Abstract
Background
The human protozoan parasite Trichomonas vaginalis is an organism of interest for understanding eukaryotic evolution. Despite having an unusually large genome and a rich gene repertoire among protists, spliceosomal introns in T. vaginalis appear rare: only 62 putative introns have been annotated in this genome, and little or no experimental evidence exists to back up these predictions.
Results
This study revisited the 62 annotated introns of T. vaginalis derived from the genome sequencing plus previous publications. After experimental validation and a new genome-wide search, we confirmed the presence of introns in 32 genes and 18 others were concluded to be intronless. Sequence analyses classified the validated introns into two types, based on distinctive features such as length and conservation of splice site motifs.
Conclusions
Our study provides an updated list of intron-containing genes in the genome of T. vaginalis. Our findings suggests the existence of two intron ‘families’ spread among T. vaginalis protein-coding genes. Additional studies are needed to understand the functional separation of these two classes of introns and to assess the existence of further introns in the T. vaginalis genome.
Similar content being viewed by others
Background
Introns, intervening non-coding sequences in genes of eukaryotes, are precisely removed from pre-mRNA by splicing, yielding mature mRNA. Splicing is achieved by a ribonucleoprotein complex (i.e. the spliceosome) through recognition of sequence elements on the introns such as the 5' and 3' splice sites (SS) and the branch site (BS) [1]. Phylogenomic analyses indicate that components of the spliceosome are conserved across eukaryotes [2], and that the last common ancestor of all eukaryotes contained an intron-rich genome [3, 4]. That said, identification of spliceosomal introns and small nuclear RNAs often requires careful experimental study [5,6,7].
One species where our understanding of introns remains particularly patchy is Trichomonas vaginalis. This is a protozoan parasite of the human urogenital tract and the causative agent of trichomoniasis, the most prevalent non-viral sexually transmitted infection worldwide [8]. As a member of the Excavata, a major eukaryotic supergroup [9, 10], T. vaginalis has experienced a recent genome expansion as a result of gene duplications, transposon activities and lateral gene transfer [11, 12]. The relatively large genome of T. vaginalis (~170 Mbp) harbours a protein-coding capacity of ~60,000 genes [11], and there is evidence of expression for about half of these [13, 14]. Despite this large gene repertoire, the number of T. vaginalis genes that contain spliceosomal introns is predicted to be very low (~0.001 introns/gene) [11, 15, 16]. Unicellular eukaryotes often have intron-poor genomes as compared to metazoans and plants but exceptions exist [17,18,19,20,21]. Kipferlia bialata, also a member of the Excavata, exhibits an average of ~7 introns/gene which is similar to the most intron-rich eukaryotic genomes [20].
While intron density appears low in T. vaginalis genome, it is not clear whether this is a result of natural variation or limited data. It is also worth noting that only a very few introns have been assigned to protein-coding genes of T. vaginalis experimentally [15, 16]. Vanacova et al. [15] were the first to demonstrate splicing activity in T. vaginalis. A 35-nt intron, modified from Giardia lamblia [22], was shown to be spliced out from a reporter gene in T. vaginalis [15]. Mutagenesis demonstrated a requirement for conserved splicing motifs and searching for this strict motif yielded the identification of 41 putative introns in T. vaginalis protein-coding genes [15]. However, searching for a strict motif, based solely on mutagenesis of a single Giardia-derived intron [15], suggests that other introns might exist. Indeed, Deng et al. [16] identified a 25nt-long intron in the gene Rab1a of T. vaginalis where the splicing motifs did not match to the strict motif used by the previous report [15]. The draft genome of T. vaginalis, released between the aforementioned studies, included the annotation of 62 introns among the ~60,000 predicted protein-coding genes [11].
Given the limited data currently available, it is therefore difficult to ascertain whether these observations indicate that T. vaginalis has an intron-poor genome and short introns with conserved SS and BS, or whether other introns may be present in the genome. As a first step towards addressing this question, we revisited all T. vaginalis introns that were predicted from the genome annotation and described in previous publications [11, 15, 16]. We applied reverse transcription and PCR to validate all introns in T. vaginalis experimentally. Splicing activity was confirmed by sequencing the exon-exon boundaries of the spliced products. Our results support the existence of two types of introns in T. vaginalis with distinctive length, SS and BS features.
