Abstract
Background
The Ca2+-independent contraction of vascular smooth muscle is a leading cause of cardiovascular and cerebrovascular spasms. In the previous study, we demonstrated the involvement of Src family protein tyrosine kinase Fyn and Rho-kinase in the sphingosylphosphorylcholine (SPC)-induced abnormal and Ca2+-independent contraction of vascular smooth muscle, but the specific mechanism has not been completely clarified.
Methods
Paxillin knockdown human coronary artery smooth muscle cells (CASMCs) and smooth muscle-specific paxillin knockout mice were generated by using paxillin shRNA and the tamoxifen-inducible Cre-LoxP system, respectively. CASMCs contraction was observed by time-lapse recording. The vessel contractility was measured by using a myography assay. Fyn, Rho-kinase, and myosin light chain activation were assessed by immunoprecipitation and western blotting. The paxillin expression and actin stress fibers were visualized by histological analysis and immunofluorescent staining.
Results
The SPC-induced abnormal contraction was inhibited in paxillin knockdown CASMCs and arteries of paxillin knockout mice, indicating that paxillin is involved in this abnormal contraction. Further study showed that paxillin knockdown inhibited the SPC-induced Rho-kinase activation without affecting Fyn activation. In addition, paxillin knockdown significantly inhibited the SPC-induced actin stress fiber formation and myosin light chain phosphorylation. These results suggest that paxillin, as an upstream molecule of Rho-kinase, is involved in the SPC-induced abnormal contraction of vascular smooth muscle.
Conclusions
The present study demonstrated that paxillin participates in the SPC-induced abnormal vascular smooth muscle contraction by regulating Rho-kinase activation.
Video Abstract
Similar content being viewed by others
Background
Myocardial infarction and cerebral infarction caused by coronary artery spasm and cerebrovascular spasm are considered to be among the main causes of morbidity and mortality worldwide. This vasospasm is an abnormal and long-lasting contraction of vascular smooth muscle (VSM), which leads to narrowing of blood vessels and reduces blood flow to tissues and organs, eventually resulting in ischemia [1,2,3,4]. Many studies have demonstrated that this abnormal contraction is characterized by the Ca2+-independent contraction or Ca2+-sensitization [5,6,7], which differs from the normal vascular tone that is characterized by the Ca2+-dependent contraction mediated through Ca2+-calmodulin (CaM)-myosin light chain kinase (MLCK) pathway [8,9,10]. Our previous study has shown that sphingosylphosphorylcholine (SPC), a bioactive sphingolipid produced by the hydrolysis of the membrane sphingolipid, triggers the Ca2+-independent contraction of VSM [6, 7]. SPC concentration in the cerebrospinal fluid significantly increases after subarachnoid hemorrhage and is considered to be the main cause of cerebrovascular spasm [11], acting as a spasmogen.
Fyn tyrosine kinase, a member of the Src family protein tyrosine kinases, and Rho-kinase have been shown to be involved in the SPC-induced abnormal contraction of VSM [5, 7]. In our previous study, we proposed that Fyn translocation from the cytosol to the cell membrane is involved in the SPC-induced contraction of VSM. Additionally, inhibition of Fyn translocation by EPA effectively inhibits the SPC-induced contraction [5]. The active Fyn regulates the SPC-induced actin stress fiber formation and constitutively active Fyn (CA-Fyn) is located at the ends of actin stress fibers anchored at focal adhesion [12, 13]. Accumulating evidence has demonstrated that Rho-kinase mediates coronary artery spasm and cerebrovascular spasm [14,15,16,17,18,19,20], including the SPC-induced contraction of VSM [5,6,7, 21]. Dominant negative Rho-kinase and Rho-kinase inhibiter Y27632 abolish the SPC-induced contraction [6, 7]. Fasudil, another Rho-kinase inhibitor, has been shown to effectively prevent coronary artery spasm and cerebral vasospasm [16, 18]. Translocation of Rho-kinase from the cytosol to the cell membrane in VSM cells plays an essential role in the SPC-induced contraction [6]. Although Rho-kinase and Fyn have been implicated in the regulation of the SPC-induced contraction of VSM, the precise mechanism(s) underlying this abnormal contraction of VSM remains largely unclear.
Focal adhesions are large and dynamic protein complexes containing paxillin and focal adhesion kinase (FAK), which connect the extracellular matrix to cytoskeletal microfilaments and play a fundamental role in cell migration. Recent research has shown that focal adhesions play a critical role in muscle contraction by acting as sites for transmitting the contractile force generated by the actin-myosin stress fiber network to the rigid extracellular matrix [22,23,24]. FAK activation facilitates smooth muscle contraction in the gastric fundus and depletion of FAK inhibits contractile activation of smooth muscle [25]. Paxillin plays an important role in cytoskeletal reorganization by recruiting various signaling molecules, including FAK [26,27,28,29]. Paxillin, as a substrate for FAK, regulates the Ca2+-dependent contraction of tracheal smooth muscle [30]. Loss of paxillin in zebrafish results in decreased cardiac contractility, leading to severe heart failure [31]. Recently, we have discovered that paxillin is a binding partner of activated Fyn [32]. Based on these findings, we hypothesized that paxillin may play a role in the SPC-induced abnormal contraction of VSM.
In this study, we aimed to investigate the involvement of paxillin in the SPC-induced abnormal contraction of VSM. We utilized paxillin knockdown human coronary artery smooth muscle cells (CASMCs) and smooth muscle-specific paxillin knockout mice to examine their SPC-induced contractions. Furthermore, we examined the mechanism(s) by which paxillin regulates the SPC-induced contraction of VSM. Our findings revealed a previously unknown role for paxillin in the SPC-induced abnormal contraction of VSM, highlighting its potential as a therapeutic target for reducing cardiovascular and cerebrovascular diseases associated with abnormal VSM contraction.
Methods and materials
Reagents and antibodies
Sphingosylphosphorylcholine (SPC) was purchased from Biomol (Plymouth Meeting, PA, USA). Tamoxifen was purchased from Funakoshi (Tokyo, Japan). Corn oil was purchased from Sigma Aldrich (St. Louis, MO, USA).
