Abstract
Background
Pulmonary arterial hypertension (PAH) is associated with oxidative stress and affects the survival and homing of transplanted mesenchymal stem cells (MSCs) as well as cytokine secretion by the MSCs, thereby altering their therapeutic potential. In this study, we preconditioned the MSCs with prostaglandin E1 (PGE1) and performed in vitro and in vivo cell experiments to evaluate the therapeutic effects of MSCs in rats with PAH.
Methods
We studied the relationship between PGE1 and vascular endothelial growth factor (VEGF) secretion, B-cell lymphoma 2 (Bcl-2) expression, and C-X-C chemokine receptor 4 (CXCR4) expression in MSCs and MSC apoptosis as well as migration through the hypoxia-inducible factor (HIF) pathway in vitro. The experimental rats were randomly divided into five groups: (I) control group, (II) monocrotaline (MCT) group, (III) MCT + non-preconditioned (Non-PC) MSC group, (IV) MCT + PGE1-preconditioned (PGE1-PC) MSC group, and (V) MCT+PGE1+YC-1-PCMSC group. We studied methane dicarboxylic aldehyde (MDA) levels, MSC homing to rat lungs, mean pulmonary artery pressure, pulmonary artery systolic pressure, right ventricular hypertrophy index, wall thickness index (%WT), and relative wall area index (%WA) of rat pulmonary arterioles.
Results
Preconditioning with PGE1 increased the protein levels of HIF-1 alpha (HIF-1α) in MSCs, which can reduce MSC apoptosis and increase the protein levels of CXCR4, MSC migration, and vascular endothelial growth factor secretion. Upon injection with PGE1-PCMSCs, the pulmonary artery systolic pressure, mean pulmonary artery pressure, right ventricular hypertrophy index, %WT, and %WA decreased in rats with PAH. PGE1-PCMSCs exhibited better therapeutic effects than non-PCMSCs. Interestingly, lificiguat (YC-1), an inhibitor of the HIF pathway, blocked the effects of PGE1 preconditioning.
Conclusions
Our findings indicate that PGE1 modulates the properties of MSCs by regulating the HIF pathway, providing insights into the mechanism by which PGE1 preconditioning can be used to improve the therapeutic potential of MSCs in PAH.
Similar content being viewed by others
Background
Pulmonary arterial hypertension (PAH) is a progressive disease characterized by pulmonary vascular remodeling, which leads to increased pulmonary vascular resistance and right ventricle dysfunction [1]. Without treatment, the average duration of survival of patients with PAH was found to be 2.8 years [2]. Furthermore, patients with PAH treated with the appropriate drugs only survived for 7 years [3].
MSCs are multipotent, non-hematopoietic, fibroblast-like adherent cells that can be isolated from different tissue sources, such as the bone marrow, placenta, adipose tissue, dental pulp, and umbilical cord blood. They can differentiate into various types of cells and show specific surface antigen expression. In addition to their ability to differentiate, MSCs exert their therapeutic effects through the secretion of paracrine factors that have angiogenic, anti-apoptotic, anti-inflammatory, and immunomodulatory effects [4].
Stem cell therapy is a novel method for the treatment of PAH [5]. Mesenchymal stem cells (MSCs) serve as novel and effective treatment agents for PAH [6]. In experimental monocrotaline-induced PAH, MSC therapy improved hemodynamics by mitigating lung vascular remodeling [7, 8].
However, at present, there are several limitations to the treatment of PAH using MSCs. Multiple mechanisms in the process of PAH induction can cause severe oxidative stress [9]. Excessive and persistent oxidative stress can reduce the functional potential of MSCs and increase MSC apoptosis [10, 11]. Following their implantation, most MSCs are degraded and eliminated through the liver and spleen, and the number of cells homing to injured tissues is considerably low [12]. MSCs secrete vascular endothelial growth factor (VEGF), which can promote endothelial cell growth and angiogenesis and play an important role in PAH treatment [13]. Thus, the use of interventions that help maximize the therapeutic potential of MSCs has garnered significant interest.
The hypoxia-inducible factor (HIF) pathway is a key pathway in MSCs. More than 1000 genes are directly or indirectly regulated by HIF. Moreover, HIF affects major MSC features including cell viability, proliferation capacity, differentiation, migration pattern, and metabolism [14, 15]. Lificiguat (YC-1) is an inhibitor of the HIF pathway that specifically inhibits HIF-1 alpha (HIF-1α) expression [16].
