Abstract
Background
Investigating the species distribution and their role in malaria transmission is important as it varies from place to place and is highly needed to design interventions appropriate to the site. The current study aimed to investigate the Anopheles mosquito species distribution and their infection rate in southwestern Ethiopia.
Methods
The study was conducted in 14 malaria-endemic kebeles (the smallest administrative unit), which were situated in eight different malaria-endemic districts and four zones in southwestern Ethiopia. Ten per cent of households in each village were visited to collect adult mosquitoes using Centers for Disease Control and Prevention (CDC) light traps. The larval and pupal collection was done from breeding sites within the villages, and reared to adults. Female mosquitoes were morphologically identified. The head and thorax of adult Anopheles mosquitoes were tested for circumsporozoite proteins (CSPs) using ELISA. At the same time, legs, wings, and abdomen were used to identify sibling species using PCR targeting the rDNA intergenic spacers region for species typing of the Anopheles funestus group and the internal transcribed spacer 2 region genes for Anopheles gambiae complex.
Results
A total of 1445 Anopheles mosquitoes comprising eight species were collected. Of 813 An. gambiae complex tested by PCR, 785 (97%) were Anopheles arabiensis, and the remaining 28 (3%) were not amplified. There were 133 An. funestus group captured and tested to identify the species, of which 117 (88%) were positive for Anopheles parensis, and 15 (11%) were not amplified. A single specimen (1%) showed a band with a different base pair length from the known An. funestus group species. Sequencing revealed this was Anopheles sergentii. Among 1399 Anopheles tested for CSPs by ELISA, 5 (0.4%) An. arabiensis were positive for Plasmodium falciparum and a single (0.07%) was positive for Plasmodium vivax.
Conclusions
Anopheles arabiensis continues to play the principal role in malaria transmission despite implementing indoor-based interventions for decades. Sequencing results suggest that An. sergentii was amplified by the An. funestus group primer, producing PCR amplicon size of different length. Therefore, relying solely on amplifying a specific gene of interest in grouping species could be misleading, as different species may share the same gene.
Similar content being viewed by others
Background
Female Anopheles mosquitoes can carry Plasmodium protozoan parasites, some of which transmit to humans. The majority of malaria caused by Plasmodium falciparum and Plasmodium vivax occurs in Africa. Between 2010 and 2017, there was a noticeable decline in malaria deaths and cases [1]. However, the decline has slowed in recent years, and in the most recent years, there has been an increase in the number of cases [2]. The presence of the most effective vectors, such as Anopheles gambiae, Anopheles arabiensis, and Anopheles funestus is the main reason for the high rate of malaria transmission in Africa [3]. Other locally important malaria vectors include Anopheles melas, Anopheles merus, Anopheles moucheti, and Anopheles nili [3].
In Ethiopia, P. falciparum and P. vivax are the predominant malaria parasites. The country contributed 9% of the P. vivax malaria cases globally in 2017 [1]. Although there are many Anopheles species in Ethiopia, only a few are known to transmit malaria. Anopheles arabiensis, one of the An. gambiae complexes are primarily responsible for transmitting malaria in the country [4, 5]. In the 1930s, Italian malariologists identified the “An. gambiae” complex as the main vector of malaria in Ethiopia [6, 7]. Ethiopian Malaria Eradication Service, in collaboration with Jolivet, identified 31 Anopheles species in 1963 and documented the predominance of “An. gambiae” (probably An. arabiensis) [7]. Anopheles gambiae remained the most common and prominent malaria vector even after extensive malaria vector control interventions were adopted in the 1960 and 1970s [7, 8]. Several recent studies have demonstrated similar contributions to malaria transmission and the dominance of the same species despite decades of malaria control efforts [4, 8,9,10,11].
Anopheles arabiensis shows flexible resting and feeding behaviour, as it rests and bites indoors and outdoors and feeds on humans and animals based on the hosts’ availability [12]. This behavioural plasticity complicates malaria control and elimination programmes [13]. Despite the plasticity of feeding and resting behaviour, insecticide-treated nets (ITN) and indoor residue spraying (IRS) have been widely used to control this species [14]. Anopheles mosquito species diversity and behaviour will likely alter due to these interventions [15,16,17]. For instance, a shift towards outdoor biting and resting vectors could result from interventions that target indoor feeding and resting vectors [13, 15]. Behavioural resistance, such as avoiding insecticides on bed nets and walls, may reduce the efficacy of IRS and ITNs [16]. Several reports have documented the outdoor and early-night human biting behaviour of An. arabiensis in Ethiopia [10, 17]. Integrated interventions may be necessary to target changes in vector species and their behaviour.
