Abstract
Background
In addition to the conventional aetiologic agents of oropharyngeal squamous cell carcinoma (OPSCC) such as tobacco usage, alcohol consumption and betel quid usage, it has been established that a proportion of OPSCC are driven by persistent oncogenic human papillomavirus (HPV) infections. Currently, there is a lack of data on the burden of HPV- associated OPSCC in Asian countries including Malaysia.
Methods
A cross-sectional multicentre study with tissue analysis of Malaysian patients diagnosed with primary OPSCC within a five-year period, from 2015 to 2019 between 01/01/2015 to 31/12/2019 was undertaken. Determination of HPV status was carried out using p16INK4a immunohistochemistry on tissue microarrays constructed from archived formalin-fixed paraffin-embedded tissue.
Results
From the cases identified, 184 cases had sufficient tissue material for analysis. Overall, median age at diagnosis was 63.0 years (IQR = 15) and 76.1% of patients were males. In our cohort, 35.3% of patients were Indian, 34.2% were Chinese, 21.2% were Malay and 9.2% were from other ethnicities. The estimated prevalence of HPV-associated OPSCC in our cohort was 31.0% (CI 24.4–38.2%). The median age for the HPV-associated OPSCC sub-group of patients was not significantly lower than the median age of patients with HPV-independent OPSCC. More than half of HPV-associated OPSCC was seen in patients of Chinese ethnicity (54.4%). Patients with HPV-associated OPSCC had a much better overall survival than patients with HPV-independent OPSCC (Log rank test; p < 0.001). Patients with HPV-associated OPSCC with no habit-related risk factors such as smoking, were found to have much better overall survival when compared to all other sub-groups.
Conclusions
The findings from our study suggests that prevalence of HPV-associated OPSCC in Malaysia, though not as high as some developed countries, is however on an upward trend. HPV-associated OPSCC appears to be more frequently encountered in patients of Chinese ethnicity. Conventional risk-factors associated with OPSCC such as smoking, alcohol consumption and betel quid chewing should still be considered when estimating prognosis of patients with HPV-associated OPSCC.
Similar content being viewed by others
Background
The recent Global Cancer Observatory (GLOBOCAN) 2020 report estimated that more than 19 million new cases of cancer were diagnosed worldwide, with cancer being the leading cause of death in approximately 57 countries [1, 2]. Head and neck cancers are comprised of a diverse group of malignancies involving the various sub-sites of the head and neck region. In 2020, it was estimated that head and neck cancers (including lip, oral cavity, nasopharynx, larynx, hypopharynx and oropharynx) accounted for approximately 878, 348 new cancer cases (4.6%), making this group of cancers one of the top ten cancers in the world [1]. Oropharyngeal cancer is one of the cancers that has been showing an increasing trend with regard to incidence. There were approximately 98.412 new cases of oropharyngeal cancers in the 2020 GLOBOCAN report, an increase of more than 5000 from the previous 2018 GLOBOCAN report. [1, 3] Interestingly, the number of new deaths resulting from oropharyngeal cancers was lower in the 2020 GLOBOCAN report when compared to the 2018 GLOBOCAN report [1, 3].
The majority of oropharyngeal cancers are squamous cell carcinomas (SCC). In addition to the conventional aetiologic agents of oropharyngeal squamous cell carcinoma (OPSCC) such as tobacco usage, alcohol consumption and betel quid usage, it has been proven that a high proportion of OPSCC are driven by oncogenic human papillomavirus (HPV) [4]. HPV are DNA viruses that are known to infect mucosal and cutaneous epithelium, with persistent infection being known to cause benign or malignant tumours. Of the more than 100 HPV genotypes, the International Agency for Research on Cancer (IARC) has classified several genotypes as being “high-risk” genotypes and some as “low-risk” genotypes [5]. The IARC has classified HPV genotypes 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58 and 59 as carcinogenic, HPV68 as “probably carcinogenic to humans” and HPV genotypes 26, 53, 66, 67, 70, 73, 82, 30, 34, 69, 85, 97 as “possibly carcinogenic” [5]. HPV genotypes 6 and 11, are considered as “low-risk” genotypes and have been known to cause anogenital warts [5, 6].