B-cell lymphoma 2 (Bcl-2) is considered an important anti-apoptosis protein [17, 18]. We studied the degree of apoptosis in MSCs by measuring the protein levels of Bcl-2 and performed flow cytometry to detect the expression of specific cell markers. The stromal cell-derived factor 1 alpha (SDF-1α)/C-X-C chemokine receptor 4 (CXCR4) axis is of significance in MSC migration. The homing of MSCs can be evaluated by analyzing the presence of CXCR4 on the surface of MSCs and the SDF-1α-induced migration of MSCs [19]. In this study, we preconditioned MSCs with prostaglandin E1 (PGE1) and performed in vitro and in vivo cell experiments to evaluate the therapeutic effects of the MSCs in rats with PAH. Our findings showed that preconditioning MSCs with PGE1 improved their homing and reduced MSC apoptosis, thereby improving the overall therapeutic potential of the MSCs in the rat model of PAH.
Methods
Isolation, expansion, and labeling of MSCs
MSCs were harvested from the femur of young Wistar rats and anesthetized with 1% pentobarbital sodium. Alpha-Minimum essential medium (α-MEM; Biological Industries, Kibbutz Beit Haemek, Israel) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) was used as the culture medium. The bone marrow was flushed using a culture medium and then cultured at 37 °C in 5% CO2. Cell purification was achieved by washing cells with phosphate-buffered saline (PBS) and changing the culture medium after 48 h. Cells from the third to fourth generation were used in the experiments. Before transplantation, the MSCs were labeled with chloromethylbenzamido dialkylcarbocyanine (CM-Dil; BestBio, Shanghai, China) for in vivo cell tracking.
Preconditioning of MSCs
The MSCs were treated with PGE1 (Sigma-Aldrich, St. Louis, MO, USA) for 24 h. Treatment with YC-1 (MedChemExpress, Monmouth Junction, NJ, USA) was conducted for 12 h before treatment with PGE1. To study the survival and migration of MSCs under oxidative stress, the culture medium was replaced after 24 h of chemical pretreatment, which was followed by treatment with 200 μM H2O2 for 12 h.
PAH rat model
All experimental protocols were approved by the Animal Protection Committee of Qilu Hospital of Shandong University. All animals were cared for according to the guidelines for the nursing and use of experimental animals.
Adult male Wistar rats weighing 250 g–300 g (n = 48) were used for the experiment. Monocrotaline (MCT) was injected subcutaneously at a dose of 60 mg/kg (MedChemExpress). After 3 days, the CM-Dil-labeled MSCs (108/mL × 0.2 mL) were transplanted by injection through the tail vein. The experimental rats were randomly divided into five groups based on the treatment administered: (I) control group (injected with 0.2 ml PBS), n = 6; (II) MCT group (MCT without transplantation of MSCs), n = 6; (III) MCT + Non-PCMSC group (MCT and transplantation of non-preconditioned MSCs), n = 12; (IV) MCT + PGE1-PCMSC group (MCT and transplantation of PGE1-preconditioned MSCs), n = 12; and (V) MCT + PGE1+YC-1-PCMSC group (MCT and transplantation of PGE1 + YC-1-preconditioned MSCs), n = 12.
One week after the MSC transplantation, three rats were randomly selected from each group. The blood methane dicarboxylic aldehyde (MDA) and VEGF levels of these rats were measured using an enzyme-linked immunosorbent assay (ELISA).
Four weeks after the transplantation, six rats from groups III, IV, and V were humanely sacrificed for evaluating MSC homing using fluorescence microscopy. At the same time, the systolic blood pressure and mean pulmonary artery pressure of the remaining rats were measured, following which they were sacrificed. Rat hearts were dissected and weighed. The ratio of the weight of the right ventricle to the left ventricle + interventricular septum [RV/(LV + S)] was calculated to evaluate the degree of right ventricular hypertrophy. Lung tissue sections were stained with hematoxylin and eosin (HE). The wall thickness index (%WT) and relative wall area index (%WA) of pulmonary arterioles were calculated.
Cell surface phenotype analysis
Flow cytometry was performed to determine the phenotype of MSCs. MSCs were washed twice with washing buffer (PBS containing 3% FBS) and incubated for 30 min on ice with the following fluorescein isothiocyanate (FITC)- and phycoerythrin (PE)-conjugated antibodies in the washing buffer: anti-CD29 (1:200,eBioscience, San Diego, CA, USA), anti-CD44 (1:300,eBioscience), anti-CD45 (1:400,eBioscience), anti-CD90 (1:400,eBioscience), anti-CD34 (1:50, Abcam, Cambridge, MA, USA), and anti-CXCR4 (1:50, Abcam) antibodies. The MSCs were rinsed with the washing buffer again, and a FACSCalibur system (BD Biosciences, San Diego, CA, USA) was used for characterizing MSCs. Data were analyzed using the FlowJo software (Treestar, Ashland, OR, USA).
