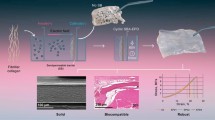
Abstract
Background
To provide a reference for clinical selection of collagen membranes by analyzing the properties of three kinds of collagen membranes widely used in clinics: Bio-Gide membrane from porcine dermis (PD), Heal-All membrane from bovine dermis (BD), and Lyoplant membrane from bovine pericardium (BP).
Methods
The barrier function of three kinds of collagen membranes were evaluated by testing the surface morphology, mechanical properties, hydrophilicity, and degradation rate of collagen membranes in collagenase and artificial saliva. In addition, the bioactivity of each collagen membrane as well as the proliferation and osteogenesis of MC3T3-E1 cells were evaluated. Mass spectrometry was also used to analyze the degradation products.
Results
The BP membrane had the highest tensile strength and Young’s modulus as well as the largest water contact angle. The PD membrane exhibited the highest elongation at break, the smallest water contact angle, and the lowest degradation weight loss. The BD membrane had the highest degradation weight loss, the highest number of proteins in its degradation product, the strongest effect on the proliferation of MC3T3-E1 cells, and the highest expression level of osteogenic genes.
Conclusions
The PD membrane is the best choice for shaping and maintenance time, while the BD membrane is good for osteogenesis, and the BP membrane is suitable for spatial maintenance. To meet the clinical requirements of guided bone regeneration, using two different kinds of collagen membranes concurrently to exert their respective advantages is an option worth considering.
Similar content being viewed by others
Introduction
Bone defects and insufficient bone remain important challenges in dental implant restoration [1, 2] because they prevent dental implants from meeting the required bone width and height, thus hindering the application of dental implants. Guided bone regeneration (GBR) uses bone grafts or osteoinduction materials to promote bone reconstruction and stable blood clot formation [3]. In GBR, a barrier membrane is used to form a secluded space in the bone defect area to prevent fast-growing connective-tissue cells and epithelial cells from entering the defect area, thus allowing slow-growing osteoblasts to proliferate, differentiate, and mineralize in the defect area for bone tissue regeneration. Therefore, barrier membranes need to exhibit biocompatibility, maneuverability, tissue integrity, and bioactivity to meet the requirements of the clinical application [4, 5].
At present, commercial barrier membranes can be divided into two types: non-resorbable membranes, such as expanded-polytetrafluoroethylene (e-PTFE) membrane, and resorbable membranes, such as collagen membranes (Bio-Gide, Heal-All, etc.) and synthetic membranes (PLGA, PLDLA, etc.) [4]. Non-resorbable membranes may damage the regenerated tissue and burden the patient owing to unavoidable exposure to the oral enviroment and the inconvenience of a subsequent operation [6, 7]. Therefore, resorbable membranes are currently the most popular material because of their excellent biocompatibility and tissue integration, and, importantly, subsequent operations are unnecessary because they are biodegradable [2].
Commercial resorbable membranes, such as Bio-Gide membrane from porcine dermis (PD), Heal-All membrane from bovine dermis (BD), and Lyoplant membrane from bovine pericardium (BP), are predominantly based on collagen. The biological properties of collagen membranes vary significantly depending on the source and structure of the collagen. The microstructure of collagen membranes, such as fiber shape and direction, in addition to the source of the collagen and hydrophilicity of the membrane can affect the migration, proliferation, and differentiation of osteoblasts [8]. Moreover, GBR osteogenesis largely depends on the patient’s potential for bone healing (such as age and general nutritional status) and local conditions (such as blood vessels and embryological origin of bone) [9]. Therefore, to achieve desirable GBR outcomes, the biological properties of the collagen membrane and the patient’s bone healing ability need to be considered when selecting the collagen membrane. A large number of clinical data show that it takes at least three months to completely regenerate the bone in the jaw, which depends on jaw-intramembranous ossification, active angiogenesis of the jaw and its surrounding soft tissue, mechanical condition of the jaw, and effective stress shielding of the barrier membrane. Ideally, the degradation rate of the collagen membrane should match the rate of bone formation [4], and the collagen membrane should be effectively shielded from the rapidly proliferating epithelium during wound healing after surgery [3]. There are many kinds of commercially available collagen membranes, which makes it difficult to choose suitable membranes for individuals with different bone healing abilities and bone defects, especially in the absence of an index. For example, to support space maintenance for patients presenting with bone defects below Class 3, selecting collagen membranes with superior mechanical properties is preferred [10]. Therefore, it is necessary to fully characterize and compare the physical, mechanical, and biological properties of commonly used collagen membranes to provide a reference for the selection of collagen membranes.