It has been estimated that approximately 690,000 cases of cancers worldwide in 2018 were attributable to HPV [7]. The vast majority of these cancers were cervical cancers (approximately 570,000 cases), followed by OPSCC (approximately 42,000 cases), anal SCC (approximately 29,000 cases), penile carcinoma (approximately 18,000 cases), and other sites making up the remaining cases [7]. Epidemiological studies have revealed that HPV16 and HPV18 genotypes are implicated in more than 70% of all cancers attributable to HPV [7]. Additionally, it is believed that the large majority (80–90%) of HPV-associated OPSCC is due to HPV16 [4, 8]. It has been hypothesized that the increase in HPV-associated OPSCC could possibly be due to changes in sexual practices and behaviour over the past few decades [8].
Patients with HPV-associated OPSCC have been shown consistently to have better survival than patients with HPV-independent OPSCC prompting the creation of distinct staging algorithm for patients with HPV-associated OPSCC [9]. However, much of the existing data regarding the prevalence, clinico-pathological features, and clinical outcome of patients with HPV-associated OPSCC has been derived from Caucasian populations [4]. The prevalence of HPV-associated OPSCC as well as clinical features of patients with this disease is not well characterized among Asian patients. At this point in time, there has only been one study looking into the prevalence of HPV- associated OPSCC in Malaysia. The study described the clinico-pathological features of HPV-associated OPSCC in a relatively small cohort of 60 patients with incomplete data that may not have been truly representative of the Malaysian population [10]. The current study was undertaken to address this gap in knowledge and to provide much needed data to inform policy making.
Methods
Patients
This was a cross-sectional multi-centre study, with tissue analysis, of Malaysian patients diagnosed with primary OPSCC within a five-year period, from 2015 to 2019. This study was performed with ethical approval from the Malaysian Medical Research and Ethics Committee (NMRR-20-49-52574) and complies with local regulations and guidelines. Individual patient consent was not sought as patients were not recruited for the study. Anonymised, archived tissue samples were analysed. The study was performed in accordance with the Declaration of Helsinki. Cases were identified from clinical and pathology databases. One representative formalin-fixed paraffin-embedded (FFPE) block, either from biopsy or resection specimen, with adequate tissue for immunohistochemistry (IHC) was selected for each patient. Cases were excluded if there was inadequate FFPE tissue for IHC. The following data were obtained from the archived records: sex; age at diagnosis; ethnicity; history of tobacco use; history of alcohol intake; history of betel-quid use; site of tumour; Tumour, Node, Metastasis (TNM) staging and date of last follow-up or death. Overall mortality status was obtained until end of June 2022. Data were link-anonymised and recorded into a standardised proforma. Determination of HPV status for each case was through assessment of surrogate marker p16INK4a IHC (70% nuclear and cytoplasmic cut-off) [9, 11, 12]. Cases were considered as HPV-associated OPSCC if p16INK4a IHC was scored as being positive and HPV-independent OPSCC if p16INK4a IHC was scored as being negative.
Histopathological assessment
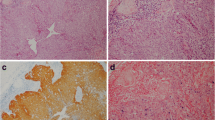
Fresh 5 µm sections were taken from each FFPE block and mounted onto microscope slides. Haematoxylin and eosin (H&E) staining was performed on the Leica ST5010 Autostainer XL (Leica Biosystems, USA). Histopathological diagnosis was confirmed through review of the H&E-stained slides by the study pathologists and areas of confluent tumour within the tissue specimens were identified for construction of tissue microarrays (TMAs) for p16INK4a IHC. For cases where TMA construction was not possible (tissue too small or unable to obtain tissue blocks), whole sections were used for p16INK4a IHC. Tissue microarrays were constructed following previously described methods [13]. Haematoxylin and eosin–stained sections of the TMAs were made and assessed to confirm sampling accuracy.
p16INK4a immunohistochemistry (IHC)
Immunohistochemistry was performed using a monoclonal p16INK4a antibody (JC2 clone; Cell Marque, Rocklin, USA). Five-micron sections of formalin-fixed paraffin-embedded (FFPE) tissues were manually stained by using a proprietary kit (EnVision FLEX+, Dako, Agilent Technologies, Santa Clara, California USA). The HPV/p16 Analyte Control block (HistoCyte Laboratories Ltd, Newcastle upon Tyne, UK) which demonstrates high homogenous, high heterogenous and negative expression of p16 was used for positive and negative control. Scoring for p16INK4a IHC were done according to previously published criteria [11]. Briefly, p16INK4a IHC was scored as being positive only if there was strong and diffuse nuclear and cytoplasmic staining present in greater than 70% of the tumour cells [9, 11, 12].