Western blotting (WB)
Proteins were extracted from the treated cells using an ice-cold lysis buffer containing phenylmethylsulfonyl fluoride. The protein content was determined using a bicinchoninic acid protein assay kit (Beyotime, Shanghai, China) and bovine serum albumin (BSA) as the standard. Proteins were separated using 10% SDS-PAGE and transferred to a PVDF membrane (Millipore Corp., Billerica, MA, USA) using the wet transfer method. Membranes were then blocked for 1 h with 5% skimmed milk or BSA in TBST and incubated overnight at 4 °C with the following primary antibodies: anti-HIF-1α (1:1000; Abcam), anti-CXCR4 (1:1000, Sigma-Aldrich), anti-Bcl-2 (1:200; Cell Signaling Technology, Danvers, MA, USA), and anti-β-tubulin (1:1000; Abcam) antibodies. β-tubulin served as the loading control. The PVDF membranes were then incubated for 1 h with the HRP-conjugated goat anti-rabbit IgG (1:1000; Beyotime). The resulting band intensity was quantified using ImageJ software (Rawak Software Inc., Stuttgart, Germany).
Apoptosis analysis
An annexin V-FITC/PI apoptosis detection kit (BD Biosciences) was used for evaluating cell apoptosis using flow cytometry.
Transwell migration assay
Migration assay was performed with a Transwell (Corning Inc., Corning, NY, USA) containing a polycarbonate membrane filter (8 μm pore size). The preconditioned MSCs (200 μL, 1.5 × 105/mL) were cultured onto the upper Transwell chamber, and medium containing SDF-1α (600 μL, 100 ng/mL; PeproTech Inc., Rocky Hill, NJ, USA) was added to the lower Transwell chamber to induce cell migration. After incubation for 12 h, the number of cells in five randomly selected microscopic fields was counted (200× magnification).
Measurement of MDA and VEGF concentrations using ELISA
Blood samples were collected from rats 1 week after the MSC transplantation. The serum MDA concentration was measured using an ELISA kit (Mlbio, Shanghai, China) to evaluate the oxidative stress levels.
MSCs were pretreated for 24 h, and the culture medium from each well was collected. The VEGF concentration in the medium was measured using an ELISA kit (Abbkine, Wuhan, China).
Immunofluorescence microscopy
The upper lobe of the right lung of the rats was removed and embedded in an optimal cutting temperature compound (SAKURA, Finetek, CA, USA). Slices (5 μm thick) were cut using a freezing microtome (Leica, Wetzlar, Germany). Nuclei were stained with 4ʹ,6-diamidino-2-phenylindole (Sigma-Aldrich). Stained sections were observed under fluorescence microscopy (Nikon, Tokyo, Japan). The homing ability of MSCs was evaluated based on the frequency of MSCs in lung tissue.
H&E staining
The left lungs were dehydrated in a graded ethanol series and embedded in paraffin. Slices (5 μm thick) were cut, deparaffinized, and stained with H&E. Sections were observed under an optical microscope (Olympus, Tokyo, Japan).
Statistical analysis
For data analysis, a one-way analysis of variance was performed, followed by Dunnett’s multiple comparisons test. Differences with P < 0.05 were considered statistically significant. Data analyses were performed using SPSS 19.0 (IBM, Armonk, NY, USA).
Results
Characterization of MSCs cultured in vitro
In vitro-cultured third-passage MSCs exhibited a uniform fibroblast-like morphology (Fig. 1A). Most CM-Dil-labeled MSCs emitted red fluorescence as observed under a fluorescence microscope (Fig. 1B). These cells tested negative for CD34 and CD45 but positive for CD90, CD44, and CD29 in the flow cytometry experiment (Fig. 1C). Therefore, the phenotype of the cell population used in our study was consistent with that in other studies [19, 20].
Characterization of MSCs. A MSCs exhibited a flat fibroblast-like morphology. B Chloromethylbenzamido dialkylcarbocyanine-labeled MSCs emitted red fluorescence. C Cultured MSCs were analyzed using flow cytometry and anti-CD90, anti-CD44, anti-CD29, anti-CD34, and anti-CD45 antibodies. MSC mesenchymal stem cell
Relationship among PGE1 administered at different concentrations and VEGF secretion, Bcl-2 expression, and CXCR4 expression in MSCs
The results of ELISA experiments showed that PGE1 (administered at different concentrations) promoted VEGF secretion from the MSCs. VEGF secretion was proportional to the PGE1 concentration and increased persistently with the increase in PGE1 concentration (Fig. 2A). The results of WB experiments showed that with the increase in PGE1 concentration, Bcl-2 and CXCR4 expression in MSCs increased. The effect was the strongest when 10 ng/mL PGE1 was administered but less pronounced when 20 ng/mL PGE1 was used (Fig. 2B). Therefore, 10 ng/mL PGE1 was used in the subsequent experiments.
