Hong Kong, Italy, Latvia, Netherlands, Norway, Poland, Russia, Singapore, Slovakia, Slovenia, South Africa, Spain, Sweden, Tajikistan, United Kingdom, United States of America, Vietnam. *One study did not report country level data and has hence not been included in the figure above. **All countries specifically identified in included multi-country studies have been depicted on the map, as such the number of countries represented is greater than the total number of included studies
Objective 3: Which interventions have been estimated to be cost-effective, and has this result been replicated in other settings/contexts?
The subgroup analysis on cost-effectiveness judgements of interventions was limited only to studies which reported a cost per QALY or cost per DALY outcome (n = 14), to allow comparison against explicit cost-effectiveness thresholds. This excluded the majority of the identified studies. A DALY was assumed to be equal to a QALY for this analysis. The results of the subgroup analysis are graphically illustrated in Figs. 3, 2. Due to the small number of studies considered in this subgroup analysis and heterogeneous characteristics of interventions, comparators and study settings, it was not possible to effectively compare, contrast and discuss the consensus of the cost-effectiveness literature for specific interventions.
Cost-Effectiveness of Interventions with an Impact on AMR (International Dollar Incremental Cost-Effectiveness Ratio). ABP antibiotic prescribing; AC/LRTI acute cough or lower respiratory tract infection; ACU acute setting; AMS Antimicrobial stewardship; CA checklist activated; CAS chromogenic agar screen; CRE carbapenem-resistant enterobacteriaceae; CRP C-reactive protein; DCN decolonisation; HIV human immune-deficiency syndrome; HRSA high-risk speciality admissions; IA intra-abdominal; ICER incremental cost-effectiveness ratio; ICU intensive care unit; MMR measles, mumps and rubella; MRSA methicillin-resistant Staphylococcus aureus; PCR polymerase chain reaction; POCCR point-of-care C-reactive protein; PrEP pre-exposure prophylaxis; SG-SDD surveillance-guided selective digestive decontamination; SPEC specialist services setting; TB tuberculosis; TEA teaching hospital setting; WHO World Health Organization. Blue dot corresponds to the ICER value on the Y axis. Green triangle means that the intervention (first mentioned technology) dominates the comparator (latter mentioned). Red square means that the intervention (first mentioned technology) was dominated by the comparator (latter mentioned). Orange diamond denotes interventions with Y axis positions adjusted to improve the presentation of the figure, the actual ICER values are listed in the footnotes. Light green lines denote the upper and lower limits of the low/middle-income country cost-effectiveness threshold ($18–600). Blue lines denote the upper and lower limits of the middle/high-income country cost-effectiveness threshold ($2,576–10,081). Adjusted ICER values: 30% coverage of viral load monitoring vs. No viral load monitoring for HIV resistance ($41,476,437/QALY); POCCR testing vs. No POCCR for ABP for AC/LRTI ($106,606)
Three of the 14 studies in this subgroup analysis considered interventions to improve antibiotic prescribing practices, namely ASPs and improved diagnostic methods. Point-of-care C-reactive (POCCR) protein testing was considered in both analyses and was estimated to be cost-effective in the analysis for Belgium, Netherlands, Poland, Spain, England and Wales but not in the analyses for Norway and Spain. The first author was the same for both articles which suggests that the cost-effectiveness conclusion is likely to be highly context-specific.
Screening strategies and treatment strategies for MRSA were analysed in three studies, two in the United Kingdom or England and Wales, and one in Singapore. The studies considering screening strategies seemed to be consistent in that it was more cost-effective to screen in the highest-risk settings (acute care settings/patients with co-morbidities). However, in Singapore, screening all patients was estimated to be dominant compared to no screening whereas in the UK, Robotham et al. 2016 at all found no screening to be cost-effective at the national cost-effectiveness threshold. However, notably this study did not consider the secondary health and cost impacts from the prevention of AMR. Robotham et al. 2011 estimated that that decolonisation was more cost-effective than an isolation for MRSA-positive patients.
Only one of the studies included in this analysis was conducted in an LMIC (Vietnam). However, as the ICERs were converted to an International Dollar scale we can infer which interventions may be cost-effective in LMICs, though locally contextualised analysis should be performed to investigate this further. These included: communications training or C-reactive protein testing to reduce inappropriate antibiotic prescribing; decolonisation of MRSA-infected patients, various screening strategies for MRSA; annual sputum PCR screening for TB in prisons; PrEP for high-risk groups or all adults in high-prevalence settings; surveillance and decontamination strategies for CRE in ICUs; comprehensive national strategies to combat drug-resistant TB; and finally, antimicrobial stewardship team consultations to improve bloodstream infection prescribing.