Methods
Trichomonas vaginalis culture and purification of nucleic acids
T. vaginalis strain G3 was cultured in Diamond’s media [23] supplemented with 10% heat-inactivated horse serum, penicillin (1000 units/ml) and streptomycin (0.1 mg/ml). Genomic DNA (gDNA) was isolated using a modified protocol [24]. Briefly, a total of 10 ml of cells (106 cells/ml) were washed and resuspended in 0.9 ml of phosphate-saline buffer. Cells were lysed by adding 0.1 ml of lysis buffer (8M urea; 2% sarkosyl; 150 mM NaCl, 1 mM EDTA and 100 mM Tris-HCl pH 7.5). Lysis was followed by phenol and chloroform extractions and the DNA/RNA from the aqueous phase was precipitated with 0.6 volumes of isopropanol. After washing with 70% ethanol and air-drying, the pellet was resuspended in 0.5 ml of TE buffer (10 mM Tris-HCl pH 8.0; 1 mM EDTA) followed by digestion with RNase A (5 μg/ml) and stored for use. Total RNA was extracted using TRIzol Reagent (Invitrogen, Waltham, USA) and treated with DNase I (New England Biolabs, Ipswich, USA) to remove gDNA contamination.
Retrieval of multi-exon genes
Sequences of the 61 T. vaginalis protein-coding genes with exon count ≥ 2 were downloaded from trichdb.org (total 62 introns). The available features of the 42 intron-containing sequences described by the two early studies (i.e. full or partial sequences, intron length, 5' and 3' SS, flanking exon sequences) were extracted from the original publications [15, 16] and used to match against the genome annotation of T. vaginalis strain G3. The old gene IDs given by the early publications were replaced with the TrichDB ID if the intron fragment was located in a protein-coding gene according to the current genome annotation.
Experimental validation of introns
Splicing activity was assessed by reverse transcription and PCR (RT-PCR). The first-strand, complementary DNA (cDNA) was synthesised from the purified total RNA using SuperScript III Reverse Transcriptase (RT) and oligo dT primer as recommended (Thermo Fisher Scientific, Waltham, USA). gDNA and cDNA were used as templates for polymerase chain reaction (PCR) using specific primers that target the flanking regions of the putative intronic sequences (Additional file 1: Table S1). As negative controls, water and a cDNA sample without reverse transcriptase (-RT) were used in the PCR reactions instead of gDNA or cDNA templates. PCR reactions were initially carried out using DreamTaq DNA Polymerase, as recommended (Thermo Fisher Scientific). Alternatively, for those PCR reactions that did not yield clear results, Phusion DNA Polymerase was used with either buffers as provided (Thermo Fisher Scientific) and by optimizing annealing temperature as detailed in Additional file 1: Table S1. Along with a 100 bp DNA ladder, PCR products from each set of primers were compared side by side by electrophoresis on a 2% (w/v) agarose in a Tris-Borate-EDTA buffer containing 0.5 μg/ml ethidium bromide (Thermo Fisher Scientific). For all newly discovered intron-containing genes in this study, the RT-PCR amplicons of the spliced products were eluted from the agarose gels and the DNA was sequenced. Each sequencing result was manually aligned to the original gene sequence to determine the exact exon-exon boundary.
