Abstract
Background
Malaria has high morbidity and mortality rates in some parts of tropical and subtropical countries. Besides respiratory and metabolic function, lung plays a role in immune system. γδT cells have multiple functions in producing cytokines and chemokines, regulating the immune response by interacting with other cells. It remains unclear about the role of γδT cells in the lung of mice infected by malaria parasites.
Methods
Flow cytometry (FCM) was used to evaluate the frequency of γδT cells and the effects of γδT cells on the phenotype and function of B and T cells in Plasmodium yoelii-infected wild-type (WT) or γδTCR knockout (γδT KO) mice. Haematoxylin-eosin (HE) staining was used to observe the pathological changes in the lungs.
Results
The percentage and absolute number of γδT cells in the lung increased after Plasmodium infection (p < 0.01). More γδT cells were expressing CD80, CD11b, or PD-1 post-infection (p < 0.05), while less γδT cells were expressing CD34, CD62L, and CD127 post-infection (p < 0.05). The percentages of IL-4+, IL-5+, IL-6+, IL-21+, IL-1α+, and IL-17+ γδT cells were increased (p < 0.05), but the percentage of IFN-γ-expressing γδT cells decreased (p < 0.05) post-infection. The pathological changes in the lungs of the infected γδT KO mice were not obvious compared with the infected WT mice. The proportion of CD3+ cells and absolute numbers of CD3+ cells, CD3+ CD4+ cells, CD3+ CD8+ cells decreased in γδT KO infected mice (p < 0.05). γδT KO infected mice exhibited no significant difference in the surface molecular expression of T cells compared with the WT infected mice (p > 0.05). While, the percentage of IFN-γ-expressing CD3+ and CD3+ CD8+ cells increased in γδT KO infected mice (p < 0.05). There was no significant difference in the absolute numbers of the total, CD69+, ICOS+, and CD80+ B cells between the WT infected and γδT KO infected mice (p > 0.05).
Conclusions
The content, phenotype, and function of γδT cells in the lung of C57BL/6 mice were changed after Plasmodium infection. γδT cells contribute to T cell immune response in the progress of Plasmodium infection.
Similar content being viewed by others
Background
Malaria is a life-threatening disease caused by Plasmodium that are transmitted to people through the bites of infected female Anopheles mosquitoes. In 2019, there were an estimated 229 million cases of malaria worldwide. The estimated number of malaria deaths stood at 409,000 in 2019 [1]. Artemisinin has now become the world’s most effective drug for fighting malaria. Recently, there was a resurgence of malaria, partly as a result of increased resistance to artemisinin [2, 3]. To date, no vaccine has been shown to provide long-lasting benefits at a population level [4,5,6,7]. So, there is still a long way to go to achieve the goal of malaria elimination.
Besides respiratory and metabolic function, lung plays a role in immune system. It contains heterogeneous populations of innate and adaptive immune cells, such as T helper cells, macrophages, natural killer cells, gamma delta T cells (γδT cells), and others [8,9,10]. Malaria-associated acute lung injury (ALI)/acute respiratory distress syndrome (ARDS) is one of the main clinical complications of severe Plasmodium infection, which is one of the main causes of death [11,12,13,14]. However, the detailed mechanism of malaria-induced lung injury is unclear. Various immune cells are reported to participate in the process of malaria-associated ALI and ARDS in mice. For example, parasite-specific CD8+ T cells promote pulmonary vascular leakage and pulmonary edema [15, 16]. The B cells can protect the host from adverse lung pathological damage by secreting the IgA [17].
γδT cells represent a minor population of innate lymphocytes that can respond to the antigen without presentation [18]. γδT cells have multiple functions, producing different types of cytokines and chemokines, regulating the immune response by interacting with other cells [19]. The study of γδT cells in malaria was first published nearly 30 years ago [20], and recent findings showed that γδT cells play an important role in the protective immune response against Plasmodium [21]. Further evidence demonstrates that γδT cells are expanded in spleen, peripheral blood, lung, and liver of mice infected with different strains of Plasmodium [22,23,24,25]. γδT cells can regulate the anti-malaria immune response by interacting with other cells. For example, they can stimulate and recruit myeloid cells, promote the differentiation of CD4+ and CD8+ T cells by producing cytokines, like IFN-γ and TNF, and chemokines upon recognizing the soluble antigens released from parasites [22, 26,27,28]. There is an increasing body of evidence to support the fact that γδ T cells could modulate humoral immunity against Plasmodium berghei infection [29]. γδT cells were reported to involve in the pulmonary immunopathological injury caused by pathogenic organisms. For example, γδT cells could mediate influenza A (H1N1) induced lung injury by secreting interleukin-17A in mice [30]. γδT cells were found to mainly regulate the Th2 immune response in the lung of the mice infected with Schistosoma japonicum [31]. However, the potential roles of γδT cells during Plasmodium infection in the lungs C57BL/6 mice remains unclear. This research try to study the phenotype and function of γδT cells in the lung of C57BL/6 mice infected by Plasmodium, as well as the effects of γδT cells on T cells and B cells after Plasmodium infection.