Objective 4: What economic evaluation methods or techniques have been used to evaluate these interventions?
As displayed in Table 2, the majority of the economic evaluations were model-based (n = 31), and used a health care system or health care payer or hospital perspective (n = 30) and > 1-year time horizons (n = 12), the maximum of which was fifty years. The most common method employed was cost-utility analysis (n = 14) and other cost-effectiveness analyses (n = 14) and cost-consequence analyses (n = 5). Decision tree, mathematical, and statistical model were the most commonly used types of models, with 14, 6, and 5 studies respectively. There were also 6 studies that used Markov or Markov microsimulation models. Table 3 summarises the methods and techniques used to evaluate the interventions in the included studies.
Objective 5: What kind of data has been used in conducting economic evaluations for these interventions? What is the quality of this data?
Thirty-five studies were assessed for the quality of data sources used in the included studies using the hierarchies proposed by Cooper et al. [31] (Fig. 4). 26 studies (74%) used observational studies for the clinical effect sizes. Meanwhile, the baseline clinical data were predominantly (63%, n = 22) sourced from recent case series or analysis of reliable administrative databases covering patients solely from the jurisdiction of interest specifically conducted for the study. In terms of resource use, 23 (66%) studies used either prospective data collection for the specific study or used published results or reliable administrative data in the same jurisdiction. As for the cost inputs, 24 studies (69%) used reliable databases of the same jurisdiction, published within 5 years of the study’s cost year. Only 40% (n = 14) of the included studies used QALYs as an outcome measure, with six of these studies sourced from high-quality data sources, classified as direct or indirect utility assessment for the specific study using validated tools.
Conclusions
This manuscript represents an addition to the limited pool of literature on systematic reviews of economic evaluations impacting AMR. It is the first review since 2002 that considers a broad set of interventions, and the first to include non-human health interventions, though only a single study of this type was identified. To meaningfully build upon the limited body of literature that exists in this area and inform future research, the review was designed with a wide scope, in terms of types of interventions, pathogens, study populations. This approach resulted in a large number of abstracts being identified and subsequently reviewed, Specifically, this review, despite its breadth, did not identify any studies from the Latin American, and Caribbean region as well as from a majority of the African countries, although this may be a result of only including English language articles. AMR is growing fastest in LMIC settings, where resources are particularly constrained, and economic evidence could help improve the efficiency response to the threat to AMR in these settings. The increased importance of global health security issues, in the context of COVID-19 and other outbreaks, serves as a reminder of the importance of investment in addressing AMR. We urge local and international researchers and funders to prioritise conducting economic evaluations focussed on reducing AMR in LMICs.
The review highlighted that although there were several economic evaluations of ASPs identified, these studies need to be replicated in other settings, particularly in LMICs as the included studies predominantly focussed on high income settings. This could in part be due to poorer data availability in LMIC countries to populate economic evaluations, which could be abated by increased collection of relevant data in LMICs. Conducting economic evaluations on AMR may also be perceived as a lower research priority in LMIC settings, and due to the complexity of evaluating some interventions comprehensively, technical capacity constraints could have contributed to the lack of research in this area.
The review has demonstrated that there were a large proportion of CEA-but-not-CUA studies (i.e. did not use QALYs or DALYs), this may be due to the difficulty researchers have found in trying to convert certain outcomes (e.g. reduced antimicrobial consumption or infections avoided) into generic quantifiable health outcomes such as QALYs. Using QALYs and DALYs in cost-effectiveness analyses is preferable as these can then be compared to existing and accepted cost-effectiveness thresholds in order to inform policy decisions and improve allocative efficiency. Alternatively, there could be distinct and accepted cost-effectiveness thresholds specific to AMR using other measures than cost per QALY (e.g. antibiotic doses avoided) in order for CEA-but-not-CUA studies to have a greater impact on policy.