In this study, the physical properties (surface morphology and hydrophilicity), mechanical properties, and degradation rate of three common collagen membranes were characterized. Additionally, the effects of degradation products (DPs) on the proliferation and differentiation of MC3T3-E1 cells were determined using assays. Furthermore, the composition of the DPs of the collagen membranes was also identified.
Materials and methods
Collagen membrane materials
The collagen membranes selected in this study meet the following criteria: The osteogenic ability of the membrane has been verified in animal experiments and clinical studies; the membrane has been widely used in clinical practice; and the collagen source or manufacturing process of the collagen membranes is different. On this basis, three kinds of collagen membranes were selected for this study: porcine dermis membrane (PD), bovine pericardium membrane (BP), and bovine dermis membrane (BD) (Table 1).
Surface morphology
The surface morphology of the three membranes was analyzed using scanning electron microscopy (SEM, Hitachi S-4800, Hitachi Co., Ltd., JPN). The membranes were cut to a size of 1 mm × 1 mm. After vacuum gold plating, the micro-morphology of the smooth and rough surfaces were observed and recorded with a scanning electron microscope operating at an acceleration voltage of 1 kV [11].
Mechanical property
Each collagen membrane was divided into two groups: dry and wet. Collagen membranes in the wet group were soaked in artificial saliva (Solarbio, Beijing, China) for 2 min before testing. The thickness of the sample was measured with a leather thickness measuring instrument (Kunshan Xiangke Testing Instrument Co., Ltd., Kunshan, China), and six test points were randomly selected on each membrane to calculate the average value. The tensile strength, elongation, and Young’s modulus of the collagen membranes were evaluated using a universal testing machine (ZwickRoell GmbH & Co.KG, Ulm, Germany). Each membrane was cut into 20 mm × 3.0 mm specimens and mounted on the gripping unit of the tester. A tensile force was applied at a crosshead speed of 5 mm/min until the specimen broke (n = 5). All measurements were performed at room temperature. The Young’s modulus of the membrane is the slope of the linear elastic part of the stress–strain curve. The tensile strength and elongation at break were calculated using the following equations: Tensile strength (MPa) = Maximum load at break (N)/Cross-sectional area (m2) [11] and Elongation (%) = (Break length − Initial length)/Initial length × 100 [12].
Hydrophilic property
The hydrophilicity of the front and back sides of the collagen membranes was evaluated by measuring the contact angles. In summary, 3 μL of distilled H2O was dropped on the surface of the collagen membrane, and then the contact angle was measured with an optical contact angle measuring instrument (Theta Flex, Biolin Technology, Gothenburg, Sweden) at room temperature. Measurements were repeated for a total of 5 times, and the results were averaged.
Degradation ratio assay
The three types of collagen membranes were sheared into circle (diameter 13 mm) and transferred to a 15-mL centrifuge tube. Then, type I collagen solution (final concentration of 10 U/mL, Sigma-Aldrich, Saint Louis, USA) and artificial saliva (Solarbio, Beijing, China) were added, with a sterilized filter, giving a final volume of 10 mL (10 specimens in each group). The reaction mixture was incubated at 37 °C, 130 rpm. The residual specimens were taken out on days 7, 14, and 21, then freeze-dried, and finally weighed [13]. The degradation ratio was expressed as the percentage of weight loss, calculated using the following equation: Weight loss (%) = (W0 − Wd)/W0, where W0 is the actual weight before degradation and Wd is the dry weight after degradation.
Preparation of Degradation Products (DPs)
For subsequent assays, the DPs of the collagen membranes were prepared as follows. Briefly, the collagen membranes (5 mm in diameter) were enzymatically hydrolyzed with or without 20 U/mL collagenase solution, and PBS controls were established (triplicate samples in each group). After incubation at 37 °C and 130 rpm for 24 h, the supernatant of each sample was collected with a centrifuge (4230 rpm for 5 min). The protein concentrations were determined with a bicinchoninic acid (BCA) assay kit (Beyotime, Shanghai, China).