Methods
Mice
Wild-type female C57BL/6 mice (6–8 weeks) were obtained from Animal Centre of Guangzhou University of Chinese Medicine (Guangzhou, China). γδT KO mice (B6.129P2-Tcrdtm1Mom/J, C57BL/6J genetic background) were acquired from JAX Stock (No. 002120). All protocols for animal use were approved to be appropriate and humane by the institutional animal care and use committee of Guangzhou Medical University (2012-11).
Parasites and infection
The NSM strain of Plasmodium yoelii was purchased from the malaria research and reference reagent resource center (MR4). Frozen P. yoelii were thawed and maintained into C57BL/6 mice until the parasitaemia up to 10–15%. 6–8 weeks female C57BL/6 mice or γδT KO mice were infected with 1 × 106 infected red blood cells (iRBCs) by intraperitoneal injection.
Isolation of lymphocyte
Mice were euthanized at 11 days post-infection. Before obtaining the lung tissue, mice were perfused with sterile saline to remove the blood. The excised lung tissue was cut into small pieces and incubated in 5 ml of digestion buffer (collagenase IV/DNase I mix, Invitrogen Corporation) for 30 min at 37 °C. Digested lung tissue was pressed through a 200-gauge stainless-steel mesh and was then suspended in Hank’s balanced salt solution (HBSS). Lymphocytes were isolated using mouse lymphocyte separation medium (Dakewe Biotech) and density gradient centrifugation. The isolated cells were washed twice in HBSS and resuspended in complete RPMI 1640 medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U/ml penicillin, 100 µg/ml streptomycin. After the lymphocytes were isolated, the cells were calculated by the blood cell counting plate with trypan blue staining.
Antibodies
A detailed description of antibodies used in this study is provided in Table 1.
Histology studies
Lungs were removed from mice and perfused three times with 0.01 M PBS (pH 7.4), fixed in 10% formalin, embedded in paraffin, and sectioned. The slice was stained by standard haematoxylin-eosin (HE) staining, and examined by light microscopy (Olympus ix71).
Cell surface staining
Cells were washed twice with PBS and blocked in PBS buffer containing 1% BSA for 30 min. Cells were then stained with specific antibodies for the cell surface antigens for 30 min at 4 °C in the dark. The phenotypes (1 × 106 cells per run) were analysed using flow cytometry (Beckman CytoFLEX) and CytExpert 1.1 (Beckman Coulter Inc.). The single nuclear cells were gated to exclude the dead cells and doublet. For gating CD3+ γδTCR+ cells, CD3+, CD3+ CD4+, CD3+ CD8+, CD3− CD19+ cells, fluorescence minus one (FMO) controls were used. For other surface makers, isotype controls were used. 1,000,000 cells were used for cell surface staining, and 300,000 events were collected for each tube.
Cell intracellular cytokine staining
1.5 × 106 cells were resuspended in complete RPMI 1640 medium, then stimulated with phorbol 12-myristate 13-acetate (PMA) (20 ng/ml, Sigma) and ionomycin (1 µg/ml, Sigma) for 1 h. Brefeldin A (BFA, 10 µg/ml, Sigma) was added and incubated for 4 h. Cells were washed twice in PBS and stained with specific antibodies for the cell surface antigens for 30 min at 4 °C in the dark. Cells were fixed with Fixation and Permeabilization Solution (BD Biosciences) for 20 min at 4 °C in the dark. Next, cells were stained with specific antibodies for each cytokine. The results were analysed using flow cytometry (Beckman CytoFLEX) and CytExpert 1.1 (Beckman Coulter Inc.). The single nuclear cells were gated to exclude the dead cells and doublet. For gating CD3+ γδTCR+ cells, CD3+, CD3+ CD4+, CD3+ CD8+, CD3− CD19+ cells, FMO controls were used. Isotype controls were used for intracellular cytokines staining. 1,500,000 cells were used for cell intracellular cytokine staining, and 300,000 events were collected for each tube.
Statistics
The differences between the two groups were analysed in Prism (GraphPad Software) using a two-tailed Student’s t-test with equal variance and normal distributions. To compare more than two groups, one-way ANOVA and LSD test by SPSS software package were used with equal variance and normal distributions. Mann-Whitney U test was used with unequal variance or abnormal distributions. The statistical significance was defined as p < 0.05.
Results
Plasmodium infection induces the accumulation of γδT cells to the lung
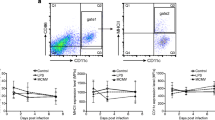
To evaluate the change of γδT cells in the lung of the Plasmodium-infected mice, C57BL/6 mice were euthanized, and the lungs were removed 11 days post-Plasmodium infection. Single cell suspensions were prepared and calculated by the blood cell counting plate with trypan blue staining. The percentage and the absolute number of CD3+ γδTCR+ cells were determined by FCM (Fig. 1a). The staining strategy is shown in Additional file 1: Table S1. All the doublet cells, dead cells, and non-lymphoid cells were excluded in this study. As shown in Fig. 1b, the percentage of CD3+ γδTCR+ cells in the infected group was significantly higher than that in the naive group (naive: 1.11 ± 0.06 %, infected: 2.98 ± 0.15%, p < 0.01); and the absolute number of CD3+ γδTCR+ cells post-infection was also significantly increased (naive: 1.46 ± 0.09 per 104 cells, infected: 19.7 ± 2.03 per 104 cells, p < 0.01).