Our quality of evidence assessment highlighted that researchers in many cases have not used, presumably due to lack of availability, high quality data for their AMR studies. This is particularly true for the impact or efficacy of the interventions themselves, which most often used non-analytic study evidence. Furthermore, our quality of evidence section overstates the average quality of evidence used, as in stations where multiple input sources were used for a single dimension (which is often the case in economic evaluations) the highest quality of evidence score was recorded in order to be consistent. This review highlighted that the One Health approach to AMR has not yet been reflected in the economic evidence; only one identified study was not focussed on human health [39], though this could be due to flaws in our search terms in identifying evidence of this type.
As stated in the methods, it was difficult to identify interventions that affect AMR, as this could be the case for a huge number of interventions and the authors may or may not have chosen to consider that facet of the intervention. Therefore, in order to have a consistent and explicit approach in the screening process, only abstracts which stated an impact on AMR, in terms of antimicrobial use or resistance were included. As such, there may have been articles that were missed which only stated an impact in the full text, however this step was necessary in order to keep the number of articles included at the full-text screening stage to a feasible amount. The inclusion criteria could be improved upon in future research. Only English language studies were considered as the research team did not have the capacity to review articles in other languages.
Only a limited review of the methodologies of the economic evaluations was performed for the purposes of this study. However one methodological recommendation is that research could make better use of recent efforts to estimate the secondary economic costs of antibiotic consumption (e.g. by Shrestha et al. [67]), which account for how changes in consumption impact the speed and therefore costs of AMR, and incorporate these into future economic analyses. There would be substantial value in a more in-depth review of the methods of economic evaluations relevant to AMR. A review of this type could also identify best practices and technical recommendations on how these studies could be improved to ensure they reflect the nuances of AMR and comprehensively reflect the costs and health outcome impacts of AMR.
A brief review of the literature of clinical trials conducted for interventions to reduce AMR highlighted numerous interventions that have not yet been the subject of an economic evaluation [68,69,70,71,72]. These included educational and behaviour change interventions to improve prescribing, or to educate patients on their treatment seeking and adherence behaviour, and even interventions to reduce antibiotic use in animals. Future research in this area should focus on economic evaluations of interventions to combat AMR that have not previously been the subject of an economic evaluation, which would be useful for broadening the literature base and facilitating future research. The most similar previous systematic review on this topic by Wilton et al. 2001 also found that most of the included studies were conducted for high-income settings, for hospital-based settings and interventions. Wilton et al. described a “paucity of evidence [which] makes definitive recommendations concerning which strategies should be pursued, when, where and how, impossible”. This review has demonstrated that this statement largely remains to be true 20 years later.
Availability of data and materials
Cooper et al. Hierarchy of Evidence Ranking Scores. Database search terms.
Change history
29 September 2023
A Correction to this paper has been published: https://doi.org/10.1186/s13756-023-01314-z
References
World Health Organisation. Ten threats to global health in 2019 [Internet]. 2019. p. 1–18. Available from: https://www.who.int/emergencies/ten-threats-to-global-health-in-2019
Jonas O, Team W. Drug-resistant infections: a threat to our economic future. 2017.
World Health Organization. What is antimicrobial resistance? 2017.
Murray CJL, Ikuta KS, Sharara F, Swetschinski L, Robles Aguilar G, Gray A, et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629–55. https://doi.org/10.1016/S0140-6736(21)02724-0.
Michael CA, Dominey-Howes D, Labbate M. The antimicrobial resistance crisis: causes, consequences, and management. Front Public Health. 2014;2:145.
Sharma C, Rokana N, Chandra M, Singh BP, Gulhane RD, Gill JPS, et al. Antimicrobial resistance: its surveillance, impact, and alternative management strategies in dairy animals. Front Vet Sci. 2018;4:237.
Sharma A, Hill A, Kurbatova E, van der Walt M, Kvasnovsky C, Tupasi TE, et al. Estimating the future burden of multidrug-resistant and extensively drug-resistant tuberculosis in India, the Philippines, Russia, and South Africa: a mathematical modelling study. Lancet Infect Dis. 2017;17(7):707–15. https://doi.org/10.1016/S1473-3099(17)30247-5.
Van Boeckel TP, Brower C, Gilbert M, Grenfell BT, Levin SA, Robinson TP, et al. Global trends in antimicrobial use in food animals. Proc Natl Acad Sci. 2015;112(18):5649–54.
Organisation for Economic Co-operation and Development. Antimicrobial Resistance: policy insights. 2016.
Klein EY, Van Boeckel TP, Martinez EM, Pant S, Gandra S, Levin SA, et al. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc Natl Acad Sci. 2018;115(15):E3463–70.
