Abstract
Malaria is a global health challenge with endemicity in sub-Saharan Africa, where there are multiple drug-resistant strains and limited access to modern health care facilities, especially in rural areas. Studies indicate that African traditional medicine could make a substantial contribution to the reduction of malaria-related deaths and achievement of universal health coverage (UHC), particularly in these regions. Thus, this study evaluated the curative antimalarial effects of Chromolaena odorata leaf extract using mouse model. Forty-five (45) albino mice weighing between 18 and 22 g were grouped into nine groups of 5 animals each. Animals in groups 2–9 were infected with the chloroquine-resistant strain of Plasmodium berghei, while animals in groups 3–9 were subsequently treated with 10 mg/kg chloroquine, a combination of 1.4 mg/kg artemether and 8.75 mg/kg lumefantrine (Coartem), and varying concentrations of the fraction from the aqueous leaf extract of C. odorata at day 3 post-infection. The findings from this study indicate that treatment with 400 mg/kg of the ethanolic fraction of the crude extract resulted in a significant decrease in parasite load (97.6%), which was comparable to the activities of the conventional drugs chloroquine (98.6%) and Coartem (98.8%). The ethyl acetate and ethanolic fractions at 400 mg/kg also ameliorated the significant alterations in the red blood cells, white blood cells, and platelets of the infected animals. The high antimalarial activity displayed by the ethanolic fraction could be due to the presence of quercetin and kaempferol, as detected by high performance liquid chromatography (HPLC) analysis. The findings suggest that the fractions from C. odorata could serve as an alternative source of malaria therapy, particularly in sub-Saharan Africa.
Similar content being viewed by others
Introduction
Despite considerable advancements in the development of antimalarial therapies, the worldwide burden of malaria persists, as the disease incidence increased from 245 million cases in 2020 to 247 million cases in 2021 [1,2,3]. Regions of sub-Saharan Africa are most plagued by malaria; this is primarily due to the climatic conditions in this region, which foster the ideal habitat for the parasite's vector, female Anopheles mosquitoes, to reproduce and propagate. In addition, there has been an increase in parasite resistance to antimalarial drugs and vector resistance to insecticides and treated nets, thereby leading to high mortality, particularly among pregnant women and young children [1]. Four sub-Saharan nations—Nigeria, the Democratic Republic of the Congo, the United Republic of Tanzania, and Niger—accounted for 51.9% of malaria-related deaths in 2021 [3].
The fact that two effective antimalarial drugs, quinine, and artemisinin, were discovered in plants provides further proof of the significance of plants and natural products in the fight against malaria. According to Soni et al. [4] and Kumar et al. [5], traditional medicine is a significant and sustainable strategy to treat malaria, especially in low-income regions where drug resistance is common and access to effective anti-malarial drugs is limited within this region. Kasilo et al. [6] also highlighted the possibility of African traditional medicine making a substantial contribution to the achievement of universal health coverage (UHC), particularly in rural areas with limited access to modern healthcare facilities.
One of the adverse effects of malarial infection is the alteration in haematological parameters as studies have reported prevalence of malaria-associated anaemia [7,8,9]. Recent studies on malaria infected patients have linked the disease to alterations in haemoglobin concentration and red blood cell counts, due to the parasite’s ability to haemolyze red blood cells and suppress erythropoiesis in vertebrate hosts [8, 10]. Experimentally, alterations in haematological parameters such as, percentage haematocrit, mean corpuscular volume, red blood cell and white blood counts have established in malaria-infected mice models [11,12,13].
Some of the major plants identified by ethno-medicinal studies as anti-malarials, particularly in Africa, are Cymbopogon citratus (S), Morinda lucida L., Azadiratchta indica, Psidium guajava, Citrus aurantifolia, Vernonia amygdalina, Magnifera indica L., Persea americana Mill., Allium sativum, Jatropha curas, Allium cepa, and Cassia fistulosa [14,15,16,17,18]. A few ethnobotanical studies, particularly from Nigeria, also identified the unwanted weed Chromolaena odorata (L.) R.M. King & H. Rob., as a plant with potential antimalarial properties [16, 17].
A plethora of studies has validated the anti-malarial activities of different plants in mice models [19,20,21,22,23]. According to Nworgu et al. [23], the ethanolic extract of combined Cassia sieberiana and Chromolaena odorata significantly reduced parasitemia in mice infected with the chloroquine sensitive strains of Plasmodium berghei. Plant extracts from Cuscuta reflexa, Maesa lanceolata and Carica papaya [19,20,21]. In addition, treatment of malaria with extracts from plants such as Salacia nitida, Artemisia annua, and Senna alata, have been reported to reverse the haematological alterations associated with malaria infection in mice models [13, 24, 25]. However, fewer studies have validated the anti-malarial properties of C. odorata [22, 23]. Therefore, the present study evaluated the curative antimalarial properties of C. odorata in a mouse model of malaria. In addition, we identified the chemical constituents of the most potent antimalarial fraction using HPLC–UV.
Materials and methods
Plant preparation and extraction
Fresh C. odorata leaves (Fig. 1) were collected from a botanical garden in Ilorin, Kwara State, between March and June 2021. The leaves were authenticated by Mr. Bolu Ajayi, at the Department of Plant Biology, Faculty of Life Sciences, University of Ilorin, Nigeria. The authentication was registered under voucher number UILH/001/1281/2021. The name of the species was fully validated on http://mpns.kew.org/mpns-portal/?_ga=1.111763972.1427522246.1459077346. The leaves of C. odorata were harvested, washed with distilled water, and air-dried for 2 weeks. The evenly dried leaves were milled to a fine powder using an electrical grinder (Excella mixer grinder by Kanchan International Limited, Dabhel, Daman-396210).