Plasmodium yoelii infection induces the accumulation of γδT cells to the lung. Pulmonary lymphocytes were stained with anti-CD3 and anti-γδTCR fluorescent mAbs. The expression of CD3 and γδT on lung lymphocytes of naive and infected mice were analysed by FCM. FMO controls for CD3 and γδTCR were included in the staining protocol. a One representative sample. All the doublet cells, dead cells, and non-lymphoid cells were excluded in this study. b Comparison of the percentage and absolute number of CD3+ γδT+ cells from the naive and infected groups. 5–7 samples were prepared for each group, and the experiments were repeated three times. **p < 0.01
Surface maker changes and cytokines released in the pulmonary γδT cells
To study the phenotypic changes of γδT cells post-infection, single pulmonary cells from the naive and infected mice were stained with the different surface markers labelled with fluorescence: CD80, MHC II, CD34, CD127, CD62L, CD11b, PD-L1, and PD-1. The staining strategy is shown in Additional file 1: Table S2. As shown in Fig. 2, more CD3+ γδT+ cells were expressing CD80, CD11b, and PD-1 post-infection (p < 0.05), while less CD3+ γδT+ cells were expressing CD34, CD127, and CD62L in the infected mice (p < 0.05). There was no significant difference in the percentages of MHC II+ and PD-L1+ γδT cells between the naive mice and infected mice (p > 0.05).
The expression of surface molecules on γδT cells. Pulmonary cells were stained with fluorescent mAbs against mice: CD3, γδTCR, CD80, CD34, CD127, CD62L, CD11b, PD-L1, and PD-1. FMO controls for CD3, γδTCR and isotype controls for CD80, CD34, CD127, CD62L, CD11b, PD-L1, and PD-1 were included in the staining protocol. a One representative FCM analysis. b Comparison of the expression of different surface molecules on CD3+ γδTCR+ cells from the naive and infected groups. 5–7 samples were prepared for each group, and the experiments were repeated three times. *p < 0.05, **p < 0.01
To investigate the cytokine expression of γδT cells, pulmonary cells were stimulated with PMA and ionomycin, then stained for intracellular cytokines labelled with fluorescence. The staining strategy is shown in Additional file 1: Table S3. As shown in Fig. 3, the percentage of IFN-γ-expressing γδT cells from the infected mice was lower than that from the naive mice (p < 0.05), while the percentages of γδT cells that were expressing IL-5, IL -6, IL-21, IL-4, IL-1α, and IL-17 in the infected mice were higher than that in the naive mice (p < 0.05).
The expression of different cytokines from γδT cells. Pulmonary cells were stimulated with PMA and ionomycin. The ability of γδT cells to secrete cytokines (IFN-γ, IL-5, IL -6, IL -21, IL -4, IL -1, and IL -17) was detected. FMO controls for CD3, γδTCR and isotype controls for IFN-γ, IL-5, IL -6, IL -21, IL -4, IL -1, and IL -17 were included in the staining protocol. a One representative FCM analysis. b Comparison of the expression of different cytokines on γδT cells from the naive and infected groups. 5–7 samples were prepared for each group, and the experiments were repeated three times. *p < 0.05, **p < 0.01
Pathological changes of the lung in γδT KO mice
To evaluate the role of γδT cells in Plasmodium infection, WT and γδT KO mice were injected with the same amount iRBCs. Mice were euthanized 11 days post-infection, and the lungs were removed. As shown in Fig. 4a, the size and colour of the lungs in the infected group is bigger and darker compared with the uninfected group, while, it was no obvious difference between the infected WT mice and infected γδT KO mice. The infected WT mice exhibited significant weight loss compared with uninfected WT mice (p < 0.05). The weight of the lung, the proportion of lung weight, and numbers of extracted lung cells from infected mice were higher than that from the uninfected mice (p < 0.05), there was no obvious difference between the infected WT and infected γδT KO mice (p > 0.05) (Fig. 4b). As shown in Fig. 4c with HE staining, the structure of the lung in the uninfected mice was clear with a uniform distribution of lung cells, but leukocyte infiltration and alveolar fusion were observed in the lung of the infected mice. However, the difference is not obvious between the infected WT and the infected γδT KO mice.
Pulmonary lesions in γδT KO mice infected with Plasmodium. a Comparison of the lung appearance in WT and γδT KO mice. Representative samples were shown for each group. b Comparison of the weight of the mice, the weight of the lung, the proportion of lung weight, and numbers of extracted lung cells from uninfected or infected WT and γδT KO mice. c HE staining of the lung tissue. The leukocyte infiltration is indicated by black arrows. Scale bar, 200 µm. *p < 0.05, **p < 0.01
The effect of γδT cells on T cells in the lung upon Plasmodium infection
To elucidate the potential regulating role of γδT cells on T cells upon Plasmodium infection. The content, surface active molecular expression, and cytokine-producing ability of T cells from the lung of infected or uninfected WT and γδT KO mice were compared. As shown in Fig. 5, the single pulmonary cells were stained with different surface markers: CD3, CD4, and CD8. For gating CD3+ cells, CD3+ CD4+ cells, CD3+ CD8+ cells, FMO controls were used. The staining strategy is shown in Additional file 1: Table S4. The proportion and absolute numbers of CD3+ cells and CD3+ CD8+ cells from infected mice were higher than that from the uninfected mice (p < 0.05). Moreover, the proportion of CD3+ cells, and absolute numbers of CD3+ cells, CD3+ CD4+ cells, CD3+ CD8+ cells were decreased in the γδT KO infected mice compared with the WT infected mice (p < 0.05). These results indicated that γδT cells could promote the production of T cells upon Plasmodium infection.