Shallcross LJ, Davies DSC. Antibiotic overuse: a key driver of antimicrobial resistance. Br J Gen Pract. 2014;64(629):604–5.
Travasso C. India draws a red line under antibiotic misuse. BMJ. 2016;352:i1202.
Afari-Asiedu S, Hulscher M, Abdulai MA, Boamah-Kaali E, Asante KP, Wertheim HFL. Every medicine is medicine; exploring inappropriate antibiotic use at the community level in rural Ghana. BMC Public Health. 2020;20(1):1103.
Sriram A, Kalanxhi E, Kapoor G, Craig J, Balasubramanian R, Brar S, et al. The State of the world’s antibiotics in 2021: a global analysis of antimicrobial resistance and its drivers. Washington D.C. 2021.
Frost I, Craig J, Joshi J, Faure K, Laxminarayan R. Access Barriers to Antibiotics. Washington D.C. 2019. Available from: https://onehealthtrust.org/wp-content/uploads/2019/04/AccessBarrierstoAntibiotics_CDDEP_FINAL.pdf
Aslam B, Khurshid M, Arshad MI, Muzammil S, Rasool M, Yasmeen N, et al. Antibiotic resistance: one health one world outlook. Front Cell Infect Microbiol. 2021;11: 771510.
Review on Antimicrobial Resistance. Antimicrobials in agriculture and the environment: reducing use and unnecessary waste. 2015.
World Health Organization. Global action plan on antimicrobial resistance. 2015.
O’Rourke B, Oortwijn W, Schuller T. The new definition of health technology assessment: a milestone in international collaboration. Int J Technol Assess Health Care. 2020;36(3):187–90.
Sulis G, Sayood S, Gandra S. Antimicrobial resistance in low- and middle-income countries: current status and future directions. Expert Rev Anti Infect Ther. 2022;20(2):147–60.
Wilton P, Smith R, Coast J. Strategies to contain the emergence of antimicrobial resistance: a systematic review of effectiveness and cost-effectiveness. J Health Serv Res Policy. 2002;7(2):111–7. https://doi.org/10.1258/1355819021927764.
Naylor NR, Atun R, Zhu N, Kulasabanathan K, Silva S, Chatterjee A, et al. Estimating the burden of antimicrobial resistance: a systematic literature review. Antimicrob Resist Infect Control. 2018;7(1):58.
Huebner C, Flessa S, Huebner N-O. The economic impact of antimicrobial stewardship programmes in hospitals: a systematic literature review. J Hosp Infect. 2019;102(4):369–76.
Ananthakrishnan A, Painter C, Teerawattananon Y. A protocol for a systematic literature review of economic evaluation studies of interventions to address antimicrobial resistance. Syst Rev. 2021;10(1):508–15.
Popay J, Roberts H, Sowden A, Petticrew M, Arai L, Rodgers M, et al. Guidance on the Conduct of Narrative Synthesis in Systematic Reviews. 2006.
The World Bank. World Bank Country and Lending Groups [Internet]. 2022. Available from: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups
Lorenz M, Aisch G, Kokkelink D. Datawrapper: create charts and maps [Software] [Internet]. 2012. Available from: https://www.datawrapper.de/
Pérez Velasco R, Praditsitthikorn N, Wichmann K, Mohara A, Kotirum S, Tantivess S, et al. Systematic review of economic evaluations of preparedness strategies and interventions against influenza pandemics. PLoS ONE. 2012;7(2): e30333.
The World Bank. World Development Indicators. PPP conversion factor, GDP (LCU per international $) [Internet]. 2022. Available from: https://data.worldbank.org/indicator/PA.NUS.PPP
Woods B, Revill P, Sculpher M, Claxton K. Country-level cost-effectiveness thresholds: initial estimates and the need for further research. Value Health. 2016;19(8):929–35.
Cooper N, Coyle D, Abrams K, Mugford M, Sutton A. Use of evidence in decision models: an appraisal of health technology assessments in the UK since 1997. J Health Serv Res Policy. 2005;10(4):245–50.
Clancy M, Graepler A, Wilson M, Douglas I, Johnson J, Price CS. Active screening in high-risk units is an effective and cost-avoidant method to reduce the rate of methicillin-resistant Staphylococcus aureus infection in the hospital. Infect Control Hosp Epidemiol. 2006;27(10):1009–17.