Several studies have been conducted to describe the species composition, feeding and resting habits, and infection rates of malaria mosquitos in various malaria-endemic areas [4, 10, 11]. Nevertheless, it has yet to be explored if these malaria control interventions affected the diversity of malaria vector species, their geographic distribution, or infection rates. Therefore, studying the species distribution and their role in malaria transmission is critical for making evidence-based decisions. The current study assessed the composition of the Anopheles mosquito species and their relative contribution to malaria transmission in southern Ethiopia.
Methods
Study area description
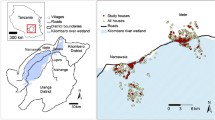
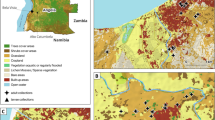
The study was conducted in four zones of Southern Nation Nationalities and Peoples Regional States (SNNPRs), southwestern Ethiopia (Fig. 1), namely Gamo-Gofa, Wolaita, South Omo and Hadiya. Hot and semi-arid areas comprise 57% of the SNNPRs, tropical sub-humid areas contribute 34%, and tropical humid areas comprise 9%. The region has an elevation of 376 to 4207 m above sea level. Annual rainfall and temperatures range between 500 and 2200 mm and 15 °C and 30 °C, respectively. Despite malaria being a health problem in the region, the incidence of malaria varies from place to place [18].
Study design and sampling technique
Multistage sampling was used to select districts, kebeles, and households. According to the zonal health department report, two high-malaria-prevalence districts in each zone were included, except the Gamo-Gofa zone, where three districts were included. Gamo-Gofa zones were initially together but became two independent zones during the study period. Of the three districts included in the Gamo-Gofa zone, two were located in the present Gamo zone and one was located within the present Gofa zone. Finally, two malarious kebeles were purposefully selected in each district based on the malaria incidence. During the study, one district in the South Omo zone was excluded because of a security concern.
Using a random sampling technique, 10% of the households in each kebele were selected for adult mosquito collection and larval surveillance close to the households. Systematic sampling was carried out using the lists of households in each kebele health post. The first household was selected from one end of the kebele, and every tenth household was included. Few houses were replaced by nearby houses when the selected household heads were absent or did not volunteer to participate in the study.
Sampling adults and larvae of Anopheles mosquitoes
Host-seeking Anopheles mosquitoes were collected indoors using CDC light traps instead of human landing catches. This was done to reduce bias due to collector skills and limit the risk of infection for collectors. Traps were installed about 1.5 m above the floor near the foot end of the bed where a person slept under an untreated bed net. The traps were set at 6:00 pm and collected at 6:00 am the following morning [19]. Except for the sibling species, collected female Anopheles were morphologically identified to the species level using a taxonomic key [20] and preserved in silica gel at room temperature.
A 300 ml standard dipper was used to collect larvae and pupae once during the study period. Pupae and larvae were moved to a temporary insectary set-up at each location. The immature Anopheles mosquitoes were collected and kept in a tray until they developed into pupae. The pupae were then transferred to cages until they emerged as adults. All reared female Anopheles mosquitoes were killed by chloroform, morphologically identified [20], and stored with silica gel at room temperature until further processing.
Molecular identification of sibling species
The species of An. gambiae complex and An. funestus group were identified through PCR at Arba Minch University’s Medical Entomology laboratory. According to the manufacturer’s instructions, the legs, abdomen, and wings were used for DNA extraction using the NucleoSpin Tissue kit (Macherey Nagel, Germany). For An. gambiae complex, the rDNA intergenic spacers (IGS) were targeted as described by Scott et al. [21], while for An. funestus group, the internal transcribed spacer 2 (ITS-2) region on the rDNA was targeted using PCR as described by Koekemoer et al. [22].