The content changes of T cells in the lung of γδT KO mice. Pulmonary cells from WT and γδT KO mice were stained with the different surface markers: CD3, CD4, and CD8. FMO controls for CD3, CD4, and CD8 were included in the staining protocol. a One representative FCM analysis for each comparison is shown. b Comparison of the percentage and the absolute number of CD3+, CD3+ CD4+, CD3+ CD8+ cells from the lung of WT and γδT KO mice. *p < 0.05, **p < 0.01
Additionally, the phenotypic changes of T cells were investigated. The single pulmonary cells were stained with different antibodies labelled with fluorescence: CD3, CD4, CD8, CD69, CD62L, and CD25. The staining strategy is shown in Additional file 1: Table S5. As shown in Fig. 6a, more CD3+ T cells were expressing CD25 and CD69 (p < 0.05) and fewer CD3+ cells were expressing CD62L (p < 0.01) in infected mice when compared with the uninfected mice. This study demonstrated that mice lacking γδT cells and the corresponding wild-type strain do not differ in the percentages of CD25+ CD3+ T cells and CD62L+ CD3+ T cells (p > 0.05). The percentage of CD69+ CD3+ T cells decreased in uninfected γδT KO mice compared with uninfected WT mice (p < 0.01). For CD3+ CD4+ cells and CD3+ CD8+ cells (Fig. 6b and c), less of them were expressing CD62L (p < 0.01) and more of them were expressing CD69 (p < 0.01) in infected mice when compared with the uninfected mice. γδTCR knockout did not make a significant difference in the percentages of CD25+, CD62L+, and CD69+ CD3+ CD4+ T cells and CD3+ CD8+ T cells (p > 0.05). These results indicated that γδTCR knockout did not make a significant difference in the surface molecular expression of T cells. Furthermore, the cytokine-producing ability of T cells from the lung of infected or uninfected WT and γδT KO mice were compared. IL-4, IFN-γ, IL-10, IL-17, IL-2 and IL-21 from CD3+, CD3+ CD4+ and CD3+ CD8+ cells were detected. The staining strategy is shown in Additional file 1: Table S6. As shown in Fig. 7a, more CD3+ T cells were expressing IFN-γ, IL-10, and IL-17 in the infected mice when compared with the uninfected mice (p < 0.05). The percentage of IFN-γ+ T cells decreased in uninfected γδT KO mice compared with uninfected WT mice (p < 0.01) (Fig. 7a–c). It implied that the deficiency of γδT cells has some effects on cytokine-producing abilities of T cells in the absence of malaria infection. However, although the difference is statistically significant, it is very small which could not have a biological impact. Moreover, the percentage of IFN-γ- expressing CD3+ and CD3+ CD8+ cells from infected γδT KO mice was higher than from the infected WT mice (p < 0.05) (Fig. 7). These indicated that γδT cells could suppress T cells to produce IFN-γ upon Plasmodium infection. When γδT cells were knockout, more T cells could produce IFN-γ upon Plasmodium infection.
The expression of active surface markers on T cells. The surface active molecules expression of T cells from the lung of WT and γδT KO mice were calculated from FCM data and compared. FMO controls for CD3, CD4, and CD8 and isotype controls for CD69, CD25, and CD62L were included in the staining protocol. The average expression of CD69, CD25, and CD62L on CD3+ cells (a), CD3+ CD4+cells (b), and CD3+ CD8+cells (c). *p < 0.05, **p < 0.01
The cytokine-producing ability of CD3+, CD3+ CD4+, CD3+ CD8+ cells. The expression of IL-4, IFN-γ, IL-10, IL-17, IL-2, and IL-21 on T cells from the lung of WT and γδT KO mice were calculated from FCM data and compared. FMO controls for CD3, CD4, and CD8 and isotype controls for IL-4, IFN-γ, IL-10, IL-17, IL-2, and IL-21 were included in the staining protocol. The average expression of IL-4, IFN-γ, IL-10, IL-17, IL-2, and IL-21 on CD3 + cells (a), CD3+ CD4+cells (b), and CD3+ CD8+cells (c). *p < 0.05, **p < 0.01
The effect of γδT cells on B cells in the lung upon Plasmodium infection
To further explore the potential modulating role of γδT cells on B cells upon Plasmodium infection. The content, surface active molecular expression of B cells from the lungs were compared. The single pulmonary cells were stained with fluorescence-labelled surface markers: CD3, CD19, CD69, ICOS, and CD80. The staining strategy is shown in Additional file 1: Table S7–8. As shown in Fig. 8, the proportion of CD19+ cells from the infected mice decreased compared with uninfected mice (p < 0.05), while it was increased in the γδT KO infected mice compared with the WT infected mice (p < 0.05). The absolute numbers of CD19+ cells were no obvious change in the infected WT and the infected γδT KO mice (p > 0.05). These results indicated that the deletion of γδT did not affect the proliferation of B cells.