After desalting with a dialysis bag (3 kDa MWCO, Genview, Beijing, China), DPs were characterized with 15.5% tricine-sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE). The tricine-SDS-PAGE gel preparation kit, tris-tricine-SDS buffer solution, and protein standard samples were all obtained from Sangon Biotech (Shanghai, China). Sample treatment, gel preparation, and electrophoresis were performed as previously described by Schagger [14]. The gel was stained with Coomassie brilliant blue and imaged using a gel imaging system (Clinx., Shanghai, China).
Cell culture
MC3T3-E1 cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and cultured in high-glucose Dulbecco’s modified Eagle’s medium (DMEM, Hyclone, Logan, USA) supplemented with 10% fetal bovine serum (FBS, Biologic Industries, Kibbutz Beit Haemek, Israel) and 1% penicillin-streptomycin (Hyclone, Logan, USA). Cells were maintained at 37 °C, 5% CO2, and 95% humidity. The cells were passaged at 75%–90% confluence using trypsin (Hyclone, Logan, USA). For all experiments, the cells were seeded on 96-well plates, 24-well plates, or 6-well plates, at a density of 2 × 103, 2 × 104, or 8 × 104 cells/well, respectively [15].
Cell proliferation
Proliferation of MC3T3-E1 cells was evaluated using the cell counting kit 8 (CCK8) method. After 1 day of incubation, MC3T3-E1 cell medium was replaced with 100 μL of complete medium containing 50 μL of DPs, acquired as described above, to serve as the treatment group. Cells cultured with complete medium only served as the control group, and medium only (without cells) served as the blank group. Triplicate samples were prepared for each group. At days 1, 3, and 5, 10 μL of CCK8 (Beyotime, Shanghai, China) was added to the well, and the absorbance (OD) at 450 nm was recorded with a microplate reader (Bio-Tek, Winooski, USA). Cell viability was calculated using the following equation: Cell viability = (Treatment Group OD−Blank Group OD)/(Control Group OD−Blank Group OD) × 100% [16].
Alkaline Phosphatase (ALP) activity
After 1 day of incubation, MC3T3-E1 cell medium was replaced with 500 μL of osteoinduction differentiation medium (ODM; 50 μg/mL ascorbic acid, 100 nM DEX, and 5 mM β-glycerolphosphate) containing 250 μL of DPs, serving as the experiment group. Cells cultured with ODM only served as the control group, and normal medium only (without cells) served as the blank group. Triplicate samples were prepared for each group. After 1, 7, and 14 days, cells were lysed with deionized water and homogenized via ultrasound at 4 °C. The cell lysates were transferred to a 96-well plate. ALP activity and total protein concentration were measured and quantified using an ALP activity kit (Jiancheng Bioengineering Institute, Nanjing, China) and a BCA assay kit. ALP activity was normalized by the corresponding total protein concentration (U/mg) following the kit instructions [15].
Real-time quantitative polymerase chain reaction (qPCR)
Total RNA was extracted with TRIzol (Invitrogen, Carlsbad, USA) and then converted to cDNA with Hifair II 1st Strand cDNA Synthesis Kit (Yeasen, Shanghai, China) following the kit instructions. SYBR green PCR Master Mix (Takara, Dalian, China) was used for real-time qPCR on an MX 3000 platform (Agilent, Boeblingen, Germany). The primers for real-time qPCR were as follows: Runx2, 5’-ATAGCAAAGGCCCTCACTAA-3’ (forward) and 5’-AACTGGCTCTTCTGCTGATT-3’ (reverse); Col 1, 5’-GAGGCATAAAGGGTCATCGTGG-3’ (forward) and 5’-CATTAGGCGCAGGAAGGTCAGC-3’ (reverse); OC, 5’-TGACCTCACAGATGCCAAGC-3’ (forward) and 5’- CGCCGGAGTCTGTTCACTAC-3’ (reverse); and GAPDH, 5’- ACCACAGTCCATGCCATCAC-3’ (forward) and 5’-TCCACCACCCTGTTGCTGTA-3’ (reverse).