Crude extraction
To simulate the traditional use of the leaves of C. odorata in the treatment of malaria in Nigeria, a crude extract of the leaves was obtained via aqueous extraction following the methods described by Yakubu et al. [26] and Sadiq et al. [27]. Using a solid to solvent ratio of 1:10 (w/v), 1000 g of the powdered C. odorata leaves were macerated in 10 L of distilled water and agitated for 24 h on a mechanical shaker at 200 rpm at 25 °C. The mixture was filtered using Whatman filter paper (No. 1), and the resulting filtrate was lyophilized in a freeze dryer (Mini Lyrotrap, LTE Scientific Limited, Greenfield, Oldham, United Kingdom).
Solvent partitioning
Solvent portioning was achieved following the method described by Zeleke et al. [28], with slight modifications. In brief, 180 g of crude aqueous extract was reconstituted in 450 mL of distilled water and stirred until a homogenous solution was obtained. The reconstituted aqueous extract was then transferred into a 1000 mL separating funnel, and 500 mL of 96.5% n-hexane was subsequently added and left to stand for 12 h to de-fat the crude extract. After 12 h, the aqueous layer was eluted, and the fatty layer was decanted and discarded. This process was repeated once, after which the defatted crude aqueous extract was transferred into a clean separating funnel, and solvent–solvent partitioning was carried out by using two solvents with different polarities: 100% ethyl acetate (4.4 polarity) and 96.5% ethanol (6.1 polarity).
Based on the polarities, the ethyl acetate fraction was obtained first, the defatted crude extract was transferred into a 1000 mL separating funnel, and 600 mL 100% ethyl acetate was added. The mixture was agitated and left for 12 h, after which the aqueous residual fraction (lower layer) and the ethyl acetate fraction (upper layer) were eluted into different beakers. This process was repeated by adding 400 mL of ethyl acetate to the aqueous residual fraction. After obtaining the ethyl acetate fraction, the ethanolic fraction was obtained using the same procedure. The three different fractions, the aqueous residual fraction (ARF), ethyl acetate fraction (EAF), and ethanolic fraction (EF), were concentrated at 40 °C using a water bath, weighed, and stored at 4 °C until further use.
Experimental animals
Forty-five (45) mice and five (5). Wistar albino rats were obtained from the animal holding facility of the Institute for Advanced Medical Research and Training (IAMRAT), University of Ibadan, Ibadan, Oyo state, Nigeria, after receiving ethical approval (LUAC/BCH/2022/005A) from the Landmark University Omu-Aran, Kwara State, Nigeria. The animals received humane care in compliance with the institution’s guidelines and criteria outlined in the National Institute of Health (NIH) Guidelines for the care and use of laboratory animals [29]. The animals were kept in well-ventilated plastic cages and housed at 27 °C and a 12-h day/night cycle. The experimental animals were fed commercial rat pellets and were given unlimited access to clean water.
Inoculum
Chloroquine-sensitive strains of P. berghei (ANKA) were obtained from the Institute for Advanced Medical Research and Training (IAMRAT), University College Hospital, Ibadan, Oyo state, Nigeria. Parasitized erythrocytes were obtained from an anaesthetized donor-infected mouse via cardiac puncture into a plain bottle containing acid citrate dextrose (ACD) and were serially diluted with normal saline to make a suspension containing 1 × 107 parasitized erythrocytes in every 2 mL of infected blood.
Standard drugs
The standard antimalarial drugs chloroquine phosphate (250 mg) and Coartem (20/120 mg Artemether and Lumefantrine) were manufactured by Jopan Pharmaceuticals Limited, Nigeria and Tuyil Pharmaceutical Limited, Nigeria, respectively.
In vitro quantitative phytochemical screening of the crude extract and solvent fractions of C. odorata
In vitro quantitative phytochemical screening of C. odorata aqueous extract, aqueous residual fraction, ethanolic and ethyl acetate fractions were evaluated by determining the total flavonoid and phenolic content; total flavonoid content; total antioxidant capacity; ferrous reducing capacity; nitric oxide scavenging capacity; and 2,2-diphenyl-1-picrylhydrazy (DPPH) radical scavenging power using the methods described by Elebiyo et al. [30]. To evaluate the In vitro bioactivity of the crude extract and fractions, stock solutions of the crude extract and fractions were prepared by dissolving 6 mg of the dried sample in 1 mL of dimethyl sulfoxide (DMSO). Varying concentrations of the plant extract (60–900 µg/mL) were then prepared from the stock solutions.
Lethality study and dose determination
The lethal dose of the crude aqueous extract of C. odorata was first established using five female Wistar rats following the up and down procedure described in OECD guideline 425 [31]. The selection of doses for the treatment was later calculated as one-fifth and one-twentieth of the LD50 (2000 mg/kg) using the formula presented below:
In vivo curative antimalarial screening of solvent fractions of the aqueous leaf extract of C. odorata
The antimalarial potential of the solvent fractions from the aqueous leaf extract of C. odorata was screened using a curative malarial test described by Baah et al. [32]. This was done to ascertain that the C. odorata leaves possess anti-malarial properties.
Experimental design and grouping
Forty-five (45) albino mice weighing between 18 and 22 g were used for curative antimalarial screening of the ethyl acetate (EAF), ethanolic (EF) and aqueous residual (ARF) fractions of the aqueous leaf extract of C. odorata using the treatment grouping presented in Table 1. Forty of the experimental animals were inoculated intraperitoneally with 0.2 mL of infected blood suspension. Seventy-two hours after inoculation with the parasite, parasitemia of the infected mice was established using blood smear microscopy. The infected animals were randomly distributed into eight (8) groups (Groups 2–9). The animals in group 2 served as the negative control group and received no form of treatment with the fractions or standard drugs throughout the experiment. From day 3 post-infection, the animals in groups 3–9 received oral administration of the different doses of fractions or standard drugs for 4 days. The animals in groups 3 and 4 served as the positive control groups and were treated with 10 mg/kg chloroquine and a combination of 1.4 mg/kg artemether and 8.75 mg/kg lumefantrine (Coartem), respectively. Group 5 animals received 100 mg/kg of the aqueous residual fraction, while groups 6 and 7 received 100 mg/kg and 400 mg/kg of the ethyl acetate fraction, respectively. Groups 8 and 9 received 100 mg/kg and 400 mg/kg ethanolic fraction, respectively (Table 1).