The content changes of B cells in the lung of γδT KO mice. Pulmonary cells from WT and γδT KO mice were stained with fluorescent mAbs against mice CD19, CD13. FMO controls for CD19 and CD3 were included in the staining protocol. a One representative FCM analysis for the comparison is shown. b Comparison of the percentage and absolute numbers of CD3− CD19+ cells from the lung of WT and γδT KO mice. *p < 0.05, **p < 0.01
As shown in Fig. 9, the percentages of CD69, ICOS, or CD80-expressing B cells increased in infected mice during the course of infection (p < 0.01). It showed that a higher percentage of ICOS-expressing B cells and a lower percentage of CD80-expressing B cells from infected γδT KO mice compared with infected WT mice (p < 0.05). However, there was no obvious difference for the absolute numbers of CD69+, ICOS+, and CD80+ B cells between infected WT and infected γδT KO mice (p > 0.05). Taken together, these results indicated that the deletion of γδT did not significantly affect B cells’ immune response upon Plasmodium infection.
The expression of active surface markers on B cells. The surface molecules expression of B cells was calculated from FCM data and compared. FMO controls for CD19 and CD3 and isotype controls for CD69, ICOS, and CD80 were included in the staining protocol. a One representative FCM analysis for each comparison is shown. b Comparison of the percentage of CD69+, ICOS+, and CD80+ B cells from the lung of WT and γδT KO mice. *p < 0.05, **p < 0.01
Discussion
γδT cells comprise a small population of T cells (3–5%) [32]. In this study, the characteristics of γδT cells from the lungs of P. yoelii infected C57BL/6 mice were explored. The percentage and the absolute number of γδT cells were significantly increased (Fig. 1) in the lungs of P. yoelii infected C57Bl/6 mice at 11 days post-infection. Similarly, Mamedov et al. also reported γδT cells are expanded in the lungs of Plasmodium chabaudi infected C57Bl/6 mice at 16 days post-infection when parasite recrudescence reached a peak in the mice whose γδT cells were silenced [22]. These results indicated that γδT cells accumulate in the lung and may play a role in the process of host anti-Plasmodium infection.
To study the potential role of CD3+ γδTCR+ cells after infection, the phenotype of γδT cells was examined. CD127, CD34 and CD62L are T cell activation-associated molecules [33,34,35]. CD34 serves as a ligand for CD62L, CD34 and CD62L primarily regulates the proliferation and migration of leukocytes to inflammatory sites and lymph nodes [33, 34]. The percentages of CD62L+, CD127+, and CD34+ γδT cells decreased significantly in the infected group (p < 0.05). PD-1 acts as an inhibitory receptor, which could reduce T cell receptor (TCR) induced cell proliferation, cytokine production, and cytolytic activity [36]. MHC II, CD80, and CD11b are the surface markers of antigen-presenting cells, which could accelerate T-cell activation [37, 38]. More γδT cells were expressing CD80, CD11b and PD-1 post-infection (p < 0.05). While the percentages of MHC II+ and PD-L1+ γδT cells did not significantly change post-infection (p > 0.05). These results further confirm that γδT cells could regulate the inflammation response in the lung of P. yoelii infected mice, and γδT cells may be beneficial for antigen presentation in the lung of infected mice.
It is reported that γδT cells could secrete numerous cytokines to mediate the immune response [19]. In this study, the results showed more γδT cells secrete Th2 cytokines (IL-4, IL-5), IL -6, IL -21, IL -1α, IL -17, and fewer γδT cells secrete IFN-γ in response to Plasmodium infection. It implied that the Th2 immune response is promoted by increased IL-4 and IL-5 secreted from γδT cells. As a pro-inflammatory cytokine, IL-17 extensively participated in host antimicrobial immunity [39,40,41]. It is commonly accepted that IL-17 is predominately produced by γδT cells upon Mycobacterium tuberculosis infection [42]. IL-21 is a pleiotropic cytokine, which is related to autoimmune diseases, allergies, and inflammatory diseases. It can enhance the body’s adaptive immune response and innate immune response [43]. IL-1α and IL-6 are required for Th17 lymphocyte differentiation upon host infected with Paracoccidioides brasiliensis [44]. In this study, the percentage of IL-6, IL-21, IL-1α, IL-17 γδT cells increased significantly after infection (p < 0.05). These data indicated that γδT cells could promote host immune response in anti-P. yoelii infection.