The PCRs were run on the Biometra T professional gradient Thermocycler (Biometra, the Netherlands). The PCR products were visualised on a 2% agarose gel (Sisco Research Laboratories, India) using a 100 bp ladder (Promega, The Netherlands), and species identification was done based on the PCR amplicon height described by Scott et al. [21] and Koekemoer et al. [22].
Amplicon sequencing of cytochrome c oxidase I (COI) was performed for the specimen that displayed varying heights in PCR amplicons. The initial amplification was performed for the ITS-2 gene, followed by COI amplification for further confirmation. The COI sequences were aligned, a consensus sequence was generated, and primers were trimmed off using Geneious software [23]. These were used to compare with reference strains that were uploaded in GenBank using BLAST.
Circum-sporozoite protein test
The head and thorax of female adult Anopheles mosquito were used to test for CSPs of P. falciparum, P. vivax- 210 and P. vivax-247 using ELISA, as described by Beier et al. [24]. The sample was prepared by grinding buffer PBS + IGPAL (Fisher Bioreagents, USA and SIGMA-ALDRICH, USA). Fifty microlitres (50 µl) of 0.20 µg/50 µl PBS Pf and 0.10 µg/50 µl PBS (Pv210 and Pv247 each) monoclonal antibodies (mAb) (KPL, USA) were coated on a plate and incubated for half an hour. Confirmation of positive results was obtained by retesting the samples. The remaining steps were carried out as specified in Beier et al. [24].
Data analysis
The Quantum Geographical Information System (qGIS, www.qgis.org openAFRICA) was used to develop the map. The circumsporozoite protein rate was calculated as the proportion of mosquitoes that tested positive for CSPs in relation to the total number screened. To create a phylogenetic tree, mega molecular evolutionary analysis software [23, 25] was used. Consensus sequences were generated for each specimen and then compared to GenBank reference sequences using BLAST. For multiple sequence alignment, we used the Clustal W tool and trimmed the primers to ensure that all sequences had equal length [23, 25]. Pairwise comparisons were made using COI sequences retrieved from GenBank. The inter-species distances were accurately calculated.
Results
Anopheles species composition
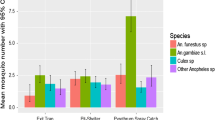
A total of 1445 Anopheles mosquitoes were collected. Based on the morphological identification, seven species were documented in the region. Overall, 1227 (85.0%) mosquitoes belonged to the An. gambiae complex (46 reared from larvae and 1181 adults collected) and 133 (9.2%) were An. funestus group. The remaining Anopheles mosquitoes were Anopheles pharoensis, Anopheles pretoriensis, Anopheles demeilloni, Anopheles kingi and Anopheles tenebrosus (Table 1). From the larval collection, only 46 An. gambiae complex were identified in three study villages.
Spatial distribution of Anopheles mosquitoes
Anopheles gambiae complex was the most common and widely distributed species in the region, occurring in each village except in Mirab Badawacho of Hadiya zone (Table 2). Anopheles pharoensis was observed in 50% of study sites. Anopheles funestus group was collected only from Humabo of Wolaita zone. Anopheles demeilloni, An. kingi, and An. tenebrosus were rare species.
Sibling species of An. gambiae complex and An. funestus group
Out of the 767 An. gambiae sensu lato (s.l.) adults tested, 97% (743) were successfully amplified for An. arabiensis, and of the 46 An. gambiae s.l. specimens that emerged from larvae, 91% (42) were amplified for An. arabiensis. The remaining specimens (4 from larval collection and 24 from adult collection) were not amplified.
Of the 133 An. funestus s.l. tested, 87.9% (117/133) were amplified as An. parensis, and 15 (11%) were not amplified. A single specimen (0.7%) collected in the Humbo district of the Wolaita zone was amplified using primers for the An. funestus group, but its PCR amplicon height was not in accordance with the existing group members. The standard PCR amplicon size for An. funestus s.l. was 505 bp for An. funestus sensu stricto (s.s.), 587 bp for An. vaneedeni, 411 pb for Anopheles rivulorum, 313 bp for An. rivulorum-like, 252 bp for An. parensis, and 146 bp for Anopheles leesoni. The species of interest had a PCR amplicon size of 200 bp. After sequencing, the specimen was identified as Anopheles sergentii (Fig. 2).