Microscopic determination of parasite burden in the blood
Parasitaemia levels in the mice were determined using a slightly modified method described Ounjaijean et al. [33]. Blood from the tails of the experimental mice at days 0, 3, 5, and 7 post-malarial infection was smeared on clean glass slides to make a thin film. The slides were then fixed with a few drops of methanol, air-dried and stained with 10% Giemsa solution prepared with phosphate buffer (pH 7.2). The slides were then air-dried for 30 min and rinsed with distilled water to remove excess stain and then air-dried for another 20 min before microscopic examination. The parasite burden in the blood of each mouse was evaluated from 5 randomly selected viewing fields under a magnification of × 100 objective lens using a light microscope. The percentage parasite burden in the blood and percentage mean parasitemia suppression were determined using the formula described by Baah et al. [32]:
Haematological screening
The full blood count of the infected animals was performed on the 7th day post-infection using an automated haematology analyser. Parameters evaluated included white blood cell (WBC), red blood cell (RBC), haemoglobin (HGB) and haematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular haemoglobin concentration (MCHC), red blood cell distribution width -CV (RDW-CV), red blood cell distribution width-SD (RDW-SD), platelet (PLT), mean platelet volume (MPV), Platelet Distribution Width (PDW), plateletocrit (PCT), lymphocytes (LYMPH) and neutrophils (NEUT).
High-performance liquid chromatography (HPLC) analysis
The phytochemical constituents of the most potent fraction of the crude extract were quantified by HPLC coupled to ultraviolet spectroscopy using the procedure described by Farag et al. [34]. A 99.8% acetonitrile (Sigma Aldrich. Burlington, Massachusetts, United States), and water were used as the mobile phase for elution on a µBONDAPAK C18 column (3.9 mm × 300 mm, 10-µm particle size, 70:30). The phytochemical constituents of 10.0 g of the ethanolic fraction of C. odorata were extracted using acetonitrile and stabilized using 99.7% ethyl acetate (Sigma Aldrich. Burlington, Massachusetts, United States), after which 5 µL was injected into the system and monitored at a flow rate of 2 mL/min. The UV detection was performed at 330 nm while peaks were labelled based on individual compounds' retention time using the Phytochemical Metabolite Library of Standards by IROA Technologies, with the technical support of Bato Chemical Laboratories Limited, Lagos State Nigeria (https://batochemlab.com/).
Data analysis
All data were analysed using GraphPad Prism 9.3. Descriptive statistics were used to examine the trends in each sample or experimental groups while one-way and two analysis of variances were used to establish the relationship across the different treatment groups. The variations in the observations across group were considered significant at p < 0.05 and 95% confidence level.
Results
Quantitative phytochemical screening of the crude extract and fractions of C. odorata
Total phenolic and total flavonoid contents
Phytochemical screening of the crude aqueous leaf extract of C. odorata and its solvent fractions revealed that the concentrations of flavonoids and phenolic compounds in the plant samples increased in a dose-dependent manner (Fig. 2). The crude extract and the EF and EAF of C. odorata had comparative phenolic contents of 6.82 ± 0.5, 7.18 ± 0.08 and 6.3 ± 0.56 g gallic acid equivalent (GAE)/100 g extract, respectively. ARF had the lowest phenolic content of 4.29 ± 0.06 g GAE/100 g extract. The total flavonoid content of the extract was 13.96 ± 0.11 g quercetin equivalent (QUE)/100 g extract in CE, 9.63 ± 0.51 g QUE/100 g extract in ARF, 13.29 ± 0.52 g QUE/100 g in EAF and 6.79 ± 0.64 g QUE/100 g in EF.
Total antioxidant capacity and ferric reducing power
The total antioxidant and ferric reducing antioxidant capacities of the crude extract and solvent fractions of C. odorata increased in a dose-dependent manner. As shown in Fig. 3, EF showed the highest total antioxidant capacity of 8.21 ± 0.07 g AA/100 g extract, followed by the crude extract, 6.19 ± 0.4 g AA/100 g extract; ARF, 4.08 ± 0.81 g AA/100 g extract; and EAF, 0.45 ± 0.05 g AA/100 g extract. In contrast, the crude extract and EAF showed comparative ferric antioxidant capacities of 2.93 ± 0.14 and 3.32 ± 0.038 g AA/100 g extract, respectively, while those of ARF and EF were lower at 1.43 ± 0.32 and 1.72 ± 0.17 g AA/100 g extract, respectively.
Scavenging properties for DPPH and nitric oxide free radicals
The DPPH scavenging power of the crude aqueous extract of C. odorata (74.41 ± 2.45% at 750 µg/mL) was significantly lower (p = 0.009) than that of ascorbic acid (99.44 ± 0.320% at 750 µg/mL). However, there was no significant difference in the DPPH scavenging power of ARF (95.07 ± 1.92, p = 0.999), EF (88.25 ± 3.38, p = 0.063) and EAF (96.64 ± 1.27, p = 0.126) relative to ascorbic acid at 750 µg/mL (Fig. 4). The nitric oxide scavenging power of EF (58.68 ± 2.63%, p = 0.999) and EAF (57.51 ± 0.835%, p = 0.069) also compared favourably with ascorbic acid (59.71 ± 0.41%) at 600 µg/mL. On the other hand, the NO scavenging properties of the crude extract (54.41 ± 0.95%, p = 0.011) and ARF (52.27 ± 1.09%, p = 0.008) were significantly lower than that of ascorbic acid.