D’Agata EMC, Tran D, Bautista J, Shemin D, Grima D. Clinical and economic benefits of antimicrobial stewardship programs in hemodialysis facilities: a decision analytic model. Clin J Am Soc Nephrol. 2018;13(9):1389–97.
Fox GJ, Oxlade O, Menzies D. Fluoroquinolone therapy for the prevention of multidrug-desistant tuberculosis in contacts: a cost-effectiveness analysis. Am J Respir Crit Care Med. 2015;192(2):229–37.
Gidengil CA, Gay C, Huang SS, Platt R, Yokoe D, Lee GM. Cost-effectiveness of strategies to prevent methicillin-resistant Staphylococcus aureus transmission and infection in an intensive care unit. Infect Control Hosp Epidemiol. 2015;36(1):17–27.
Gurieva T, Bootsma MCJ, Bonten MJM. Cost and effects of different admission screening strategies to control the spread of methicillin-resistant Staphylococcus aureus. PLoS Comput Biol. 2013;9(2):e1002874.
Harding-Esch EM, Huntington SE, Harvey MJ, Weston G, Broad CE, Adams EJ, et al. Antimicrobial resistance point-of-care testing for gonorrhoea treatment regimens: cost-effectiveness and impact on ceftriaxone use of five hypothetical strategies compared with standard care in England sexual health clinics. Eur Surveill. 2020;25(43):1900402.
Ho K, Ng W, Ip M, You JHS. Active surveillance of carbapenem-resistant Enterobacteriaceae in intensive care units: Is it cost-effective in a nonendemic region? Am J Infect Control. 2016;44(4):394–9.
Höjgård S, Aspevall O, Bengtsson B, Hæggman S, Lindberg M, Mieziewska K, et al. Preventing introduction of livestock associated MRSA in a pig population–benefits, costs, and knowledge gaps from the Swedish perspective. PLoS ONE. 2015. https://doi.org/10.1371/journal.pone.0122875.
Hubben G, Bootsma M, Luteijn M, Glynn D, Bishai D, Bonten M, et al. Modelling the Costs and effects of selective and universal hospital admission screening for methicillin-resistant Staphylococcus aureus. PLoS ONE. 2011;6(3):e14783.
Jakab Z, Acosta CD, Kluge HH, Dara M. Consolidated action plan to prevent and combat multidrug- and extensively drug-resistant tuberculosis in the WHO European Region 2011–2015: cost-effectiveness analysis. Tuberculosis. 2015;95(Suppl 1):S212–6.
Jansen JP, Kumar R, Carmeli Y. Cost-effectiveness evaluation of ertapenem versus piperacillin/tazobactam in the treatment of complicated intraabdominal infections accounting for antibiotic resistance. Value Health. 2009;12(2):234–44.
Jayaraman SP, Jiang Y, Resch S, Askari R, Klompas M. cost-effectiveness of a model infection control program for preventing multi-drug-resistant organism infections in critically ill surgical patients. Surg Infect. 2016;17(5):589–95.
Kip MMA, Kusters R, IJzerman MJ, Steuten LMG. A PCT algorithm for discontinuation of antibiotic therapy is a cost-effective way to reduce antibiotic exposure in adult intensive care patients with sepsis. J Med Econ. 2015;18(11):944–53.
Meropol SB. Valuing reduced antibiotic use for pediatric acute otitis media. Pediatrics. 2008;121(4):669–73.
Mewes JC, Pulia MS, Mansour MK, Broyles MR, Nguyen HB, Steuten LM. The cost impact of PCT-guided antibiotic stewardship versus usual care for hospitalised patients with suspected sepsis or lower respiratory tract infections in the US: a health economic model analysis. PLoS ONE. 2019;14(4):e0214222.
Oppong R, Jit M, Smith RD, Butler CC, Melbye H, Moelstad S, et al. Cost-effectiveness of point-of-care C-reactive protein testing to inform antibiotic prescribing decisions. Br J Gen Pract. 2013;63(612):E465–71.
Oppong R, Smith RD, Little P, Verheij T, Butler CC, Goossens H, et al. Cost effectiveness of amoxicillin for lower respiratory tract infections in primary care: an economic evaluation accounting for the cost of antimicrobial resistance. Br J Gen Pract. 2016;66(650):e633–9.
Oppong R, Smith RD, Little P, Verheij T, Butler CC, Goossens H, et al. Cost-effectiveness of internet-based training for primary care clinicians on antibiotic prescribing for acute respiratory tract infections in Europe. J Antimicrob Chemother. 2018;73(11):3189–98.