Phylogenetic tree of Anopheles funestus group and two other species collected the study sites. The variance of the strain An. sergentii species identified from the Humbo district of the Wolaita zone of southwest Ethiopia and other An. sergentii strains KT160247 from Egypt: Fayoum, and KT160246 from Egypt: Aswan. Based on a pairwise comparison of the COI sequence of An. funestus and An. sergentii retrieved from GenBank, it can be inferred that the current species belong to the An. sergentii group, rather than the An. funestus group. The comparison revealed that the percentage relation between the COI sequence of An. funestus and the species of interest was 82%, while for An. sergentii, it was more than 95%
Plasmodium sporozoite infection rate of Anopheles species
Of the 1399 Anopheles mosquitoes tested for Plasmodium CSPs, six An. arabiensis were positive for CSPs with an overall prevalence of 0.43% (95% Confidence Interval (CI): 0.16–0.93). Of the six positive An. arabiensis, five were positive for P. falciparum CSP (0.36%; 95% CI 0.11–0.83), and 1 was positive for P. vivax (0.07%; 95% CI 0.002–0.40)—none of the other mosquito species tested positive for CSPs.
Discussion
Eight Anopheles species, namely An. arabiensis, An. parensis, An. pharoensis, An. pretoriensis, An. demeilloni, An. kingi, An. tenebrosus and An. sergentii, were identified in southwest Ethiopia. Anopheles arabiensis was the most common and primary malaria vector in the region. There were no documented cases of Anopheles stephensi in the region.
Anopheles arabiensis was the region’s most widely distributed and predominantly vectoring P. falciparum malaria. Many years ago, O’Connor [26] documented 34 Anopheles species in Ethiopia. Among these Anopheles species recorded at that time, An. gambiae (presumably An. arabiensis) was the dominant and primary malaria vector in wide geographic areas. Several studies have also shown that An. arabiensis contribution as a primary vector in different parts of the country [5, 10, 11]. Despite implementing a wide range of vector control interventions for decades, the species remained the dominant malaria vector in the region. For example, the P. falciparum CSP infection rate of An. arabiensis was 0.36%, comparable to the CSP rate of 0.77% reported from Gambella in 1994 [27]. Krafsur reported a somewhat higher CSP rate (1.87%) of the same species from Gambella in 1977, although the method was a microscopic dissection of salivary glands [6]. Using the HLC technique, the P. falciparum CSP rate of An. arabiensis was 0.5% in Sille, southern Ethiopia [28]. In the Central Rift Valley of Ethiopia, a CSP rate of An. arabiensis was 1.18% from the CDC light traps [29]. The P. falciparum CSP rate of An. arabiensis from CDC light traps, as used in the current investigation, was 0.3% in the south-central and southwest Ethiopia [4, 30, 31]. This implies that An. arabiensis does not respond well to the current malaria vector control interventions. To effectively eliminate malaria in the country, it is necessary to use additional tools to control the species [32, 33]. For example, interventions such as larval source management can be used to target the aquatic stages of malaria and other vectors [34]. Toxic bait traps could be used to control mosquitoes seeking hosts outdoor [32], while animal-based interventions can address the problem related to zoophagic mosquitoes [35].
A challenge with An. arabiensis is its plasticity in feeding and resting habits. For example, the species’ anthropophagic, zoophagic, and outdoor and indoor resting behaviour has been documented in Ethiopia [12, 36, 37]. These adaptive characteristics allow the species to avoid interventions mainly targeting those that rest and feed indoors.
The majority of An. funestus group was identified as An. parensis. Similarly, An. funestus group from Jimma was analysed and verified as An. parensis using the PCR technique [38], suggesting that An. parensis is the predominant species of An. funestus group in the region. During the 1930 and 1960s, the An. funestus group was prevalent and widespread in Ethiopia [39]. However, none of the 339 An. funestus group mosquitoes from Zwai and Awasa tested positive for sporozoite [39]. In contrast, Krafsur reported a sporozoite rate of 1.23% in Gambella using PSCs in 1977 [8]. However, identifying the specific species within the group was challenging due to the limited identification tools available. It’s worth noting that An. funestus s.s. tends to bite and rest indoors, making it a potential candidate for DDT IRS, as seen in East African countries [39]. Following the widespread use of DDT IRS, the outdoor biting and resting species An. rivulorum substituted the primary vector An. funestus s.s. in East Africa [40]. This species was not identified at the current study sites. During the 1960s, the IRS projects carried out in southern Africa resulted in the elimination of the same species [41]. This suggests that indoor-based intervention methods could be effective in reducing malaria vectors if the vectors are completely resting and biting indoors. However, in the presence of opportunistic feeders like An. arabiensis, the current interventions may need to be revised to achieve the intended objective of malaria control and elimination.