Lethality dose of C. odorata aqueous leaf extract
The results from the lethality test revealed that 2000 mg/kg of the crude extract did not induce adverse reactions or significant death (80% survival rates) in the experimental animals throughout the 14-day observation period (Table 2).
Curative antimalarial activity of the aqueous residual, ethyl acetate and ethanolic fractions of C. odorata aqueous leaf extract
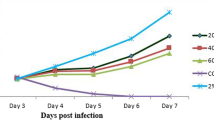
The data presented in Fig. 5A revealed no significant difference in the parasite burden of all the animals infected with P. berghei at day 3 post-infection. However, after a 4-day treatment with the different fractions of C. odorata or standard drugs, a significant decrease in parasite burden was observed in the RBCs of infected mice treated with chloroquine (0.6 ± 0.10%, p = 0.019), Coartem (0.47 ± 0.29%, p = 0.011)), ARF 100 mg/kg (10.23 ± 1.38%, p = 0.0007), EAF 100 mg/kg (11.07 ± 2.47%, p = 0.0044), EAF 400 mg/kg (5.5 ± 0.50, p = 0.0025), EF 100 mg/kg (8.25 ± 1.95%, p = 0.024) and EF 400 mg/kg (1.50 ± 0.5%, p = 0.003) compared with the negative control (62.0 ± 1.0%). Chloroquine, Coartem and EF 400 mg/kg showed the highest parasite suppression activity of 98.6%, 98.8% and 97.6%, respectively (Fig. 5B). The percentages of mean parasite suppression for the other treatment groups were 75.8%, 70.9%, 86.9% and 86.7% for ARF 100 mg/kg, EAF 100 mg/kg, EAF 400 mg/kg and EF 100 mg/kg, respectively.
Effect of the aqueous residual, ethyl acetate and ethanolic fraction on the haematological indices of P. berghei-infected mice
Effect of C. odorata fractions on red blood cell indices of malaria-infected mice
The data presented in Fig. 6A show that treatment with the C. odorata fractions or standard drugs significantly mitigated the effect of malaria infection on the red blood cells of infected mice; the RBC count increased in infected animals treated with chloroquine (5.75 ± 0.00, p = 0.0053), ARF 100 mg/kg (5.27 ± 0.77, p = 0.03), EAF 100 mg/kg (5.65 ± 0.21, p = 0.0079), EAF 400 mg/kg (6.38 ± 0.15, p = 0.0008) and EF 400 mg/kg (5.47 ± 0.07, p = 0.015) compared with the untreated infected animals (3.67 ± 0.06). There was no significant difference in the RBC count of the noninfected mice (7.070 ± 0.18) and the animals treated with chloroquine (p = 0.084), EAF 100 mg/kg (0.058) and EAF 400 mg/kg (p = 0.647). In addition, the effect of all the plant fractions on the RBC count of malaria-infected mice also compared favourably with chloroquine.
Effect of standard drugs and C. odorata fractions on the red blood cell indices of P. berghei-infected mice. The data are the mean of three replicates ± standard deviation (SD). All measures were considered significant at p < 0.05. * represents outcomes significantly different from control; #represents outcomes significantly different from negative control; +represents outcomes significantly different from chloroquine-treated group; &represents significantly different from Coartem-treated group. ARF represents the aqueous residual fraction; EAF represents the ethyl acetate fraction; EF represents the ethanolic fraction
There was a significant reduction in the haematocrit levels in the untreated animals infected with P. berghei (18.95 ± , 0.07, p = 0.0004) compared with the uninfected animals (36.40 ± 1.41), as shown in Fig. 6C. This alteration was significantly mitigated by the administration of chloroquine (29.95 ± 0.34, p = 0.01), 100 mg/kg EAF (28.00 ± 1.41, p = 0.033) and 400 mg/kg EAF (29.45 ± 1.768, p = 0.0137). The negative control group (5.95 ± 0.35, p = 0.009) also showed a significant decrease in haemoglobin count when compared with the uninfected control group (11.15 ± 0.35). However, there was no significant difference in the haemoglobin count of the animals treated with chloroquine (9.45 ± 0.07, p = 0.718), Coartem (7.3 ± 0.57, p = 0.052), ARF 100 mg/kg (7.3 ± 2.3, p = 0.052), EAF 100 mg/kg (8.5 ± 0.98, p = 0.265), EAF 400 mg/kg (9.2 ± 0.99, p = 0.582) and EF 400 mg/kg (8.4 ± 0.14, p = 0.233) when compared with the uninfected control group (Fig. 6D). The data presented in Fig. 6F indicated that malaria infection caused a significant increase in the mean corpuscular volume in the negative control group (50.1 ± 1.27 p = 0.0014) compared with the uninfected control group (41.2 ± 1.34). However, treatment with standard drugs or the fractions of the aqueous extract of C. odorata did not lead to any significant ameliorative effect on the MCV of the infected animals. In addition, there was no significant alteration in the red blood cell distribution width –CV (RDW-CV) and mean corpuscular haemoglobin concentration (MCHC) of all the animals in the treatment groups when compared with the control (Fig. 6B and E).