Statistics
Statistical analysis was performed using IBM SPSS for Windows version 26 (IBM Corp., Armonk, N.Y., USA). Parametric and non-parametric tests were used for initial analysis of variables. Time-to-event analysis was performed to assess the influence of HPV status on overall survival using the Log Rank test. Statistical significance was defined at the 5% level.
Results
From the cases identified, 184 cases had sufficient formalin-fixed paraffin-embedded tissue material for analysis. Overall, median age at diagnosis was 63.0 years (IQR = 15) and 76.1% of patients were males. In our cohort, 35.3% of patients were Indian, 34.2% were Chinese, 21.2% were Malay and 9.2% were from other ethnicities. The tonsils were the most frequently encountered sub-site for OPSCC (41.8%) followed by base of tongue tumours (30.4%). Smoking history was only available for 146 patients, with 59.6% of these patients having a positive smoking history (either current or former smokers). The majority of OPSCC patients presented with high-stage disease (Stage III or IV) when TNM staging was based on the 7th Edition of American Joint Committee on Cancer (AJCC) Staging system. Demographic and clinico-pathological features of these patients stratified according to p16 status are displayed in Table 1.
In our study, 57 patients were found to have p16INK4a IHC positivity, giving an estimated prevalence of 31.0% (95% confidence interval of 24.4 to 38.2%) for HPV-associated OPSCC in our cohort. The median age for the HPV-associated OPSCC sub-group of patients was slightly lower at 63.0 years (IQR = 11), this however was not statistically significant. The majority of HPV-associated OPSCC was seen in Chinese patients (54.4%) whilst the majority of HPV-independent OPSCC was seen in Indian patients (44.9%) (Table 1; Fisher-Freeman-Halton Exact Test; p < 0.001). The tonsils were the most frequently site for HPV-associated OPSCC (57.9%) (Table 1; Fisher-Freeman-Halton Exact Test; p = 0.002).
Sub-analysis of cases with available habit related history showed the following results; smoking history and alcohol consumption history were not statistically associated with HPV status of OPSCC whilst history of betel quid usage was significantly associated with HPV status of OPSCC (Fisher’s exact test; p << 0.001). None of the HPV-associated OPSCC patients had a history of betel quid usage.
Survival analysis showed that patients with HPV-associated OPSCC had a much better overall survival than patients with HPV-independent OPSCC (Fig. 1; Log rank test; p < 0.001). Sub-analysis on habit-related risk factors revealed that patients who were never smokers and had HPV-associated OPSCC were found to have much better overall survival when compared to all other sub-groups. Patients who had a positive smoking history (current/former) and HPV-independent OPSCC had the worst overall survival (Fig. 2; Log-rank test; p < 0.001; n = 146). Similarly, HPV-associated OPSCC patients with no history of betel quid usage had much better overall survival rates compared to the other sub-groups (Fig. 3; Log-rank test; p < 0.001; n = 117). However, the overall survival of patients with HPV-independent OPSCC with or without a history of betel quid use were rather similar (Fig. 3; Log-rank test; p = 0.738; n = 117). Interestingly, in the sub-analysis for alcohol consumption status, non-drinkers appeared to do slightly better than drinkers in both sub-groups, however, the findings were not statistically significant (Fig. 4; Log-rank test; p > 0.05; n = 131).