In this study, after a detailed analysis of the sequence, a single Anopheles mosquito specimen was confirmed to be An. sergentii. Two species of An. sergentii have been documented in Africa: An. sergentii sergentii, found in several countries, and An. sergentii macmahoni, mainly found in East Africa, including Ethiopia [42]. Adult An. sergentii macmahoni are rarely found indoors in human dwellings and typically feed on animals [3]. Anopheles sergentii sergentii is an important malaria vector in the Saharan belt, spanning from northern Africa to the Middle East [3]. The species is morphologically related to An. parensis, An. funestus, An. demeilloni and An. cameroni [43]. It was also grouped under An. demeilloni in the report of “Phylogeny and Classification of Anopheles” [44]. It is important to note that although the primers of the An. funestus group amplify An. sergentii, it is insufficient evidence to classify it as a member of the An. funestus group. However, the results from sequencing suggest that the two species share the gene of interest used to identify species in the An. funestus group. Therefore, it is crucial to provide detailed information on the degree of species divergence and percentage relationship by comparing the sequence of the species of interest with the sequence of the species obtained from GenBank [45] in addition to primer amplification. By comparing COI sequences from GenBank, it is evident that the current species belongs to An. sergentii and does not belong to An. funestus group.
The cross-sectional method used in this study has limitations because it cannot account for monthly and seasonal variations in species composition, which could lead to an incomplete list of species. Additionally, no confirmatory molecular test or boiling was performed on CSP ELISA positive samples, which could have led to false positives. However, the positive samples were repeated using the ELISA assay.
Conclusions
Despite several decades of efforts to deploy indoor-based interventions, An. arabiensis continues to play a dominant role in transmission. Based on the sequencing data, the Anopheles species amplified for An. funestus group primers were identified as An. sergentii. It is important to note that there were no documented cases of An. stephensi in the study sites.
Availability of data and materials
The article contains the data that was utilised to draw the conclusion.
References
WHO. World malaria report 2019. Geneva: World Health Organization; 2019.
WHO. World malaria report: 20 years of global progress and challenges. Geneva: World Health Organization; 2020.
Sinka ME, Bangs MJ, Manguin S, Coetzee M, Mbogo CM, Hemingway J, et al. The dominant Anopheles vectors of human malaria in Africa, Europe and the Middle East: occurrence data, distribution maps and bionomic précis. Parasit Vectors. 2010;3:117.
Massebo F, Balkew M, Gebre-Michael T, Lindtjørn B. Entomologic inoculation rates of Anopheles arabiensis in southwestern Ethiopia. Am J Trop Med Hyg. 2013;89:466–73.
Abraham M, Massebo F, Lindtjørn B. High entomological inoculation rate of malaria vectors in area of high coverage of interventions in southwest Ethiopia: implication for residual malaria transmission. Parasite Epidemiol Control. 2017;2:61–9.
National Malaria Control Team, Ethiopian Public Health Institute, World Health Organization, Addis Ababa University and the INFORM Project. An epidemiological profile of malaria in Ethiopia. A report prepared for the Federal. Ministry of Health, Ethiopia, the Roll Back Malaria Partnership and the Department for International Development, UK. 2013.
Schaller KF. Medizinische Landerkunde Athiopien-Ethiopia. Vol 3 of Geomedical Monograph Series. 1972. p. 159.
Krafsur ES. The bionomics and relative prevalence of Anopheles species with respect to the transmission of Plasmodium to man in western Ethiopia. J Med Entomol. 1977;14:180–94.
Taffese HS, Hemming-Schroeder E, Koepfli C, Tesfaye G, Lee MC, Kazura J, et al. Malaria epidemiology and interventions in Ethiopia from 2001 to 2016. Infect Dis Poverty. 2018;7:103.