Effect of C. odorata fractions on white blood cell indices of malaria-infected mice
The data presented in Fig. 7A show a significant increase (p < 0.0001) in the white cell count of animals infected with P. bergehi (4.55 ± 0.21) in comparison with the control (14.50 ± 2.1). This alteration was significantly ameliorated by treatment with chloroquine (3.9 ± 0.14, p < 0.0001), Coartem (9.35 ± 0.25, p = 0.013), EAF 400 mg/kg (9.05 ± 0.79, p = 0.009) and EF 400 mg/kg (10.0 ± 0.40, p = 0.03). The percentage of lymphocytes was also significantly increased (p = 0.03) in animals infected with P. berghei (76.0 ± 4.00) compared to the control animals (56.50 ± 2.12). Treatment with chloroquine (60.00 ± 0.0, p = 0.07), Coartem (63.00 ± 9.8, p = 0.19), ARF 100 mg/kg (69.00 ± 1.4, p = 0.78), EAF 400 mg/kg (62.50 ± 3.53, p = 0.16) and EF 400 mg/kg (65.00 ± 0.0, p = 0.33) caused a slight decrease in the percentage of lymphocytes in the infected animals (Fig. 7B). As shown in Fig. 7C, in contrast to the observed percentage of lymphocytes, malarial infection resulted in a significant decrease in neutrophils (21.50 ± 4.95, p = 0.03) compared with the control (42.00 ± 2.28), which was significantly improved by treatment with chloroquine (44.45 ± 6.36, p = 0.01).
Effect of standard drugs and C. odorata fractions on the white blood cell indices of P. berghei-infected mice. The data are the mean of three replicates ± standard deviation (SD). All measures were considered significant at p < 0.05. * represents outcomes significantly different from control; #represents outcomes significantly different from negative control; +represents outcomes significantly different from chloroquine-treated group; &represents significantly different from Coartem-treated group. ARF represents the aqueous residual fraction; EAF represents the ethyl acetate fraction; EF represents the ethanolic fraction
Effect of C. odorata fractions on platelet indices of malaria-infected mice
The data presented in Fig. 8A show that the platelet counts of animals infected with malaria parasites were significantly reduced relative to the control. However, the effects were significantly improved by treatment with chloroquine, Coartem, 100 mg/kg EAF and 400 mg/kg EF. Similarly, the plateletcrit of animals infected with malaria parasites was significantly reduced compared with the control (Fig. 8B). Treatment with chloroquine, coartem and EAF 400 mg/kg significantly ameliorated the PCT reduction. There was no significant alteration in the mean platelet volume (Fig. 8C) or the platelet distribution width (Fig. 8D) following infection with the malaria parasite.
Effect of standard drugs and C. odorata fractions on the platelet indices of P. berghei-infected mice. The data are the mean of three replicates ± standard deviation (SD). All measures were considered significant at p < 0.05. * represents outcomes significantly different from control; #represents outcomes significantly different from negative control; +represents outcomes significantly different from chloroquine-treated group; &represents significantly different from Coartem-treated group. ARF represents the aqueous residual fraction; EAF represents the ethyl acetate fraction; EF represents the ethanolic fraction
Chemical fingerprint of the ethanolic fraction of C. odorata aqueous leaf extract
HPLC screening of the ethanolic fraction of the C. odorata aqueous extract identified thirteen bioactive components, with quercetin (41.5%), kaempferol (15.9%), naringin (14.4%) and chalcone having the highest concentrations (Fig. 9 and Table 3).
Discussion
Malaria is a global health challenge with endemicity in sub-Saharan Africa, where there are multiple drug-resistant strains and limited access to modern health care facilities, especially in rural areas [1, 2]. These factors underscore efforts to integrate botanical antimalarial molecules into the health care system, as this could facilitate sustainable development goal 3.8, which targets attaining universal health coverage for all [6]. Considering this, the present study evaluated the curative antimalarial properties of C. odorata, a relatively abundant but underexplored plant in Africa.
The quantitative phytochemical screening of the aqueous crude extract of C. odorata and its solvent fractions revealed richness in flavonoids rather than phenols, as observed from the abundance of quercetin, kaempferol, naringenin and luteolin in the ethanolic fraction of the plant extract. This finding corresponds with previous studies, which suggest that the methanolic leaf extract of C. odorata contains higher concentrations of flavonoids than phenolics [35]. The lethality study conducted showed that the median lethal dose of the crude extract was higher than 2000 mg/kg. However, due to the extremely low yield of the aqueous residual fraction obtained during solvent partitioning (8.66%), the highest dosage for this fraction was pegged at 100 mg/kg to prevent any adverse reactions in the animals.
Furthermore, our findings in this study revealed that the ethyl acetate, ethanolic and aqueous residual fractions of the aqueous extract of C. odorata possess highly active antimalarial properties with a percentage of chemo-suppression between 75.8–97.6%. According to Gathirwa et al. [36] and Afolayan et al. [37], botanical samples showing chemo-suppression of the Plasmodium parasite of 60% or more are strongly active, 30% to 60% are moderately active, and 30% or less are weakly active. Among the plant extracts, treatment with 400 mg/kg ethanolic fraction resulted in the highest decrease in parasite load (97.6%), which was comparable to the activities of the conventional drugs chloroquine (98.6%) and Coartem (98.8%). A similar study conducted by Nworgu et al. [22] revealed that 250, 500 and 1000 mg/kg ethanolic extracts of C. odorata induced 84.81%, 90.70%, and 95.63% reductions in the parasite burden of mice infected with P. berghei, respectively.
The anti-malarial activities elicited by the solvent fractions of C. odorata leaf extract is likely due to the abundance of flavonoids such as quercetin, kaempferol, naringenin and luteolin. Quercetin and kaempferol has been reported to possess moderate anti-malarial activity which is elicited by the compounds ability to downregulate the activity of tumour necrotic factor -α, thus, reducing malaria severity and malaria-associated tissue damage [38,39,40].