Kaplan Meier estimates of overall survival stratified according to smoking status in combination with HPV status. Never smokers with HPV-associated OPSCC had the best overall survival rates compared to other sub-groups (Log-rank test, pooled over strata; p < 0.001; n = 146). Pairwise statistical comparisons between patients with HPV-independent OPSCC who had no history of smoking and those that did, showed statistically significant difference between the two (Log-rank test, pairwise over strata; p = 0.021; n = 146). However, pairwise statistical comparisons between patients with HPV-associated OPSCC who had no history of smoking and those that did, showed no statistically significant difference (Log-rank test, pairwise over strata; p = 0.074; n = 146). Interestingly, pairwise statistical comparisons between patients with HPV-associated OPSCC with history of smoking and non-smokers with HPV-independent OPSCC had a rather similar prognosis with no statistically significant difference between them (Log-rank test, pairwise over strata; p = 0.286; n = 146)
Kaplan Meier estimates of overall survival stratified according to history of betel quid use in combination with HPV status. All HPV-associated OPSCC patients were found to have never used (chewed) betel quid and had much better overall survival rates compared to the other sub-groups (Log-rank test, pooled over strata; p < 0.001; n = 117). However, pairwise statistical comparisons between patients with HPV-independent OPSCC who had no history of betel quid usage and those that did, showed no statistically significant difference (Log-rank test, pairwise over strata; p = 0.738; n = 117)
Kaplan Meier estimates of overall survival stratified according to history of alcohol consumption in combination with HPV status. Patients with HPV-associated OPSCC were found to have better overall survival rates compared to HPV-independent OPSCC patients (Log-rank test, pooled over strata; p < 0.001; n = 131). Pairwise statistical comparisons between patients with HPV-associated OPSCC who had no history of alcohol consumption and those that did, showed no statistically significant difference (Log-rank test, pairwise over strata; p = 0.612; n = 131). Similarly, pairwise statistical comparisons between patients with HPV-independent OPSCC who had no history of alcohol consumption and those that did, showed no statistically significant difference (Log-rank test, pairwise over strata; p = 0.222; n = 131)
Overall survival for patients with HPV-associated OPSCC was not influenced by ethnicity (Fig. 5; Log-rank test, pooled over strata; p = 0.234; n = 57), however for patients with HPV-independent OPSCC, patients of Malay ethnicity were seen to have poorer overall survival when compared to patients of other ethnicity (Fig. 5; Log-rank test, pooled over strata; p = 0.028; n = 127).
Kaplan Meier estimates of overall survival stratified according to ethnicity of patients. a There was no statistically significant difference in overall survival for patients with HPV-associated OPSCC of different ethnic groups (Log-rank test, pooled over strata; p = 0.234; n = 57); b Patients stratified according to ethnicity of patients. There was statistically significant difference in overall survival for patients with HPV-independent OPSCC of different ethnic groups, with patients of Malay ethnicity having poorer overall survival when compared to patients of other ethnicity (Log-rank test, pooled over strata; p = 0.028; n = 127)
Discussion
The incidence of OPSCC in Malaysia has been on an increasing trend based on the last two Malaysian National Cancer Registry Reports [14, 15]. The estimated prevalence of HPV-associated OPSCC of 31.0% from our study was slightly higher than the 25% prevalence rate found by a previous Malaysian study published in 2018 [10]. The aforementioned study looked at 60 cases of OPSCC curated over a 12-year period (2004–2015). Aside from possible increase in the HPV-associated fraction of OPSCC over the years, the vast difference in sample size between the current study and the previous study by Yap et al. could also be a contributing factor to this difference in prevalence rates.
Although the prevalence rates from our study are comparable to reported rates from Singapore and lower than that reported in other developed Asian countries such as Japan, South Korea and China, cross comparison between epidemiological studies are hampered by the criteria used to define HPV status in OPSCC [16,17,18,19,20]. Numerous definitions have been used to define the HPV status of OPSCC in the literature with some investigators using additional molecular methods to determine if a tumour is truly “HPV-driven” [21,22,23,24,25,26,27]. The prognostic utility of such dual or tiered testing methods that involve more complex molecular methods has been quite promising, though it may be difficult to roll out such assays as readily as p16INK4a IHC to the general, hospital-based histopathology laboratories around the world [23,24,25,26,27]. However, such dual- or three-tiered testing may be appropriate in clinical trials that involve treatment de-escalation so that allocation of patients to treatment arms is done with better accuracy. Although a surrogate marker, the utilization of p16INK4a IHC staining as a defining criteria for HPV-associated OPSCC has been included in the 8th Edition of the AJCC cancer staging manual as well as the latest edition of the World Health Organization (WHO) reference text on Head and Neck Tumours and as such it is advisable to include assessment of p16INK4a IHC in any study involving OPSCC to enable cross-comparisons [9, 12].