Using RNA sequencing data to evaluate splicing activity of undetermined introns
A strategy based on the available RNA sequencing (RNA-Seq) data was used to evaluate the splicing activity of 13 introns that could not be determined experimentally, as described above (i.e. undetermined introns). The two largest and most recent T. vaginalis RNA-Seq datasets (SRX2311573 and SRX2311572) were downloaded from the Sequence Read Archive at the National Center for Biotechnology Information (https://www.ncbi.nlm.nih.gov/sra). These datasets contain expressed sequences of T. vaginalis strains G3 and B7RC2, respectively. Over 180 and 160 million sequencing reads of 125 bp long from these 2 datasets were imported to Geneious 11.1.2 separately and mapped to the 13 genes carrying the undetermined introns. The genes TVAG_416520 and TVAG_337250, containing a functional and a non-functional intron, were chosen as positive and negative controls, respectively. The number of sequencing reads from the RNA-Seq data covering each nucleotide position was determined for each gene. A marked drop in sequencing depth on the intron regions as compared to their respective flanking exons, and in reference to controls above, was considered as evidence of splicing activity.
Analyses of the intron features and the search for additional intron-containing genes
Sequence properties of the newly discovered introns in this study were described using a Python script which reads FASTA sequences one by one and retrieves all intron-related information as presented here. Introns were classified here into two different families, based on sequence characteristics and putative splicing motifs. The consensus sequences of the two intron families in T. vaginalis were updated, according to the previous studies on T. vaginalis introns [15, 16]. These two newly emerged patterns ('GYAYGYN{41,178}RCTAACACAYAG' and 'GTWYWDN{7}TCTAACH{1,2}AACAG', regular expressions correspond to Python syntax) were used to search de novo for matches on T. vaginalis genome segments using the DNA motif pattern search tool at TrichDB (http://trichdb.org/trichdb/showQuestion.do?questionFullName=SpanQuestions.DynSpansByMotifSearch). Potential hits found by this search were subjected to the same RT-PCR validation, as described above.
Results
An overview of the putative list of intron-containing genes in T. vaginalis
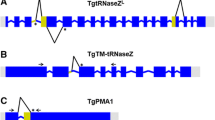
In this study, we interrogated two datasets of intron-containing genes in T. vaginalis (Fig. 1). The first dataset is represented by 61 genes, derived from the genome annotation. These genes contain 62 putative introns and are labelled as multi-exon genes by TrichDB (trichdb.org). The second dataset consists of 42 gene sequences, each carrying a single putative intron that were described in earlier publications [15, 16]. The available sequences of these 42 putative intron-containing genes, extracted from earlier publications [15, 16], were used to search for annotated genes in the T. vaginalis genome. Nineteen of the 41 putative intron fragments predicted by Vanacova et al. [15] were located in the transcribed region of protein-coding genes and annotated as multi-exon genes by TrichDB (Fig. 1). The remaining 22 fragments were exclusively present in non-transcribed intergenic regions (as supported by the EST data available at TrichDB), hence were not considered further. TvRab1a, the only intron-containing gene confirmed by Deng et al. [16], was annotated in TrichDB as one of the 61 multi-exon genes (Fig. 1). The strict motif sequence search used in the genome-wide search by Vanacova et al. [15] did not identify this intron (Fig. 1). The gene IDs were updated where applicable (Additional file 2: Table S2).
Experimental validation of T. vaginalis introns
Primers flanking all 62 putative introns were designed to assess splicing activity using RT-PCR, gel electrophoresis and DNA sequencing (Additional file 1: Table S1). PCR reactions were performed on gDNA and cDNA templates. Negative PCR controls (water and -RT) were included to ensure that reactions were free of DNA contamination. While a single amplicon of expected size from the gDNA demonstrated the reliability of PCR, amplification from the cDNA demonstrates that the gene is transcribed. Size comparison between the amplicons produced from cDNA and gDNA should indicate if the intron has been spliced out. We also considered that both unspliced and spliced mRNAs can be represented in a cDNA sample, hence double bands may be observed.
Based on the expectations above, we classified introns into three mutually exclusive categories: (A) functional; (B) non-functional or (C) undetermined (Additional file 3: Figure S1). We identified 31 functional introns based on the detection of the spliced amplicon from the cDNA, always followed by unspliced amplicon from the gDNA. The cDNA may also produce the unspliced amplicon, indicated by double bands in some examples (Additional file 3: Figure S1a). Contrastingly, although the unspliced amplicon was detected from both the gDNA and cDNA, 18 introns were categorized as non-functional because the cDNA did not produce the spliced amplicon (Additional file 3: Figure S1b). The remaining 13 introns were classified as undetermined (Additional file 3: Figure S1c). This is because either the mRNA transcript was not detected (i.e. no amplicon from cDNA) or there were no size-reliable amplicons from cDNA and/or gDNA templates (such as a lack of amplification, smeary amplification or multiple bands), despite PCR optimization (as described in Methods and Additional file 1: Table S1).
