Abstract
A stochastic susceptible–exposed–infected–recovered (SEIR) model with nonlinear incidence rate is investigated. Under suitable conditions, existence and uniqueness of a global solution, stationary distribution with ergodicity, persistence in the mean, and extinction of the disease are obtained. Numerical simulations and conclusions are separately carried out at the end of this paper.
Similar content being viewed by others
1 Model formulation
The spread and control of infectious diseases [1–3] was described by governing the mathematical models, aiming to investigate the dynamical properties. Since the pioneering work was established by Kermack and McKendrick [4], the epidemic models have been paid more attention at present. Usually, epidemic models included three states within a total population: the susceptible S, the infected I, and the recovered R. For instance, Cai et al. [5] introduced an SIS model incorporating media coverage to investigate the effects of environment fluctuations. However, for some diseases, such as Hepatitis B, Hepatitis C, and AIDS, the exposed hosts E took a vital role when dynamical behaviors were expected to be discussed. As mentioned in recent literature, a class of epidemic models was considered by some scholars; it was called susceptible–exposed–infected–recovered model (short for SEIR model, see [6–18]).
During the development of epidemic models, incidence rates which describe the relationship between the susceptible and the infected/the exposed play an important role, and meanwhile change its form ranging from bilinear case to nonlinear case when investigation of epidemic models is conducted. For example, [19–22] governed bilinear incidence rate \(\beta SI\) to explore epidemic models with fluctuation in epidemic models. If the number of the infected within a population was large, then three types of saturated incidence rates were usually used in epidemiological models: the mixing standard incidence rate \(\frac{\beta SI}{N}\) [23–25], the nonlinear incidence rate \(\beta S^{q} I^{p}\) [26, 27], and the saturation incidence rate \(\frac{\beta SI}{1+\alpha S}\) [28–30]. Later, some generalized and nonlinear incidence rates were studied in recent literature (see [31–33]).
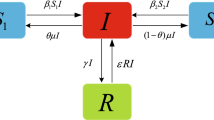
In this paper, we still use four states of epidemic models, that is, the susceptible S, the exposed E, the infected I, and the recovered R, to describe our model with environmental fluctuations. Motivated by the above-mentioned discussions, we assume that individuals within total population are well mixed and settle in the same environment. Our model goes with the idea that the susceptible and the infected contact with constant rate β; after contacting with the infected, the susceptible turns into the exposed when time exceeds incubation period (also called the latent period, see [8, 11, 18]); that the exposed individuals become the infected and then the recovered; and that part of recovered individuals enter again into the susceptible state. According to this spread cycle, we establish our model by equations, and we start with an equation of the susceptible as follows:
where A and μ respectively denote the new recruitment rate and the disease-free death rate, δ is the rate at which the recovered individuals become susceptible, \(\frac{\beta S I}{\varphi (I)}\) is a nonlinear incidence rate with property that \(\varphi (I)\) is increasing and \(\varphi (0)=1\), and that, for some constant \(l>0\), the following property is valid:
For the exposed, we have that
where σ is the rate at which exposed individuals become infected individuals. Further, the changes of infected and recovered individuals at time t are assumed to follow two ordinary differential equations:
and
where ρ is the death rate caused by diseases and γ is the recovery rate of infected individuals. Now, we derive a system that consists of four ordinary differential equations:
It follows from (1) that
Then \(\limsup_{t\rightarrow +\infty }(S(t)+E(t)+I(t)+R(t))\leq \frac{A}{\mu }\). Thus the feasible region for system (1) is
Let IntΩ denote the interior of Ω. It is easy to verify that the region Ω is positively invariant with respect to system (1) (i.e., the solutions with initial conditions in Ω remain in Ω). Hence, system (1) will be considered mathematically and epidemiologically well posed in Ω.