Malaria infection and subsequent treatment have been associated with significant alterations in haematological parameters [11,12,13, 41]. The results of this study showed that mice infected with the chloroquine-resistant strain of P. berghei had considerably lower levels of anaemia-related indices: haemoglobin count, haematocrit level, and red blood cell count. According to the study conducted by Mwaiswelo et al. [41], most adult and paediatric patients infected with P. vivax and P. falciparum frequently have low RBC, haematocrit, and haematology counts. Similar haematological alterations in have also been observed in mouse models [11,12,13]. The Plasmodium spp. elicits its infectious activity by invading red blood cells causing the premature destruction of RBCs resulting in anaemia [42]. In this study, treatment with the different solvent fractions of C. odorata mitigated the alterations in the red blood cell indices of the infected animals which suggests that the solvent fractions of C. odorata elicited its anti-malarial activity by preventing the invasion of red blood cells by malarial parasite.
Malaria infection also significantly increased the white blood cell count and percentage of lymphocytes and decreased the percentage of neutrophils. This is similar to the study conducted by Ounjaijean et al. [43], which showed that WBC counts are elevated in mice infected with P. berghei. The increased lymphocytes seen in the current study could be a result of the immune system's efforts to combat the malaria parasite. On the other hand, a depletion of neutrophils may indicate compromise of the immune system by malaria infection, as neutrophils are involved in the phagocytosis of Plasmodium spp. [44]. The mitigative effect of treatment of malarial infected mice with the solvent fractions of C. odorata suggests that the plant inhibits the proliferation of plasmodium spp. in the infected host which reduces the immune responses associated with malaria infection.
Platelets have been identified as one of the major host defence systems against the malaria parasite [45]. The current study noted a substantial decline in platelet and plateletcrit counts in P. berghei-infected animals. Series of literature have shown that platelet counts is usually depleted in both humans and mouse models during infection with Plasmodium spp. [46,47,48]. According to the studies conducted by Zhou et al. [49], infection with Plasmodium spp. in mice inhibits the secretion of anti-platelet antibody which prevents the destruction of the malaria parasite by the platelet sells of the host organism which explains the depletion in the platelet count of the mice infected with P. berghei as observed from this study. The improvement of these haematological alterations following administration of the various fractions of the aqueous extract of C. odorata, particularly the ethanolic fraction at 400 mg.kg, suggests that the bioactive compounds of the plant extract and fraction could have the ability to stimulate platelet production or prevent the production of anti-platelet antibody during malaria infection.
This could be due to the synergistic interactions among the bioactive components of the extract. HPLC screening of the ethanolic fraction revealed that the extract was most abundant in quercetin, kaempferol, naringenin and chalcone. Numerous studies have associated the antimalarial activities of botanical drugs with the presence of quercetin and kaempferol [38, 50,51,52].
Conclusion
The findings from the current study suggest that C. odorata fractions could be used as an adjuvant and/or alternative to treat malaria, particularly in sub-Saharan Africa, where most of the population cannot afford or do not have access to primary health care services. The ethanolic fraction of C. odorata had a comparative antimalarial activity to standard drugs chloroquine and artermether-lumenfantrine, which suggests that the fraction contains bioactive molecules with potent anti-malarial properties. The fractions of C. odorata leaf extract also mitigated the haematological alterations induced by malaria infection. Additionally, due to synergistic interaction between among the bioactive compounds in the fractions using botanical drug may be a more effective treatment option given that the malaria parasite is constantly evolving and acquiring resistance to available drugs. The synergistic effects of these bioactive components could facilitate attack of multiple parasitic pathways, making it difficult for the parasite to survive within its host.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
References
Oladipo HJ, Tajudeen YA, Oladunjoye IO, Yusuff SI, Yusuf RO, Oluwaseyi EM, et al. Increasing challenges of malaria control in sub-Saharan Africa: Priorities for public health research and policymakers. Ann Med Surg. 2022;81:104366.
Sarpong E, Acheampong DO, Fordjour GNR, Anyanful A, Aninagyei E, Tuoyire DA, et al. Zero malaria: a mirage or reality for populations of sub-Saharan Africa in health transition. Malar J. 2022;21(1):314.
W.H.O. World malaria report 2022: World Health Organization. 2022.
Soni R, Shankar G, Mukhopadhyay P, Gupta V. A concise review on Artemisia annua L.: A major source of diverse medicinal compounds. Ind Crops and Prod. 2022;184:115072.
Kumar V, Garg V, Dureja H. Role of traditional herbal medicine in the treatment of malaria. TMR Modern Herb Med. 2022;5(4):20.
Kasilo OMJ, Wambebe C, Nikiema J-B, Nabyonga-Orem J. Towards universal health coverage: advancing the development and use of traditional medicines in Africa. BMJ Glob Health. 2019;4(Suppl 9):e001517.
Nwaneri D, Oladipo O, Ifebi E, Oviawe O, Asemota O, Ogboghodo B, et al. Haematological parameters and spleen rate of asymptomatic and malaria negative children in Edo south district, Nigeria. Annals of Global Health. 2020;86(1):62.
Saleh AG, Shehab AY, Abdul-Ghani R, Allam AF, Osman MM, Ibrahim HS, et al. Key cytokines and hematological parameters in patients with uncomplicated falciparum malaria in Hodeidah. Yemen Cytokine. 2023;164:156156.
Yusuf F, Sadau Y, Natasha P, Lai I. Effects of malaria parasitemia on haematological parameters of pregnant women attending gombe specialist hospital. Bima J Sci Technol. 2023;7(01):190–9.
White NJ. Anaemia and malaria. Malar J. 2018;17(1):1–17.
Azizah IH, Bahi SF, Arwati H, Mufassirin M. Correlation of Parasitaemia and Anemia in Mice Infected with Plasmodium berghei ANKA. Med Lab Technol J. 2023;9(1):22–8.
Asemota K, Omoirri M, Iloh S, Obi E. Haematological Changes in Co-Administration of Artemisinin and Anti-Oxidants to Immuno-Compromised Mice. South Asian J Parasitol. 2019;2(2):1–10.