The following antibodies were used: mouse monoclonal anti-paxillin antibody (Cat#610620, BD Biosciences, San Jose, CA, USA), anti-paxillin polyclonal antibody (H85, Santa Cruz, Dallas, CA, USA), anti-GAPDH monoclonal antibody (Wako, Osaka, Japan), anti-phospho-MYPT1 (T850) with phosphorylation site corresponding to T853 in human MYPT1 (Milipore, Burlington, CA, USA), anti-MYPT1 antibody (H130, Santa Cruz, Dallas, CA, USA), anti-phospho-myosin light chain 2 (S19) monoclonal antibody (Cell Signaling, Danvers, MA, USA), anti-myosin light chain (20 kDa) monoclonal antibody (Sigma Aldrich, St. Louis, MO, USA), anti-α-smooth muscle actin antibody (Thermo Fisher, Waltham, MA, USA), anti-mouse Alexa Fluor 488 (Thermo Fisher, Waltham, MA, USA), and anti-rabbit Alexa Fluor 546 (Thermo Fisher,Waltham, MA, USA). Secondary HRP-labeled antibodies (anti-mouse and anti-rabbit) were purchased from Promega (Madison, WI, USA).
Cell culture
Human coronary artery smooth muscle cells (CASMCs) were cultured in HuMedia SG2 (Kurabo, Osaka, Japan) supplemented with 5% fetal bovine serum (FBS), 0.5 ng/ml human epidermal growth factor (hEGF), 2 ng/ml human fibroblast growth factor-B (hFGF-B), 5 µg/ml insulin, 50 µg/ml gentamycin, and 50 ng/ml amphotericin B. Cells were maintained at 37 °C in a humidified atmosphere of 5% CO2 and 95% air. Human CASMCs at passage numbers < 10 splitting cycles were used for experiments.
To generate paxillin-downregulated cells, we infected human CASMCs with paxillin shRNA lentiviral particles (sc-29439-V, Santa Cruz, Dallas, CA, USA) following the manufacturer’s instructions. Prior to infection, we optimized the infection conditions using copGFP control lentiviral particles (sc-108084, Santa Cruz, Dallas, CA, USA) as previously reported [32]. Using the optimal condition, we successfully generated a paxillin-downregulated cell line. In brief, cells were seeded in a 12-well plate and were infected with paxillin shRNA or control shRNA (sc-108080, Santa Cruz, Dallas, CA, USA) lentiviral particles mixed with complete medium and Polybrene (sc-134220, Santa Cruz, Dallas, CA, USA) when they reached approximately 50% confluency. After infection, stable clones were selected using puromycin dihydrochloride (sc-108071, Santa Cruz, Dallas, CA, USA) at the concentration of 2 µg/mL.
Immunoprecipitation and western blotting assay
Serum-starved cells were treated with SPC (30 µM) for the indicated times. The cells were washed with PBS and lysed using an immunoprecipitation assay buffer (50 mM Tris, pH 7.4, 150 mM NaCl, 10 mM NaF, 0.2 mM Na3VO4, 0.1% Triton X-100, 0.5% NP-40, 1 mM phenylmethylsulphonyl fluoride [PMSF], 1 µg/mL of leupeptin, and 1 µg/mL of aprotinin). Protein concentrations were determined using a protein assay kit (Bio-Rad, Hercules, CA, USA) with bovine serum albumin as the standard. The cell lysates containing equal amounts of proteins were centrifuged at 4 °C for 10 min at 10,000×g and the supernatants were precleared by incubation with protein A/G agarose beads (Santa Cruz, Dallas, CA, USA) at 4 °C for 1 h, followed by centrifugation at 4 °C for 5 min at 10,000×g. The supernatants were then incubated with 1 µg of Fyn antibody and protein A/G agarose beads overnight at 4 °C. After washing the beads four times with ice-cold immunoprecipitation assay buffer, the bound proteins were eluted with 2 × SDS-PAGE sample buffer (2% SDS, 20 mM DTT, 20% glycerol, 20 mM Tris pH 6.8, and 0.1% bromophenol blue). The samples were separated using 10% SDS-PAGE and transferred onto Amersham Hybond-P PVDF membrane (GE Healthcare Life Sciences, Baie d’Urfé, QC, Canada), followed by the blocking of the membrane with 5% non-fat milk in TBS-T for 60 min at 25 °C. The membranes were incubated with primary antibodies for 1 h at 25 °C or overnight at 4 °C, followed by their incubation with peroxidase-conjugated secondary antibodies for 1 h at 25 °C. Immunoreactive bands were detected using SuperSignal West Pico chemiluminescent substrate kit (Thermo Fisher Scientific, Waltham, MA, USA) and digitally quantified using Bio-Rad ChemiDoc XRS-J detection system.
Tissue samples were collected from mice as described in our previous report [33]. Briefly, the smooth muscle layer of the aorta was isolated after the removal of adipose tissue and adventitia under an optical microscope. Subsequently, the layer was sectioned into strips and subjected to treatment either in the absence (vehicle control) or in the presence of SPC (10 µM for 15 min at 37 °C). The tissues were then promptly frozen in a solution of 10% trichloroacetic acid (TCA) and 10 mM DTT in acetone, which had been pre-chilled on ice, and subsequently rinsed twice with cold 10 mM DTT/acetone. The tissues were then placed in liquid nitrogen and pulverized using the SK-Mill Freeze-Crush Apparatus. Protein was extracted from the freeze-dried tissues by adding 100 µL of RIPA buffer (Wako, Osaka, Japan) along with a protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, CA), and the samples were heated at 95 °C for 5 min for western blot analysis.
Time-lapse recording of VSM cell contraction
Human CASMCs infected with lentiviral control shRNA or paxillin shRNA were grown in 35-mm glass-based dishes (Iwaki, Osaka, Japan). When cell confluence reached 90–100%, FBS and growth factor-free HuMedia SB2 (Kurabo, Osaka, Japan) medium was changed to induce hypercontractile type of CASMCs following previously established methods [33]. After treatment with HuMedia SB2 for 48 h, 30 µM SPC was added to the medium and time-lapse recording of VSM cell contraction was performed under a fluorescent microscope (Keyence Biorevo BZ-9000, Osaka, Japan). Cell images were recorded for 30 min every 30 s.
Animals
C57BL/6 mice at 6–18 weeks old were used for experiments. SMMHC-CreERT2 transgenic mice were purchased from The Jackson Laboratory (RRID:IMSR_JAX:019079). The offspring homozygous mice were produced in the Institute of Life Science and Medicine of Yamaguchi University. Mice were kept with a 12 h light-dark border circulation system (25℃) under a pathogen-free condition with free access to food and water. All mice utilized in this study and all experiments were conducted in accordance with the Animal Care and Use Committee of the Model Animal Research Center at Yamaguchi University. Animal welfare and experimental programs strictly comply with the guidelines for the care and use of laboratory animals.