One concern for further investigation is to find out an expression for the basic reproduction number \(R_{0}\) of model (1) by using of the next generation matrix (see [34]). The basic reproduction number, sometimes called basic reproductive rate or basic reproductive ratio, is one of the most useful threshold parameters that characterize mathematical problems concerning infectious diseases. This metric is useful because it helps determine whether or not an infectious disease will spread through a population. Next, we calculate the basic reproduction number of system (1). Let \(x=(S,E,I,R)^{T}\), where T denotes the transpose of matrix (or vector). Then model (1) can be written as
where
So the infected classes can be referred to as \(m=2\), that is, the exposed compartment (E) and the infected compartment (I), and the disease-free equilibrium of model (1) is \(x_{0}=(\frac{A}{\mu },0,0,0)^{T}\). Based on the detailed documentations in [34–36], we can easily get
and the inverse matrix of V is
Therefore \(FV^{-1}\) is the next generation matrix for model (1). It follows that the spectral radius of matrix \(FV^{-1}\) is \(\rho (FV^{-1})= \frac{A\beta \sigma }{\mu (\mu +\sigma )(\mu +\rho +\gamma )}\). According to Theorem 2 in [34], the basic reproduction number is
When diseases attack total population in real circumstances, the effects of comprehensive and external fluctuation are inevitable and distinct. We here assume that the effects are proportional to states of models. For instance, the temperature, air humidity, and other factors normally are regarded as comprehensive and external fluctuations. Therefore we consider the following epidemic model with environmental fluctuations and nonlinear incidence rate:
where \(B_{i}(t)\) are standard one-dimensional independent Wiener processes, \(\sigma _{i}\) are the intensities of white noise for \(\sigma _{i}>0\) and \(i=1,2,3,4\). Throughout the paper, unless otherwise specified, let \((\varOmega , \{\mathcal{F}{_{t}}\}_{t\geq 0}, \mathbb{P})\) be a complete probability space with a filtration \({\{\mathcal{F}}{_{t}}\}_{t\geq 0}\) satisfying the usual conditions, that is, it is increasing and right continuous, while \(\mathcal{F}{_{0}}\) contains all \(\mathbb{P}\)-null sets.
The rest of this paper is organized as follows. In Sect. 2, we show that model (2) admits a unique global positive solution with any initial value. In Sect. 3, we establish sufficient conditions for extinction of the disease. In Sect. 4, we verify the persistence in the mean under some conditions. Finally, we prove that there is an ergodic stationary distribution of model (2) by constructing suitable Lyapunov functions.
2 Existence and uniqueness of positive solution
Theorem 2.1
There is a unique solution\((S(t),E(t),I(t),R(t))\)to model (2) on\(t\geq 0\)for any given initial value\((S(0),E(0),I(0),R(0))\)in\(\mathbb{R}^{4}_{+}\)with probability one.
Proof
Because the coefficients of system (2) satisfy locally Lipschitz continuity, there exists a unique local solution \((S(t),E(t),I(t),R(t))\) on \(t\in [0,\tau _{e})\), where \(\tau _{e}\) is the explosion time. To show that the solution is global, we just need to prove that \(\tau _{e}=\infty \). Let \(m_{0}>1\) be a sufficiently large integer such that each component of \((S(0),E(0),I(0),R(0))\) lies within the interval \([\frac{1}{m_{0}},m_{0}]\). For each integer \(m\geq m_{0}\), we define the stopping time
Throughout this paper, we set \(\inf \emptyset =\infty \). It is obvious that \(\tau _{m}\) is increasing as \(m\rightarrow \infty \) (details can be seen in [37]). We also denote \(\lim_{m\rightarrow \infty }\tau _{m}=\tau _{\infty }\). Obviously, there is \(\tau _{\infty }\leq \tau _{e}\). If we confirm that \(\tau _{\infty }=\infty \), then we get \(\tau _{e}=\infty \) for all \(t\geq 0\). The proof goes by contradiction. Assuming that \(\tau _{\infty }\neq \infty \), then there exists a pair of constants \(T>0\) and \(\varepsilon \in (0,1)\) such that \(\mathbb{P}\{\tau _{\infty }\leq T\}\geq \varepsilon \). Hence there exists an integer \(m_{1}\geq m_{0}\) such that \(\mathbb{P}\{\tau _{m}\leq T\}\geq \varepsilon \) for each integer \(m\geq m_{1}\). We define a \(C^{2}\)-function \(V:\mathbb{R}^{4}_{+}\rightarrow \mathbb{R}_{+}\) as follows:
