Abstract
Natural ageing of organisms and corresponding age-related diseases result mainly from stem cell ageing and “inflammaging”. Mesenchymal stem cells (MSCs) exhibit very high immune-regulating capacity and are promising candidates for immune-related disease treatment. However, the effect of MSC application is not satisfactory for some patients, especially in elderly individuals. With ageing, MSCs undergo many changes, including altered cell population reduction and differentiation ability, reduced migratory and homing capacity and, most important, defective immunosuppression. It is necessary to explore the relationship between the “inflammaging” and aged MSCs to prevent age-related diseases and increase the therapeutic effects of MSCs. In this review, we discuss changes in naturally ageing MSCs mainly from an inflammation perspective and propose some ideas for rejuvenating aged MSCs in future treatments.
Similar content being viewed by others
Background
Ageing greatly increases the risk for many chronic diseases that profoundly threaten the health of elderly individuals [1]. Most age-related diseases, such as atherosclerosis, nonalcoholic steatohepatitis (NASH), osteoarthritis, Alzheimer’s disease (AD), cardiovascular disorders and several cancers [2,3,4,5,6,7,8], result from continuous low-level inflammation called “inflammaging” [9]. Normal levels of inflammation can help organisms defend against microbial invasion and repair tissues, and the inflammatory response is attenuated when it is no longer needed [10]. In aged organisms, inflammation appears to be difficult to control, and therefore, inflammatory responses persists at low levels. Multiple mechanisms have been reported to contribute to age-related inflammation, including redox stress responses, glycation, mitochondria dysfunction, and immune system deregulation [11]. However, altered negative regulation of inflammation deserves attention.
Ageing of stem cells (SCs) is involved in age-related diseases. MSCs are endowed with a potent immune suppression capacity in the inflammatory environment due to their ability to inhibit T, B, dendritic, and natural killer cell functions and to favour macrophage polarization no acquisition of an anti-inflammatory phenotype [12]. Gene expression profiling of bone marrow-derived MSCs isolated from the femoral heads of elderly and middle-aged donors revealed differentially expressed genes in pathways related to ageing, such as oxidative stress-induced DNA damage, telomere attrition, differentiation and epigenetic regulatory pathways [13,14,15,16,17]. Here, we focus on alterations to aged MSCs that are associated with inflammaging.
Cell therapy based on MSCs [18,19,20,21,22,23] is a promising treatment for many diseases, especially immune-related diseases [24,25,26]. However, the effects of MSC therapy are not always satisfactory in some elderly patients, such as those who undergo autologous transplantation [27,28,29,30]. This observation indicates that ageing probably affects the therapeutic efficacy of MSCs. Quality control of aged MSCs and improves to the ageing microenvironment of patients are probably both important strategies for realizing the highest therapeutic efficacy. Ageing has indeed been demonstrated to affect various functions of MSCs. Thus, development of strategies to increase the therapeutic effect of aged MSCs is an urgent need. Furthermore, the undesirable role of senescent cells can be minimized, even in cases in which young MSCs are employed. In this review, we discuss changes to naturally ageing MSCs and how these alterations contribute to inflammaging. Furthermore, we provide perspective on ways to increase the therapeutic effects of aged MSCs.
Characteristics of aged MSCs
In studies of aged MSCs, senescence and ageing are often conflated, but these processes are very different. Generally, senescence is a cellular programme mainly activated by stress, including stress caused by DNA damage, oncogene activation, and replication-related telomere shortening [31] both in vivo and in vitro. Ageing is a natural condition of individuals caused by the passage of time [32]. Thus, aged MSCs are chronologically ageing cells that are retained in old organisms (in humans, older than 60 years and, in transgenic mice, 18 months old or older) (Fig. 1).
Characteristics of aged MSCs
With ageing, MSCs express many senescence markers, including an enlarged and flattened cell shape, upregulation of p53, p21, and p16 expression, increased activation of beta-galactosidase, and increased miR-335 expression in MVs. Ageing suppressed some basic functions of MSCs, including their proliferation, CFU formation, migration and homing. Aged MSCs also present a skewed trilineage differentiation pattern, with enhanced adipogenesis and reduced osteogenesis and chondrogenesis rates
Senescent phenotype and impaired contribution to the stem cell pool
To some extent, aged MSCs share some common features with senescent cells [33]. Both present with an enlarged, flat morphology instead of a spindle shape. Similar to those in senescent cells, the activation of the p53 pathway and the expression of its target genes, p21 and p16Ink4α, are commonly used as markers, in addition to the increased activation of β-galactosidase, to distinguish aged MSCs [34, 35]. During ageing, the number of MSCs in vivo decreases, and this outcome is probably due to a decline in proliferative and cell colony-formation ability [36, 37]. These are among the reasons why autologous MSC therapy in ageing patients is hampered [38], and stem cell exhaustion has been suggested to be a hallmark of ageing [39, 40]. In parallel to the inhibited-proliferation phenotype, the expression of genes involved in apoptosis and cell cycle inhibition, including p53, p21, p16Ink4α and Bax, is upregulated in bone marrow MSCs isolated from 23- to 24-month-old mice [41]. Mesenchyme homeobox 2 (MEOX2), which functions as a negative regulator of proliferation, has been found to be upregulated in aged MSCs [42]. Extracellular microvesicles have been proposed to be novel biomarkers of ageing MSCs [43]. For instance, the expression of miR-335 in extracellular microvesicles has been found to correlate with the age of donors of human MSCs, and overexpression of miR-335 caused the acquisition of a senescence phenotype and reduced the differentiation potential of MSCs [44].
Skewed differentiation
Regarding the effect of ageing on the differentiation potential of MSCs, particular attention has been given to adipogenesis, osteogenesis and chondrogenesis. Decreased osteogenic and chondrogenic differentiation potential in aged MSCs has been reported in several studies [37, 45,46,47]. In addition, during MSC ageing, adipogenesis is favoured, while osteogenesis is simultaneously disfavoured, impairing bone formation capacity [48,49,50] and subsequent osteoporosis [48, 51, 52]. These findings are consistent with the effects of increased adipose tissue accumulation in bone marrow during ageing. This shift in cell differentiation programmes is likely due to changes in gene expression. For instance, ageing triggers changes in the transcription of the nuclear receptor peroxisome proliferator-activated receptor gamma (PPARγ) and CCAAT/enhancer binding protein alpha [C/EBPα] and inhibits the expression of Runt-related transcription factor2 (RUNX2) [53, 54].
In contrast, a study by Khanh and colleagues reported that the brown/beige adipocyte differentiation capacity of adipose MSCs derived from young and aged donors was inversely correlated with age. Impaired expression of Sirt1 was shown to be critical for repressed beige adipocyte differentiation [55]. These data, which seem contradictory, suggested that the effect of ageing on adipocyte differentiation ability is probably dependent on the specific type of adipocyte involved. In addition, MSCs constitute a population of adult stem cells with obvious heterogeneity, which means that analyses of MSCs from different subgroups and different donors might lead to different results.
In addition to the altered differentiation potential governed by gene expression changes during ageing, MSCs also show the potential to transdifferentiate into endothelial-like cells in both in vivo and in vitro systems [56,57,58]. Duscher et al. discovered age-related depletion of a pro-vascular subpopulation of MSCs via single-cell transcription analysis, and these results indicated the reduced ability of aged MSCs to support vessel formation [59].
Decreased migration and homing properties
Migration and homing to injury and inflammatory sites are necessary for MSCs to facilitate tissue injury repair and immunosuppression. The release of signalling factors such as cytokines and chemokines results in MSC recruitment. Stromal derived factor-1 [SDF-1] is one of the most important MSC-recruiting chemokines secreted at an injury site, and this effect is mediated by SDF-1 binding to its receptor, C-X-C motif chemokine receptor 4 (CXCR4), which is expressed on the surface of MSCs. In addition, various other chemokine receptors, such as C-C chemokine receptor type 2 (CCR2), C-C chemokine receptor type 27 (CCR7), C-X-C motif chemokine receptor 5 (CXCR5), and C-X3-C Motif Chemokine Receptor 1 (CX3CR1) [60,61,62,63], are involved in MSC migration. Proteinases such as matrix metalloproteinases (MMPs) are also crucial for degrading components of the extracellular matrix and generating space to allow MSC migration [64,65,66,67]. Several studies have reported that aged MSCs display a lower ability to home to wound sites relative to the ability of young MSCs [36, 68]. Moreover, by measuring the migration rate in vitro, Geibler et al. showed that young MSCs showed significantly higher migratory potential than aged MSCs throughout several passages in culture [69]. Consistent with these observations, the expression of CXCR4 and C-X-C motif chemokine receptor 7 (CXCR7) on the surface of aged MSCs has been found to be significantly reduced compared to that in young counterparts [70, 71]. Tumour necrosis factor receptor (TNFR), interferon-γ receptor (IFNGR) and CCR7 level reduction was also involved in the age-dependent decrease in the migratory capacity and the activation of bone marrow MSCs [68]. Impaired migration of aged MSCs suggests an attenuated response of aged MSCs to injury signals.