A The PGE1-induced VEGF secretion from MSCs was proportional to the PGE1 concentration. B PGE1 treatment increased the protein levels of Bcl-2 and CXCR4 in MSCs, with the strongest effect observed upon treatment with 10 ng/mL PGE1 (P < 0.05). Bcl-2 B-cell lymphoma 2, CXCR4 C-X-C chemokine receptor 4, MSC mesenchymal stem cell, PGE1 prostaglandin E1, VEGF vascular endothelial growth factor
PGE1 inhibits H2O2-induced MSC apoptosis through the HIF-1α pathway
Flow cytometry revealed that MSC apoptosis increased in response to treatment with 200 μM H2O2. PGE1 reduced the apoptosis of MSCs when administered at 10 ng/mL, whereas 100 μM YC-1 reversed the protective effect of PGE1 (Fig. 3A; Additional file 1: Fig. S1). The WB experiment showed that the expression of Bcl-2 in MSCs decreased in response to treatment with H2O2. PGE1 increased HIF-1α expression, and YC-1 blocked this effect by inhibiting HIF-1α (Fig. 3B). To summarize, PGE1 increased the expression of Bcl-2 and reduced the apoptosis of MSCs by activating the HIF-1α pathway.
A Results of the flow cytometry experiment showed that MSC apoptosis increased in response to treatment with H2O2. PGE1 treatment reduced MSC apoptosis, whereas YC-1 blocked the protective effect of PGE1. B Results of the western blotting experiment showed that the protein levels of Bcl-2 in MSCs decreased in response to treatment with H2O2. Moreover, PGE1 increased the protein levels of HIF-1α, Bcl-2, and CXCR4, and YC-1 reversed the effects of PGE1 (P < 0.05). Bcl-2 B-cell lymphoma 2, CXCR4 C-X-C chemokine receptor 4, HIF-1α hypoxia-inducible factor 1 alpha, MSC mesenchymal stem cell, PGE1 prostaglandin E1
CXCR4 expression on the surface of MSCs and MSC migration
The results of the flow cytometry and Transwell migration assays showed that H2O2 treatment inhibited the expression of CXCR4 on the surface of MSCs and SDF-1α-induced MSC migration; however, there were no statistically significant differences. PGE1 increased the expression of CXCR4 on the surface of MSCs and MSC migration, with both effects being blocked by YC-1. This finding indicates that PGE1 increased the expression of CXCR4 on the surface of MSCs and promoted MSC migration through the HIF pathway (Fig. 4; Additional file 2: Fig. S2).
A As observed using flow cytometry, PGE1 increased the protein levels of CXCR4 on the surface of MSCs, an effect that was blocked by YC-1. B PGE1 increased SDF-1α-induced MSC migration in the Transwell migration assay, an effect that was blocked by YC-1 (P < 0.05). MSC mesenchymal stem cell, PGE1 prostaglandin E1, SDF-1α stromal cell-derived factor 1 alpha
PGE1 enhances VEGF secretion from MSCs through the HIF pathway
As observed using ELISA, H2O2 treatment marginally suppressed VEGF secretion from MSCs without any statistically significant difference. PGE1 increased VEGF secretion from MSCs, whereas YC-1 reversed this effect.
This indicates that PGE1 increased VEGF secretion from MSCs through the HIF pathway (Fig. 5; Additional file 3: Fig. S3).
Serum MDA levels in rats
ELISA using rat serum samples showed that the MDA levels increased 1 week after the MCT injection, which reflected the considerably high levels of oxidative stress in rats with PAH. The MDA levels decreased slightly in rats transplanted with non-preconditioned MSCs, which indicated a reduction in MCT-induced oxidative stress in vivo. Moreover, the MDA levels decreased significantly in rats transplanted with PGE1-preconditioned MSCs, meaning that the latter reduced the MCT-induced oxidative stress in vivo more potently than non-preconditioned MSCs. In rats transplanted with PGE1 + YC-1-preconditioned MSCs, the protective effects of PGE1 were reversed (Fig. 6).