Generation of smooth muscle tissue-specific paxillin knockout mice (paxillin SMKO mice)
Major experimental tasks (design and construction of targeting vector, establishment of targeted ES cells, generation of the chimera and F1 mice, and removal of the neo cassette) to generate the paxillin SMKO mice were performed by Unitech, Co. (Kashiwa, Japan). Briefly, bacterial artificial chromosome (BAC)-retrieval methods were used for constructing the targeting vector (Additional file 1: Figure S1). Exons 2–5, which encode paxillin, were flanked by 2 loxP sites and an frt-Neo-frt cassette as a positive selection marker. The “FRT-Neo-FRT-loxP” sequence was inserted upstream of Exon 2, and the loxP sequence was inserted downstream of Exon 5. Therefore, when the region containing Exons 2 to 5 flanked by loxP sequences was removed by the Cre-loxP system, the region after Exon 2 of the target gene was knocked out by frameshifting.
To specifically ablate the expression of paxillin in smooth muscle cells, we generated mice with two floxed paxillin alleles and the SMMHC-CreERT2 transgene by crossing paxillin floxed mice with SMMHC-CreERT2 transgenic mice, as shown in Additional file 2: Figure S2A. DNA isolated from mice tail tissues were genotyped by PCR for LoxP+/+ sites and the presence of the Cre-recombinase using specific primers (For LoxP+/+, forward primer sequence: 5′-AAACCTTTCTTCATAAATTGGAAGG-3′, the revers primer sequence: 5′-GGAAGGTATAGATGTGTATCAGCAC-3′; For Cre, the forward primer sequence: 5′-TGACCCCATCTCTTCACTCC-3′, the reverse primer sequence: 5′-AGTCCCTCACATCCTCAGGTT-3′). A representative genotyping result showed Lox P+/+ sites (350 bp), Cre control (287 bp) and wild type (190 bp) in Additional file 2: Figure S2B, S2C, and S2D. Male mice received intraperitoneal injections of tamoxifen at a dose of 1 mg tamoxifen solution in 100 µl corn oil for five consecutive days [34, 35]. Tamoxifen (T5648, Sigma-Aldrich, St. Louis, MO, USA) was dissolved in corn oil (42 °C) at a concentration of 10 mg/mL and incubated at 37 °C for 1 h before use. After four weeks of tamoxifen injections, the mice were used for experiments. Only male mice were used in this study, as the SMMHC-CreERT2 transgene is located on the Y-chromosome.
Myography assay for vessel contractility
Male mice injected with corn oil (control mice) and tamoxifen (paxillin SMKO mice) were sacrificed by cervical dislocation, and thoracic aorta was excised. Connective tissue and fat adjacent to the aorta were carefully removed without causing any damage to the endothelium. The aorta was cut into several 2 mm rings and suspended in a multi-wire myograph system (DMT610M, Lab Tech, Japan) and bathed in Krebs solution (123 mM NaCl, 4.7 mM KCl, 15.5 mM NaHCO3, 1.2 mM KH2PO4, 1.2 mM MgCl2, 1.25 mM CaCl2, and 11.5 mM D-glucose) gassed with 95% O2/5% CO2. After equilibration for 30 min at 37℃, the rings were contracted with 80 mM K+-physiological salt solution (48.9 mM NaCl, 78.8 mM KCl, 15.5 mM NaHCO3, 1.2 mM KH2PO4, 1.2 mM MgCl2, 1.25 mM CaCl2, and 11.5 mM D-glucose) and washed three times with Krebs solution. After recording steady responses to repeated applications of 80 mM K+-depolarization, endothelium inhibitor L-NAME was added to inhibit eNOS for 15 min, and then 80 mM K+-depolarization was used to induce contraction again. After re-adding L-NAME for another 15 min, VSM contraction was induced using different concentrations of SPC (1, 3, 10, and 30 µM) or noradrenaline (0.001, 0.003, 0.01, 0.03, 0.1, 0.3, 1, 3, 10, and 30 µM).
Histological analysis and immunofluorescent staining
For the histological analysis, tissues were fixed overnight using 4% paraformaldehyde and then embedded in paraffin. Transverse Sect. (5 μm) were stained with hematoxylin/eosin and examined under a microscope (Keyence Biorevo BZ-9000, Osaka, Japan). For the immunofluorescence analysis, sections were blocked with NanoBio blocker solution (Nano Bio Tech Co., Ltd) diluted in PBS for 30 min at 25℃. After washing three times with PBS, primary antibodies and fluorescent secondary antibodies were used. The fluorescent staining was examined with a fluorescent microscope (Keyence Biorevo BZ-9000, Osaka, Japan).
The staining of F-actin was carried out using rhodamine-conjugated phalloidin (1:100, Thermo Fisher Scientific, Waltham, MA, USA) and visualized under a fluorescence microscope (Keyence Biorevo BZ-9000, Osaka, Japan). The formation of actin stress fibers was quantified by counting the number of cells containing a thick linear structure of actin stress fibers.
Statistical analysis
Data are presented as mean ± SEM. Statistical analysis was performed using GraphPad Prism 8.0. Student’s t-test was used for analyzing data between two groups. Significant differences between groups were considered when p < 0.05.
Results
Paxillin knockdown inhibits the SPC-induced contraction in human CASMCs
To investigate the role of paxillin in the SPC-induced contraction, we first constructed paxillin knockdown human CASMCs using paxillin shRNA lentiviral transduction (Fig. 1a, b). We then recorded the SPC-induced contraction in control and paxillin knockdown human CASMCs through time-lapse observation. As shown in Fig. 1c, SPC induced a noticeable contraction in control CASMCs, with the elongated cells becoming short or round (Additional file 5: Video S1). In contrast, paxillin knockdown human CASMCs showed little change in morphology after stimulation with SPC for 30 min (Additional file 6: Video S2). We quantified cellular area in control and paxillin knockdown CASMCs with and without SPC stimulation. As shown in Fig. 1d, after stimulation with SPC for 30 min, control CASMCs exhibited a significant decrease in cell area compared to the non-stimulated cells. In contrast, the area of paxillin knockdown cells remained relatively unchanged following SPC stimulation for 30 min. These results suggest that paxillin knockdown attenuates the SPC-induced contraction in CASMCs.