The expression levels of each target gene were normalized to the corresponding GAPDH threshold cycle (CT) values using the 2−▵▵CT comparative method [16].
High-performance Liquid Chromatography (HPLC) and mass spectrometry
The three types of collagen membranes were subjected to enzymatic hydrolysis for 21 days. DPs were collected by filtration–centrifugation using a Millipore centrifugal device (10 kDa MWCO, Millipore, Milford, MA). The resulting fractions were desalted using a C18 column (Thermo Scientific, San Jose, USA), then lyophilized, and finally stored at −20 °C until analyzed. Mass spectrometric analyses were performed as previously described [17]. The lyophilized peptides were separated using an Ultimate 3000 RSLCnano system coupled to a Q Exactive (Thermo Scientific, USA). Samples were loaded into a trap column (C18, 3 μm, 120 Å, 100 μm × 2 cm) and separated with a reversed-phase analytical column (C18, 2μm, 100 Å, 75 μm × 150 mm, Thermo Scientific, USA). Peptides were separated using a gradient of mobile phase A (3% dimethyl sulfoxide, 0.1% formic acid, and 97% H2O) and B (3% dimethyl sulfoxide, 0.1% formic acid, and 97% acetonitrile). The flow rate was set to 300 nL/min. The mass spectrometer was operated in data-dependent acquisition (DDA) mode with the following settings: Full MS scan (R = 70 K, AGC = 3e6, max IT = 20 ms, scan range = 350–1800 m/z) followed by up to 15 MS/MS scans (R = 17.5 K, AGC = 2e5, max IT = 100 ms). The isolation window was set to 1.6 m/z, and 28% normalized collision energy was used for higher-energy collisional dissociation (HCD). The dynamic exclusion time of repeated ion acquisition was set to 35 s [17].
The acquired raw mass spectrometric data were processed in MaxQuant (v1.6.2.10) [18] and searched against UniProt for taxonomy: “Sus scrofa” (TaxID 9823) for PD and “Bos taurus”(TaxID 9913) for the other two collagen membranes. All data and search results have been deposited to the iProX database ( http://www.iprox.org) with the iProX accession: IPX0006335000.
Statistical analysis
Data analysis and figure construction was performed using GraphPad Prism 6 (GraphPad, San Diego, USA). Values represent mean ± standard deviation (SD). Comparison among different groups was made by two-way analysis of variance (ANOVA). P values <0.05 were considered statistically significant.
Results
Physical characteristics of collagen membranes
Gross appearance was determined: The smooth surface of all collagen membranes was soft and elastic, while the texture of the rough surface was irregular (upper part, Fig. 1). SEM images revealed marked differences in the microstructures of the collagen membranes (lower part, Fig. 1). On the smooth side, collagen bundles of the PD and BP membranes were regular and arranged closely, while collagen bundles of the BD membrane were slightly irregular and arranged loosely. On the rough side, collagen bundles of BP interweaved with each other to form a three-dimensional (3D) grid with regular pores, while collagen bundles of PD and BD were arranged loosely.
The mechanical properties (tensile strength, elongation, and Young’s modulus) of the three kinds of collagen membranes in both the dry and wet states were compared (Fig. 2a–c). The results indicated that the tensile strength and Young’s modulus of the BP membrane in both the dry and wet states were significantly higher than those in other groups (P < 0.0001), although no significant difference was found between the BD and PD membranes (P > 0.05) except the Young’s modulus in the wet state (P = 0.034). Elongation was the highest for the PD membrane, irrespective of the state, intermediate for the BD membrane, and lowest for the BP membrane. Moreover, for all collagen membranes, the tensile strength and Young’s modulus were lower in the wet group than in the dry group, while elongation showed the opposite trend.