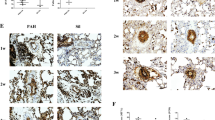
MSC homing to rat lungs
The proportion of MSCs that exhibited homing to lung tissues was estimated using fluorescence microscopy. Cells emitting red fluorescence were homing MSCs, whereas those emitting blue fluorescence were lung cells. The proportion of homing MSCs was the highest in the MCT + PGE1-PCMSC group, while that in the MCT + PGE1+YC-1-PC MSC group was similar to that in the MCT + Non-PCMSC group (Fig. 7).
The proportion of MSCs homing to the lungs in the MCT + PGE1-PCMSC group was higher than that in the MCT + PGE1+YC-1-PC MSC and MCT + Non-PCMSC groups (P < 0.05). MCT monocrotaline, MSC mesenchymal stem cell, Non-PC non-preconditioned, PGE1-PC prostaglandin E1-preconditioned, PGE1 + YC-1 prostaglandin E1-preconditioned plus YC-1-treated
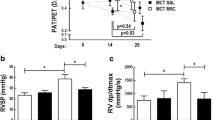
Mean pulmonary artery pressure, pulmonary artery systolic pressure, and right ventricular hypertrophy index in rats
The mean pulmonary artery pressure, pulmonary artery systolic pressure, and right ventricular hypertrophy index increased significantly in the control group. These parameters decreased marginally in the MCT + Non-PCMSC group and significantly in the MCT + PGE1-PCMSC group. The parameters obtained in the MCT + PGE1+YC-1-PCMSC group were similar to those obtained in the MCT + Non-PCMSC group (Fig. 8).
%WT and %WA of rat pulmonary arterioles
Lung tissue sections were subjected to HE staining, and the %WT and %WA of pulmonary arterioles were calculated. These parameters increased significantly in the control group but were slightly lower in the MCT + Non-PCMSC group and decreased significantly in the MCT + PGE1-PCMSC group. The parameters obtained in the MCT + PGE1+YC-1-PCMSC group were similar to those obtained in the MCT + Non-PCMSC group (Fig. 9).
%WT and %WA of rats from the five groups. A Control group. B MCT group. C MCT + Non-PCMSC group. D MCT + PGE1-PCMSC group. E MCT + PGE1+YC-1-PCMSC group (P < 0.05). MCT monocrotaline, MSC mesenchymal stem cell, Non-PC non-preconditioned, PGE1-PC prostaglandin E1-preconditioned, PGE1 + YC-1 prostaglandin E1-preconditioned plus YC-1-treated, %WA relative wall area index, %WT wall thickness index
Discussion
PAH is a difficult disease to treat, as traditional treatment methods mostly exhibit limited effectiveness. The application of stem cell therapy to PAH treatment is a novel approach [4]. Clinical trials have shown that the transplantation of endothelial progenitor cells (EPCs) can exert beneficial effects on the exercise ability and pulmonary hemodynamics of patients with PAH [21, 22]. At present, there are no ongoing clinical trials on MSC treatment. However, the therapeutic potential of MSCs has been demonstrated in preclinical studies. Compared with treatment with EPCs, treatment with MSCs is potentially advantageous and allows the allogeneic transplantation of cells without the need for immunosuppression. Therefore, MSCs are an attractive source of donor cells for PAH therapy [1].
The application of MSCs in PAH treatment is based on the homing of MSCs to the site of injury, differentiation into endothelial cells, repair of injured endothelial cells, and concurrent secretion of paracrine factors, including various cytokines and mediators (such as VEGF) that promote endothelial cell repair and angiogenesis, thereby improving the symptoms of PAH [1, 23]. Here, we tested a novel strategy for reducing apoptosis after MSC implantation, improving the homing rate of MSCs, and increasing the production of cytokines, including VEGF, which would help to treat PAH.
HIF-1α and its target genes are associated with apoptosis, cell migration, and secretion of various factors from cells. MSCs subjected to hypoxia and expressing increased levels of HIF-1α exhibited higher cell migration and transplant survival rates than untreated MSCs [15]. The pharmacological stabilization of HIF-1α can increase CXCR4 secretion and enhance the homing and implantation of stem cells [24]. VEGF is an important target gene of HIF-1α; therefore, HIF-1α activation increases VEGF expression [25, 26].
The use of PGE1, a prostaglandin commonly produced in the human body, was approved by the Food and Drug Administration in 1981 for the treatment of infants with catheter-dependent heart disease (CHD) who require the maintenance of an unobstructed catheter until palliative or corrective surgery can be performed. PGE1 is often used in newborns diagnosed with CHD immediately after birth [27].
It was shown that treatment with MSCs and PGE1 in rats significantly reduced apoptosis induced by serum deprivation, decreased the protein levels of Bax and caspase-3, and increased that of Bcl-2; however, the underlying mechanism remained unclear [28]. Prostaglandin E2 (PGE2) can increase HIF-1α expression in hematopoietic stem/progenitor cells (HSPCs), thereby increasing CXCR4 expression and promoting the homing of HSPCs [29]. Treatment of human prostate cancer cells with PGE2 enhanced VEGF expression by regulating HIF-1α expression [30].