Ekam V, Ukpanukpong R, Odio S, Ihe S, Ekam N. Haematological changes in malaria infested Wistar albino rats treated with the crude extract of Artemisia Annua and Artemisinin combination therapy. Intern J Med Res. 2019;4(3):80–3.
Babalola AS, Idowu OA, Ademolu KO, Olukunle J, Rahman SA. Antiplasmodial activities and abortifacient properties of three commonly used African indigenous anti-malarial plants in Plasmodium berghei infected pregnant mice: implication for maternal and fetal health. Bull Nat Res Centre. 2020;44(1):1–12.
Nigussie G, Wale M. Medicinal plants used in traditional treatment of malaria in Ethiopia: a review of ethnomedicine, anti-malarial and toxicity studies. Malar J. 2022;21(1):262.
Oladeji OS, Oluyori AP, Bankole DT, Afolabi TY. Natural Products as Sources of Antimalarial Drugs: Ethnobotanical and Ethnopharmacological Studies. Scientifica (Cairo). 2020;2020:7076139.
Olorunnisola OS, Adetutu A, Balogun EA, Afolayan AJ. Ethnobotanical survey of medicinal plants used in the treatment of malaria in Ogbomoso Southwest Nigeria. J Ethnopharmacol. 2013;150(1):71–8.
Tonny A-A, Heron RB, Susana OM, Mary-Ann A, Frederick A, Alexis CS, et al. Ethnobotanical studies of medicinal plants used in traditional treatment of malaria by some herbalists in Ghana. J Med Plants Res. 2019;13(16):370–83.
Eze CC, Agbo MC, Ozioko CA, Ugwu PC, Obayiuwana AC. Antimicrobial Activities and Malaria Parasite Clearance of Crude Extract of Carica papaya Seeds in Mice Infected with Plasmodium berghei. Trop J Nat Prod Res. 2022;6(9):1492–6.
Ojha SB, Sah RK, Madan E, Bansal R, Roy S, Singh S, et al. Cuscuta reflexa Possess Potent Inhibitory Activity Against Human Malaria Parasite: An In vitro and In Vivo Study. Curr Microbiol. 2023;80(5):189.
Tekalign E, Tadege G, Fisseha N, Nureye D. Suppressive, Curative, and Prophylactic Effects of <i>Maesa lanceolata</i> Forssk. against Rodent Malaria Parasite <i>Plasmodium berghei</i>. BioMed Res Intern. 2022;2022:8901555.
Nworgu C, Elele K, Adikwu E. Toxicity and Antiplasmodial Assessments of Chromolaena odorata Leaf Extract on Plasmodium berghei-Infected Mice. J Infect Dis Epidemiol. 2022;8:276.
Nworgu CO, Nyong A, Needom PB. Antimalarial activity of the combined cassia sieberiana and chromolaena odorata ethanolic leaf extract on plasmodium berghei infected mice. Ignatius Ajuru Univ J Appl Environ Biol. 2023;1(1):50–8.
Nwiloh BI, Uwakwe AA. Ameliorating effects of ethanol extract of root bark of Salacia nitida on blood electrolyte and renal perturbations in Plasmodium berghei-infected mice. Braz J Pharm Sci. 2023;59:e20229.
Atanu FO, Rotimi D, Ilesanmi OB, Al Malki JS, Batiha GE, Idakwoji PA. Hydroethanolic Extracts of Senna alata Leaves Possess Antimalarial Effects and Reverses Haematological and Biochemical Pertubation in Plasmodium berghei-infected Mice. J Evid Based IntegMed. 2022;27:2515690X221116407.
Yakubu MT, Akanji MA, Oladiji AT. Evaluation of antiandrogenic potentials of aqueous extract of Chromolaena odoratum (L.) K. R. leaves in male rats. Andrologia. 2007;39(6):235–43.
Sadiq MB, Tharaphan P, Chotivanich K, Tarning J, Anal AK. In vitro antioxidant and antimalarial activities of leaves, pods and bark extracts of Acacia nilotica (L.) Del. BMC Complement Altern Med. 2017;17(1):372.
Zeleke G, Kebebe D, Mulisa E, Gashe F. In Vivo Antimalarial Activity of the Solvent Fractions of Fruit Rind and Root of Carica papaya Linn (Caricaceae) against Plasmodium berghei in Mice. J Parasitol Res. 2017;2017:3121050.
National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals. 8th ed. Washington: National Academies Press; 2011.
Elebiyo TC, Olori OO, Rotimi DE, Al-Megrin WAI, De Waard M, Alkhuriji AF, et al. Chemical fingerprinting, comparative In vitro antioxidant properties, and biochemical effects of ginger and bitterleaf infusion. Biomed Pharmacother. 2022;155:113788.
OECD. Test No. 425: Acute oral toxicity: Up-and-down procedure. OECD Guidelines for the Testing of Chemicals, Section 4: Health Effects. 2008:1–27.
Baah MK, Mensah AY, Asante-Kwatia E, Amponsah IK, Forkuo AD, Harley BK, et al. In Vivo Antiplasmodial Activity of Different Solvent Extracts of Myrianthus libericus Stem Bark and Its Constituents in Plasmodium berghei-Infected Mice. Evid Based Complement Alternat Med. 2020;2020:8703197.
Ounjaijean S, Rattanatham R, Somsak V, Boonhoh W, Surinkaew S. Gymnema inodorum Leaf Extract Improves Cardiac Function in Experimental Mice Infected with Plasmodium Berghei. J Evid Based IntegMed. 2023;28:2515690X221150526.
Farag RS, Abdel-Latif MS, Abd El Baky HH, Tawfeek LS. Phytochemical screening and antioxidant activity of some medicinal plants’ crude juices. Biotechnol Rep. 2020;28:e00536.