where b is a positive constant that will be determined later. Then, making use of Itô’s formula on \(V(S,E,I,R)\), we obtain that
where
We choose \(b=\frac{\mu +\rho }{\beta }\) and denote \(K:=A+\mu b+\sigma +3\mu +\rho +\gamma +\delta + \frac{(\mu +\rho )\sigma ^{2}_{1}+\beta \sigma ^{2}_{2}+\beta \sigma ^{2}_{3}+ \beta \sigma ^{2}_{4}}{2\beta }\), so we have
where K is a positive constant. Then
For any \(t\in [0,T]\) and \(m\geq m_{1}\), we take integration from 0 to \(\tau _{m}\wedge T\), and then take expectation on both sides, which gives
We set \(\varOmega _{m}=\{\tau _{m}\leq T\}\) for \(m\geq m_{1}\), then \(\mathbb{P}(\varOmega _{m})\geq \varepsilon \). Each component of \((S(\tau _{m}\wedge T),E(\tau _{m}\wedge T),I(\tau _{m}\wedge T),R( \tau _{m}\wedge T))\) equals either m or \(\frac{1}{m}\) for all \(w\in \varOmega _{m}\). Therefore, we get
Let \(m\rightarrow \infty \), which implies the contradiction
as a consequence, we have \(\tau _{\infty }=\infty \). The proof is complete. □
3 Extinction of diseases
Extinction and persistence are two most important issues in the study of epidemic models. For the sake of simplicity, we denote
Lemma 3.1
For any initial value\((S(0),E(0),I(0),R(0))\in \mathbb{R}_{+}^{4}\), the solution\((S(t),E(t),I(t), R(t))\)has the following properties:
and
Moreover, if\(\mu > \frac{\sigma ^{2}_{1}\vee \sigma ^{2}_{2}\vee \sigma ^{2}_{3}\vee \sigma ^{2}_{4}}{2}\), then
The proof of Lemma 3.1 is similar to the approach used in [38, 39], and we omit the proof here.
Lemma 3.2
(Strong law of large numbers [37])
Let\(M=\{M_{t}\}_{t\geq 0}\)be a real-value continuous local martingale vanishing at\(t=0\). Then
and also
Theorem 3.1
Let\((S(t),E(t),I(t),R(t))\)be the solution of model (2) with any initial value in\(\mathbb{R}^{4}_{+}\). If the basic reproduction number satisfies
and
then
then the density of the infected individuals reaches extinction.
Proof
Let
we obtain that
Taking integration on both sides of (3) from 0 to t and according to Lemma 3.1, we see that
Now we define a \(C^{2}\)-function \(W:{\mathbb{R}^{2}_{+}}\rightarrow {\mathbb{R}_{+}}\) as follows:
where
Making use of Itô’s formula, we have
Based on the fundamental inequality \((a^{2}+b^{2})(c^{2}+d^{2})\geq (ac+bd)^{2}\) for positive a, b, c, and d, we obtain that
therefore
Now we take integration on both sides of (7) and divide it by t, which then implies the following expression:
where
are local martingales, whose quadratic variations are \(\langle M_{1}(t),M_{1}(t) \rangle \leq \sigma ^{2}_{2}t\), \(\langle M_{2}(t),M_{2}(t) \rangle \leq \sigma ^{2}_{3}t\) respectively. Applying Lemma 3.2, we conclude that
Then taking the upper limit on both sides, from (7), (8), and (9), we can get
If \(\nu <0\), we obtain that
which suggests that \(\lim_{t\rightarrow \infty }I(t)=0\). This indicates that the disease would tend to extinction. The proof is complete. □
4 Persistence in the mean
In this section, we will demonstrate some useful results about the persistence of the diseases.
Theorem 4.1
Let\((S(t),E(t),I(t),R(t))\)be a solution of system (2) with any initial value in\(\mathbb{R}^{4}_{+}\). If
then system (2) has the following property:
where
That is to say, the disease will be prevalent.
Proof
In order to testify the persistence, we establish a \(C^{2}\)-function \({V_{1}}:\mathbb{R}^{4}_{+}\rightarrow \mathbb{R}\) as follows:
where \(c_{1}\), \(c_{2}\), \(c_{3}\) are positive constants to be determined later. Next we apply Itô’s formula to (10). Then we get the following result:
where
Let
Then
where
Hence, from (11) and (12) we get
where
is a local martingale. Using Lemma 3.2 yields
Therefore
The proof is complete. □
5 Stationary distribution
In this section, we will establish sufficient conditions for the existence of a unique ergodic stationary distribution. First of all, we present a lemma which will be used later.