Altered properties dominated by p53
p53 has attracted much attention in the context of MSC ageing because of its crucial role in cell cycle arrest and regulated expression of other age-related genes. In addition, it has been demonstrated that p53 knock-in mice showed obvious signs of organismal ageing [72,73,74].
Role of p53 in the osteogenesis of aged MSCs
As a transcription factor, p53 not only regulates the cell cycle [75,76,77,78] but is also involved in bone formation [79,80,81]. Upregulation of p53 pathway activity in the context of ageing may play a critical role in mediating the reduction in osteoblastogenesis by human MSCs, indicating that intrinsic alterations in human MSCs with ageing may contribute to skeletal ageing in humans [81]. Osteosclerosis has been detected in p53-knockout mice, and there is also evidence suggesting that p53 regulates osteoblast differentiation through the action of the transcription factors Runx2 and Osterix [82]. p53 induces the transcription of several miRNAs, including miR-29 and miR-34a, to regulate stemness and differentiation [83, 84]. Another study identified an additional miRNA, miR-145a, targeted by p53. The authors found that the p53/miR-145a/Cbfb axis inhibited the osteogenic differentiation of MSCs [80]. Overexpression of p53 inhibited osteogenesis in young MSCs in culture and in those implanted into NOD/SCID mice by inhibiting the transcription of the miR-17‐92 cluster, which is decreased in aged mice. More importantly, Smurf, a direct target gene of miR‐17, plays an important role in the p53/miR‐17 cascade during osteogenesis [85].
Preventive role of p53 in the tumor cell transformation of aged MSCs
As a cancer suppressor gene [86, 87], p53 plays a key role in the prevention of the tumoral cell transformation of MSCs. Fibrosarcomas frequently develop in aged mice. Hanchen et al. used a genetically tagged bone marrow (BM) transplantation model to show that aged mice develop MSC-derived fibrosarcomas. They also showed that transplantation of aged MSCs recapitulated the development of naturally occurring fibrosarcomas in old mice, with gene expression changes or p53 mutations similar to those identified in an in vivo model [88]. A study verified the effect of p53 on the tumoral transformation of AT-MSCs by using p53-knockout mice. The authors showed that wild-type or p21−/−p53+/+ MSCs did not show any sign of tumor cell transformation of the MSCs. However, loss of p53 favoured the fibrosarcoma formation by MSCs after either subcutaneous or intrafemoral injection in immunodeficient mice [89]. Inactivation of Rb and p53 in BM-MSC-derived osteogenic progenitors has been proven to give rise to osteosarcoma-like tumours [90].
Mechanistically, p53 mutations drive the tumoral transformation of aged MSCs, which is probably dependent on the loss of p53 binding to the survivin gene promoter, leading to abnormally upregulated survivin expression, which ultimately results in unlimited cell proliferation [91]. The mutation of p53 in aged mice is the main reason for the tumoral transformation of aged MSCs. Thus, upregulation of p53 in aged MSCs may be an underlying mechanism of inherent self-protection and homeostatic maintenance.
Involvement of Aged MSCs in inflammaging
Inflammaging has been described as the persistence of long-term and low-grade systemic chronic inflammation [92]. For two decades, inflammaging has been widely studied and has emerged as an important concept to enable dynamic reassessments of the immune responses in elderly people [93, 94]. Inflammaging is a strong risk factor for many chronic diseases in elderly individuals. Unbalanced inflammation can lead to severe organ damage and disrupt homeostasis. Multiple mechanisms have been reported to contribute to inflammaging [11], including redox stress, glycation, dysfunction of mitochondria, and deregulation of the immune system [95,96,97]. All of these mechanisms are related to accelerated inflammation. Increasing evidence indicates that aged MSCs can also contribute to inflammaging.
Activation of the innate immune system
The accumulation of damaged macromolecules and cellular debris can trigger inflammaging because both conditions activate innate immunity via damage-associated molecular patterns (DAMPs) [98, 99]. The inflammaging process involves macrophages that secrete high amounts of proinflammatory cytokines and chemokines [100]. These factors, in turn, activate inflammatory signalling pathways, including the NF-κB and STAT pathways [101, 102].
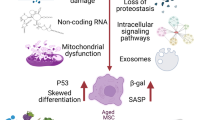
The debris of aged MSCs are relevant sources of DAMPs. During ageing, MSCs are enlarged and heterogeneously shaped and granules and cell inclusions accumulate in the cytoplasm, forming cellular debris [103]. Debris from MSCs can induce innate immune responses through the activation of innate immune cell receptors, including NOD-like receptor 4 (NLR4) and Toll-like receptor 4 (TLR4) [104,105,106,107]. Chemokines and cytokines secreted by cells in the activated innate immune system further promote inflammation cascade activation that may contribute to inflammaging (Fig. 2).
Involvement of aged MSCs in the development of inflammaging.
Ageing MSCs secrete a large amount of SASP factors, favouring the formation of an inflammatory microenvironment. Debris produced in aged MSCs can be considered a kind of DAMP and induces macrophage activation through receptors expressed on these cells; for example,, TLR4, NLR4 and RAGE. Aged MSCs promote monocyte recruitment by secreting ligands of CCR2 and induce the shift of macrophages from the M2 to the M1 phenotype. The immunosuppressive capacity of MSCs decreases with ageing and manifests by the reduced inhibitory effects of T cells mediated through IL-6, IL-8, CCL2, and miRNA carried by EVs and ROS production
Secretion of inflammatory factors by MSCs with SASP
Aged MSCs share similar phenotypical traits with senescent cells, including the senescence-associated secreted phenotype (SASP), which characterizes high levels of secreted inflammatory bioactive factors, including cytokines, chemokines and proteinases. The SASP plays an essential role in mediating MSC conversion from an anti-inflammatory cell type to a proinflammatory cell type. A significant increase in the expression levels of several SASP molecules, such as monocyte chemoattractant protein-1 (MCP-1), interleukin 6 (IL6), interleukin 8 (IL8), interleukin 1β (IL1β) and interleukin 1α (IL1α), have been revealed in aged MSCs compared to young MSCs (Fig. 2). Protein analysis of conditioned medium obtained from cultures of young and aged MSCs showed a marked increase in the secretion of MCP-1, IL6, IL8, IL1α, C-X-C motif ligand 2 (CXCL2) and C-C motif ligand 4 (CCL4) in aged MSCs relative to young MSCs [108]. Other studies have also reported that aged MSCs release excessive secretome factors, including IL6, IL8, IFN-γ, MCP-1, C-C motif ligand 12 (CCL12), C-C motif ligand 11 (CCL11), WNT1-inducible-signalling pathway protein 1 (WISP-1) and matrix metalloproteinases (e.g., MMP2) [109,110,111]. IL6 is considered the most important cytokine in inflammaging. Augmented IL6 expression in elderly people has been associated with reduced physical abilities, cognitive dysfunction, the onset of cancers, and disease progression of general degenerative disorders [111].
The SASP of aged MSCs is probably related to the persistent increase in the activation of TLR signalling [112]. Enhanced adipogenesis with ageing might contribute to acquisition of the SASP [113, 114]. Exosomes, which are secreted cellular microvesicles, also play important roles in the inflammatory secretome of MSCs [115].
Aged MSCs facilitate monocyte recruitment and a proinflammatory macrophage polarization shift
Martini and colleagues reported that cardiac MSCs acquire the ability to express chemokines, such as CCL2, CCL8, CXCL12, and CX3CL1, which are ligands of the CCR2 receptor and play major roles in monocyte recruitment. By using an inhibitor of CCR2, the authors proved that aged cMSCs promoted monocyte recruitment through the action of CCR2, thus contributing to inflammaging [116].