An interesting observation from our study is the relatively similar age at diagnosis between patients with HPV-associated OPSCC and HPV-independent OPSCC. The median values for each sub-group showed that the HPV-associated OPSCC patients were only slightly younger than the HPV-independent OPSCC patients. Though most earlier epidemiological studies on HPV-associated OPSCC have suggested that these patients are usually younger that patients with HPV-independent OPSCC patients, this trend may be shifting and HPV-associated OPSCC cases may no longer be frequently encountered in younger patients [12, 17, 21, 28,29,30,31]. This trend has serious implications as the survival advantage demonstrated in patients with HPV-associated OPSCC may be less pronounced in older patients especially those with more co-morbidities.
Although OPSCC was most frequently seen among patients of the Indian ethnic group, the vast majority of the Indian patients had HPV-independent OPSCC (57/65 cases). More than half of all the HPV-associated OPSCC patients in our study were from the Chinese ethnic group. Almost 50% (31/63 cases) of Chinese patients with OPSCC had HPV-associated OPSCC and in fact more than half of the HPV-associated OPSCC disease burden is experienced by the Chinese ethnic group. It is worth noting that when compared to the findings from the earlier study by Yap et al. [10] the proportion of HPV-associated OPSCC in both the Chinese and Indian ethnic groups appears to be increasing. This difference in prevalence rates between ethnic groups warrants further research into risk factors that may predispose to HPV-associated OPSCC such as sexual behaviour and other lifestyle-related factors. [32, 33] Sexual transmission of high-risk HPV between partners/couples has already been implicated in cases of concurrent HPV-associated OPSCC in couples [34, 35]. However, no such cases were seen in our cohort of patients. In patients with HPV-independent OPSCC, patients from the Malay ethnic group appeared to have worse overall survival when compared to other ethnic groups. This difference was not seen in patients with HPV-associated OPSCC. This may be due to other patient-related prognostic factors such as diet, habits (smoking, etc.) and other co-morbidities.
Consistent with other studies on OPSCC, our patients with HPV-associated OPSCC were shown to have better overall survival when compared to patients with HPV-independent OPSCC [12, 17, 36,37,38]. The prognosis of patients with OPSCC, regardless of whether HPV-associated or otherwise, appears to be adversely affected by smoking history, history of betel quid usage as well as history of alcohol consumption. Our findings are similar to that reported by others [36, 39]. As such, it is imperative to always take into account other patient-related prognostic factors such as smoking history, betel quid chewing and alcohol consumption, just to name a few. A limitation in our study is the lack of in-depth data on patient-related (such as smoking pack-years, etc.) and treatment-related (treatment provided, etc.) prognostic factors. Future studies should aim to obtain more detailed accounts of patient-related and treatment-related prognostic factors.
Another possible limitation in our study relates to the utilisation of TMAs for p16INK4a IHC. The small cores sampled from the tissue block may not be completely representative of whole tumours as tumours tend to be heterogenous and may express proteins at varying levels in different regions [40]. However, evaluation of p16INK4a IHC on TMAs has been performed successfully by several research groups [27, 41, 42].