To sum up, the experimental validation confirmed the presence of functional introns in all of the 20 protein-coding genes claimed by previous studies [15, 16] (Table 1, Fig. 1). Additionally, we were able to experimentally confirm the presence of 11 additional introns in protein-coding genes of T. vaginalis. Together, all 31 functional introns were found to be located in the coding sequence (CDS) of the protein-coding genes as one intron per gene. Finally, 18 introns in 17 protein-coding genes were assigned as non-functional as they were certainly transcribed but the predicted intronic sequences were not removed (Table 1).
Characterisation of the newly discovered intron-containing genes
To confirm splicing activity of the 11 newly-discovered introns, the PCR products corresponding to the spliced amplicons were gel-purified and sequenced. DNA sequencing confirmed the predicted exon boundaries precisely for all 11 protein-coding genes interrupted by these introns (Fig. 2). The main features of these intron-containing genes are summarized in Table 2. They code for hypothetical proteins (6/11), kinases (4/11) and a eukaryotic protein belonging to the Mob1/phocein family (1/11). Eight of 11 introns were very short in length (25–26 nt), a feature shared with the short intron found in the TvRab1a gene [16]. Intronic sequences were shown to exhibit a lower GC content than exonic sequences, without exception (Table 2). Nine of 11 introns were in phases 1 or 2 (i.e. the intron interrupts a codon). Also, 9 of 11 introns were found in the first quarter of their open reading frames (Table 2).
The exon boundaries of the 11 newly discovered intron-containing genes in T. vaginalis. Gel images, cropped from Additional file 3: Figure S1, show the unspliced and spliced amplicons in the first and second lanes, respectively followed by the 100 bp DNA ladder (New England Biolabs). The TrichDB ID of the genes is shown on the top of each gel image followed by the expected bp size of unspliced | spliced amplicons. Each gel image is accompanied by part of the DNA sequencing chromatogram where the line indicates the precise boundary between exon 1 and 2. The actual DNA sequence is shown under the chromatogram with the arrow indicating the nucleotide boundary between exons. The image in the box contains the 100 bp DNA ladder for reference
Manual inspection of the new 11 introns allowed us to classify them into two distinct types, named here types A and B, based on their sequence properties and splicing motifs (Fig. 3). Types A and B were represented by 3 and 8 introns and were closely related to the existing introns previously reported by Vanacova et al. [15] and Deng et al. [16], respectively. Type A introns show a one nucleotide mismatch to the strict consensus used for the genome-wide search from the previous study [15], precisely the first nucleotide on the 12nt-motif that encompasses the BS and the 3' SS. A guanosine instead of an adenosine at position one in this motif, just before the yeast consensus BS sequence ACTAAC [25], was found in the gene TVAG_217460. The intron length, however, was within the range of 41–178 nt as previously reported [15]. All three new introns of type A display the conserved 7-nt distance between the branch adenosine and the 3' SS, reiterating the previous observation [15] (Fig. 3).