Pham QD, Wilson DP, Nguyen TV, Do NT, Truong LX, Nguyen LT, et al. Projecting the epidemiological effect, cost-effectiveness and transmission of HIV drug resistance in Vietnam associated with viral load monitoring strategies. J Antimicrob Chemother. 2016;71(5):1367–79.
Phillips A, Cambiano V, Johnson L, Nakagawa F, Homan R, Meyer-Rath G, et al. Potential impact and cost-effectiveness of condomless-sex-concentrated PrEP in KwaZulu-Natal accounting for drug resistance. J Infect Dis. 2021. https://doi.org/10.1093/infdis/jiz667.
Puzniak LA, Gillespie KN, Leet T, Kollef M, Mundy LM. A cost-benefit analysis of gown use in controlling vancomycin-resistant Enterococcus transmission: Is it worth the price? Infect Control Hosp Epidemiol. 2004;25(5):418–24.
Robotham JV, Graves N, Cookson BD, Barnett AG, Wilson JA, Edgeworth JD, et al. Screening, isolation, and decolonisation strategies in the control of meticillin resistant Staphylococcus aureus in intensive care units: cost effectiveness evaluation. BMJ. 2011;343:d5694.
Robotham JV, Deeny SR, Fuller C, Hopkins S, Cookson B, Stone S. Cost-effectiveness of national mandatory screening of all admissions to English National Health Service hospitals for meticillin-resistant Staphylococcus aureus: a mathematical modelling study. Lancet Infect Dis. 2016;16(3):348–56.
Ruiz-Ramos J, Frasquet J, Roma E, Poveda-Andres JL, Salavert-Leti M, Castellanos A, et al. Cost-effectiveness analysis of implementing an antimicrobial stewardship program in critical care units. J Med Econ. 2017;20(6):652–9.
Scheetz MH, Bolon MK, Postelnick M, Noskin GA, Lee TA. Cost-effectiveness analysis of an antimicrobial stewardship team on bloodstream infections: a probabilistic analysis. J Antimicrob Chemother. 2009;63(4):816–25.
Smith RD, Yago M, Millar M, Coast J. A macroeconomic approach to evaluating policies to contain antimicrobial resistance: a case study of methicillin-resistant Staphylococcus aureus (MRSA). Appl Health Econ Health Policy. 2006;5(1):55–65.
Tran D, Grima D, Bautista JE, D’Agata E. Use of a decision analytic model to assess the clinical and economic impact of an educational program for antimicrobial use in a dialysis clinic. Value Health. 2016;19(3):A223. https://doi.org/10.1016/j.jval.2016.03.1198.
Voermans AM, Mewes JC, Broyles MR, Steuten LMG. Cost-effectiveness analysis of a procalcitonin-guided decision algorithm for antibiotic stewardship using real-world U.S. hospital data. OMICS. 2019;23(10):508–15.
Wang Y, Yuan Y, Lin L, Tan X, Tan Y. Determining the ideal prevention strategy for multidrug-resistance organisms in resource-limited countries: a cost-effectiveness analysis study. Epidemiol Infect. 2020;148:e176.
Wassenberg MWM, de Wit GA, van Hout BA, Bonten MJM. Quantifying cost-effectiveness of controlling nosocomial spread of antibiotic-resistant bacteria: the case of MRSA. PLoS ONE. 2010;5(7):e11562.
Wilton P, Smith RD, Coast J, Millar M, Karcher A. Directly observed treatment for multidrug-resistant tuberculosis: an economic evaluation in the United States of America and South Africa. Int J Tuberc Lung Dis. 2001;5(12):1137–42.
Win M-K, Soliman TAA, Lee LK, Wong CS, Chow A, Ang B, et al. Review of a two-year methicillin-resistant Staphylococcus aureus screening program and cost-effectiveness analysis in Singapore. BMC Infect Dis. 2015;15:391.
Winetsky DE, Negoescu DM, DeMarchis EH, Almukhamedova O, Dooronbekova A, Pulatov D, et al. Screening and rapid molecular diagnosis of tuberculosis in prisons in Russia and Eastern Europe: a cost-effectiveness analysis. PLoS Med. 2012;9(11):e1001348.