To explore the role of γδT cells in Plasmodium infection-induced lung injury, γδT KO mice were infected with P. yoelii. γδT cells, one functional group of cells, has some effects on phenotypes and cytokine-producing abilities of T and B cells. For example, the percentages of CD69+ CD3+ T cells, IFN-γ- expressing T cells, IL-17 expressing CD3+ T cells, ICOS+ B cells decreased in γδT KO mice in the absence of malaria infection (p < 0.05) (Figs. 6, 7, 8 and 9). Even so, the γδT KO mice are still the best model to study the role of γδT cells. There was no obvious difference in the lung between the WT and γδT KO mice in either the uninfected or the infected group (Fig. 4). One potential reason for this phenomenon may be the lower percentage of γδT cells in the lung. Although it plays a certain role in anti-P. yoelii infection, the deletion of γδ TCR is not enough to alter the pathological damage of the lung. T cell response was studied in the P. yoelii-infected WT and γδT KO mice. T cell-mediated immunity is the key for the host to defense against malaria parasite infection [45]. Parasite-specific CD8+ T cells participate in the process of malaria-associated ALI and ARDS by promoting pulmonary vascular leakage and pulmonary oedema [15, 16]. There was no significant difference for the proportion and absolute numbers of T cells between uninfected WT mice and the uninfected γδT KO mice (p > 0.05). However, the proportion of CD3+ cells and the absolute numbers of CD3+ cells, CD3+ CD4+ cells, CD3+ CD8+ cells were decreased in γδT KO infected mice compared with the WT infected mice (p < 0.05). These results indicated that γδT cells could promote the recruitment of T cells upon P. yoelii infection. The possible reason maybe the secretion of chemokines by γδT cells, like M-CSF. It is commonly accepted that the chemokine system plays critical role in the recruitment of lymphocytes [22, 46].
The deficiency in γδT cells did not make a significant difference on the surface molecular expression of T cells for the infected mice (p > 0.05), suggesting that γδT cells were not associated with the activation of T cells. IFN-γ is the central molecule in mediating host protective immune responses against malaria parasites [47]. The percentage of IFN-γ- expressing CD3+ and CD3+ CD8+ cells increased in γδT KO infected mice compared with the WT infected mice (p < 0.05). These results indicated that γδT cells could suppress the production of IFN-γ in CD3+ and CD3+ CD8 + cells upon P. yoelii infection. Taken together, γδT cells played double effects on T cells, especially CD3+ CD8 + cells, mediated anti-malarial response in the lung.
Additionally, B cell response was also investigated, the absolute number of B cells was not affected by γδ TCR knockout. The B cells expressed ICOS could induce regulatory T cells [48]. Many types of antigen-presenting cells can express CD80 [49] and the expression of CD80 in B cells plays a critical role in regulating B-T interactions in both early and late germinal center responses [50]. Although the percentages of ICOS and CD80-expressing B cells differed in infected γδT KO mice compared with the infected WT mice (p < 0.05). The absolute numbers of ICOS+, CD69+, and CD80+ B cells were not significantly changed in the infected γδT KO mice compared with the infected WT mice (p > 0.05). It is suggested that γδT cells may not contribute to the proliferation and phenotype changes of B cells upon P. yoelii infection.
Conclusions
This study explored the phenotypic and functional characteristics of γδT cells in the lung of P. yoelii-infected mice, and found that Plasmodium infection can induce significant changes in the content, phenotype, and function of the lung γδT cells, γδT cells contribute to T cell immune response in the lungs of mice infected with Plasmodium. Although there are differences in tissue distributions and TCR development between mice and humans, the primary theme for γδ T cells in protecting against disease and tissue damage is basically conserved [51]. This study is beneficial for understanding the roles of γδT cells in malaria patients.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- WT:
-
Wild-type
- TCR:
-
T cell receptor
- iRBCs:
-
Infected red blood cells
- HE:
-
Haematoxylin-eosin
- HBSS:
-
Hank’s balanced salt solution
- γδT cells:
-
Gamma delta T cells
- ALI:
-
Acute lung injury
- ARDS:
-
Acute respiratory distress syndrome
- γδT KO:
-
γδTCR knockout
- FMO:
-
Fluorescence minus one
- PMA:
-
Phorbol 12-myristate 13-acetate
References
WHO. Malaria. Geneva WH, Organization. 2020. https://www.who.int/news-room/fact-sheets/detail/malaria. Accessed 4 Dec 2020.
Maxmen A. How to defuse malaria’s ticking time bomb. Nature. 2018;559:458–65.
Ashley EA, Pyae PA, Woodrow CJ. Malaria. Lancet. 2018;391:1608–21.
Fried M, Duffy PE. Designing a VAR2CSA-based vaccine to prevent placental malaria. Vaccine. 2015;33:7483–8.
Tinto H, Otieno W, Gesase S, Sorgho H, Otieno L, Liheluka E, et al. Long-term incidence of severe malaria following RTS,S/AS01 vaccination in children and infants in Africa: an open-label 3-year extension study of a phase 3 randomised controlled trial. Lancet Infect Dis. 2019;19:821–32.
Stiepel RT, Batty CJ, MacRaild CA, Norton RS, Bachelder E, Ainslie KM. Merozoite surface protein 2 adsorbed onto acetalated dextran microparticles for malaria vaccination. Int J Pharm. 2020;593:120168.
Sissoko MS, Healy SA, Katile A, Omaswa F, Zaidi I, Gabriel EE, et al. Safety and efficacy of PfSPZ vaccine against Plasmodium falciparum via direct venous inoculation in healthy malaria-exposed adults in Mali: a randomised, double-blind phase 1 trial. Lancet Infect Dis. 2017;17:498–509.
Snyder ME, Farber DL. Human lung tissue resident memory T cells in health and disease. Curr Opin Immunol. 2019;59:101–8.
Segawa S, Goto D, Yoshiga Y, Horikoshi M, Sugihara M, Hayashi T, et al. Involvement of NK 1.1-positive γδT cells in interleukin-18 plus interleukin-2-induced interstitial lung disease. Am J Respir Cell Mol Biol. 2011;45:659–66.