Kenea O, Balkew M, Tekie H, Gebre-Michael T, Deressa W, Loha E, et al. Human-biting activities of Anopheles species in south-central Ethiopia. Parasit Vectors. 2016;9:527.
Esayas E, Woyessa A, Massebo F. Malaria infection clustered into small residential areas in lowlands of southern Ethiopia. Parasite Epidemiol Control. 2020;10: e00149.
Massebo F, Balkew M, Gebre-Michael T, Lindtjørn B. Zoophagic behaviour of anopheline mosquitoes in southwest Ethiopia: opportunity for malaria vector control. Parasit Vectors. 2015;8:645.
Kreppel KS, Viana M, Main BJ, Johnson PCD, Govella NJ, Lee Y, et al. Emergence of behavioural avoidance strategies of malaria vectors in areas of high LLIN coverage in Tanzania. Sci Rep. 2020;10:14527.
Bhatt S, Weiss DJ, Cameron E, Bisanzio D, Mappin B, Dalrymple U, et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature. 2015;526:207–11.
Mwangangi JM, Mbogo CM, Orindi BO, Muturi EJ, Midega JT, Nzovu J, et al. Shifts in malaria vector species composition and transmission dynamics along the Kenyan coast over the past 20 years. Malar J. 2013;12:13.
Killeen GF. Characterizing, controlling and eliminating residual malaria transmission. Malar J. 2014;13:330.
Tomas T, Eligo N, Tamiru G, Massebo F. Outdoor and early hour human biting activities of malaria mosquitoes and the suitability of clay pot for outdoor resting mosquito collection in malaria endemic villages of southern Rift valley, Ethiopia. Parasite Epidemiol Control. 2022;19: e00278.
Loha E, Lindtjørn B. Model variations in predicting incidence of Plasmodium falciparum malaria using 1998–2007 morbidity and meteorological data from south Ethiopia. Malar J. 2010;9:166.
WHO. Manual on practical entomology in malaria: methods and techniques. Geneva: World Health Organization. Division of Malaria and Other Parasitic Diseases; 1975. p. 191.
Coetzee M. Key to the females of Afrotropical Anopheles mosquitoes (Diptera: Culicidae). Malar J. 2020;19:70.
Scott JA, Brogdon WG, Collins FH. Identification of single specimens of the Anopheles gambiae complex by the polymerase chain reaction. Am J Trop Med Hyg. 1993;49:520–9.
Koekemoer LL, Kamau L, Hunt RH, Coetzee M. A cocktail polymerase chain reaction assay to identify members of the Anopheles funestus (Diptera: Culicidae) group. Am J Trop Med Hyg. 2002;66:804–11.
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28:2731–9.
Beier JC, Perkins PV, Wirtz RA, Whitmire RE, Mugambi M, Hockmeyer WT. Field evaluation of an enzyme-linked immunosorbent assay (ELISA) for Plasmodium falciparum sporozoite detection in anopheline mosquitoes from Kenya. Am J Trop Med Hyg. 1987;36:459–68.
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997;25:4876–82.
O’Connor CT. The distribution of anopheline mosquitoes in Ethiopia. Mosq News. 1967;27:42–54.
Nigatu W, Petros B, Lulu M, Adugna N, Wirtz R. Species composition, feeding and resting behaviour of the common anthropophilic anopheline mosquitoes in relation to malaria transmission in Gambella, south west Ethiopia. Int J Trop Insect Sci. 1994;15:371–7.
Taye A, Hadis M, Adugna N, Tilahun D, Wirtz RA. Biting behavior and Plasmodium infection rates of Anopheles arabiensis from Sille, Ethiopia. Acta Trop. 2006;97:50–4.
Kibret S, Alemu Y, Boelee E, Tekie H, Alemu D, Petros B. The impact of a smallscale irrigation scheme on malaria transmission in Ziway area, Central Ethiopia. Trop Med Int Health. 2010;15:41–50.
Animut A, Balkew M, Gebre-Michael T, Lindtjørn B. Blood meal sources and entomological inoculation rates of anophelines along a highland altitudinal transect in south-central Ethiopia. Malar J. 2013;12: 76.
Assa A, Eligo N, Massebo F. Anopheles mosquito diversity, entomological indicators of malaria transmission and challenges of morphological identification in southwestern Ethiopia. Trop Med Health. 2023;51:38.