Paxillin knockdown inhibits the SPC-induced contraction in human CASMCs. a, Western blot showing paxillin expression in control shRNA or paxillin shRNA lentiviral particles infected human CASMCs in the absence and presence of SPC. b, Statistical analysis of paxillin expression in control shRNA and paxillin shRNA lentiviral particles infected human CASMCs (n = 4, **p < 0.01). c, Representative live cell imaging of cellular contraction at various time points following SPC stimulation (30 µM) in control shRNA and paxillin shRNA lentiviral particles infected human CASMCs. Scale bar = 100 µm. d, Statistical analysis of the cell area of control shRNA and paxillin shRNA lentiviral particles infected human CASMCs after SPC stimulation (30 µM) for 30 min. Three independent experiments were performed, and 100 cells were measured for the cell area using ImageJ. **p < 0.01; ns, no significant
Paxillin deficiency inhibits the SPC-induced contraction in mice
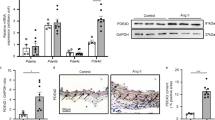
To further investigate whether paxillin deficiency in VSM tissue leads to the weakening or loss of the SPC-induced contraction, we generated the paxillin smooth muscle knockout (SMKO) mice using a tamoxifen-inducible Cre-LoxP system. Western blots confirmed the absence of paxillin expression in the medial smooth muscle layer of the thoracic aorta from paxillin SMKO mice (Fig. 2a). Additionally, immunofluorescence staining showed no paxillin expression in the medial smooth muscle layer of the thoracic aorta in paxillin SMKO mice (Fig. 2b).
Establishment of paxillin smooth muscle-specific knockout mice. a, Representative western blot of paxillin expression in heart muscle, straited muscle layer of esophagus, and the medial smooth muscle layer of thoracic aorta, from control mice (Cont) and paxillin smooth muscle knockout (SMKO) mice (PXN CKO). Note that we removed the endothelial layer and connective tissue, leaving only the medial smooth muscle layer of thoracic aorta. GAPDH is shown as a loading control. b, Immunofluorescent staining showing that paxillin is knockout in the medial smooth muscle layer of thoracic aorta of paxillin SMKO mice. α-smooth muscle actin (α-SMA, red) as a smooth muscle-specific marker is detected in both control mice (Cont) and paxillin SMKO mice (PXN CKO). Paxillin (Green) is detected in the medial smooth muscle layer of control mice (Cont) but not in that of paxillin SMKO mice (PXN CKO), as shown in the yellow arrow. Scale Bar = 100 µm in 10× images, Scale Bar = 20 µm in 40× images
Prior to investigating the SPC-induced contraction of VSM using paxillin SMKO mice, we assessed the structure and function of blood vessels in both control and paxillin SMKO mice. Hematoxylin and eosin (HE) staining revealed no significant difference in the structure of VSM between paxillin SMKO mice and control mice (Fig. 3a). In addition, we examined high K+ depolarization-induced and noradrenaline (NA)-induced contractions, which showed no significant differences between control and paxillin SMKO mice (Fig. 3b, c). These results indicate that paxillin deletion in VSM does not affect the structure or contractile function of the artery under normal physiological conditions.
Paxillin knockout has no effect on the physiological structure and function of thoracic aorta. a, HE staining of transverse sections of the thoracic aorta in control and paxillin SMKO mice. Scale bar = 200 µm. b, 80 mM K+ depolarization-induced contraction in thoracic aorta shows no difference between control (n = 9) and paxillin SMKO mice (n = 9). c, Noradrenaline (NA)-induced contraction in thoracic aorta shows a dose-dependent manner from control (n = 3) and paxillin SMKO mice (n = 3).Cont: control mice; PXN KO: paxillin SMKO mice; ns: no significant
We then examined the SPC-induced contraction in the thoracic aorta of control and paxillin SMKO mice. The results, shown in Fig. 4a, depict representative contraction tracings of thoracic aorta strips of control mice and paxillin SMKO mice that were stimulated with varying concentrations of SPC. Figure 4b shows that the SPC-induced contractions (3 µM and 10 µM) were significantly weaker in paxillin SMKO mice compared to the control mice. We also compared the relative value of the SPC-induced contraction to the maximal K+-induced contraction. In line with the findings in Fig. 4b, paxillin SMKO mice showed a decrease relative to the control mice when stimulated with SPC at 3 µM and 10 µM concentrations. These results suggest that paxillin plays an important role in the SPC-induced contraction.
Paxillin knockout attenuates the SPC-induced abnormal contraction of thoracic aorta. a, Representative contraction traces are shown for thoracic aorta from control and paxillin SMKO mice after stimulation with SPC at the different concentrations (1, 3, 10, and 30 µM). b, The SPC-induced contraction (mN) at the concentration of 3 µM and 10 µM from control (n = 10) and paxillin SMKO mice (n = 11). *p<0.05. c, Ratio of 3 µM and 10 µM SPC-induced contraction compared to 80 mM K+ depolarization-induced contraction in control (n= 9) and paxillin SMKO mice (n= 9). *p<0.05. Cont: control mice; PXN KO: paxillin SMKO mice
Paxillin deficiency inhibits the SPC-induced Rho-kinase activation, but not Fyn activation
Our recent study demonstrated the association between paxillin and the active Fyn [32], indicating that paxillin may act as a downstream molecule of Fyn. To further investigate this, we examined the activity of Fyn in paxillin knockdown cells. Fyn activity is regulated by Y420 phosphorylation [36]. However, the specific antibody for Fyn Y420 phosphorylation is currently unavailable. Therefore, we utilized an immunoprecipitation method using Fyn antibody to obtain Fyn and then detected Y420 phosphorylation with the use of the Src Y416 phosphorylated antibody that cross-reacts with Y420 phosphorylated Fyn. As shown in Fig. 5a, b, Y420 phosphorylated Fyn increased in both control and paxillin knockdown CASMCs after stimulation of SPC, indicating that paxillin knockdown could not affect the SPC-induced activation of Fyn.