MSCs are able to promote the polarization of macrophages from the M1 to the M2 phenotype to exert anti-inflammatory effects [117]. However, SASP factors have been demonstrated to polarize macrophages by shifting the M2 phenotype towards the M1 phenotype [118] (Fig. 2). Thus, aged MSCs affect macrophage polarization in opposite ways [119]. Interestingly, although macrophages cocultured with young MSCs expressed M2 phenotype markers, such as arginase 1 (Arg1) and interleukin 10 (IL10), those cocultured with aged MSCs showed increased expression of M1 phenotype-related tumour necrosis factor-α (TNF-α). In addition, macrophages cocultured with aged MSCs exhibited increased migratory ability, which is a property typical of classically activated M1 macrophages [120]. As described above, aged MSCs can produce essential inducers of M1 macrophage differentiation, including IFN-γ and IL1, and activate the NF-κB signalling pathway [121].
Impaired inhibition effect of T cells
The immunosuppressive effects of MSCs are mainly directed towards T cells. However, aged MSCs display a diminished capacity to suppress T cell proliferation and activation [122, 123]. Allogeneic coculture systems show that young MSCs were able to effectively inhibit phytohemagglutinin (PHA)-induced PBMC proliferation, while aged MSCs exhibited a significantly reduced ability to impair PHA-PBMC proliferation [109]. Another study reported that the suppressive ability of aged MSCs on both CD4+ and CD8+ T-cell proliferation was impaired compared with that of young MSCs. The diminished ability to suppress T cells is probably due to SASP factor secretion, since neutralization of IL6, IL8 and CCL2 enhances the immunomodulatory function of elderly MSCs [123]. Priming MSCs with proinflammatory factors stimulates the production of extracellular vesicles (EVs), which exhibit potent anti-inflammatory effects and enhanced therapeutic potential [124, 125]. Alteration of EVs and related miRNAs may explain the impaired immunomodulation properties of aged MSCs. EVs secreted by MSCs obtained from an aged donor showed reduced immunosuppression activity relative to MSC-EVs obtained from a younger donor [126]. These differences were ascribed to ectopic levels of MSC-EV miRNAs, including miR-223-5p, miR-125b-5p and miR-127-3p. These data support previous findings highlighting that age-related alterations in miRNA amounts in MSCs-EVs were associated with altered immunomodulatory properties of aged MSCs [127,128,129]. AT-MSCs obtained from elderly people were characterized by increased oxidative stress compared to MSCs obtained from younger people, and ROS decreased the ability of MSCs to suppress T cells [130].
A series of immunosuppressive molecules, such as indoleamine-2,3-dioxygenase (IDO) (in humans), inducible nitric oxide synthase (iNOS) (in rodents), prostaglandin E2 (PGE2), transforming growth factor β (TGF-β), tumour necrosis factor-inducible gene 6 protein (TSG6), and IL10, have been demonstrated to exert immunosuppressive effects on MSCs [131]. In particular, IDO has been found to be downregulated in replication-associated senescent MSCs [132, 133]. However, there is an urgent need to expand this research to identify the mechanisms underlying the decreased immunosuppression of naturally aged MSCs.
Potential strategies to rejuvenate aged MSCs
MSCs have been widely used for treating immune-related diseases, and their immunosuppressive capacity has been evaluated in clinical trials. More than 12% of clinical trials of MSC-based treatments have been performed for immune-related diseases, such as graft-versus-host disease (GvHD), Crohn’s disease, psoriasis, urticaria, and arthritis (https://www.clinicaltrials.gov/). However, the therapeutic effects have been unsatisfactory, probably due to the quality of the MSCs. Ageing is an important factor affecting the therapeutic effects of MSCs, especially for use in autologous transplantation in elderly individuals. Thus, determining how to improve the therapeutic efficacy of aged MSCs is an urgent need for promoting the clinical applications of MSCs (Fig. 3).
Strategies to rejuvenate aged MSCs
Aged MSCs suitable for treatment can be obtained by sorting surface markers. Senescent MSCs can be deleted with senolytics. Pretreatment can be performed with several reagents able to target the AMPK signalling pathway, ROS and autophagy factors to rejuvenate aged MSCs. Gene modification for inducing the upregulation or knocking down some target genes favours aged MSC rejuvenation
Identifying superior cells
Based on many studies, the heterogeneity of MSCs increases with ageing. Lise Lefèvre et al. identified a CD73+ kidney mesenchymal stromal cell (kMSC) population that was increased in the kidneys of ageing organisms. Aged CD73+ kMSCs displayed senescence-associated hallmarks, including a low proliferation rate and DNA damage accumulation. In addition, these cells produced niche factors required to recruit monocytes, ultimately promoting a positive regulatory loop in response to local inflammation [134]. Another study showed that CD271+CD146+ MSCs were predominant in children, while CD271+CD146− MSCs were most common in adults [135], with the number of CD271+ cells declining with increasing donor age [136]. CD71+, CD146+ and CD274+ BM-MSCs are reported to be negatively correlated with donor age [137]. Based on the heterogeneity of aged MSCs, depletion of a subgroup of aged MSCs or enrichment of a subgroup with young features maintained in vitro before transplantation may be alternative cell to increase the therapeutic effect of aged MSCs.
Elimination of senescent cells
Eliminating senescent cells delays the onset of several pathologies and allegedly promotes a healthy lifestyle and increased lifespan [138, 139]. Many studies have identified agents that specifically eliminate senescent cells through the production of senolytic compounds [140]. Most of these products function mainly through apoptosis induction and immune system activation to eliminate senescent cells. Senolytics can be categorized into several classes according to their different targets; they include the B-cell lymphoma 2 (BCL-2) family, HSP90 and the p53 pathway compounds [141]. Some senolytic effects are produced via the combination of two drugs, such as dasatinib plus quercetin (D + Q), which have been proven to remove naturally occurring senescent cells from human adipose tissue [142]. D + Q has also been evaluated for use in treating age-associated diseases in clinical trials (https://www.clinicaltrials.gov/), and D + Q allegedly showed significant beneficial effects [143], indicating it exhibits some potential as a combination senolytic agent.
Pretreatment
Several studies have indicated that aged MSCs can be rejuvenated. MSCs derived from aged donors showed a lower level of fibroblast growth factor 21 (FGF21), and overexpression of FGF21 in aged MSCs inhibited senescence via the AMP-activated protein kinase (AMPK) signalling pathway. Thus, targeting FGF21 might represent a novel strategy to increase the quality of aged MSCs [144]. The expression of miR-155-5p was much higher in MSCs obtained from aged donors than in MSCs obtained from young donors, and miR-155-5p downregulation decreases senescence of aged MSCs by activating AMPK signalling. Hence, an AMPK activator, AICAR, may be used to renew aged MSCs [145]. Other AMPK activators, such as melatonin [146], C1q/tumour necrosis factor-related protein 9 (CTRP9) [147], and macrophage migration inhibitory factor (MIF) [148], have also been reported to exert an antiaging effect on MSCs.
ROS production is the major contributor to MSC ageing and age-related diseases. Compounds with antioxidant activity, such as lactoferrin [149], acetovanillone, N-acetyl cysteine (NAC), NAC and l-ascorbic acid 2-phosphate, vitamin E, metformin, fullerol, fucoidan, carvedilol, nicorandil and 5-azacytidine, might be able to rejuvenate aged MSCs [150]. β-Carotene is another candidate for rejuvenation of aged MSCs. It has been reported that β-carotene can relieve ageing in MSCs, as evidenced by the reduced expression of p16 and p21. β-Carotene has also been shown to reduce ageing rates in tissues and organs in vivo and appeared to inhibit ageing caused by antioxidative stress by regulating KAT7-P15 signalling [151].
Hypoxia has been demonstrated to suppress the senescence of aged MSCs in several reports. Hypoxic preconditioning enhanced the in vivo angiogenic capacities of human AT-MSCs obtained from older donors. Hypoxia also repressed the expression of ageing-associated gene p16Ink4α and ageing inducer aminoacyl-tRNA synthetase‐interacting multifunctional protein 3 (AIMP3) [152]. Hypoxia probably reverses the ageing of MSCs partially by activating autophagy [153]. Rapamycin, an autophagy activator that inhibits the mammalian target of rapamycin (mTOR) pathway, has also been reported to exert an antiaging effect on MSCs and to enhance the immunomodulatory potency of MSCs [154]. Interestingly, another study reported that young MSC-derived apoptotic vesicles were able to restore nuclear alterations, as well as the self-renewal and osteogenic and adipogenic lineage differentiation capacity of aged bone marrow MSCs via autophagy activation [155].