The 11 newly discovered T. vaginalis introns are classified into two types based on their sequence properties. Types A (top) and B (bottom) fit closely with the introns described by Vanacova et al. [15] and Deng et al. [16], respectively. The nucleotides of the newly discovered introns that are identical to the previously identified introns [15, 16] are shaded in grey. The branch site sequence, initially described as identical to the yeast consensus [26], is underlined with the red arrowhead indicating the branch adenosine. The distance in nucleotides (nt) between the 5' SS and the motif that encompasses the BS and 3' SS is indicated. Based on the intron nucleotide sequences, the consensus sequences for intron types A and B are shown below each alignment. Nucleotide ambiguity represents: ‘W’ to A or T; ‘Y’ to T or C; ‘H’ to A, C or T and ‘{1,2}’ specifies that ‘H’ can be one or two nucleotides
Type B introns are notably short, between 25–26 nt long. In comparison to type A, they show a lower sequence conservation at the 5' SS. Also, the consensus BS sequence of type B introns places a T as the first nucleotide (TCTAAC) instead of A or G as in type A introns (RCTAAC). In contrast to type A introns, a space flexibility between the branch adenosine and the 3' SS is apparently allowed for type B introns. The type B intron found in the gene TVAG_416520 shows that this distance can be either 7 or 8 nt (Fig. 3).
A genome-wide search for additional intron-containing genes
With a consensus sequences for intron types A and B (Fig. 3), we searched for additional intron-containing genes in the T. vaginalis genome. As a result, 52 and 32 genomic segments were found to contain the consensus sequences for introns types A and B, respectively (Additional file 4: Table S3, Additional file 5: Table S4). We identified 10 new sequences that resembled type A introns, but none were present in protein-coding genes (Additional file 4: Table S3). The other 22 segments, found in protein-coding genes, had already been validated by PCR in this study (Additional file 3: Figure S1, Additional file 4: Table S3). Therefore, no additional type A introns could be assigned to protein-coding genes from this new genome-wide search.
On the search for type B introns, however, a novel 25nt-long intron was found in a gene coding for a hypothetical protein (TVAG_269270). As this intron had not been experimentally validated (Additional file 3: Figure S1, Additional file 5: Table S4), we applied the same experimental approach to test the splicing activity of this intron (Fig. 4). PCR of gDNA and cDNA and sequencing of the spliced amplicon confirmed that this intron is spliced and enabled us to identify the exon-exon boundary (Fig. 4a). In contrast to all others, this intron is located within the 5' untranslated region (UTR) of the transcript (Fig. 4b).
A newly identified type B intron in the gene TVAG_269270. a Experimental validation by RT-PCR and DNA sequencing. Left, the gel image of the RT-PCR is labelled with the gene ID on the top followed by the expected bp size of unspliced vs spliced amplicons. The lanes A-D were loaded with PCR products obtained from water, RNA, gDNA and cDNA templates, respectively. The lane L contains a molecular weight marker (100 bp DNA ladder by New England Biolabs) with band sizes indicated. Right, part of the DNA sequencing chromatogram where the line and arrow indicate the precise exon-exon boundary. The actual DNA sequence is shown under the chromatogram with the arrow indicating the nucleotide boundary between exons. b Partial sequence of gene TVAG_269270. The nucleotide sequence shows the entire 5' UTR where this 25nt-intron, highlighted in grey, was found. This is followed by part of the CDS sequence (in red) which is accompanied by its translation into one-letter amino acids. Primers surrounding the intron, which were used for the PCR here, are shown as underlined sequences. In contrast to the other characterized introns (Table 1), this type-B intron is found in the 5' UTR of the transcript
Discussion
This study confirmed the existence of 32 introns in protein-coding genes of T. vaginalis, as one intron per gene. Besides the 20 introns previously reported [15, 16], we validated another 12 introns in T. vaginalis experimentally. When revisiting introns in the T. vaginalis genome, we found that more than half of the putative intron segments reported previously [15] were not annotated in TrichDB. Similarly, we could not find experimental evidence for splicing activity for 31 putative introns in T. vaginalis multi-exon genes. More specifically, 18 of these could be classified as intronless genes (i.e. which do not appear to carry functional introns) since cDNA samples indicate expression, but only yielded full-length PCR products, consistent with the absence of splicing activity. We could not ascertain the splicing status for the remaining 13 putative introns experimentally. However, by mapping available T. vaginalis transcriptome reads to these genes in silico, we observed that all 13 genes are transcribed and that five of them show read mapping patterns consistent with functional introns (Additional file 6: Figure S2).