Conclusion
The findings from our study suggests that prevalence of HPV-associated OPSCC in Malaysia, though not as high as some developed countries, is however on an upward trend. HPV-associated OPSCC appears to be more frequently encountered in patients of Chinese ethnicity. The shift of HPV-associated OPSCC from “younger” to “older” patients is also of concern with regard to prognosis and burden on the healthcare system. Conventional risk-factors associated with OPSCC such as smoking, alcohol consumption and betel quid chewing should still be considered when estimating prognosis of patients with HPV-associated OPSCC as they may attenuate the positive prognostic effects of HPV status.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AJCC:
-
American Joint Committee on Cancer
- FFPE:
-
Formalin-fixed paraffin-embedded
- GLOBOCAN:
-
Global Cancer Observatory
- HPV:
-
Human papillomavirus
- H&E:
-
Haematoxylin and eosin
- IARC:
-
International Agency for Research on Cancer
- IHC:
-
Immunohistochemistry
- IQR:
-
Interquartile range
- NRD:
-
National Registration Department
- OPSCC:
-
Oropharyngeal squamous cell carcinoma
- SCC:
-
Squamous cell carcinoma
- TMA:
-
Tissue microarray
- TNM:
-
Tumour, Node, Metastasis
- WHO:
-
World Health Organization
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209–49.
Bray F, Laversanne M, Weiderpass E, Soerjomataram I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer. 2021;127(16):3029–30.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018.
Castellsagué X, Alemany L, Quer M, Halec G, Quirós B, Tous S, Clavero O, Alòs L, Biegner T, Szafarowski T, et al. HPV involvement in head and neck cancers: comprehensive assessment of biomarkers in 3680 patients. J Natl Cancer Inst. 2016;108(6):djv403.
Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, Ghissassi FE, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L, et al. A review of human carcinogens—Part B: biological agents. Lancet Oncol. 2009;10(4):321–2.
Burd EM. Human papillomavirus and cervical cancer. Clin Microbiol Rev. 2003;16(1):1–17.
de Martel C, Georges D, Bray F, Ferlay J, Clifford GM. Global burden of cancer attributable to infections in 2018: a worldwide incidence analysis. Lancet Glob Health. 2020;8(2):e180–90.
Kreimer AR, Chaturvedi AK, Alemany L, Anantharaman D, Bray F, Carrington M, Doorbar J, D’Souza G, Fakhry C, Ferris RL, et al. Summary from an international cancer seminar focused on human papillomavirus (HPV)-positive oropharynx cancer, convened by scientists at IARC and NCI. Oral Oncol. 2020;108:104736.
Lydiatt WM, Patel SG, O'Sullivan B, Brandwein MS, Ridge JA, Migliacci JC, Loomis AM, Shah JP. Head and neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;67(2), 122–137.
Yap LF, Lai SL, Rhodes A, Sathasivam HP, Abdullah MA, Pua KC, Rajadurai P, Cheah PL, Thavaraj S, Robinson M, et al. Clinico-pathological features of oropharyngeal squamous cell carcinomas in Malaysia with reference to HPV infection. Infect Agent Cancer. 2018;13:21.
Singhi AD, Westra WH. Comparison of human papillomavirus in situ hybridization and p16 immunohistochemistry in the detection of human papillomavirus-associated head and neck cancer based on a prospective clinical experience. Cancer. 2010;116(9):2166–73.
WHO Classification of Tumours Editorial Board. Head and neck tumours [Internet; beta version ahead of print]. In: Vol. 9, 5th edn. Lyon (France): International Agency for Research on Cancer; 2022.
Parsons M, Grabsch H. How to make tissue microarrays. Diagn Histopathol. 2009;15(3):142–50.
Malaysian National Cancer Registry Report 2007–2011. In. Putrajaya, Malaysia: National Cancer Institute, Ministry of Health, Malaysia; 2016.
Malaysian National Cancer Registry Report 2012–2016. In. Putrajaya, Malaysia: National Cancer Institute, Ministry of Health, Malaysia; 2020.
Fu EWZ, Chilagondanahalli N, Bundele MM, Gan JY, Li H, Lim MY. Trends in HPV-related oropharyngeal cancers in Singapore. Ann Acad Med Singap. 2021;50(6):508–9.
Tan LS, Fredrik P, Ker L, Yu FG, Wang Y, Goh BC, Loh KS, Lim CM. High-risk HPV genotypes and P16INK4a expression in a cohort of head and neck squamous cell carcinoma patients in Singapore. Oncotarget. 2016;7(52):86730–9.
Ni G, Huang K, Luan Y, Cao Z, Chen S, Ma B, Yuan J, Wu X, Chen G, Wang T, et al. Human papillomavirus infection among head and neck squamous cell carcinomas in southern China. PLoS ONE. 2019;14(9):e0221045.