Paxillin knockdown inhibits SPC-induced Rho-kinase activation but not SPC-induced Fyn activation. a, Fyn activity in paxillin knockdown human CASMCs in the absence and presence of SPC (30 µM, 5 min), using immunoprecipitation and Western blot. We used Fyn antibody to obtain Fyn and then detected Y420 phosphorylation of Fyn (pY420Fyn) with the Src Y416 phosphorylated antibody that cross-reacts with pY420Fyn. b, Statistical analysis of Fyn activation (Ratio of pY420Fyn to total Fyn after Fyn immunoprecipitation) in (a). n = 3, *p<0.05, ns: no significant. c and d, The phosphorylation of MYPT1 at site T853 was analyzed by Western blot in paxillin knockdown human CASMCs (c) and paxillin SMKO mice (d). e and f, Statistical analysis of Rho-kinase activation (Ratio of phosphorylated MYPT1 at site T853 to total MYPT1) in paxillin knockdown human CASMCs (e,n = 3) and paxillin SMKO mice (f,n = 4). *p<0.05. Cont: control mice; PXN KO: paxillin SMKO mice
Then we investigated the impact of paxillin deficiency on the SPC-induced Rho-kinase activation. As previously reported, we evaluated the phosphorylation of myosin phosphatase targeting subunit 1 (MYPT1) at T853 [33], which is directly related to Rho-kinase activation. In both control CASMCs and control mice, SPC stimulation significantly increased MYPT1 phosphorylation (Fig. 5c-f). However, in paxillin knockdown CASMCs and paxillin SMKO mice, even in the presence of SPC, MYPT1 phosphorylation did not increase (Fig. 5c-f). These results indicate that paxillin is involved in the SPC-induced Rho-kinase activation and that paxillin serves as an intermediate regulatory molecule linking Fyn and Rho-kinase in SPC-induced contraction.
Paxillin deficiency attenuates the SPC-induced actin stress fiber formation and myosin light chain (MLC) phosphorylation
Smooth muscle contraction requires myosin activation and actin cytoskeleton remodeling [33, 37,38,39,40,41]. Rho-kinase activation has two effects: (1) Directly phosphorylating MLC at S19 [42, 43] or phosphorylating MYPT1, which is a subunit of myosin light chain phosphatase (MLCP), inactivates MLCP and increases phosphorylation of MLC; (2) Promoting the formation of actin stress fibers [44, 45]. Since paxillin is an upstream molecule of Rho-kinase, it is hypothesized that paxillin deficiency could attenuate MLC phosphorylation and actin stress fiber formation. To investigate this, we first observed the effect of paxillin on SPC-induced actin stress fiber formation in human CASMCs. Compared to control cells, the SPC-induced actin stress fiber formation was inhibited in paxillin knockdown cells (Fig. 6a, b). Next, we examined MLC phosphorylation in control cells, which showed that SPC stimulation induced MLC phosphorylation. However, the SPC-induced MLC phosphorylation was significantly decreased in paxillin knockdown cells (Fig. 6c, e). Similarly, the SPC-induced MLC phosphorylation was significantly reduced in paxillin SMKO mice compared to control mice (Fig. 6d, f). These results suggest that paxillin regulates two contractile factors, actin cytoskeleton remodeling and myosin activation, by regulating the activation of Rho-kinase, which in turn regulates the SPC-induced contraction of VSM.
Paxillin knockdown inhibits SPC-induced actin stress fiber formation and myosin light chain phosphorylation. a, Immunofluorescence staining of the SPC-induced actin stress fiber formation in control and paxillin knockdown human CASMCs. Scale bar = 20 µm. b, Statistical analysis of the SPC-induced actin stress fiber formation in control and paxillin knockdown human CASMCs. Data were obtained from three independent experiments, and at least 44 cells were counted. c and d, Western blot showing the myosin light chain (MLC) phosphorylation in paxillin knockdown human CASMCs (c) and paxillin SMKO mice (d). e and f, Statistical analysis of MLC phosphorylation in paxillin knockdown human CASMCs (e, n = 4) and paxillin SMKO mice (f, n = 4). *p<0.05; **p<0.01. Cont: control mice; PXN KO: paxillin SMKO mice
Discussion
In this study, we generated paxillin knockout mice targeting smooth muscle for the first time and reported the following key findings: (1) paxillin deficiency attenuates abnormal contraction of VSM induced by SPC but not K+ depolarization-induced or NA-induced contraction; (2) paxillin acts as an intermediate molecule between Fyn and Rho-kinase to participate in the SPC-induced abnormal contraction; (3) paxillin deficiency inhibits the SPC-induced actin stress fiber formation; (4) paxillin deficiency inhibits the SPC-induced MLC phosphorylation; (5) in human CASMCs, paxillin knockdown also inhibits the SPC-induced contraction, Rho-kinase activation, and MLC phosphorylation. These key findings indicate that paxillin controls Rho-kinase activation and participates in the SPC-induced abnormal contraction of VSM. The revealed mechanism will provide new insights into cardiovascular and cerebrovascular diseases caused by abnormal contraction of VSM.
In the present study, we discovered a novel function of paxillin, which is involved in regulating abnormal contraction of VSM. While previous research has extensively studied the role of paxillin in cell migration, there is limited information regarding its effect on muscle contraction. Several studies have reported that paxillin is necessary for active tension development during Ca2+-dependent contraction of canine tracheal smooth muscle [30, 46]. However, to our knowledge, no research has yet investigated the role of paxillin in abnormal contraction of VSM, which occurs in a Ca2+-independent manner. So far, no paxillin inhibitor is currently available for in vitro and in vivo studies. Therefore, we employed a loss-of-function approach to investigate the role of paxillin in abnormal contraction of VSM induced by SPC. Initially, we generated paxillin knockdown human CASMCs through the use of paxillin shRNA and observed a significant inhibition of SPC-induced contraction in these cells (Fig. 1c, d). We then sought to investigate the role of paxillin in animal vascular tissue. However, mice with a whole-body knockout of paxillin displayed embryonic lethality at E9.5 due to defects in the amnion and allantois, as well as impaired growth, abnormal heart development, and impaired somite development [47]. As a result, we utilized a tamoxifen-inducible Cre-LoxP system to generate temporally selectable and spatially specific paxillin SMKO mice in this study. In this system, Cre recombinase is placed downstream of the smooth muscle myosin heavy chain (SMMHC) promoter, ensuring smooth muscle cell (SMC)-specific expression of Cre recombinase [48]. Because Cre is fused to a mutated human estrogen receptor (ERT2), its activity can be induced by tamoxifen [49]. When mice are administrated with tamoxifen, Cre is expressed and then enters into the nucleus. Cre catalyzes site-specific recombination events between loxP sites, thus smooth muscle tissue-specific paxillin knockout mice are obtained [34]. On the other hand, the expression of paxillin in cardiomyocytes and skeletal muscle cells are not affected (Fig. 2 and Additional file 3: Figure S3). Paxillin deficiency in VSM of mice also resulted in a decreased contraction induced by SPC (at 3 µM and 10 µM, Fig. 4), further supporting the critical role of paxillin in the abnormal VSM contraction. However, at a higher dose of SPC (30 µM), no significant difference in vasoconstriction was observed between control and paxillin SMKO mice (Additional file 4: Figure S4). It should be noted that the concentration of SPC in human plasma and serum is typically reported to be approximately 50 nM and 130 nM, respectively [50]. Although the exact concentration of SPC in mice plasma is unknown, the concentration of 30 µM SPC used in this experiment may significantly exceed physiological levels, potentially inducing vasoconstriction in paxillin-deficient mice.