In examining the sequence properties of the 32 functional introns, confirmed here experimentally, these could be categorized into two types (named here A and B). Introns of type A conform to those previously reported by Vanacova et al. [15], except for the first nucleotide of the 12nt-motif that encompasses the BS and 3' SS. The change from ‘A’ to ‘G’ at this position was not considered in the original mutagenesis study [15]. Therefore, flexibility on this position may be allowed for splicing. Consistent with this, type B introns contain a ‘T’ at this position. Although the BS consensus for T. vaginalis does not seem as degenerate as for metazoans, it may be represented as a shorter consensus than initially observed [15], i.e. simply ‘CTAAC’ as seen in some Hemiacsomycete yeasts [26]. Despite re-considering this flexibility, no new introns of type A were found in protein-coding genes of T. vaginalis following our genome-wide search.
We confirmed the original observation of the positional conservation of the branch adenosine, precisely 7 nt away from the 3' SS [15], among type A introns. This feature of spliceosomal introns is shared between T. vaginalis [15, 16] and G. lamblia [27]. However, among type B introns, we identified one intron that challenged this positional conservation. Instead, the branch adenosine of the type B intron in TVAG_269270 was 8 nt away from the 3' SS. Mutagenesis studies are necessary to ascertain the exact proximity of the branch adenosine, relatively to the 3' SS of the pre-mRNA, that is necessary for splicing of type B introns in T. vaginalis.
In addition to the short length of type B introns, the low conservation of the 5' SS is another feature that separates them from type A introns according to the previous mutagenesis study [15]. Three of 10 type B introns contain the dinucleotide ‘AC’ in front of the canonical ‘GT’ at the 5' SS. This should prevent these introns from being spliced, according to the type A intron consensus and as experimentally supported [15]. However, based on our splicing activity assays, this is clearly not the case. In contrast to type A, a degenerate 5' SS seems to be allowed for type B introns. This feature was unexpected as a degenerate 5' SS is common to metazoans but not to yeast and protists [26]. Genomes of all protists that have experienced major intron losses had undergone strengthening of the 5' SS [21].
The type B intron found in TVAG_269270 after new genome search was the only one found in the UTR of a T. vaginalis transcript. All other introns were found in CDS of T. vaginalis protein-coding genes. The 5' UTRs of T. vaginalis mRNAs are known to be short in length, based on the close proximity between conserved core promoter elements and the ATG start codon [28]. These core promoter elements, which dictate transcription to initiate near the ATG start codon, are found in the large majority of T. vaginalis protein-coding genes [28]. The 5' UTR of this transcript, even after intron removal, does not seem to conform to this model. Although the distinctive features between intron types A and B described here suggest the existence of two intron families in T. vaginalis, further investigation is necessary to confirm the functional differences on sequence conservation of their splicing motifs.
In summary, we have examined evidence for splicing for a number of intron-containing genes in T. vaginalis. This study is by no means an exhaustive screen: however, it indicates that more introns might be present in this genome. As expected, standard intron-prediction models potentially fail when applied to genetically divergent eukaryotes such as T. vaginalis. Transcriptomic-based surveys, such as the one recently conducted for G. lamblia and Spironucleus salmonicida [27], will be useful to reveal the potential number of introns in the genome of T. vaginalis. These studies together should help close our gaps of understanding in the evolution of splicing, a signature of eukaryotic life.
Conclusions
Our study provided an updated list of intron-containing genes in the T. vaginalis genome. A large number of misannotated introns indicates the inaccuracy of intron prediction algorithms used by genome projects when applied to a non-model eukaryotic organism. From this updated list, we were able to identify two potential intron families carrying distinctive features. A new intron consensus allowed us to discover one additional intron-containing gene, suggesting that further studies may expand the list of introns in the genome of this evolutionarily divergent eukaryote.
Abbreviations
- BS:
-
Branch site;
- cDNA:
-
Complementary DNA
- CDS:
-
Coding sequence
- gDNA:
-
Genomic DNA
- IP:
-
Intron position
- RIP:
-
Relative intron position
- RNA-Seq:
-
RNA Sequencing
- RT:
-
Reverse transcriptase
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- SS:
-
Splice site
- UTR:
-
Untranslated region
References
Carmel L, Chorev M. The function of introns. Front Genet. 2012;3:55.