Liu J, Wang H, Yu Q, Zheng S, Jiang Y, Liu Y, et al. Aberrant frequency of IL-10-producing B cells and its association with Treg and MDSC cells in non small cell lung carcinoma patients. Hum Immunol. 2016;77:84–9 Retracted.
Pereira M, Marinho C, Epiphanio S. Could heme oxygenase-1 be a new target for therapeutic intervention in malaria-associated acute lung injury/acute respiratory distress syndrome? Front Cell Infect Microbiol. 2018;8:161.
Taylor WR, White NJ. Malaria and the lung. Clin Chest Med. 2002;23:457–68.
Lozano F, Leal M, Lissen E, Munoz J, Bautista A, Regordan C. P. falciparum and P. malariae malaria complicated by pulmonary edema with disseminated intravascular coagulation. Press Med. 1983;12:3004–5. (in French)
Dondorp AM, Fanello CI, Hendriksen IC, Gomes E, Seni A, Chhaganlal KD, et al. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet. 2010;376:1647–57.
Claser C, Nguee S, Balachander A, Wu HS, Becht E, Gunasegaran B, et al. Lung endothelial cell antigen cross-presentation to CD8(+)T cells drives malaria-associated lung injury. Nat Commun. 2019;10:4241.
Pham TT, Verheijen M, Vandermosten L, Deroost K, Knoops S, VandenEynde K, et al. Pathogenic CD8(+) T cells cause increased levels of VEGF-A in experimental malaria-associated acute respiratory distress syndrome, but therapeutic VEGFR inhibition is not effective. Front Cell Infect Microbiol. 2017;7:416.
Deore S, Kumar A, Kumar S, Mittal E, Lotke A, Musti K. Erythrocyte binding ligand region VI specific IgA confers tissue protection in malaria infection. Mol Biol Rep. 2019;46:3801–8.
Wu YL, Ding YP, Tanaka Y, Shen LW, Wei CH, Minato N, et al. γδ T cells and their potential for immunotherapy. Int J Biol Sci. 2014;10:119–35.
Beetz S, Wesch D, Marischen L, Welte S, Oberg HH, Kabelitz D. Innate immune functions of human gammadelta T cells. Immunobiology. 2008;213:173–82.
Goerlich R, Häcker G, Pfeffer K, Heeg K, Wagner H. Plasmodium falciparum merozoites primarily stimulate the V gamma 9 subset of human gamma/delta T cells. Eur J Immunol. 1991;21:2613–6.
Deroost K, Langhorne J. Gamma/Delta T cells and their role in protection against malaria. Front Immunol. 2018;9:2973.
Mamedov MR, Scholzen A, Nair RV, Cumnock K, Kenkel JA, Oliveira J, et al. A macrophage colony-stimulating-factor-producing γδ T cell subset prevents malarial parasitemic recurrence. Immunity. 2018;48:350–63.
Inoue SI, Niikura M, Asahi H, Iwakura Y, Kawakami Y, Kobayashi F. Preferentially expanding Vγ1(+) γδ T cells are associated with protective immunity against Plasmodium infection in mice. Eur J Immunol. 2017;47:685–91.
Ho M, Tongtawe P, Kriangkum J, Wimonwattrawatee T, Pattanapanyasat K, Bryant L, et al. Polyclonal expansion of peripheral gamma delta T cells in human Plasmodium falciparum malaria. Infect Immun. 1994;62:855–62.
Taniguchi T, Tachikawa S, Kanda Y, Kawamura T, Tomiyama-Miyaji C, Li C, et al. Malaria protection in beta 2-microglobulin-deficient mice lacking major histocompatibility complex class I antigens: essential role of innate immunity, including gamma delta T cells. Immunology. 2007;122:514–21.
Roussilhon C, Agrapart M, Guglielmi P, Bensussan A, Brasseur P, Ballet JJ. Human TcR gamma delta + lymphocyte response on primary exposure to Plasmodium falciparum. Clin Exp Immunol. 1994;95:91–7.
Roussilhon C, Agrapart M, Ballet JJ, Bensussan A. T lymphocytes bearing the gamma delta T cell receptor in patients with acute Plasmodium falciparum malaria. J Infect Dis. 1990;162:283–5.
Tran TM, Jones MB, Ongoiba A, Bijker EM, Schats R, Venepally P, et al. Transcriptomic evidence for modulation of host inflammatory responses during febrile Plasmodium falciparum malaria. Sci Rep. 2016;6:31291.
Inoue SI, Niikura M, Asahi H, Kawakami Y, Kobayashi F. γδ T cells modulate humoral immunity against Plasmodium berghei infection. Immunology. 2018;155:519–32.
Xue C, Wen M, Bao L, Li H, Li F, Liu M, et al. Vγ4(+)γδT cells aggravate severe H1N1 influenza virus infection-induced acute pulmonary immunopathological injury via secreting interleukin-17A. Front Immunol. 2017;8:1054.
Cha H, Xie H, Jin C, Feng Y, Xie S, Xie A, et al. Adjustments of γδ T cells in the lung of Schistosoma japonicum-infected C56BL/6 mice. Front Immunol. 2020;11:1045.