Gene modifications
In addition to pretreatment, gene modification is a widely used method to modify MSCs. There have only been a few studies using gene modification as a strategy to rejuvenate MSCs and recover their function. In a study of MSC reprogramming, which was similar to an approach used for cell rejuvenation, the expression of GATA-binding protein 6 (GATA6) was found to be attenuated, and that of Forkhead box P1 (FOXP1) was upregulated. Thus, knocking down GATA6 and inducing FOXP1 overexpression showed the potential to ameliorate the expression of cellular hallmarks of ageing [156]. Liu et al. found that leucine-rich repeat-containing 17 (LRRc17) expression in BM-MSCs was highly positively correlated with age. LRRc17 knockdown rejuvenated aged MSCs and increased their therapeutic efficacy in the context of osteoporosis [157]. In another study on MSC treatment in OP, it was reported that reactivating optineurin or inhibiting FABP3 activity rescued osteoporotic phenotypes [49]. AIMP3-induced senescence was negatively regulated by hypoxia‐inducible factor 1α (HIF1α) and positively regulated by Notch3, which means that HIF1α overexpression and Notch3 knockdown inhibited the senescence of MSCs [152].
Conclusions
MSCs undergo numerous changes with natural ageing, including acquisition of a senescence phenotype and reductions in differentiation, migratory and homing ability, and they contribute to inflammaging in the ageing body. All these alterations are probably due to changes at the molecular level, including gene expression changes and epigenetic modifications [33]. These changes support the view that stem cell ageing results in body ageing and age-related diseases. Therefore, aged MSCs can be considered targets for therapy developed to prevent or attenuated inflammaging. A considerable number of senolytics have been identified for targeting and eliminating senescent cells in vivo.
The changes in MSC property with ageing not only favour inflammaging but also affect their therapeutic potential. Indeed, impaired osteogenic differentiation, migration or immune regulation reduces the therapeutic effect of MSCs in tissue injury repair and inflammation-related disease treatment. The undesirable effects of senescence in aged MSCs can be minimized in several ways, as previously discussed. The same approaches might also be employed to increase therapeutic effects, even on young MSCs. Notably, a study on human gingival tissue MSCs (GMSCs) reported that irrespective of donor age, GMSCs displayed effective neurogenesis, immunoregulation and regenerative potential [158,159,160,161]. These findings suggest a possible option for treating elderly patients who need autologous transplantation of MSCs. However, MSC therapy was less effective in an elderly cohort than in a young cohort, indicating that old plasma might carry factors that inhibit the function of MSCs [162]. Murphy and coauthors proposed that growth differentiation factor 11 (GDF11), mTOR, CCL11, and the insulin/insulin-like growth 1 (IGF1) signalling pathways may be relevant to stem cell function [163, 164]. Therefore, in addition to MSC quality, the microenvironment of patients also needs to be considered or improved for successful MSC therapy.
Based on the mechanisms underlying MSC function that are affected by ageing, the development of develop novel strategies to ameliorate inflammaging and to increase the therapeutic efficacy of aged MSCs is promising.
References
Vitale I, Pietrocola F, Guilbaud E, Aaronson SA, Abrams JM, Adam D et al. Apoptotic cell death in disease-current understanding of the NCCD 2023. Cell Death Differ. 2023;30[5]:1097 – 154.
Pickup JC. Inflammation and activated innate immunity in the pathogenesis of type 2 diabetes. Diabetes Care. 2004;27[3]:813 – 23.
Stuart MJ, Baune BT. Depression and type 2 diabetes: inflammatory mechanisms of a psychoneuroendocrine co-morbidity. Neurosci Biobehav Rev. 2012;36[1]:658 – 76.
Libby P, Ridker PM, Hansson GK. Leducq Transatlantic Network on A. inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol. 2009;5423:2129–38.
Wallin R, Wajih N, Greenwood GT, Sane DC. Arterial calcification: a review of mechanisms, animal models, and the prospects for therapy. Med Res Rev. 2001;214:274–301.
Hirose K, Tomiyama H, Okazaki R, Arai T, Koji Y, Zaydun G et al. Increased pulse wave velocity associated with reduced calcaneal quantitative osteo-sono index: possible relationship between atherosclerosis and osteopenia. J Clin Endocrinol Metab. 2003;88[6]:2573–8.
Manolagas SC. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr Rev. 2000;21[2]:115 – 37.
Koyama A, O’Brien J, Weuve J, Blacker D, Metti AL, Yaffe K. The role of peripheral inflammatory markers in dementia and Alzheimer’s disease: a meta-analysis. J Gerontol A Biol Sci Med Sci. 2013;68[4]:433 – 40.
Franceschi C, Capri M, Monti D, Giunta S, Olivieri F, Sevini F, et al. Inflammaging and anti-inflammaging: a systemic perspective on aging and longevity emerged from studies in humans. Mech Ageing Dev. 2007;1281:92–105.
Bektas A, Schurman SH, Sen R, Ferrucci L. Aging, inflammation and the environment. Exp Gerontol. 2018;105:10–8.
Fougere B, Boulanger E, Nourhashemi F, Guyonnet S, Cesari M. Chronic inflammation: Accelerator of Biological Aging. J Gerontol A Biol Sci Med Sci. 2017;729:1218–25.
Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8[9]:726 – 36.
Wu Y, Yang J, Ai Z, Yu M, Li J, Li S. Identification of key genes and transcription factors in aging mesenchymal stem cells by DNA microarray data. Gene. 2019;692:79–87.
Xu L, Zhu Y, Li C, Wang Q, Ma L, Wang J et al. Small extracellular vesicles derived from Nrf2-overexpressing human amniotic mesenchymal stem cells protect against lipopolysaccharide-induced acute lung injury by inhibiting NLRP3. Biol Direct. 2022;17[1]:35.
Chen Z, Tang W, Zhou Y, He Z. The role of LINC01419 in regulating the cell stemness in lung adenocarcinoma through recruiting EZH2 and regulating FBP1 expression. Biol Direct. 2022;17[1]:23.
Fazi B, Melino S, De Rubeis S, Bagni C, Paci M, Piacentini M, et al. Acetylation of RTN-1 C regulates the induction of ER stress by the inhibition of HDAC activity in neuroectodermal tumors. Oncogene. 2009;2843:3814–24.
Melino S, Nepravishta R, Bellomaria A, Di Marco S, Paci M. Nucleic acid binding of the RTN1-C C-terminal region: toward the functional role of a reticulon protein. Biochemistry. 2009;48[2]:242 – 53.
Priami C, Montariello D, De Michele G, Ruscitto F, Polazzi A, Ronzoni S et al. Aberrant activation of p53/p66Shc-mInsc axis increases asymmetric divisions and attenuates proliferation of aged mammary stem cells. Cell Death Differ. 2022;29[12]:2429–44.
Fang J, Feng C, Chen W, Hou P, Liu Z, Zuo M et al. Redressing the interactions between stem cells and immune system in tissue regeneration. Biol Direct. 2021;16[1]:18.
Hao RH, Guo Y, Wang C, Chen F, Di CX, Dong SS, et al. Lineage-specific rearrangement of chromatin loops and epigenomic features during adipocytes and osteoblasts commitment. Cell Death Differ. 2022;2912:2503–18.
Jeong J, Jung I, Kim JH, Jeon S, Hyeon DY, Min H et al. BAP1 shapes the bone marrow niche for lymphopoiesis by fine-tuning epigenetic profiles in endosteal mesenchymal stromal cells. Cell Death Differ. 2022;29[11]:2151–62.
Huang Y, Wan S, Yang M. Circ_0067680 expedites the osteogenic differentiation of human bone marrow-derived mesenchymal stem cells through miR-4429/CTNNB1/Wnt/beta-catenin pathway. Biol Direct. 2021;16[1]:16.
Pieknell K, Sulistio YA, Wulansari N, Darsono WHW, Chang MY, Ko JY et al. LIN28A enhances regenerative capacity of human somatic tissue stem cells via metabolic and mitochondrial reprogramming. Cell Death Differ. 2022;29[3]:540 – 55.