We speculated that PGE1 may affect the function of MSCs through the HIF pathway. Using ELISA, we confirmed that the serum MDA concentration increased significantly 1 week after MCT injection, which indicated that the oxidative stress level in rats with PAH was considerably high. Oxidative stress is closely related to many heart diseases, such as atherosclerosis and clinical aortic valve disease [31].
Furthermore, we used 200 μM H2O2 to simulate oxidative stress in vivo [20], and MSC apoptosis increased observably upon this treatment. The expression of Bcl-2 in MSCs was low under oxidative stress. Transplantation of PGE1-pretreated MSCs activated the HIF-1α pathway, increased Bcl-2 expression, and suppressed MSC apoptosis. Our hypothesis was further validated when treatment with YC-1 suppressed the protective effects of PGE1-preconditioned MSCs.
After MSC transplantation, the number of MSCs homing to injured tissues is considerably small, and the percentage of MSCs detected in the lungs 21 days after transplantation is estimated to be 0.25%–0.375% of the MSCs initially administered; the homing of a low number of MSCs severely affects the functions of MSCs [12]. In this experiment, the Non-PCMSC group exhibited low expression of intracellular and cell surface CXCR4, and only a few cells underwent SDF-1α-induced migration in vitro. In vivo experiments in the rats indicated that only a small number of non-preconditioned MSCs exhibited homing to the lung tissue. PGE1 treatment increased CXCR4 expression on the MSC surface and SDF-1α-induced MSC migration, whereas YC-1 treatment suppressed the aforementioned effects. This indicates the role of the HIF pathway in the protective effects of PGE1 on MSCs. Moreover, MSCs can secrete VEGF, which plays an important role in the treatment of PAH [13]. PGE1 preconditioning increased VEGF secretion in these cells through the HIF-1α pathway.
In vivo experiments in the rats indicated that the therapeutic effects of PGE1-preconditioned MSC transplantation were better than those of the non-preconditioned MSC transplantation. Moreover, the pulmonary artery systolic pressure, mean pulmonary artery pressure, right ventricular hypertrophy index, %WT, and %WA were lower in rats transplanted with PGE1-preconditioned MSCs than in the other groups.
Conclusions
In our study, we confirmed that the pretreatment of MSCs with PGE1 can activate the HIF-1α pathway, reduce MSC apoptosis, and increase MSC migration to the site of injury and VEGF secretion. Rats implanted with PGE1-preconditioned MSCs experienced better therapeutic effects than those implanted with non-preconditioned MSCs. Our study indicated that PGE1 preconditioning can be used to improve the therapeutic potential of MSCs in PAH.
Availability of data and materials
All the data generated or analyzed during this study are included in this published article.
Abbreviations
- PAH:
-
Pulmonary arterial hypertension
- MSCs:
-
Mesenchymal stem cells
- VEGF:
-
Vascular endothelial growth factor
- HIF:
-
Hypoxia-inducible factor
- HIF-1α:
-
HIF 1 alpha
- YC-1:
-
Lificiguat
- Bcl-2:
-
B-cell lymphoma 2
- SDF-1α:
-
Stromal cell-derived factor 1 alpha
- CXCR4:
-
C-X-C chemokine receptor 4
- PGE1:
-
Prostaglandin E1
- FBS:
-
Fetal bovine serum
- PBS:
-
Phosphate-buffered saline
- CM-Dil:
-
Chloromethylbenzamido dialkylcarbocyanine
- MCT:
-
Monocrotaline
- MDA:
-
Methane dicarboxylic aldehyde
- ELISA:
-
Enzyme-linked immunosorbent assay
- HE:
-
Hematoxylin and eosin
- FITC:
-
Fluorescein isothiocyanate
- PE:
-
Phycoerythrin
- WB:
-
Western blotting
- BSA:
-
Bovine serum albumin
- EPCs:
-
Endothelial progenitor cells
- CHD:
-
Catheter-dependent heart disease
- PGE2:
-
Prostaglandin E2
- HSPCs:
-
Hematopoietic stem/progenitor cells
- α-MEM:
-
Alpha-Minimum essential medium
References
Fukumitsu M, Suzuki K. Mesenchymal stem/stromal cell therapy for pulmonary arterial hypertension: comprehensive review of preclinical studies. J Cardiol. 2019;74:304–12.