Our recent study showed that paxillin binds to the active Fyn, but not the inactive Fyn [32], indicating that Fyn is activated by SPC and subsequently combines with paxillin. Here, we further showed that paxillin deficiency did not impact the Fyn activation (Fig. 5a, b), suggesting that paxillin, as a downstream molecule of active Fyn, is involved in the SPC-induced contraction of VSM. In addition, paxillin deficiency in human CASMCs or in mice inhibited the Rho-kinase activation, indicating that paxillin, as an upstream molecule of Rho-kinase, regulates the Rho-kinase-mediated contraction of VSM. Thus, paxillin acts as an intermediate regulatory molecule between Fyn and Rho-kinase to mediate abnormal contraction of VSM induced by SPC.
Smooth muscle contraction results from the interaction between myosin filament and actin filament. Actin filament polymerization and myosin activation are two parallel cellular processes that are essential for regulating smooth muscle contraction [39]. There are two types of contractions in vascular smooth muscle: Ca2+-dependent and Ca2+-independent contractions. Ca2+-dependent contraction, including high K+ depolarization-induced or NA-induced contraction, maintains physiological contraction through the Ca2+/CaM-MLCK signaling pathway. On the other hand, Ca2+-independent contraction induced by SPC causes pathological vasospasm via the Fyn/Rho-kinase signaling pathway. Our experiments on paxillin SMKO mice showed that the high K+ depolarization-induced or NA-induced Ca2+-dependent contractions were not inhibited, indicating that paxillin may not play an important role in physiological, Ca2+-dependent contraction (Fig. 3b,c). However, we observed that paxillin deficiency reduced SPC-induced Ca2+-independent contraction (Fig. 4). Furthermore, our results have shown that paxillin plays a dual regulatory role, which is involved in regulation of actin polymerization and myosin light chain phosphorylation by controlling of Rho-kinase activation. Rho-kinase activation not only promotes actin polymerization [45] but also increases MLC phosphorylation by inhibiting MLC phosphatase [51] or by directly phosphorylating MLC [43]. Paxillin deletion inhibited the SPC-induced Rho-kinase activation, suggesting that paxillin could be involved in two processes of abnormal VSM contraction, actin polymerization (Fig. 6a, b) and myosin activity (Fig. 6c-f), through Rho-kinase activation.
Conclusions
Collectively, we have provided evidence for the first time that paxillin, a novel regulatory molecule, participates in the abnormal contraction of VSM mediated by the SPC/Fyn/paxillin/Rho-kinase pathway (Fig. 7). Our findings suggest that targeting paxillin could be a promising therapeutic strategy for treating cardiovascular and cerebrovascular diseases caused by abnormal VSM contraction.
A schematic diagram showing that paxillin participates in the SPC-induced vascular smooth muscle contraction mediated by the Fyn/paxillin/Rho-kinase pathway. When stimuli factor SPC acts on vascular smooth muscle cells, Fyn is activated and translocated to the focal adhesion to bind to paxillin, resulting in Rho-kinase activation, which increases actin stress fiber formation and phosphorylation of MLC, triggering vascular smooth muscle abnormal contraction (vasospasm)
Availability of data and materials
Data can be made available upon request of the corresponding author.
Abbreviations
- SPC:
-
Sphingosylphosphorylcholine
- CASMCs:
-
Coronary artery smooth muscle cells
- VSM:
-
Vascular smooth muscle
- CaM:
-
Calmodulin
- MLCK:
-
Myosin light chain kinase
- CA-Fyn:
-
Constitutively active Fyn
- FAK:
-
Focal adhesion kinase
- MYPT1:
-
Myosin phosphatase targeting subunit 1
- SMKO:
-
Smooth muscle knockout
- NA:
-
Noradrenaline
- MLC:
-
Myosin light chain
- MLCP:
-
Myosin light chain phosphatase
References
Campe C, Neumann J, Sandalcioglu IE, Rashidi A, Luchtmann M. Vasospasm and delayed cerebral ischemia after uneventful clipping of an unruptured intracranial Aneurysm - a case report. BMC Neurol. 2019;19:226.
Yasue H, Nakagawa H, Itoh T, Harada E, Mizuno Y. Coronary artery spasm–clinical features, diagnosis, pathogenesis, and treatment. J Cardiol. 2008;51:2–17.
Shakur SF, Farhat HI. Cerebral vasospasm with ischemia following a spontaneous spinal subarachnoid Hemorrhage. Case Rep Med. 2013;2013:934143.
Slavich M, Patel RS. Coronary artery spasm: current knowledge and residual uncertainties. Int J Cardiol Heart Vasc. 2016;10:47–53.
Nakao F, Kobayashi S, Mogami K, Mizukami Y, Shirao S, Miwa S, Todoroki-Ikeda N, Ito M, Matsuzaki M. Involvement of src family protein tyrosine kinases in ca(2+) sensitization of coronary artery contraction mediated by a sphingosylphosphorylcholine-rho-kinase pathway. Circ Res. 2002;91:953–60.
Shirao S, Kashiwagi S, Sato M, Miwa S, Nakao F, Kurokawa T, Todoroki-Ikeda N, Mogami K, Mizukami Y, Kuriyama S, et al. Sphingosylphosphorylcholine is a novel messenger for rho-kinase-mediated Ca2 + sensitization in the bovine cerebral artery: unimportant role for protein kinase C. Circ Res. 2002;91:112–9.
Todoroki-Ikeda N, Mizukami Y, Mogami K, Kusuda T, Yamamoto K, Miyake T, Sato M, Suzuki S, Yamagata H, Hokazono Y, Kobayashi S. Sphingosylphosphorylcholine induces ca(2+)-sensitization of vascular smooth muscle contraction: possible involvement of rho-kinase. FEBS Lett. 2000;482:85–90.