Ciocci M, Mochi F, Carotenuto F, Di Giovanni E, Prosposito P, Francini R, et al. Scaffold-in-Scaffold potential to induce growth and differentiation of Cardiac Progenitor cells. Stem Cells Dev. 2017;26:1438–47. [19].
Ganini C, Amelio I, Bertolo R, Bove P, Buonomo OC, Candi E et al. Global mapping of cancers: the Cancer Genome Atlas and beyond. Mol Oncol. 2021;15[11]:2823–40.
Hu M, Lu Y, Wang S, Zhang Z, Qi Y, Chen N et al. CD63 acts as a functional marker in maintaining hematopoietic stem cell quiescence through supporting TGFbeta signaling in mice. Cell Death Differ. 2022;29[1]:178 – 91.
Wu Y, Chen K, Li L, Hao Z, Wang T, Liu Y, et al. Plin2-mediated lipid droplet mobilization accelerates exit from pluripotency by lipidomic remodeling and histone acetylation. Cell Death Differ. 2022;2911:2316–31.
Xing G, Liu Z, Huang L, Zhao D, Wang T, Yuan H, et al. MAP2K6 remodels chromatin and facilitates reprogramming by activating Gatad2b-phosphorylation dependent heterochromatin loosening. Cell Death Differ. 2022;295:1042–54.
Augusto-Oliveira M, Verkhratsky A. Lifestyle-dependent microglial plasticity: training the brain guardians. Biol Direct. 2021;16[1]:12.
Manic G, Musella M, Corradi F, Sistigu A, Vitale S, Soliman Abdel Rehim S et al. Control of replication stress and mitosis in colorectal cancer stem cells through the interplay of PARP1, MRE11 and RAD51. Cell Death Differ. 2021;28[7]:2060-82.
Gorgoulis V, Adams PD, Alimonti A, Bennett DC, Bischof O, Bishop C et al. Cellular Senescence: defining a path Forward. Cell. 2019;179[4]:813 – 27.
Chen H, Liu O, Chen S, Zhou Y. Aging and mesenchymal stem cells: Therapeutic Opportunities and Challenges in the Older Group. Gerontology. 2022;68[3]:339 – 52.
Zupan J, Strazar K, Kocijan R, Nau T, Grillari J, Marolt Presen D. Age-related alterations and senescence of mesenchymal stromal cells: implications for regenerative treatments of bones and joints. Mech Ageing Dev. 2021;198:111539.
Hall BM, Balan V, Gleiberman AS, Strom E, Krasnov P, Virtuoso LP, et al. p16[Ink4a] and senescence-associated beta-galactosidase can be induced in macrophages as part of a reversible response to physiological stimuli. Aging. 2017;98:1867–84.
Rufini A, Tucci P, Celardo I, Melino G. Senescence and aging: the critical roles of p53. Oncogene. 2013;3243:5129–43.
Lin H, Sohn J, Shen H, Langhans MT, Tuan RS. Bone marrow mesenchymal stem cells: aging and tissue engineering applications to enhance bone healing. Biomaterials. 2019;203:96–110.
Stolzing A, Jones E, McGonagle D, Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mech Ageing Dev. 2008;129[3]:163 – 73.
Baker N, Boyette LB, Tuan RS. Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone. 2015;70:37–47.
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153[6]:1194 – 217.
Partridge L, Deelen J, Slagboom PE. Facing up to the global challenges of ageing. Nature. 2018;5617721:45–56.
Choudhery MS, Khan M, Mahmood R, Mehmood A, Khan SN, Riazuddin S. Bone marrow derived mesenchymal stem cells from aged mice have reduced wound healing, angiogenesis, proliferation and anti-apoptosis capabilities. Cell Biol Int. 2012;36[8]:747 – 53.
Wagner W, Bork S, Horn P, Krunic D, Walenda T, Diehlmann A et al. Aging and replicative senescence have related effects on human stem and progenitor cells. PLoS ONE. 2009;4[6]:e5846.
Terlecki-Zaniewicz L, Lammermann I, Latreille J, Bobbili MR, Pils V, Schosserer M, et al. Small extracellular vesicles and their miRNA cargo are anti-apoptotic members of the senescence-associated secretory phenotype. Aging. 2018;105:1103–32.
Tome M, Sepulveda JC, Delgado M, Andrades JA, Campisi J, Gonzalez MA, et al. miR-335 correlates with senescence/aging in human mesenchymal stem cells and inhibits their therapeutic actions through inhibition of AP-1 activity. Stem Cells. 2014;328:2229–44.
Guang LG, Boskey AL, Zhu W. Age-related CXC chemokine receptor-4-deficiency impairs osteogenic differentiation potency of mouse bone marrow mesenchymal stromal stem cells. Int J Biochem Cell Biol. 2013;458:1813–20.
Kanawa M, Igarashi A, Ronald VS, Higashi Y, Kurihara H, Sugiyama M, et al. Age-dependent decrease in the chondrogenic potential of human bone marrow mesenchymal stromal cells expanded with fibroblast growth factor-2. Cytotherapy. 2013;159:1062–72.
Umbayev B, Masoud AR, Tsoy A, Alimbetov D, Olzhayev F, Shramko A et al. Elevated levels of the small GTPase Cdc42 induces senescence in male rat mesenchymal stem cells. Biogerontology. 2018;19[3–4]:287–301.
Maredziak M, Marycz K, Tomaszewski KA, Kornicka K, Henry BM. The influence of aging on the regenerative potential of human adipose derived mesenchymal stem cells. Stem cells international. 2016;2016:2152435.
Liu ZZ, Hong CG, Hu WB, Chen ML, Duan R, Li HM, et al. Autophagy receptor OPTN [optineurin] regulates mesenchymal stem cell fate and bone-fat balance during aging by clearing FABP3. Autophagy. 2021;1710:2766–82.
Li H, Liu P, Xu S, Li Y, Dekker JD, Li B et al. FOXP1 controls mesenchymal stem cell commitment and senescence during skeletal aging. J Clin Investig. 2017;127[4]:1241–53.
Bonyadi M, Waldman SD, Liu D, Aubin JE, Grynpas MD, Stanford WL. Mesenchymal progenitor self-renewal deficiency leads to age-dependent osteoporosis in Sca-1/Ly-6A null mice. Proc Natl Acad Sci USA. 2003;100[10]:5840–5.
Infante A, Rodriguez CI. Osteogenesis and aging: lessons from mesenchymal stem cells. Stem Cell Res Ther. 2018;9[1]:244.
Moerman EJ, Teng K, Lipschitz DA, Lecka-Czernik B. Aging activates adipogenic and suppresses osteogenic programs in mesenchymal marrow stroma/stem cells: the role of PPAR-gamma2 transcription factor and TGF-beta/BMP signaling pathways. Aging Cell. 2004;3[6]:379 – 89.
Kim J, Ko J. A novel PPARgamma2 modulator sLZIP controls the balance between adipogenesis and osteogenesis during mesenchymal stem cell differentiation. Cell Death Differ. 2014;2110:1642–55.
Khanh VC, Zulkifli AF, Tokunaga C, Yamashita T, Hiramatsu Y, Ohneda O. Aging impairs beige adipocyte differentiation of mesenchymal stem cells via the reduced expression of Sirtuin 1. Biochem Biophys Res Commun. 2018;500[3]:682 – 90.
Oswald J, Boxberger S, Jorgensen B, Feldmann S, Ehninger G, Bornhauser M et al. Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells. 2004;22[3]:377 – 84.
Vittorio O, Jacchetti E, Pacini S, Cecchini M. Endothelial differentiation of mesenchymal stromal cells: when traditional biology meets mechanotransduction. Integrative biology: quantitative biosciences from nano to macro. 2013;5[2]:291–9.
Nassiri SM, Khaki Z, Soleimani M, Ahmadi SH, Jahanzad I, Rabbani S et al. The similar effect of transplantation of marrow-derived mesenchymal stem cells with or without prior differentiation induction in experimental myocardial infarction. J Biomed Sci. 2007;14[6]:745 – 55.
Duscher D, Rennert RC, Januszyk M, Anghel E, Maan ZN, Whittam AJ, et al. Aging disrupts cell subpopulation dynamics and diminishes the function of mesenchymal stem cells. Sci Rep. 2014;4:7144.
Hemeda H, Jakob M, Ludwig AK, Giebel B, Lang S, Brandau S. Interferon-gamma and tumor necrosis factor-alpha differentially affect cytokine expression and migration properties of mesenchymal stem cells. Stem Cells Dev. 2010;195:693–706.