D’Alonzo GE, Barst RJ, Ayres SM, Bergofsky EH, Brundage BH, Detre KM, et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med. 1991;115:343–9.
Benza RL, Miller DP, Barst RJ, Badesch DB, Frost AE, McGoon MD. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the REVEAL Registry. Chest. 2012;142:448–56.
Nammian P, Asadi-Yousefabad SL, Daneshi S, Sheikhha MH, Tabei SMB, Razban V. Comparative analysis of mouse bone marrow and adipose tissue mesenchymal stem cells for critical limb ischemia cell therapy. Stem Cell Res Ther. 2021;12:58.
Loisel F, Provost B, Haddad F, Guihaire J, Amsallem M, Vrtovec B, et al. Stem cell therapy targeting the right ventricle in pulmonary arterial hypertension: Is it a potential avenue of therapy? Pulm Circ. 2018;8:2045893218755979.
Cruz FF, Rocco PRM. The potential of mesenchymal stem cell therapy for chronic lung disease. Expert Rev Respir Med. 2020;14(1):31–9.
de Mendonça L, Felix NS, Blanco NG, Da Silva JS, Ferreira TP, Abreu SC, et al. Mesenchymal stromal cell therapy reduces lung inflammation and vascular remodeling and improves hemodynamics in experimental pulmonary arterial hypertension. Stem Cell Res Ther. 2017;8:220.
Zhang Q, Cheng X, Zhang H, Zhang T, Wang Z, Zhang W, et al. Dissecting molecular mechanisms underlying H2O2-induced apoptosis of mouse bone marrow mesenchymal stem cell: role of Mst1 inhibition. Stem Cell Res Ther. 2020;11:526.
Tabima DM, Frizzell S, Gladwin MT. Reactive oxygen and nitrogen species in pulmonary hypertension. Free Radic Biol Med. 2012;52:1970–86.
Kawashiri MA, Nakanishi C, Tsubokawa T, Shimojima M, Yoshida S, Yoshimuta T, et al. Impact of enhanced production of endogenous heme oxygenase-1 by pitavastatin on survival and functional activities of bone marrow-derived mesenchymal stem cells. J Cardiovasc Pharmacol. 2015;65:601–6.
Kim JY, Lee JS, Han YS, Lee JH, Bae I, Yoon YM, et al. Pretreatment with lycopene attenuates oxidative stress-induced apoptosis in human mesenchymal stem cells. Biomol Ther. 2015;23:517–24.
Jiang L, Song XH, Liu P, Zeng CL, Huang ZS, Zhu LJ, et al. Platelet-mediated mesenchymal stem cells homing to the lung reduces monocrotaline-induced rat pulmonary hypertension. Cell Transplant. 2012;21:1463–75.
Smadja DM, Levy M, Huang L, Rossi E, Blandinières A, Israel-Biet D, et al. Treprostinil indirectly regulates endothelial colony forming cell angiogenic properties by increasing VEGF-A produced by mesenchymal stem cells. Thromb Haemost. 2015;114:735–47.
Ejtehadifar M, Shamsasenjan K, Movassaghpour A, Akbarzadehlaleh P, Dehdilani N, Abbasi P, et al. The effect of hypoxia on mesenchymal stem cell biology. Adv Pharm Bull. 2015;5:141–9.
Meng SS, Xu XP, Chang W, Lu ZH, Huang LL, Xu JY, et al. LincRNA-p21 promotes mesenchymal stem cell migration capacity and survival through hypoxic preconditioning. Stem Cell Res Ther. 2018;9:280.
Wu K, Zhou K, Wang Y, Zhou Y, Tian N, Wu Y, et al. Stabilization of HIF-1α by FG-4592 promotes functional recovery and neural protection in experimental spinal cord injury. Brain Res. 2016;1632:19–26.
Liu L, Liu L, Shi J, Tan M, Xiong J, Li X, et al. MicroRNA-34b mediates hippocampal astrocyte apoptosis in a rat model of recurrent seizures. BMC Neurosci. 2016;17:56.
He G, Li LI, Guan E, Chen J, Qin YI, Xie Y. Fentanyl inhibits the progression of human gastric carcinoma MGC-803 cells by modulating NF-κB-dependent gene expression in vivo. Oncol Lett. 2016;12:563–71.
Bing W, Pang X, Qu Q, Bai X, Yang W, Bi Y, et al. Simvastatin improves the homing of BMSCs via the PI3K/AKT/miR-9 pathway. J Cell Mol Med. 2016;20:949–61.
Bai X, Xi J, Bi Y, Zhao X, Bing W, Meng X, et al. TNF-α promotes survival and migration of MSCs under oxidative stress via NF-κB pathway to attenuate intimal hyperplasia in vein grafts. J Cell Mol Med. 2017;21:2077–91.