Jun HW, Ji YB, Song CM, Myung JK, Park HJ, Tae K. Positive rate of human papillomavirus and its trend in head and neck cancer in South Korea. Front Surg. 2021;8:833048.
Hama T, Tokumaru Y, Fujii M, Yane K, Okami K, Kato K, Masuda M, Mineta H, Nakashima T, Sugasawa M, et al. Prevalence of human papillomavirus in oropharyngeal cancer: a multicenter study in Japan. Oncology. 2014;87(3):173–82.
Nauta IH, Rietbergen MM, van Bokhoven AAJD, Bloemena E, Lissenberg-Witte BI, Heideman DAM, Baatenburg de Jong RJ, Brakenhoff RH, Leemans CR. Evaluation of the eighth TNM classification on p16-positive oropharyngeal squamous cell carcinomas in the Netherlands and the importance of additional HPV DNA testing. Ann Oncol. 2018;29(5):1273–9.
Sathasivam HP, Santambrogio A, Andoniadou CL, Robinson M, Thavaraj S. Prognostic utility of HPV specific testing in addition to p16 immunohistochemistry in oropharyngeal squamous cell carcinoma. Ann Oncol. 2018;29(10):2144–5.
Mirghani H, Amen F, Moreau F, Guigay J, Ferchiou M, Melkane AE, Hartl DM, Lacau St Guily J. Human papilloma virus testing in oropharyngeal squamous cell carcinoma: what the clinician should know. Oral Oncol. 2014;50(1):1–9.
Mirghani H, Casiraghi O, Amen F, He M, Ma X-J, Saulnier P, Lacroix L, Drusch F, Ben Lakdhar A, Saint Guily JL, et al. Diagnosis of HPV-driven head and neck cancer with a single test in routine clinical practice. Mod Pathol. 2015;28(12):1518–27.
Thavaraj S, Stokes A, Guerra E, Bible J, Halligan E, Long A, Okpokam A, Sloan P, Odell E, Robinson M. Evaluation of human papillomavirus testing for squamous cell carcinoma of the tonsil in clinical practice. J Clin Pathol. 2011;64(4):308–12.
Robinson M, Schache A, Sloan P, Thavaraj S. HPV specific testing: a requirement for oropharyngeal squamous cell carcinoma patients. Head Neck Pathol. 2012;6 Suppl 1(Suppl 1):S83-90.
Craig SG, Anderson LA, Schache AG, Moran M, Graham L, Currie K, Rooney K, Robinson M, Upile NS, Brooker R, et al. Recommendations for determining HPV status in patients with oropharyngeal cancers under TNM8 guidelines: a two-tier approach. Br J Cancer. 2019;120(8):827–33.
Rettig EM, Zaidi M, Faraji F, Eisele DW, El Asmar M, Fung N, D’Souza G, Fakhry C. Oropharyngeal cancer is no longer a disease of younger patients and the prognostic advantage of Human Papillomavirus is attenuated among older patients: analysis of the National Cancer Database. Oral Oncol. 2018;83:147–53.
Del Mistro A, Frayle H, Menegaldo A, Favaretto N, Gori S, Nicolai P, Spinato G, Romeo S, Tirelli G, da Mosto MC, et al. Age-independent increasing prevalence of Human Papillomavirus-driven oropharyngeal carcinomas in North-East Italy. Sci Rep. 2020;10(1):9320.
Rettig EM, Fakhry C, Khararjian A, Westra WH. Age profile of patients with oropharyngeal squamous cell carcinoma. JAMA Otolaryngol Head Neck Surg. 2018;144(6):538–9.
Windon MJ, D’Souza G, Rettig EM, Westra WH, van Zante A, Wang SJ, Ryan WR, Mydlarz WK, Ha PK, Miles BA, et al. Increasing prevalence of human papillomavirus-positive oropharyngeal cancers among older adults. Cancer. 2018;124(14):2993–9.
D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356(19):1944–56.
Gillison ML, D’Souza G, Westra W, Sugar E, Xiao W, Begum S, Viscidi R. Distinct risk factor profiles for human papillomavirus type 16-positive and human papillomavirus type 16-negative head and neck cancers. J Natl Cancer Inst. 2008;100(6):407–20.