Baek SJ, Kang SK, Ra JC. In vitro migration capacity of human adipose tissue-derived mesenchymal stem cells reflects their expression of receptors for chemokines and growth factors. Exp Mol Med. 2011;4310:596–603.
Song CH, Honmou O, Furuoka H, Horiuchi M. Identification of chemoattractive factors involved in the migration of bone marrow-derived mesenchymal stem cells to brain lesions caused by prions. J Virol. 2011;8521:11069–78.
Hung SC, Pochampally RR, Hsu SC, Sanchez C, Chen SC, Spees J et al. Short-term exposure of multipotent stromal cells to low oxygen increases their expression of CX3CR1 and CXCR4 and their engraftment in vivo. PLoS ONE. 2007;2[5]:e416.
Park MJ, Shin JS, Kim YH, Hong SH, Yang SH, Shin JY et al. Murine mesenchymal stem cells suppress T lymphocyte activation through IL-2 receptor alpha [CD25] cleavage by producing matrix metalloproteinases. Stem cell reviews and reports. 2011;7[2]:381 – 93.
Mannello F, Tonti GA, Bagnara GP, Papa S. Role and function of matrix metalloproteinases in the differentiation and biological characterization of mesenchymal stem cells. Stem Cells. 2006;24[3]:475 – 81.
De Becker A, Riet IV. Homing and migration of mesenchymal stromal cells: how to improve the efficacy of cell therapy? World J stem cells. 2016;83:73–87.
Kolf CM, Cho E, Tuan RS. Mesenchymal stromal cells. Biology of adult mesenchymal stem cells: regulation of niche, self-renewal and differentiation. Arthritis Res therapy. 2007;9[1]:204.
Bustos ML, Huleihel L, Kapetanaki MG, Lino-Cardenas CL, Mroz L, Ellis BM et al. Aging mesenchymal stem cells fail to protect because of impaired migration and antiinflammatory response. Am J Respir Crit Care Med. 2014;189[7]:787 – 98.
Geissler S, Textor M, Kuhnisch J, Konnig D, Klein O, Ode A et al. Functional comparison of chronological and in vitro aging: differential role of the cytoskeleton and mitochondria in mesenchymal stromal cells. PLoS ONE. 2012;7[12]:e52700.
Liu M, Lei H, Dong P, Fu X, Yang Z, Yang Y, et al. Adipose-derived mesenchymal stem cells from the Elderly exhibit decreased Migration and differentiation abilities with senescent Properties. Cell Transplant. 2017;269:1505–19.
Shao H, Xu Q, Wu Q, Ma Q, Salgueiro L, Wang J et al. Defective CXCR4 expression in aged bone marrow cells impairs vascular regeneration. Journal of cellular and molecular medicine. 2011;15[10]:2046-56.
Zhao Y, Wu L, Yue X, Zhang C, Wang J, Li J et al. A polymorphism in the tumor suppressor p53 affects aging and longevity in mouse models. eLife. 2018;7.
Panatta E, Butera A, Celardo I, Leist M, Melino G, Amelio I. p53 regulates expression of nuclear envelope components in cancer cells. Biol Direct. 2022;17[1]:38.
Rozenberg JM, Zvereva S, Dalina A, Blatov I, Zubarev I, Luppov D et al. The p53 family member p73 in the regulation of cell stress response. Biol Direct. 2021;16[1]:23.
Yuan J, Zhu Q, Zhang X, Wen Z, Zhang G, Li N et al. Ezh2 competes with p53 to license lncRNA Neat1 transcription for inflammasome activation. Cell Death Differ. 2022;29[10]:2009-23.
Panatta E, Zampieri C, Melino G, Amelio I. Understanding p53 tumour suppressor network. Biol Direct. 2021;16[1]:14.
Mammarella E, Zampieri C, Panatta E, Melino G, Amelio I. NUAK2 and RCan2 participate in the p53 mutant pro-tumorigenic network. Biol Direct. 2021;16[1]:11.
Lindstrom MS, Bartek J, Maya-Mendoza A. p53 at the crossroad of DNA replication and ribosome biogenesis stress pathways. Cell Death Differ. 2022;29[5]:972 – 82.
Velletri T, Xie N, Wang Y, Huang Y, Yang Q, Chen X et al. P53 functional abnormality in mesenchymal stem cells promotes osteosarcoma development. Cell Death Dis. 2016;7[1]:e2015.
Xia C, Jiang T, Wang Y, Chen X, Hu Y, Gao Y. The p53/miR-145a Axis promotes Cellular Senescence and inhibits osteogenic differentiation by Targeting Cbfb in Mesenchymal Stem cells. Front Endocrinol. 2020;11:609186.
Zhou S, Greenberger JS, Epperly MW, Goff JP, Adler C, Leboff MS et al. Age-related intrinsic changes in human bone-marrow-derived mesenchymal stem cells and their differentiation to osteoblasts. Aging Cell. 2008;7[3]:335 – 43.
Artigas N, Gamez B, Cubillos-Rojas M, Sanchez-de Diego C, Valer JA, Pons G et al. p53 inhibits SP7/Osterix activity in the transcriptional program of osteoblast differentiation. Cell Death Differ. 2017;24[12]:2022-31.
Lin CP, Choi YJ, Hicks GG, He L. The emerging functions of the p53-miRNA network in stem cell biology. Cell Cycle. 2012;11[11]:2063–72.
Ugalde AP, Ramsay AJ, de la Rosa J, Varela I, Marino G, Cadinanos J, et al. Aging and chronic DNA damage response activate a regulatory pathway involving miR-29 and p53. EMBO J. 2011;3011:2219–32.
Liu W, Qi M, Konermann A, Zhang L, Jin F, Jin Y. The p53/miR-17/Smurf1 pathway mediates skeletal deformities in an age-related model via inhibiting the function of mesenchymal stem cells. Aging. 2015;7[3]:205 – 18.
Thomas AF, Kelly GL, Strasser A. Of the many cellular responses activated by TP53, which ones are critical for tumour suppression? Cell Death Differ. 2022;29[5]:961 – 71.
Levine AJ. Exploring the future of research in the Tp53 field. Cell Death Differ. 2022;295:893–4.
Li H, Fan X, Kovi RC, Jo Y, Moquin B, Konz R, et al. Spontaneous expression of embryonic factors and p53 point mutations in aged mesenchymal stem cells: a model of age-related tumorigenesis in mice. Cancer Res. 2007;6722:10889–98.
Rodriguez R, Rubio R, Masip M, Catalina P, Nieto A, de la Cueva T, et al. Loss of p53 induces tumorigenesis in p21-deficient mesenchymal stem cells. Neoplasia. 2009;114:397–407.
Rubio R, Gutierrez-Aranda I, Saez-Castillo AI, Labarga A, Rosu-Myles M, Gonzalez-Garcia S, et al. The differentiation stage of p53-Rb-deficient bone marrow mesenchymal stem cells imposes the phenotype of in vivo sarcoma development. Oncogene. 2013;3241:4970–80.
He L, Zhao F, Zheng Y, Wan Y, Song J. Loss of interactions between p53 and survivin gene in mesenchymal stem cells after spontaneous transformation in vitro. Int J Biochem Cell Biol. 2016;75:74–84.
Franceschi C, Bonafe M, Valensin S, Olivieri F, De Luca M, Ottaviani E, et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci. 2000;908:244–54.
Fransen F, van Beek AA, Borghuis T, Aidy SE, Hugenholtz F, van der Gaast C, et al. Aged gut microbiota contributes to Systemical Inflammaging after transfer to germ-free mice. Front Immunol. 2017;8:1385.
Calcada D, Vianello D, Giampieri E, Sala C, Castellani G, de Graaf A et al. The role of low-grade inflammation and metabolic flexibility in aging and nutritional modulation thereof: a systems biology approach. Mech Ageing Dev. 2014;136–137:138 – 47.
Angelucci S, Sacchetta P, Moio P, Melino S, Petruzzelli R, Gervasi P et al. Purification and characterization of glutathione transferases from the sea bass [Dicentrarchus labrax] liver. Arch Biochem Biophys. 2000;373[2]:435 – 41.