Wang XX, Zhang FR, Shang YP, Zhu JH, Xie XD, Tao QM, et al. Transplantation of autologous endothelial progenitor cells may be beneficial in patients with idiopathic pulmonary arterial hypertension: a pilot randomized controlled trial. J Am Coll Cardiol. 2007;49:1566–71.
Granton J, Langleben D, Kutryk MB, Camack N, Galipeau J, Courtman DW, et al. Endothelial NO-synthase gene-enhanced progenitor cell therapy for pulmonary arterial hypertension: the PHACeT trial. Circ Res. 2015;117:645–54.
Xiao Y, Chen PP, Zhou RL, Zhang Y, Tian Z, Zhang SY. Pathological mechanisms and potential therapeutic targets of pulmonary arterial hypertension: a review. Aging Dis. 2020;11:1623–39.
Speth JM, Hoggatt J, Singh P, Pelus LM. Pharmacologic increase in HIF1α enhances hematopoietic stem and progenitor homing and engraftment. Blood. 2014;123:203–7.
Lin CJ, Lan YM, Ou MQ, Ji LQ, Lin SD. Expression of miR-217 and HIF-1α/VEGF pathway in patients with diabetic foot ulcer and its effect on angiogenesis of diabetic foot ulcer rats. J Endocrinol Invest. 2019;42:1307–17.
Zhang PC, Liu X, Li MM, Ma YY, Sun HT, Tian XY, et al. AT-533, a novel Hsp90 inhibitor, inhibits breast cancer growth and HIF-1α/VEGF/VEGFR-2-mediated angiogenesis in vitro and in vivo. Biochem Pharmacol. 2020;172: 113771.
Akkinapally S, Hundalani SG, Kulkarni M, Fernandes CJ, Cabrera AG, Shivanna B, et al. Prostaglandin E1 for maintaining ductal patency in neonates with ductal-dependent cardiac lesions. Cochrane Database Syst Rev. 2018;2:CD011417.
Zeng K, Deng BP, Jiang HQ, Wang M, Hua P, Zhang HW, et al. Prostaglandin E1 protects bone marrow-derived mesenchymal stem cells against serum deprivation-induced apoptosis. Mol Med Rep. 2015;12:5723–9.
Speth JM, Hoggatt J, Pelus LM. Enhanced homing of HSPCs after treatment with prostaglandin E2 (PGE2) may be an effect of transcriptional regulation of CXC chemokine receptor 4 (CXCR4) through hypoxia inducible factor 1α (HIF1α). Blood. 2011;118:918.
Liu XH, Kirschenbaum A, Lu M, Yao S, Dosoretz A, Holland JF, et al. Prostaglandin E2 induces hypoxia-inducible factor-1alpha stabilization and nuclear localization in a human prostate cancer cell line. J Biol Chem. 2002;277:50081–6.
Dutta P, Lincoln J. Calcific aortic valve disease: a developmental biology perspective. Curr Cardiol Rep. 2018;20:21.
Acknowledgments
Not applicable.
Funding
Supported by Qingdao Key Health Discipline Development Fund.
Author information
Authors and Affiliations
Contributions
D-TJ participated in the conception and design of the study and manuscript writing. LT, XB, W-DB, and Q-XQ participated in the acquisition, analysis, and interpretation of the data. XZ and G-MS participated in the design of the study and coordination and in reviewing the intellectual content. Y-WB and W-YS performed the statistical analysis and helped to draft the manuscript. No work was outsourced. All data were collected by the authors. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All experimental protocols were approved by the Animal Protection Committee of Qilu Hospital of Shandong University. All animals were cared for according to the guidelines for the nursing and use of experimental animals.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1. Fig. S1
: Results of the flow cytometry experiment showing that MSC apoptosis increases in response to treatment with H2O2. PGE1 treatment reduces MSC apoptosis, whereas YC-1 blocks the protective effect of PGE1.
Additional file 2. Fig. S2
: PGE1 increases SDF-1α-induced MSC migration in the Transwell migration assay; this effect was blocked by YC-1 treatment.
Additional file 3. Fig. S3
: PGE1 increases VEGF secretion from MSCs, which was blocked by YC-1 treatment.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Jiang, DT., Tuo, L., Bai, X. et al. Prostaglandin E1 reduces apoptosis and improves the homing of mesenchymal stem cells in pulmonary arterial hypertension by regulating hypoxia-inducible factor 1 alpha. Stem Cell Res Ther 13, 316 (2022). https://doi.org/10.1186/s13287-022-03011-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13287-022-03011-x