You JHS, Chan CY, Wong MY, Ip M. Active surveillance and decolonization of methicillin-resistant Staphylococcus aureus on admission to neonatal intensive care units in Hong Kong: a cost-effectiveness analysis. Infect Control Hosp Epidemiol. 2012;33(10):1024–30.
You JHS, Li H-K, Ip M. Surveillance-guided selective digestive decontamination of carbapenem-resistant Enterobacteriaceae in the intensive care unit: a cost-effectiveness analysis. Am J Infect Control. 2018;46(3):291–6.
Shrestha P, Cooper BS, Coast J, Oppong R, Do Thi Thuy N, Phodha T, et al. Enumerating the economic cost of antimicrobial resistance per antibiotic consumed to inform the evaluation of interventions affecting their use. Antimicrob Resist Infect Control. 2018;7(1):98. https://doi.org/10.1186/s13756-018-0384-3.
Crayton E, Richardson M, Fuller C, Smith C, Liu S, Forbes G, et al. Interventions to improve appropriate antibiotic prescribing in long-term care facilities: a systematic review. BMC Geriatr. 2020;20(1):237. https://doi.org/10.1186/s12877-020-01564-1.
Lambert M, Smit CCH, De Vos S, Benko R, Llor C, Paget WJ, et al. A systematic literature review and meta-analysis of community pharmacist-led interventions to optimise the use of antibiotics. Br J Clin Pharmacol. 2022;88(6):2617–41. https://doi.org/10.1111/bcp.15254.
Parveen S, Garzon-Orjuela N, Amin D, McHugh P, Vellinga A. Public health interventions to improve antimicrobial resistance awareness and behavioural change associated with antimicrobial use: a systematic review exploring the use of social media. Antibiotics. 2022;11
Tang KL, Caffrey NP, Nobrega DB, Cork SC, Ronksley PE, Barkema HW, et al. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: a systematic review and meta-analysis. Lancet Planet Health. 2017;1(8):e316–27.
Behavioural Economics and Research Team (Department of Health) and the Behavioural Economics Team of the Australian Government (Department of the Prime Minister and Cabinet). Nudge vs Superbugs: A behavioural economics trial to reduce the overprescribing of antibiotics [Internet]. 2018. Available from: https://behaviouraleconomics.pmc.gov.au/projects/nudge-vs-superbugs-behavioural-economics-trial-reduce-overprescribing-antibiotics
Acknowledgements
The authors wish to thank Saudamini Dabak and Sarin KC for their support (Health Intervention and Technology Assessment Program, Thai Ministry of Public Health).
Study registration
This study has been registered with the PROSPERO International Prospective Register of Systematic Reviews (CRD42020190310).
Funding
Aparna Ananthakrishnan and Chris Painter received support from the Fleming Fund and the Overseas Development Institute as well as a grant from the Royal Society of Tropical Medicine and Hygiene (RSTMH) Small Grants programme towards this project [16109569]. The funders have not been directly involved in any stage of developing this protocol or decision to publish. Aparna Ananthakrishnan, Chris Painter and Yot Teerawattananon are or were at the time of this work employed by the Health Intervention and Technology Assessment Program (HITAP), Ministry of Public Health, Thailand. HITAP is part of the International Decision Support Initiative (iDSI) which provides technical assistance on health intervention and technology assessment to governments in low and middle-income countries. iDSI is funded by the Bill and Melinda Gates Foundation [OPP1202541], the UK’s Department for International Development, and the Rockefeller Foundation. This research was funded in whole, or in part, by the Wellcome Trust [220211]. For the purpose of Open Access, the author has applied a CC BY public copyright licence to any Author Accepted Manuscript version arising from this submission.
Author information
Authors and Affiliations
Contributions
AA and CP conceptualised the study, identified the research question and developed the methodology. AA, CP and DF conducted the screening of articles and analysis of the data. KKC and ENS also assisted with the analysis of the data. YT provided support with reviewing the search strategy and research questions and provided strategic input in the analysis of the results. KA provided strategic input to the analysis approach. All authors reviewed and provided input on the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors consent to publication.
Competing interests
The authors do not have any competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
. Cooper et al. Hierarchy of Evidence Ranking Scores.
Additional file 2
. Database search terms.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Painter, C., Faradiba, D., Chavarina, K.K. et al. A systematic literature review of economic evaluation studies of interventions impacting antimicrobial resistance. Antimicrob Resist Infect Control 12, 69 (2023). https://doi.org/10.1186/s13756-023-01265-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13756-023-01265-5