Collins L, Penny D. Complex spliceosomal organization ancestral to extant eukaryotes. Mol Biol Evol. 2005;22:1053–66.
Csuros M, Rogozin IB, Koonin EV. A detailed history of intron-rich eukaryotic ancestors inferred from a global survey of 100 complete genomes. PLoS Comput Biol. 2011;7:e1002150.
Koonin EV, Csuros M, Rogozin IB. Whence genes in pieces: reconstruction of the exon-intron gene structures of the last eukaryotic common ancestor and other ancestral eukaryotes. Wiley Interdiscip Rev RNA. 2013;4:93–105.
Simoes-Barbosa A, Meloni D, Wohlschlegel JA, Konarska MM, Johnson PJ. Spliceosomal snRNAs in the unicellular eukaryote Trichomonas vaginalis are structurally conserved but lack a 5'-cap structure. RNA. 2008;14:1617–31.
Hudson AJ, Moore AN, Elniski D, Joseph J, Yee J, Russell AG. Evolutionarily divergent spliceosomal snRNAs and a conserved non-coding RNA processing motif in Giardia lamblia. Nucleic Acids Res. 2012;40:10995–1008.
Hinas A, Larsson P, Avesson L, Kirsebom LA, Virtanen A, Soderbom F. Identification of the major spliceosomal RNAs in Dictyostelium discoideum reveals developmentally regulated U2 variants and polyadenylated snRNAs. Eukaryot Cell. 2006;5:924–34.
Schwebke JR, Hobbs MM, Taylor SN, Sena AC, Catania MG, Weinbaum BS, et al. Molecular testing for Trichomonas vaginalis in women: results from a prospective US clinical trial. J Clin Microbiol. 2011;49:4106–11.
Baldauf SL. The deep roots of eukaryotes. Science. 2003;300:1703–6.
Roger AJ, Simpson AG. Evolution: revisiting the root of the eukaryote tree. Curr Biol. 2009;19:R165–7.
Carlton JM, Hirt RP, Silva JC, Delcher AL, Schatz M, Zhao Q, et al. Draft genome sequence of the sexually transmitted pathogen Trichomonas vaginalis. Science. 2007;315:207–12.
Hirt RP, Alsmark C, Embley TM. Lateral gene transfers and the origins of the eukaryote proteome: a view from microbial parasites. Curr Opin Microbiol. 2015;23:155–62.
Huang KY, Chen YY, Fang YK, Cheng WH, Cheng CC, Chen YC, et al. Adaptive responses to glucose restriction enhance cell survival, antioxidant capability, and autophagy of the protozoan parasite Trichomonas vaginalis. Biochim Biophys Acta. 2014;1840:53–64.
Gould SB, Woehle C, Kusdian G, Landan G, Tachezy J, Zimorski V, et al. Deep sequencing of Trichomonas vaginalis during the early infection of vaginal epithelial cells and amoeboid transition. Int J Parasitol. 2013;43:707–19.
Vanacova S, Yan W, Carlton JM, Johnson PJ. Spliceosomal introns in the deep-branching eukaryote Trichomonas vaginalis. Proc Natl Acad Sci USA. 2005;102:4430–5.
Deng XL, Xu MY, Xu XY, Ba-Thein W, Zhang RL, Fu YC. A 25-bp ancient spliceosomal intron in the TvRab1a gene of Trichomonas vaginalis. Int J Biochem Cell Biol. 2009;41:417–23.
Rogozin IB, Carmel L, Csuros M, Koonin EV. Origin and evolution of spliceosomal introns. Biol Direct. 2012;7:11.
Rogozin IB, Sverdlov AV, Babenko VN, Koonin EV. Analysis of evolution of exon-intron structure of eukaryotic genes. Brief Bioinform. 2005;6:118–34.