Sathasivam HP, Bhatia R, Bradley P, Robson A, Paleri V, Cocks H, Oozeer N, Milne D, Sloan P, Robinson M. Concurrent HPV-related oropharyngeal carcinoma in four couples. Oral Oncol. 2018;86:33–7.
Haddad R, Crum C, Chen Z, Krane J, Posner M, Li Y, Burk R. HPV16 transmission between a couple with HPV-related head and neck cancer. Oral Oncol. 2008;44(8):812–5.
Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tân PF, Westra WH, Chung CH, Jordan RC, Lu C, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363(1):24–35.
Fakhry C, Westra WH, Wang SJ, van Zante A, Zhang Y, Rettig E, Yin LX, Ryan WR, Ha PK, Wentz A, et al. The prognostic role of sex, race, and human papillomavirus in oropharyngeal and nonoropharyngeal head and neck squamous cell cancer. Cancer. 2017;123(9):1566–75.
Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, Forastiere A, Gillison ML. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst. 2008;100(4):261–9.
Weiss BG, Anczykowski MZ, Küffer S, Spiegel JL, Bertlich M, Canis M, Ihler F, Kitz J, Jakob M. Prognostic impact of additional HPV diagnostics in 102 patients with p16-stratified advanced oropharyngeal squamous cell carcinoma. Eur Arch Otorhinolaryngol. 2021;278(6):1983–2000.
Khouja MH, Baekelandt M, Sarab A, Nesland JM, Holm R. Limitations of tissue microarrays compared with whole tissue sections in survival analysis. Oncol Lett. 2010;1(5):827–31.
Lesnikova I, Lidang M, Hamilton-Dutoit S, Koch J. p16 as a diagnostic marker of cervical neoplasia: a tissue microarray study of 796 archival specimens. Diagn Pathol. 2009;4(1):22.
Schache AG, Liloglou T, Risk JM, Jones TM, Ma XJ, Wang H, Bui S, Luo Y, Sloan P, Shaw RJ, et al. Validation of a novel diagnostic standard in HPV-positive oropharyngeal squamous cell carcinoma. Br J Cancer. 2013;108(6):1332–9.
Acknowledgements
The authors would like to thank the Director-General of Health Malaysia for his permission to publish this article. We would also like to thank the Biostatistics & Data Repository Sector of The National Institutes of Health for death verification services, and the provision of study data for this study. We are also grateful to the National Registration Department (NRD) of Malaysia for allowing NRD death data to be used for research purposes. We would like to acknowledge the contributions of all the members of the Malaysian Head & Neck Cancer Research Group.
Funding
This project received funding from the Ministry of Health, Malaysia (NMRR-20-49-52574). The funding body had no role in the design of the study or the analysis of the data.
Author information
Authors and Affiliations
Consortia
Contributions
HPS: Proposed the idea, designed the study, contributed towards data collection, contributed towards curation of samples, performed laboratory analysis, performed pathological assessment, performed statistical analysis, and wrote major parts of the manuscript. SPD, CSM, RJ, ZZ, ARA, HH, SGM, YTL, YDJ, ASMS, BGHB, ZY, KASAD, AH, MM, MZI, SQMAK, WEWM & MHH: Contributed to collection & quality assurance of data, curation of samples and critically reviewed the manuscript. PV: Contributed to data collection, performed pathological assessment, and critically reviewed the manuscript. RFR: Contributed to laboratory work, quality assurance of data, and critically reviewed the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study has ethical approval from the Malaysian Medical Research and Ethics Committee (NMRR-20-49-52574) and complies with local legislation and guidelines. The study was performed in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Competing interests
HPS has been funded by a pharmaceutical company for a separate investigator-initiated study pertaining to human papillomavirus. Aside from that, the authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Sathasivam, H.P., Davan, S.P., Chua, S.M. et al. Findings from a Malaysian multicentre study on oropharyngeal squamous cell carcinoma. Infect Agents Cancer 18, 74 (2023). https://doi.org/10.1186/s13027-023-00557-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13027-023-00557-0