Let \(x(t)\) be a homogeneous Markov process in \(E_{l}\) (\(E_{l}\) denotes an l-dimensional Euclidean space) and be described by the following stochastic differential equation:
The diffusion matrix is defined as follows:
Lemma 5.1
([40])
The Markov process\(x(t)\)has a unique ergodic stationary distribution\(\mu (\cdot )\)if there exists a bounded domain\(U\subset E_{l}\)with regular boundaryΓand
- (A1)
There is a positive numberMsuch that\(\sum^{l}_{i,j=1}a_{ij}(x)\xi _{i}\xi _{j}\geq M|\xi |^{2}\), \(x\in U\), \(\xi \in {\mathbb{R}}^{l}\).
- (A2)
There exists a nonnegative\(C^{2}\)-functionVsuch that\(\mathcal{L}V\)is negative for any\(E_{l}\setminus U\).
Then
for all\(x\in E_{l}\), where\(f(\cdot )\)is a function integrable with respect to the measureμ.
Theorem 5.1
Assume that\(\widetilde{R}_{0}>1\). Then, for any initial value\((S(0),E(0),I(0),R(0))\in {\mathbb{R}}^{4}_{+}\), there is a stationary distribution\(\mu (\cdot )\)for system (2) and the ergodicity holds.
Proof
The diffusion matrix of system (2) is given by \(\widetilde{A}= \operatorname{diag}(\sigma ^{2}_{1}S^{2},\sigma ^{2}_{2}E^{2}, \sigma ^{2}_{3}I^{2},\sigma ^{2}_{4}R^{2})\). We choose \(M=\min_{(S,E,I,R)\in D_{k}\subset {\mathbb{R}}^{4}_{+}}\{\sigma ^{2}_{1}S^{2}, \sigma ^{2}_{2}E^{2},\sigma ^{2}_{3}I^{2},\sigma ^{2}_{4}R^{2}\}\). We get
for any \((S,E,I,R)\in D_{k}\), \(\xi =(\xi _{1},\xi _{2},\xi _{3},\xi _{4})\in {\mathbb{R}}^{4}_{+}\), where \(D_{k}=[\frac{1}{k},k]\times [\frac{1}{k},k]\times [\frac{1}{k},k] \times [\frac{1}{k},k]\) and \(k>1\) is a sufficiently large integer. Then condition (A1) holds, where \(E_{l}=\mathbb{R}^{4}_{+}\), \(U=D_{k}\).
Next we construct a nonnegative \(C^{2}\)-function \(\bar{V}: {\mathbb{R}}^{4}_{+} \rightarrow {\mathbb{R}}\) in the following form:
It is easy to check that
where \(U_{k}=(\frac{1}{k},k)\times (\frac{1}{k},k)\times (\frac{1}{k},k) \times (\frac{1}{k},k)\). Besides, \(\widetilde{V}(S,E,I,R)\) is a continuous function. Hence, \(\widetilde{V}(S,E,I,R)\) must admit a minimum point \((S^{*},E^{*},I^{*},R^{*})\) in the interior of \({\mathbb{R}}^{4}_{+}\). Then we define a nonnegative \(C^{2}\)-function V̄ as follows:
where \(V_{1}\) is presented in (10), and
where n is a sufficiently small constant and \(M>0\) satisfying the following condition:
where
According to similar discussions as shown in Theorem 4.1, we have
and
Therefore
where \(N=5\mu +\delta +\rho + \frac{2\sigma ^{2}_{1}+\sigma ^{2}_{2}+\sigma ^{2}_{3}+2\sigma ^{2}_{4}}{2}+B\).
Now we are in a position to construct a compact subset D such that condition (A2) in Lemma 5.1 holds. Define the bounded closed set
where \(\varepsilon _{i}>0\) (\(i=1,2,3,4\)) are sufficiently small constants satisfying the following conditions:
where P, Q, T, F, G, H, L are presented in (21), (22), (23), (24), (25), (26), (27), respectively. For convenience, we divide \({\mathbb{R}^{4}_{+}}\setminus D\) into eight domains:
Obviously, \(D^{C}=D_{1}\cup D_{2}\cup \cdots \cup D_{8}\). Next we only need to show that \(\mathcal{L}\bar{V}(S,E,I,R)\leq -1\) on \(D^{C}\).