Sougoufara S, Ottih EC, Tripet F. The need for new vector control approaches targeting outdoor biting Anopheline malaria vector communities. Parasit Vectors. 2020;13:295.
Gatton ML, Chitnis N, Churcher T, Donnelly MJ, Ghani AC, Godfray HCJ, et al. The importance of mosquito behavioural adaptations to malaria control in Africa. Evolution. 2013;67:1218–30.
Gu W, Regens JL, Beier JC, Novak RJ. Source reduction of mosquito larval habitats has unexpected consequences on malaria transmission. Proc Natl Acad Sci USA. 2006;103:17560–3.
Franco AO, Gomes MGM, Rowland M, Coleman PG, Davies CR. Controlling malaria using livestock-based interventions: a one health approach. PLoS ONE. 2014;9: e101699.
Habtewold T, Walker AR, Curtis CF, Osir EO, Thapa N. The feeding behaviour and Plasmodium infection of Anopheles mosquitoes in southern Ethiopia in relation to use of insecticide-treated livestock for malaria control. Trans R Soc Trop Med Hyg. 2001;95:584–6.
Tirados I, Costantini C, Gibson G, Torr SJ. Blood-feeding behaviour of the malarial mosquito Anopheles arabiensis: implications for vector control. Med Vet Entomol. 2006;20:425–37.
Weeto MM, Koekemoer LL, Kamau L, Hunt RH, Coetzee M. Evaluation of a species-specific PCR assay for the Anopheles funestus group from eleven African countries and Madagascar. Trans R Soc Trop Med Hyg. 2004;98:142–7.
Rishikesh N. Observations on anopheline vectors of malaria in an upland valley in Ethiopia. Geneva: World Health Organization; 1966.
Gillies MT, Smith A. The effect of a residual house-spraying campaign in East Africa on species balance in the Anopheles funestus group. The replacement of An. funestus Giles by A. rivulorum Leeson. Bull Entomol Res. 1959;51:243–52.
Mabaso MLH, Sharp B, Lengeler C. Historical review of malarial control in southern African with emphasis on the use of indoor residual house-spraying. Trop Med Int Health. 2004;9:846–56.
Kyalo D, Amratia P, Mundia CW, Mbogo CM, Coetzee M, Snow RW. A geo-coded inventory of anophelines in the Afrotropical Region south of the Sahara: 1898–2016. Wellcome Open Res. 2017;2:57.
Gillies MT, Coetzee M. A supplement to the Anophelinae of Africa South of the Sahara. S Afr Inst Med Res. 1987;55:1–143.
Harbach RE. The phylogeny and classification of Anopheles. In: Manguin S, editor. Anopheles mosquitoes—new insights into malaria vectors. London: InTechOpen; 2013.
Dahan-Moss Y, Hendershot A, Dhoogra M, Julius H, Zawada J, Kaiser M, et al. Member species of the Anopheles gambiae complex can be misidentified as Anopheles leesoni. Malar J. 2020;19:89.
Acknowledgements
We thank the health departments of the Gamo, Gofa, Wolayita, South Omo, and Hadiya zones, as well as the districts and community members, for their assistance. We thank Natalie Van Houtte and Professor Herwig Leirs of Antwerp University for granting us access to the molecular laboratory.
Funding
The Norwegian Programme for Capacity Development in Higher Education and Research for Development (project code: ETH-13/0025) funded this study. The funding agency was not engaged in any of the research activities.
Author information
Authors and Affiliations
Contributions
NE, TE and FM conceived and designed the study; NE conducted the fieldwork; NE, GT, FM and MP conducted data analysis; FM, MP and BL wrote the manuscript, FM, MP and BL revised the manuscript. The manuscript was reviewed and approved by all authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was reviewed and approved by Arba Minch University Institutional Review Board (CMHS/12033592/111). Verbal and written consents were obtained from all household heads before mosquito collection.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Eligo, N., Wegayehu, T., Pareyn, M. et al. Anopheles arabiensis continues to be the primary vector of Plasmodium falciparum after decades of malaria control in southwestern Ethiopia. Malar J 23, 14 (2024). https://doi.org/10.1186/s12936-024-04840-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12936-024-04840-2