Wu J, Xiao J, Wang L, Zhong J, Yin H, Wu S, et al. Systematic analysis of intron size and abundance parameters in diverse lineages. Sci China Life Sci. 2013;56:968–74.
Tanifuji G, Takabayashi S, Kume K, Takagi M, Nakayama T, Kamikawa R, et al. The draft genome of Kipferlia bialata reveals reductive genome evolution in fornicate parasites. PLoS One. 2018;13:ee194487.
Irimia M, Penny D, Roy SW. Coevolution of genomic intron number and splice sites. Trends Genet. 2007;23:321–5.
Nixon JE, Wang A, Morrison HG, McArthur AG, Sogin ML, Loftus BJ, et al. A spliceosomal intron in Giardia lamblia. Proc Natl Acad Sci USA. 2002;99:3701–5.
Diamond LS. The establishment of various trichomonads of animals and man in axenic cultures. J Parasitol. 1957;43:488–90.
Horner DS, Hirt RP, Kilvington S, Lloyd DG, Embley TM. Molecular data suggest an early acquisition of the mitochondrion endosymbiont. Proc R Soc Lond B. 1996;263:1053–9.
Sharp PA. Split genes and RNA splicing. Cell. 1994;77:805–15.
Irimia M, Roy SW. Evolutionary convergence on highly-conserved 3' intron structures in intron-poor eukaryotes and insights into the ancestral eukaryotic genome. PLoS Genet. 2008;4:e1000148.
Roy SW. Transcriptomic analysis of diplomonad parasites reveals a trans-spliced intron in a helicase gene in Giardia. PeerJ. 2017;5:e2861.
Smith AJ, Chudnovsky L, Simoes-Barbosa A, Delgadillo-Correa MG, Jonsson ZO, Wohlschlegel JA, et al. Novel core promoter elements and a cognate transcription factor in the divergent unicellular eukaryote Trichomonas vaginalis. Mol Cell Biol. 2011;31:1444–58.
Acknowledgements
We are grateful to the Centre for eResearch, the University of Auckland, for access to their high-performance Virtual Machine.
Funding
This work was funded by the Faculty of Science (University of Auckland) in support to AS-B as an applicant to the Marsden programme in New Zealand.
Availability of data and materials
Data supporting the conclusions of this article are included within the article and its additional files.
Author information
Authors and Affiliations
Contributions
AS-B conceived and designed the study. AS-B secured the funding, managed and coordinated the staff and laboratory where experiments were executed and supervised the students. AS-B drafted the final manuscript. All three research students (SEW, ABS and TN) conducted the experiments in this study. The PhD student SEW mentored the Hons students ABS and TN in the laboratory, did the data analysis, prepared final figures and wrote the first draft of the manuscript. AMP co-supervised SEW, ABS and TN and helped with the writing and revision of the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional files
Additional file 1:
Table S1. Primers used in this study to validate the putative introns by RT-PCR. Nucleotides that target 5' and 3' UTRs were indicated with lower-case letters. Underlined bases are located outside the transcribed region of these genes. (PDF 36 kb)
Additional file 3:
Figure S1. Experimental validation of the 62 T. vaginalis putative introns by RT-PCR. These introns were categorised as (a) functional, (b) non-functional or (c) undetermined. (PDF 256 kb)
Additional file 4:
Table S3. T. vaginalis genome segments that match the consensus sequence of type A intron (GYAYGYN{41,178}RCTAACACAYAG). (PDF 84 kb)
Additional file 5:
Table S4. T. vaginalis genome segments that match the consensus sequence of type B intron (GTWYWDN{7}TCTAACH{1,2}AACAG). (PDF 85 kb)
Additional file 6:
Figure S2. Evidence of gene transcription and mRNA splicing for the 13 putative introns that could not be experimentally determined in this study. (PDF 1422 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Wang, S.E., Amir, A.S., Nguyen, T. et al. Spliceosomal introns in Trichomonas vaginalis revisited. Parasites Vectors 11, 607 (2018). https://doi.org/10.1186/s13071-018-3196-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13071-018-3196-7