Case 1. If \((S,E,I,R)\in D_{1}\), by (13) we get that
where
Case 2. If \((S,E,I,R)\in D_{2}\), by (14) we have that
where
Case 3. If \((S,E,I,R)\in D_{3}\), by (15) we have that
where
Case 4. If \((S,E,I,R)\in D_{4}\), by (16) we get that
Case 5. If \((S,E,I,R)\in D_{5}\), by (17) we get that
where
Case 6. If \((S,E,I,R)\in D_{6}\), by (18) we get that
where
Case 7. If \((S,E,I,R)\in D_{7}\), by (19) we get that
where
Case 8. If \((S,E,I,R)\in D_{8}\), by (20) we get that
where
The proof is complete. □
6 Examples and simulations
For the numerical simulation, we use Milstein’s higher order method mentioned in [41] to obtain the following discretization transformation of system (2):
where the time increment \(\Delta t>0\), \(\sigma ^{2}_{i}>0\) (\(i=1,2,3,4\)) are the intensities of the white noise, \(\xi _{i}\) (\(i=1,2,3,4\)) are independent Gaussian random variables which follow the distribution \(N(0,1)\) for \(j=0,1,2, \ldots, n\).
We take the parameters of model (2) as follows: \(A=0.7\), \(\beta =0.055\), \(\mu =0.1\), \(\gamma =0.2\), \(\rho =0.15\), \(\sigma =0.3\), \(\Delta t=0.001\), \(\varphi (I)=1+0.6I\), \(\delta =0.04\), \(\sigma _{1}=0.009\), \(\sigma _{2}=0.1\), \(\sigma _{3}=0.1\), \(\sigma _{4}=0.1\), \(S(0)=0.8\), \(E(0)=0.7\), \(I(0)=0.6\), \(R(0)=0.5\). By condition of Theorem 3.1, we have
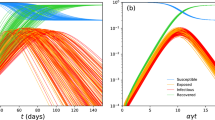
which indicates the extinction of infected individuals. The corresponding realizations of model (2) demonstrate their properties in Fig. 1, when \(n=25\text{,}000\). At the same time, we get that the disease will reach extinction faster as the environmental disturbance increases. For example, when \(\sigma _{1}=0.054\), \(\sigma _{2}=0.6\), \(\sigma _{3}=0.6\), \(\sigma _{4}=0.6\), for the corresponding dynamics see Fig. 2, when \(n=5000\).
A realization of extinction of the exposed and infected to model (2)
A realization of extinction of the exposed and infected to model (2)
We further assume that the parameters of model (2) are \(A=0.5\), \(\beta =0.1\), \(\mu =0.01\), \(\gamma =0.2\), \(\rho =0.1\), \(\sigma =0.2\), \(\sigma _{1}=0.008\), \(\sigma _{2}=0.06\), \(\sigma _{3}=0.008\), \(\sigma _{4}=0.006\), \(\delta =0.08\), \(\varphi (I)=1+0.15I\). By the condition of Theorem 4.1 and Theorem 5.1, we have \(\widetilde{R_{0}}=6.0725>1\), and \(\liminf_{t\rightarrow \infty }\langle I\rangle _{t}\geq 0.1625\), model (2) admits a unique stationary distribution as shown in Fig. 3, and the solution of model (2) is persistent in the mean as shown in Fig. 4, when \(n=200\text{,}000\).
Histogram of the susceptible, the exposed, the infected, and the recovered to model (2)
A realization of persistence in the mean for the susceptible, the exposed, the infected, and the recovered to model (2)
7 Conclusions
In this paper, we intend to investigate an epidemic model of having four stages: the susceptible, the exposed, the infected, and the recovered. And we focus on extinction, persistence, and stationary distribution of a positive solution to epidemic model with nonlinear incidence rate and independent environmental fluctuations.
We firstly, by constructing an appropriate function, show that model (2) admits a unique global positive solution with any initial value. Moreover, we also find that the extinction of disease depends on the basic reproduction number \(R_{0}\) (a threshold for its corresponding deterministic model). When \(R_{0}<1\) and \(\nu <0\), the disease under independent environmental fluctuations dies out as demonstrated in Theorem 3.1, and its corresponding dynamics could be found in Fig. 1. While, by constructing several \(C^{2}\)-functions, under the condition \(\widetilde{R_{0}}>1\), we derive sufficient conditions for persistence and existence of a unique ergodic stationary distribution to model (2), the corresponding realizations could be found in Fig. 2 and Fig. 3, respectively.