Peters C, Kabelitz D, Wesch D. Regulatory functions of γδ T cells. Cell Mol Life Sci. 2018;75:2125–35.
Choudhary D, Hegde P, Voznesensky O, Choudhary S, Kopsiaftis S, Claffey KP, et al. Increased expression of L-selectin (CD62L) in high-grade urothelial carcinoma: a potential marker for metastatic disease. Urol Oncol. 2015;33:387.e17-27.
Lanza F, Healy L, Sutherland DR. Structural and functional features of the CD34 antigen: an update. J Biol Regul Homeost Agents. 2001;15:1–13.
Ho LP, Yit PS, Ng LH, Linn YC, Zhao Y, Sun L, et al. The road to memory: an early rest for the long journey. J Immunol. 2013;191:5603–14.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15:486–99.
Sandilands GP, Ahmed Z, Perry N, Davison M, Lupton A, Young B. Cross-linking of neutrophil CD11b results in rapid cell surface expression of molecules required for antigen presentation and T-cell activation. Immunology. 2005;114:354–68.
Anderson AC, Chandwaskar R, Lee DH, Kuchroo VK. Cutting edge: the Idd3 genetic interval determines regulatory T cell function through CD11b + CD11c- APC. J Immunol. 2008;181:7449–52.
Rahal EA, Hajjar H, Rajeh M, Yamout B, Abdelnoor AM. Epstein-Barr virus and human herpes virus 6 Type A DNA enhance IL-17 production in mice. Viral Immunol. 2015;28:297–302.
Da MGP, Gutierrez FR, Maia FL, Milanezi CM, Silva GK, Pavanelli WR, et al. IL-17 produced during Trypanosoma cruzi infection plays a central role in regulating parasite-induced myocarditis. PLoS Negl Trop Dis. 2010;4:e604.
Bagheri N, Azadegan-Dehkordi F, Shirzad H, Rafieian-Kopaei M, Rahimian G, Razavi A. The biological functions of IL-17 in different clinical expressions of Helicobacter pylori-infection. Microb Pathog. 2015;81:33–8.
Lockhart E, Green AM, Flynn JL. IL-17 production is dominated by gammadelta T cells rather than CD4 T cells during Mycobacterium tuberculosis infection. J Immunol. 2006;177:4662–9.
Davis MR, Zhu Z, Hansen DM, Bai Q, Fang Y. The role of IL-21 in immunity and cancer. Cancer Lett. 2015;358:107–14.
Ketelut-Carneiro N, Souza COS, Benevides L, Gardinassi LG, Silva MC, Tavares LA, et al. Caspase-11-dependent IL-1α release boosts Th17 immunity against Paracoccidioides brasiliensis. PLoS Pathog. 2019;15:e1007990.
Kurup SP, Butler NS, Harty JT. T cell-mediated immunity to malaria. Nat Rev Immunol. 2019;19:457–71.
Oo YH, Shetty S, Adams DH. The role of chemokines in the recruitment of lymphocytes to the liver. Dig Dis. 2010;28:31–44.
McCall MB, Sauerwein RW. Interferon-γ--central mediator of protective immune responses against the pre-erythrocytic and blood stage of malaria. J Leukoc Biol. 2010;88:1131–43.
Chien CH, Chiang BL. Regulatory T cells induced by B cells: a novel subpopulation of regulatory T cells. J Biomed Sci. 2017;24:86.
Shirasaka K, Morikawa H. The kinetics of CD80 and CD86 expression on antigen-presenting cells in cedar pollinosis subjects . Nihon Jibiinkoka Gakkai Kaiho. 2003;106:823–30 (in Japanese).
Good-Jacobson KL, Song E, Anderson S, Sharpe AH, Shlomchik MJ. CD80 expression on B cells regulates murine T follicular helper development, germinal center B cell survival, and plasma cell generation. J Immunol. 2012;188:4217–25.
Holderness J, Hedges JF, Ramstead A, Jutila MA. Comparative biology of γδ T cell function in humans, mice, and domestic animals. Annu Rev Anim Biosci. 2013;1:99–124.
Acknowledgements
Not applicable.
Funding
This research was supported by grants from the Natural Science Foundation of China (81771696, 81802024), the Guangdong provincial education department (2016KZDXM033), the the Natural Science Foundation of Guangdong province (2020A1515010251, 2018A030313217), and Guangzhou science and technology project (202002030082).
Author information
Authors and Affiliations
Contributions
HW, CJ, and AP: Lymphocyte isolation, flow cytometry analysis, figure drafting, and manuscript drafting, funding support. HX, SX, YF, AX, JL, and CF: Animal feeding and infection, HE staining. QY, HQ, and YQ: statistical analysis. XW and JH: study conception and design, supervision of the research group, funding support, and manuscript drafting. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All animal experiments were performed following the guidelines for the Administration of Affairs Concerning Experimental Animals (1988.11.1). All protocols for animal use were approved to be appropriate and humane by the institutional animal care and use committee of Guangzhou Medical University (2012-11). Every effort was made to minimize suffering.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Additional Tables.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wei, H., Jin, C., Peng, A. et al. Characterization of γδT cells in lung of Plasmodium yoelii-infected C57BL/6 mice. Malar J 20, 89 (2021). https://doi.org/10.1186/s12936-021-03619-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12936-021-03619-z