Saravanan K, Sivaramakrishnan T, Praveenraj J, Kiruba-Sankar R, Suma D, Devi V, et al. Phytochemical, Antioxidant and Antibacterial Activity of Chromolaena odorata from Andaman Islands. India Andaman Sci Assoc. 2019;24(2):123–30.
Gathirwa JW, Rukunga GM, Mwitari PG, Mwikwabe NM, Kimani CW, Muthaura CN, et al. Traditional herbal antimalarial therapy in Kilifi district. Kenya J Ethnopharmacol. 2011;134(2):434–42.
Afolayan FID, Adegbolagun O, Mwikwabe NN, Orwa J, Anumudu C. Cytokine modulation during malaria infections by some medicinal plants. Sci African. 2020;8:e00428.
Ali AH, Sudi S, Shi-Jing N, Hassan WRM, Basir R, Agustar HK, et al. Dual Anti-Malarial and GSK3β-Mediated Cytokine-Modulating Activities of Quercetin Are Requisite of Its Potential as a Plant-Derived Therapeutic in Malaria. Pharmaceuticals. 2021;14(3):248.
Alkandahri MY, Berbudi A, Subarnas A. Evaluation of Experimental Cerebral Malaria of Curcumin and Kaempferol in Plasmodium berghei ANKA-Infected Mice. Pharmacogn J. 2022;14(6s):905–11.
Liana L, Rizal R, Widowati W, Fioni F, Akbar K, Fachrial E, et al. Antioxidant and anti-hyaluronidase activities of dragon fruit peel extract and kaempferol-3-o-rutinoside. Jurnal Kedokteran Brawijaya. 2019;30(4):247–52.
Mwaiswelo RO, Mmbando BP, Chacky F, Molteni F, Mohamed A, Lazaro S, et al. Malaria infection and anemia status in under-five children from Southern Tanzania where seasonal malaria chemoprevention is being implemented. PLoS ONE. 2021;16(12):e0260785.
Lakkavaram A, Lundie RJ, Do H, Ward AC, de Koning-Ward TF. Acute Plasmodium berghei mouse infection elicits perturbed erythropoiesis with features that overlap with anemia of chronic disease. Front Microbiol. 2020;11:702.
Ounjaijean S, Sukati S, Somsak V, Sarakul O. The Potential Role of <i>Gymnema inodorum</i> Leaf Extract Treatment in Hematological Parameters in Mice Infected with <i>Plasmodium berghei</i>. J Trop Med. 2021;2021:9989862.
Aitken EH, Alemu A, Rogerson SJ. Neutrophils and malaria. Front Immunol. 2018;9:3005.
Darling TK, Schenk MP, Zhou CC, Maloba FM, Mimche PN, Gibbins JM, et al. Platelet α-granules contribute to organ-specific pathologies in a mouse model of severe malaria. Blood Adv. 2020;4(1):1–8.
Mohamedahmed KA, Ahmed ZA, Nour BYM, Abakar AD, Babker AM. Impact of sever Plasmodium falciparum infection on platelets parameters among Sudanese children living in Al-Jazira state. 2020.
Okpokam DC, Echonwere-Uwikor BE, Umoumo RI, Ochigbo SO. Platelet count and platelet indices of symptomatic and asymptomatic children infected by malaria in Calabar Metropolis, Nigeria. Intern J Res Med Sci. 2022;10(5):1016.
Abubakar F, Oloyede O. Prosopis africana stem bark extract: Effects on parasitaemia and haematological parameters of Plasmodium berghei-infected mice. Biokemistri. 2021;32(3):177–84.
Zhou M, Xie J, Kawase O, Nishikawa Y, Ji S, Zhu S, et al. Characterization of anti-erythrocyte and anti-platelet antibodies in hemolytic anemia and thrombocytopenia induced by Plasmodium spp. and Babesiaspp. infection in mice. Front Cell Infect Microbiol. 2023;13:1143138
Ounjaijean S, Benjasak N, Sae-Lao S, Somsak V. Kaempferol addition increases the antimalarial activity of artesunate in experimental mice. J Trop Med. 2020;2020:6165928.
Somsak V, Damkaew A, Onrak P. Antimalarial activity of kaempferol and its combination with chloroquine in Plasmodium berghei infection in mice. J Pathogens. 2018;2018:3912090.
Wong PP, Demircioglu F, Ghazaly E, Alrawashdeh W, Stratford MR, Scudamore CL, et al. Dual-action combination therapy enhances angiogenesis while reducing tumor growth and spread. Cancer Cell. 2015;27(1):123–37.
Acknowledgements
The authors would like to thank the technical personnel of Landmark University's Department of Biochemistry in Omu-Aran, Kwara State, Nigeria, for their assistance. In addition, the authors appreciate Mrs. Thomas Victoria, Drug and Cancer Research Unit, Institute for Advanced Medical Research and Training (IAMRAT), University College Hospital, Ibadan, Oyo State for technical assistance.
Funding
None.
Author information
Authors and Affiliations
Contributions
TCE conceived the study, carried out the experiment, gathered and analysed the data, and wrote the first draft of manuscript. OMO and OSA conceived study, supervised the experiments, data collection and analysis, edited manuscript and approved the final draft.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Ethical approval was obtained from the Landmark University ethics committee in Omu-Aran, Kwara State, Nigeria, with the approval number LUAC/BCH/2022/005A. All methods involving animal housing and handling were in accordance with the guidelines and regulations of the ARRIVE guidelines (https://arriveguidelines.org). The methods of plant collection were also in accordance with institutional, national, and international guidelines.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Elebiyo, T.C., Oluba, O.M. & Adeyemi, O.S. Anti-malarial and haematological evaluation of the ethanolic, ethyl acetate and aqueous fractions of Chromolaena odorata. BMC Complement Med Ther 23, 402 (2023). https://doi.org/10.1186/s12906-023-04200-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12906-023-04200-8