We further present numerical simulations on ergodicity of model (2) at the end of this paper and point out that extinction time of infected individuals decreases when intensities of environmental fluctuations \(\sigma _{i}\) (\(i=1,2,3,4\)) increase. These results provide readers a biological perspective when understanding an epidemic model with fluctuated environments.
References
Jin, Z., Haque, M., Liu, Q.: Pulse vaccination in the periodic infection rate SIR epidemic model. Int. J. Biomath. 4(1), 409–432 (2008)
Huo, H., Ma, Z.: Dynamics of a delayed epidemic model with non-monotonic incidence rate. Commun. Nonlinear Sci. Numer. Simul. 15(2), 459–468 (2010)
Van Den Driessche, P., Watmough, J.: A simple SIS epidemic model with a backward bifurcation. J. Math. Biol. 40, 525–540 (2000)
Kermack, W., McKendric, A.: A contribution to the mathematical theory of epidemics. Proc. R. Soc. Ser. A 115(1), 700–721 (1927)
Cai, Y., Kang, Y., Banerjee, M., Wang, W.: A stochastic epidemic model incorporating media coverage. Commun. Math. Sci. 14(4), 893–910 (2016)
Zhao, D., Sun, J., Tan, Y., Wu, J., Dou, Y.: An extended SEIR model considering homepage effect for the information propagation of online social networks. Physica A 512, 1019–1031 (2018)
Fan, X., Wang, Z.: Stability analysis of an SEIR epidemic model with stochastic perturbation and numerical simulation. Int. J. Nonlinear Sci. Numer. Simul. 14(2), 113–121 (2013)
Britton, T., Ouedraogo, D.: SEIRS epidemics with disease fatalities in growing populations. Math. Biosci. 296, 45–59 (2018)
Wang, X., Peng, H., Shi, B., Jiang, D., Zhang, S.: Optimal vaccination strategy of a constrained time-varying SEIR epidemic model. Commun. Nonlinear Sci. Numer. Simul. 67, 37–48 (2019)
Li, X., Gupur, G., Zhu, G.: Threshold and stability results for an age-structured SEIR epidemic model. Comput. Math. Appl. 42(6), 883–907 (2015)
Khan, A., Zaman, G.: Global analysis of an age-structured SEIR endemic model. Chaos Solitons Fractals 108, 154–165 (2018)
Liu, Q., Jiang, D., Hayat, T., Alsaedi, A.: Stationary distribution of a stochastic delayed SVEIR epidemic model with vaccination and saturation incidence. Physica A 512, 849–863 (2018)
Wang, L., Liu, Z., Zhang, X.: Global dynamics of an SVEIR epidemic model with distributed delay and nonlinear incidence. Appl. Math. Comput. 284, 47–65 (2016)
Liu, Q., Jiang, D., Hayat, T., Ahmad, B.: Stationary distribution and extinction of a stochastic SEIR epidemic model with standard incidence. Physica A 476, 58–69 (2017)
Wei, F., Xue, R.: Stability and extinction of SEIR epidemic models with generalized nonlinear incidence. Math. Comput. Simul. 170, 1–15 (2020)
Wei, F., Lin, Q.: Dynamical behavior for a stochastic epidemic model with nonlinear incidence. Acta Math. Sinica (Chin. Ser.) 61(1), 157–166 (2018)
Khan, M., Khan, Y., Islam, S.: Complex dynamics of an SEIR epidemic model with saturated incidence rate and treatment. Physica A 493, 210–227 (2018)
Liu, J., Wei, F.: Dynamics of stochastic SEIS epidemic model with varying population size. Physica A 464, 241–250 (2016)
Cao, B., Shan, M., Zhang, Q., Wang, W.: A stochastic SIS epidemic model with vaccination. Physica A 486, 127–143 (2017)
Liu, Q., Chen, Q., Jiang, D.: The threshold of a stochastic delayed SIR epidemic model with temporary immunity. Physica A 450, 115–125 (2016)
Gray, A., Greenhalgh, D., Hu, L., Mao, X., Pan, J.: A stochastic differential equation SIS epidemic model. SIAM J. Appl. Math. 71, 876–902 (2011)
Liu, J., Chen, L., Wei, F.: The persistence and extinction of a stochastic SIS epidemic model with logistic growth. Adv. Differ. Equ. 2018, 68 (2018)
Agaba, G., Kyrychko, Y., Blyuss, K.: Dynamics of vaccination in a time-delayed epidemic model with awareness. Math. Biosci. 294, 92–99 (2017)