Gallo M, Paludi D, Cicero DO, Chiovitti K, Millo E, Salis A, et al. Identification of a conserved N-capping box important for the structural autonomy of the prion alpha 3-helix: the disease associated D202N mutation destabilizes the helical conformation. Int J ImmunoPathol Pharmacol. 2005;181:95–112.
Favaloro B, Tamburro A, Angelucci S, Luca AD, Melino S, di Ilio C, et al. Molecular cloning, expression and site-directed mutagenesis of glutathione S-transferase from Ochrobactrum anthropi. Biochem J. 1998;335:573–9. [ Pt 3][Pt 3].
Franceschi C, Campisi J. Chronic inflammation [inflammaging] and its potential contribution to age-associated diseases. J Gerontol A Biol Sci Med Sci. 2014;69(Suppl 1):4–9.
Lee BC, Yu KR. Impact of mesenchymal stem cell senescence on inflammaging. BMB Rep. 2020;532:65–73.
Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat reviews Endocrinol. 2018;14[10]:576 – 90.
Salminen A, Huuskonen J, Ojala J, Kauppinen A, Kaarniranta K, Suuronen T. Activation of innate immunity system during aging: NF-kB signaling is the molecular culprit of inflamm-aging. Ageing Res Rev. 2008;72:83–105.
Chazaud B, Mouchiroud G. Inflamm-aging: STAT3 signaling pushes muscle stem cells off balance. Cell Stem Cell. 2014;154:401–2.
Bonab MM, Alimoghaddam K, Talebian F, Ghaffari SH, Ghavamzadeh A, Nikbin B. Aging of mesenchymal stem cell in vitro. BMC Cell Biol. 2006;7:14.
de Witte SFH, Luk F, Sierra Parraga JM, Gargesha M, Merino A, Korevaar SS et al. Immunomodulation by therapeutic mesenchymal stromal cells [MSC] is triggered through phagocytosis of MSC by Monocytic cells. Stem Cells. 2018;36[4]:602 – 15.
Ramasamy R, Vannucci SJ, Yan SS, Herold K, Yan SF, Schmidt AM. Advanced glycation end products and RAGE: a common thread in aging, diabetes, neurodegeneration, and inflammation. Glycobiology. 2005;15[7]:16R-28R.
Pisetsky DS, Erlandsson-Harris H, Andersson U. High-mobility group box protein 1 [HMGB1]: an alarmin mediating the pathogenesis of rheumatic disease. Arthritis Res therapy. 2008;10[3]:209.
Salminen A, Ojala J, Kaarniranta K, Kauppinen A. Mitochondrial dysfunction and oxidative stress activate inflammasomes: impact on the aging process and age-related diseases. Cell Mol Life Sci. 2012;6918:2999–3013.
Gnani D, Crippa S, Della Volpe L, Rossella V, Conti A, Lettera E et al. An early-senescence state in aged mesenchymal stromal cells contributes to hematopoietic stem and progenitor cell clonogenic impairment through the activation of a pro-inflammatory program. Aging Cell. 2019;18[3]:e12933.
Zhang Y, Ravikumar M, Ling L, Nurcombe V, Cool SM. Age-related changes in the inflammatory status of human mesenchymal stem cells: implications for cell therapy. Stem cell reports. 2021;164:694–707.
Chanda D, Rehan M, Smith SR, Dsouza KG, Wang Y, Bernard K et al. Mesenchymal stromal cell aging impairs the self-organizing capacity of lung alveolar epithelial stem cells. eLife. 2021;10.
Coppe JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118.
Lepperdinger G. Inflammation and mesenchymal stem cell aging. Curr Opin Immunol. 2011;23[4]:518 – 24.
Cartwright MJ, Tchkonia T, Kirkland JL. Aging in adipocytes: potential impact of inherent, depot-specific mechanisms. Exp Gerontol. 2007;42[6]:463 – 71.
Starr ME, Evers BM, Saito H. Age-associated increase in cytokine production during systemic inflammation: adipose tissue as a major source of IL-6. J Gerontol A Biol Sci Med Sci. 2009;64[7]:723 – 30.
Mokarizadeh A, Delirezh N, Morshedi A, Mosayebi G, Farshid AA, Mardani K. Microvesicles derived from mesenchymal stem cells: potent organelles for induction of tolerogenic signaling. Immunol Lett. 2012;147[1–2]:47–54.
Martini H, Iacovoni JS, Maggiorani D, Dutaur M, Marsal DJ, Roncalli J et al. Aging induces cardiac mesenchymal stromal cell senescence and promotes endothelial cell fate of the CD90 + subset. Aging Cell. 2019;18[5]:e13015.
Shin TH, Kim HS, Kang TW, Lee BC, Lee HY, Kim YJ et al. Human umbilical cord blood-stem cells direct macrophage polarization and block inflammasome activation to alleviate rheumatoid arthritis. Cell Death Dis. 2016;7[12]:e2524.
Oishi Y, Manabe I. Macrophages in age-related chronic inflammatory diseases. NPJ aging and mechanisms of disease. 2016;2:16018.
Pajarinen J, Lin T, Gibon E, Kohno Y, Maruyama M, Nathan K, et al. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials. 2019;196:80–9.
Yin Y, Wu RX, He XT, Xu XY, Wang J, Chen FM. Influences of age-related changes in mesenchymal stem cells on macrophages during in-vitro culture. Stem Cell Res Ther. 2017;8[1]:153.
Mattiucci D, Maurizi G, Leoni P, Poloni A. Aging- and senescence-associated changes of mesenchymal stromal cells in myelodysplastic syndromes. Cell Transplant. 2018;27[5]:754 – 64.
Yu KR, Kang KS. Aging-related genes in mesenchymal stem cells: a mini-review. Gerontology. 2013;59[6]:557 – 63.
Kizilay Mancini O, Lora M, Shum-Tim D, Nadeau S, Rodier F, Colmegna I. A proinflammatory secretome mediates the impaired immunopotency of human mesenchymal stromal cells in Elderly patients with atherosclerosis. Stem cells translational medicine. 2017;6[4]:1132–40.
Ti D, Hao H, Tong C, Liu J, Dong L, Zheng J, et al. LPS-preconditioned mesenchymal stromal cells modify macrophage polarization for resolution of chronic inflammation via exosome-shuttled let-7b. J translational Med. 2015;13:308.
Yao M, Cui B, Zhang W, Ma W, Zhao G, Xing L. Exosomal miR-21 secreted by IL-1beta-primed-mesenchymal stem cells induces macrophage M2 polarization and ameliorates sepsis. Life Sci. 2021;264:118658.
Huang R, Qin C, Wang J, Hu Y, Zheng G, Qiu G, et al. Differential effects of extracellular vesicles from aging and young mesenchymal stem cells in acute lung injury. Aging. 2019;1118:7996–8014.
Davis C, Dukes A, Drewry M, Helwa I, Johnson MH, Isales CM et al. MicroRNA-183-5p increases with age in bone-derived extracellular vesicles, suppresses bone marrow stromal [Stem] cell proliferation, and induces stem cell senescence. Tissue Eng Part A. 2017;23[21–22]:1231–40.
Fafian-Labora J, Lesende-Rodriguez I, Fernandez-Pernas P, Sangiao-Alvarellos S, Monserrat L, Arntz OJ, et al. Effect of age on pro-inflammatory miRNAs contained in mesenchymal stem cell-derived extracellular vesicles. Sci Rep. 2017;7:43923.
Wang Y, Fu B, Sun X, Li D, Huang Q, Zhao W, et al. Differentially expressed microRNAs in bone marrow mesenchymal stem cell-derived microvesicles in young and older rats and their effect on tumor growth factor-beta1-mediated epithelial-mesenchymal transition in HK2 cells. Stem Cell Res Ther. 2015;6:185.
Denu RA, Hematti P. Effects of oxidative stress on Mesenchymal Stem Cell Biology. Oxidative Med Cell Longev. 2016;2016:2989076.
Betancourt AM. New Cell-Based therapy paradigm: induction of bone marrow-derived multipotent mesenchymal stromal cells into pro-inflammatory MSC1 and anti-inflammatory MSC2 phenotypes. Adv Biochem Eng Biotechnol. 2013;130:163–97.
Loisel S, Dulong J, Menard C, Renoud ML, Meziere N, Isabelle B, et al. Brief report: Proteasomal Indoleamine 2,3-Dioxygenase degradation reduces the immunosuppressive potential of clinical Grade-Mesenchymal stromal cells undergoing replicative senescence. Stem Cells. 2017;355:1431–6.