Brozovich FV, Nicholson CJ, Degen CV, Gao YZ, Aggarwal M, Morgan KG. Mechanisms of vascular smooth muscle contraction and the basis for pharmacologic treatment of smooth muscle disorders. Pharmacol Rev. 2016;68:476–532.
Raina H, Zacharia J, Li M, Wier WG. Activation by Ca2+/calmodulin of an exogenous myosin light chain kinase in mouse arteries. J Physiol. 2009;587:2599–612.
Touyz RM, Alves-Lopes R, Rios FJ, Camargo LL, Anagnostopoulou A, Arner A, Montezano AC. Vascular smooth muscle contraction in Hypertension. Cardiovasc Res. 2018;114:529–39.
Kurokawa T, Yumiya Y, Fujisawa H, Shirao S, Kashiwagi S, Sato M, Kishi H, Miwa S, Mogami K, Kato S, et al. Elevated concentrations of sphingosylphosphorylcholine in cerebrospinal fluid after subarachnoid Hemorrhage: a possible role as a spasmogen. J Clin Neurosci. 2009;16:1064–8.
Xu D, Kishi H, Kawamichi H, Kajiya K, Takada Y, Kobayashi S. Involvement of fyn tyrosine kinase in actin stress fiber formation in fibroblasts. FEBS Lett. 2007;581:5227–33.
Xu D, Kishi H, Kawamichi H, Kajiya K, Takada Y, Kobayashi S. Sphingosylphosphorylcholine induces stress fiber formation via activation of Fyn-RhoA-ROCK signaling pathway in fibroblasts. Cell Signal. 2012;24:282–9.
Hizume T, Morikawa K, Takaki A, Abe K, Sunagawa K, Amano M, Kaibuchi K, Kubo C, Shimokawa H. Sustained elevation of serum cortisol level causes sensitization of coronary vasoconstricting responses in pigs in vivo: a possible link between stress and coronary vasospasm. Circ Res. 2006;99:767–75.
Kandabashi T, Shimokawa H, Miyata K, Kunihiro I, Kawano Y, Fukata Y, Higo T, Egashira K, Takahashi S, Kaibuchi K, Takeshita A. Inhibition of myosin phosphatase by upregulated rho-kinase plays a key role for coronary artery spasm in a porcine model with interleukin-1beta. Circulation. 2000;101:1319–23.
Masumoto A, Mohri M, Shimokawa H, Urakami L, Usui M, Takeshita A. Suppression of coronary artery spasm by the rho-kinase inhibitor fasudil in patients with vasospastic angina. Circulation. 2002;105:1545–7.
Shimokawa H, Seto M, Katsumata N, Amano M, Kozai T, Yamawaki T, Kuwata K, Kandabashi T, Egashira K, Ikegaki I, et al. Rho-kinase-mediated pathway induces enhanced myosin light chain phosphorylations in a swine model of coronary artery spasm. Cardiovasc Res. 1999;43:1029–39.
Naraoka M, Munakata A, Matsuda N, Shimamura N, Ohkuma H. Suppression of the Rho/Rho-kinase pathway and prevention of cerebral vasospasm by combination treatment with statin and fasudil after subarachnoid Hemorrhage in rabbit. Transl Stroke Res. 2013;4:368–74.
Sato M, Tani E, Fujikawa H, Kaibuchi K. Involvement of rho-kinase-mediated phosphorylation of myosin light chain in enhancement of cerebral vasospasm. Circ Res. 2000;87:195–200.
Wang CJ, Lee PY, Wu BN, Wu SC, Loh JK, Tsai HP, Chung CL, Kassell NF, Kwan AL. Alteration of basilar artery rho-kinase and soluble guanylyl cyclase protein expression in a rat model of cerebral vasospasm following subarachnoid Hemorrhage. Biomed Res Int. 2014;2014:531508.
Morikage N, Kishi H, Sato M, Guo F, Shirao S, Yano T, Soma M, Hamano K, Esato K, Kobayashi S. Cholesterol primes vascular smooth muscle to induce Ca2 sensitization mediated by a sphingosylphosphorylcholine-rho-kinase pathway: possible role for membrane raft. Circ Res. 2006;99:299–306.
Burridge K, Guilluy C. Focal adhesions, stress fibers and mechanical tension. Exp Cell Res. 2016;343:14–20.
Livne A, Geiger B. The inner workings of stress fibers - from contractile machinery to focal adhesions and back. J Cell Sci. 2016;129:1293–304.
Martino F, Perestrelo AR, Vinarsky V, Pagliari S, Forte G. Cellular Mechanotransduction: from tension to function. Front Physiol. 2018;9:824.
Tang DD, Gunst SJ. Depletion of focal adhesion kinase by antisense depresses contractile activation of smooth muscle. Am J Physiol Cell Physiol. 2001;280:C874–883.
Deakin NO, Turner CE. Paxillin comes of age. J Cell Sci. 2008;121:2435–44.
Hu YL, Lu S, Szeto KW, Sun J, Wang Y, Lasheras JC, Chien S. FAK and paxillin dynamics at focal adhesions in the protrusions of migrating cells. Sci Rep. 2014;4:6024.
Martin-Garcia R, Arribas V, Coll PM, Pinar M, Viana RA, Rincon SA, Correa-Bordes J, Ribas JC, Perez P. Paxillin-mediated recruitment of Calcineurin to the Contractile Ring is required for the correct progression of Cytokinesis in Fission yeast. Cell Rep. 2018;25:772–783e774.
Ripamonti M, Liaudet N, Azizi L, Bouvard D, Hytonen VP, Wehrle-Haller B. Structural and functional analysis of LIM domain-dependent recruitment of paxillin to alphavbeta3 integrin-positive focal adhesions. Commun Biol. 2021;4:380.
Tang DD, Wu MF, Opazo Saez AM, Gunst SJ. The focal adhesion protein paxillin regulates contraction in canine tracheal smooth muscle. J Physiol. 2002;542:501–13.
Hirth S, Buhler A, Buhrdel JB, Rudeck S, Dahme T, Rottbauer W, Just S. Paxillin and focal adhesion kinase (FAK) regulate Cardiac Contractility in the zebrafish heart. PLoS ONE. 2016;11:e0150323.