Chen, L., Wei, F.: Persistence and distribution of a stochastic susceptible–infected–recovered epidemic model with varying population size. Physica A 483, 386–397 (2017)
Stolerman, L., Coombs, D., Boatto, S.: SIR-network model and its application to dengue fever. SIAM J. Appl. Math. 75, 2581–2609 (2015)
Hethcote, H., Van Den Driessche, P.: Some epidemiological models with nonlinear incidence. J. Math. Biol. 29, 271–287 (1991)
Hui, J., Chen, L.: Impulsive vaccination of SIR epidemic models with nonlinear incidence rates. Discrete Contin. Dyn. Syst., Ser. B 4, 595–605 (2004)
Anderson, R., May, R.: Infectious Diseases of Humans: Dynamics and Control. Oxford University Press, Oxford (1990)
May, R., Anderson, R.: Regulation and stability of host-parasite population interactions II: destabilizing process. J. Anim. Ecol. 47, 219–247 (1978)
Wei, F., Chen, F.: Asymptotic behaviors of a stochastic SIRS epidemic model with saturated incidence. J. Syst. Sci. Math. Sci. 36(12), 2444–2453 (2016)
Gan, S., Wei, F.: Study on a susceptible–infected–vaccinated model with delay and proportional vaccination. Int. J. Biomath. 11(8), 1850102 (2018)
Lu, R., Wei, F.: Persistence and extinction for an age-structured stochastic SVIR epidemic model with generalized nonlinear incidence rate. Physica A 513, 572–587 (2019)
Zhao, Y., Wei, F.: Impact of random perturbations with state-dependent on an epidemic model. J. Northeast Norm. Univ., Nat. Sci. Ed. 49(1), 9–14 (2017)
Van Den Driessche, P., Watmough, J.: Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 180, 29–48 (2002)
Diekmann, O., Heesterbeek, J.A.P.: On the definition and the computation of the basic reproduction ratio \(R_{0}\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28, 365–382 (1990)
Van Den Driessche, P., Watmough, J.: A simple SIS epidemic model with a backward bifurcation. J. Math. Biol. 40, 525–540 (2000)
Mao, X.R.: Stochastic Differential Equations and Applications, 2nd edn., pp. 11–26. Horwood, Chichester (2007)
Zhao, Y., Jiang, D.: Dynamics of stochastically perturbed SIS epidemic model with vaccination. Abstr. Appl. Anal. 2013, Article ID 517439 (2013)
Zhao, Y., Jiang, D.: The threshold of a stochastic SIS epidemic model with vaccination. Appl. Math. Comput. 243, 718–727 (2014)
Hasminskij, R.: Stochastic Stability of Differential Equations. Sijthoff & Noordhoff, Alphen aan den Rijn (1980)
Higham, D.J.: An algorithmic introduction to numerical simulation of stochastic differential equations. SIAM Rev. 43, 525–546 (2001)
Acknowledgements
The authors would like to thank the anonymous referees for their helpful and valuable comments and suggestions to improve our paper.
Availability of data and materials
Please contact one of all authors for any data requests if needed. E-mail addresses for all authors: chenlijun101086@163.com (L. Chen) and weifengying@fzu.edu.cn (F. Wei).
Funding
This work is supported by the National Natural Science Foundation of China (Grant No. 11201075), the Natural Science Foundation of Fujian Province of China (Grant No. 2016J01015), the Science and Technology Project of Education Department of Fujian Province of China (Grant No. JT180838).
Author information
Authors and Affiliations
Contributions
We declare that all the authors conceived of the study and carried out the proof. All authors of this paper read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors state that they have no competing interests in the manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, L., Wei, F. Study on a susceptible–exposed–infected–recovered model with nonlinear incidence rate. Adv Differ Equ 2020, 206 (2020). https://doi.org/10.1186/s13662-020-02662-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13662-020-02662-5