Jeske R, Yuan X, Fu Q, Bunnell BA, Logan TM, Li Y. Vitro Culture Expansion shifts the Immune phenotype of human adipose-derived mesenchymal stem cells. Front Immunol. 2021;12:621744.
Lefevre L, Iacovoni JS, Martini H, Belliere J, Maggiorani D, Dutaur M et al. Kidney inflammaging is promoted by CCR2[+] macrophages and tissue-derived micro-environmental factors. Cell Mol Life Sci. 2021;78[7]:3485 – 501.
Maijenburg MW, Kleijer M, Vermeul K, Mul EP, van Alphen FP, van der Schoot CE et al. The composition of the mesenchymal stromal cell compartment in human bone marrow changes during development and aging. Haematologica. 2012;97[2]:179 – 83.
Josephson AM, Bradaschia-Correa V, Lee S, Leclerc K, Patel KS, Muinos Lopez E, et al. Age-related inflammation triggers skeletal stem/progenitor cell dysfunction. Proc Natl Acad Sci USA. 2019;11614:6995–7004.
Siegel G, Kluba T, Hermanutz-Klein U, Bieback K, Northoff H, Schafer R. Phenotype, donor age and gender affect function of human bone marrow-derived mesenchymal stromal cells. BMC Med. 2013;11:146.
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;4797372:232–6.
Baker DJ, Childs BG, Durik M, Wijers ME, Sieben CJ, Zhong J, et al. Naturally occurring p16[Ink4a]-positive cells shorten healthy lifespan. Nature. 2016;5307589:184–9.
Jeon OH, Kim C, Laberge RM, Demaria M, Rathod S, Vasserot AP et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat Med. 2017;23[6]:775 – 81.
Zhang L, Pitcher LE, Prahalad V, Niedernhofer LJ, Robbins PD. Recent advances in the discovery of senolytics. Mech Ageing Dev. 2021;200:111587.
Xu M, Pirtskhalava T, Farr JN, Weigand BM, Palmer AK, Weivoda MM et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24[8]:1246–56.
Hickson LJ, Langhi Prata LGP, Bobart SA, Evans TK, Giorgadze N, Hashmi SK, et al. Senolytics decrease senescent cells in humans: preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019;47:446–56.
Li X, Hong Y, He H, Jiang G, You W, Liang X, et al. FGF21 mediates mesenchymal stem cell senescence via regulation of mitochondrial Dynamics. Oxidative Med Cell Longev. 2019;2019:4915149.
Hong Y, He H, Jiang G, Zhang H, Tao W, Ding Y et al. Mir-155-5p inhibition rejuvenates aged mesenchymal stem cells and enhances cardioprotection following infarction. Aging Cell. 2020;19[4]:e13128.
Lee S, Le NH, Kang D. Melatonin alleviates oxidative stress-inhibited osteogenesis of human bone marrow-derived mesenchymal stem cells through AMPK activation. Int J Med Sci. 2018;15[10]:1083–91.
Li Q, Zhu Z, Wang C, Cai L, Lu J, Wang Y, et al. CTRP9 ameliorates cellular senescence via PGC–1alpha/AMPK signaling in mesenchymal stem cells. Int J Mol Med. 2018;422:1054–63.
Xia W, Zhang F, Xie C, Jiang M, Hou M. Macrophage migration inhibitory factor confers resistance to senescence through CD74-dependent AMPK-FOXO3a signaling in mesenchymal stem cells. Stem Cell Res Ther. 2015;6[1]:82.
Park SY, Jeong AJ, Kim GY, Jo A, Lee JE, Leem SH, et al. Lactoferrin protects human mesenchymal stem cells from oxidative Stress-Induced Senescence and apoptosis. J Microbiol Biotechnol. 2017;2710:1877–84.
Hu C, Zhao L, Peng C, Li L. Regulation of the mitochondrial reactive oxygen species: strategies to control mesenchymal stem cell fates ex vivo and in vivo. J Cell Mol Med. 2018;22[11]:5196 – 207.
Zheng WV, Xu W, Li Y, Qin J, Zhou T, Li D, et al. Anti-aging effect of beta-carotene through regulating the KAT7-P15 signaling axis, inflammation and oxidative stress process. Cell Mol Biol Lett. 2022;27(1):86.
Kim C, Park JM, Song Y, Kim S, Moon J. HIF1alpha-mediated AIMP3 suppression delays stem cell aging via the induction of autophagy. Aging Cell. 2019;18[2]:e12909.
Ma Y, Qi M, An Y, Zhang L, Yang R, Doro DH et al. Autophagy controls mesenchymal stem cell properties and senescence during bone aging. Aging Cell. 2018;17[1].
Gu Z, Tan W, Ji J, Feng G, Meng Y, Da Z, et al. Rapamycin reverses the senescent phenotype and improves immunoregulation of mesenchymal stem cells from MRL/lpr mice and systemic lupus erythematosus patients through inhibition of the mTOR signaling pathway. Aging. 2016;85:1102–14.
Lei F, Huang Z, Ou Q, Li J, Liu M, Ma L et al. Apoptotic vesicles rejuvenate mesenchymal stem cells via Rab7-mediated autolysosome formation and alleviate bone loss in aging mice. Nano Res. 2022.
Jiao H, Walczak BE, Lee MS, Lemieux ME, Li WJ. GATA6 regulates aging of human mesenchymal stem/stromal cells. Stem Cells. 2021;391:62–77.
Liu F, Yuan Y, Bai L, Yuan L, Li L, Liu J, et al. LRRc17 controls BMSC senescence via mitophagy and inhibits the therapeutic effect of BMSCs on ovariectomy-induced bone loss. Redox Biol. 2021;43:101963.
Dave JR, Chandekar SS, Behera S, Desai KU, Salve PM, Sapkal NB et al. Human gingival mesenchymal stem cells retain their growth and immunomodulatory characteristics independent of donor age. Sci Adv. 2022;8[25]:eabm6504.
Verkhratsky A, Li Q, Melino S, Melino G, Shi Y. Can COVID-19 pandemic boost the epidemic of neurodegenerative diseases? Biol Direct. 2020;15[1]:28.
Li Q, Wang Y, Sun Q, Knopf J, Herrmann M, Lin L et al. Immune response in COVID-19: what is next? Cell Death Differ. 2022;29[6]:1107–22.
Melino S, Leo S, Toska Papajani V. Natural hydrogen Sulfide Donors from Allium sp. as a Nutraceutical Approach in type 2 diabetes Prevention and Therapy. Nutrients. 2019;11[7].
Gao X, Lu A, Tang Y, Schneppendahl J, Liebowitz AB, Scibetta AC et al. Influences of donor and host age on human muscle-derived stem cell-mediated bone regeneration. Stem Cell Res Ther. 2018;9[1]:316.
Zhang DY, Wang HJ, Tan YZ. Wnt/beta-catenin signaling induces the aging of mesenchymal stem cells through the DNA damage response and the p53/p21 pathway. PLoS ONE. 2011;6[6]:e21397.
Murphy T, Thuret S. The systemic milieu as a mediator of dietary influence on stem cell function during ageing. Ageing Res Rev. 2015;19:53–64.
Acknowledgements
The authors thank Massimiliano Agostini for helpful and constructive criticisms.
Funding
This study has received funding from the European Union NextGenerationEU through the Italian Ministry of University and Research under PNRR-MAC2-II.3 project PE6 “Heal Italia" to GM, EC, MP and FB) CUP E83C22004670001; and partially by Associazione Italiana per la Ricerca contro il Cancro [AIRC] to GM [IG 2022 ID 27366; 2023–2027], or to EC [IG#22206; 2019–2023]. Work has been also partially supported by Regione Lazio through LazioInnova Progetto Gruppo di Ricerca n 85-2017-14986; n 33 & 55-2021-T0002E0001.
Author information
Authors and Affiliations
Contributions
XY, GM, FB and EC conceived the project; XY prepared the first draft; XY, GM, FB and EC wrote the manuscript; XY prepared figures. All Authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
GM is members of the Editorial Board of Cell Death Differentiation/Disease. The authors declare no other conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yang, X., Wang, Y., Rovella, V. et al. Aged mesenchymal stem cells and inflammation: from pathology to potential therapeutic strategies. Biol Direct 18, 40 (2023). https://doi.org/10.1186/s13062-023-00394-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13062-023-00394-6