Zhang Y, Kishi H, Morita T, Kobayashi S. Paxillin controls actin stress fiber formation and migration of vascular smooth muscle cells by directly binding to the active Fyn. FASEB J. 2021;35:e22012.
Zhang Y, Zhang M, Lyu B, Kishi H, Kobayashi S. Omega-3 and omega-6 DPA equally inhibit the sphingosylphosphorylcholine-induced ca(2+)-sensitization of vascular smooth muscle contraction via inhibiting rho-kinase activation and translocation. Sci Rep. 2017;7:36368.
Steffensen LB, Stubbe J, Overgaard M, Larsen JH. Tamoxifen-independent cre-activity in SMMHC-CreER (T2) mice. Atheroscler Plus. 2022;48:8–11.
Reinert RB, Kantz J, Misfeldt AA, Poffenberger G, Gannon M, Brissova M, Powers AC. Tamoxifen-Induced cre-loxp recombination is prolonged in pancreatic islets of adult mice. PLoS ONE. 2012;7:e33529.
Xu J, Kurup P, Foscue E, Lombroso PJ. Striatal-enriched protein tyrosine phosphatase regulates the PTPalpha/Fyn signaling pathway. J Neurochem. 2015;134:629–41.
Carvalho K, Lemiere J, Faqir F, Manzi J, Blanchoin L, Plastino J, Betz T, Sykes C. Actin polymerization or myosin contraction: two ways to build up cortical tension for symmetry breaking. Philos Trans R Soc Lond B Biol Sci. 2013;368:20130005.
Gerthoffer WT, Gunst SJ. Invited review: focal adhesion and small heat shock proteins in the regulation of actin remodeling and contractility in smooth muscle. J Appl Physiol (1985). 2001;91:963–72.
Gunst SJ, Zhang W. Actin cytoskeletal dynamics in smooth muscle: a new paradigm for the regulation of smooth muscle contraction. Am J Physiol Cell Physiol. 2008;295:C576–587.
Sutherland C, Walsh MP. Myosin regulatory light chain diphosphorylation slows relaxation of arterial smooth muscle. J Biol Chem. 2012;287:24064–76.
Sweeney HL. Regulation and tuning of smooth muscle myosin. Am J Respir Crit Care Med. 1998;158:95–9.
Amano M, Ito M, Kimura K, Fukata Y, Chihara K, Nakano T, Matsuura Y, Kaibuchi K. Phosphorylation and activation of myosin by rho-associated kinase (Rho-kinase). J Biol Chem. 1996;271:20246–9.
Kureishi Y, Kobayashi S, Amano M, Kimura K, Kanaide H, Nakano T, Kaibuchi K, Ito M. Rho-associated kinase directly induces smooth muscle contraction through myosin light chain phosphorylation. J Biol Chem. 1997;272:12257–60.
Katoh K, Kano Y, Ookawara S. Rho-kinase dependent organization of stress fibers and focal adhesions in cultured fibroblasts. Genes Cells. 2007;12:623–38.
Amano M, Chihara K, Kimura K, Fukata Y, Nakamura N, Matsuura Y, Kaibuchi K. Formation of actin stress fibers and focal adhesions enhanced by rho-kinase. Science. 1997;275:1308–11.
Opazo Saez A, Zhang W, Wu Y, Turner CE, Tang DD, Gunst SJ. Tension development during contractile stimulation of smooth muscle requires recruitment of paxillin and vinculin to the membrane. Am J Physiol Cell Physiol. 2004;286:C433–447.
Moik D, Bottcher A, Makhina T, Grashoff C, Bulus N, Zent R, Fassler R. Mutations in the paxillin-binding site of integrin-linked kinase (ILK) destabilize the pseudokinase domain and cause embryonic lethality in mice. J Biol Chem. 2013;288:18863–71.
Miano JM, Cserjesi P, Ligon KL, Periasamy M, Olson EN. Smooth muscle myosin heavy chain exclusively marks the smooth muscle lineage during mouse embryogenesis. Circ Res. 1994;75:803–12.
Feil R, Wagner J, Metzger D, Chambon P. Regulation of cre recombinase activity by mutated estrogen receptor ligand-binding domains. Biochem Biophys Res Commun. 1997;237:752–7.
Liliom K, Sun G, Bunemann M, Virag T, Nusser N, Baker DL, Wang DA, Fabian MJ, Brandts B, Bender K, et al. Sphingosylphosphocholine is a naturally occurring lipid mediator in blood plasma: a possible role in regulating cardiac function via sphingolipid receptors. Biochem J. 2001;355:189–97.
Sward K, Mita M, Wilson DP, Deng JT, Susnjar M, Walsh MP. The role of RhoA and rho-associated kinase in vascular smooth muscle contraction. Curr Hypertens Rep. 2003;5:66–72.
Acknowledgements
We would like to express our gratitude for the technical support of Yamaguchi University Center for Gene Research and Institute of Life Science and Medicine of Yamaguchi University in carrying out our research. We would like to thank Hiroko Kishi and Dan Cui for their generous supports for this study.
Funding
This work was supported by Grants-in-Aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (JSPS KAKENHI Grant Numbers 21K06782 to Y. Zhang).
Author information
Authors and Affiliations
Contributions
Y.Z. and S.K. designed experiments. Y.Z. and N.L. performed experiments, summarized data, and wrote a manuscript. S.K. drafted the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All studies involving animals were approved by the Animal Policy and Welfare Committee of Yamaguchi University.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Additional file 1: Figure S1.
Targeting vector design of smooth muscle-specific paxillin knock out mice.
Additional file 2: Figure S2.
Establishment of paxillin SMMHC-CreERT2 transgenic mice.
Additional file 3: Figure S3.
The expression of paxillin in heart muscle, skeletal muscle, and vascular smooth muscle from paxillin SMKO mice.
Additional file 4: Figure S4.
30 μM SPC-induced contraction in control and paxillin SMKO mice.
Additional file 5: Video S1. Time-lapse video of the SPC-induced contraction in control shRNA human CASMCs.
Additional file 6: Video S2. Time-lapse video of the SPC-induced contraction in paxillin shRNA human CASMCs.
Additional file 7.
Supplementary uncropped images of western blot.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, Y., Li, N. & Kobayashi, S. Paxillin participates in the sphingosylphosphorylcholine-induced abnormal contraction of vascular smooth muscle by regulating Rho-kinase activation. Cell Commun Signal 22, 58 (2024). https://doi.org/10.1186/s12964-023-01404-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-023-01404-w