Comparison of the physical characteristics of different collagen membranes. a Tensile strength, b elongation at break, and c Young’s modulus of collagen membranes in the dry and wet state. The error bars represent ± SD. Two-way analysis of variance analysis. ns: not significant; ****P < 0.0001 vs PD; #### P < 0.0001 vs BP. n = 3 for all samples. d Water contact angle of collagen membranes. Mean ± SD shown. n = 5 for all samples
To assess the hydrophilicity of the three kinds of collagen membranes, static water-contact angles were measured. As shown in Fig. 2d, the water contact angle was significantly higher on the smooth surface than on the rough surface of the PD and BP membranes. However, the water contact angle was not significantly different between the smooth and rough surfaces of the BD membrane. On both smooth and rough surfaces, the water contact angle was the smallest for the PD membrane and the largest for the BP membrane.
Degradation ratio of collagen membranes
To determine the degradation ratio of the collagen membranes, the oral environment was mimicked using artificial saliva and collagenase. As shown in Fig. 3a, the PD and BP membranes became translucent within 21 days of treatment with collagenase, and, unexpectedly, the BD membrane was completely degraded. The degradation weight loss also confirmed the above findings. For the artificial saliva group, there was no significant difference among the three types of membranes on day 7. Moreover, the degradation weight loss increased gradually with treatment time up to day 21. The BD membrane suffered the highest degradation weight loss, followed by the BP membrane and then the PD membrane. By contrast, the degradation weight loss decreased in the collagenase group on day 7. On day 21, the PD membrane suffered the lowest degradation weight loss. The degradation weight loss of the BD membrane was up to 100%, indicating complete degradation of the BD membrane (Fig. 3b, c). The results demonstrate that the BD membrane is the most susceptible to degradation among the three membranes, irrespective of the treatment: artificial saliva or collagenase.
Comparison of the degradation of different collagen membranes. a Photographs of collagen membranes before and 21 days after degradation using collagenase. Degradation weight loss (%) of the collagen membranes incubated with artificial saliva (b) and collagenase (c) after 7, 14, and 21 days. The error bars represent ± SD. Two-way analysis of variance analysis. ns: not significant, *P < 0.05, ***P <0.001, ****P < 0.0001 vs PD. n = 3 for all samples. d SDS-PAGE-Tricine gel electrophoresis. M: low molecular weight marker. Lane 1 and 2: PD incubated with or without collagenase; Lane 3 and 4: BP incubated with or without collagenase; Lane 5 and 6: BD incubated with or without collagenase; Lane 7: Collagenase solution
On the basis of the above results, collagenase was used to degrade the collagen membranes in subsequent experiments.
After incubating for 24 h, DPs were analyzed to determine the protein concentration, as shown in Table 2. The protein concentration of degraded collagen membranes in the collagenase group was significantly higher than that in the PBS group (P < 0.0001). As expected, the BD membrane had the highest protein concentration of DPs, specifically it was 1.4 times than those of the other membranes, followed by the BP membrane and then the PD membrane. However, DPs from the three collagen membranes treated with collagenase had a similar composition. Eight protein bands were observed in the electrophoretic gel with molecular weights ranging from 25 to 116 kDa (Fig. 3d).
Effects of DPs on MC3T3-E1 cells
The proliferation of MC3T3-E1 cells was measured after MC3T3-E1 cells were cultured with collagen membrane DPs for 1, 3, and 5 days. The effect of DPs on the proliferation of MC3T3-E1 cells was correlated with the incubation time. In general, differences in cell proliferation were not significant between the control and groups with added DPs until day 5 (P < 0.05, Fig. 4a). Cell proliferation was the highest in the presence of BD-DPs, intermediate in the presence of PD-DPs, and lowest in the presence of BP-DPs.
Effects of collagen membranes degradation products on the proliferation and differentiation of MC3T3-E1 cells. CCK-8 proliferation assay performed with and without collagen membranes degradation products after 1, 3, and 5 days. b Alkaline phosphatase (ALP) activity of MC3T3-E1 cells incubated with or without collagen membranes degradation products for 1, 7, and 14 days. ALP activity was normalized by total cellular protein amounts. qPCR analysis of osteoblastic marker (c) runt-related transcription factor 2 (Runx2), (d) osteocalcin (OC), and (e) collagen type I (Col-1) relative expression in MC3T3-E1 cells incubated with or without collagen membranes degradation products for 7 and 14 days. The error bars represent ± SD. Two-way analysis of variance analysis. ns: not significant, *P < 0.05, **P < 0.01, ***P < 0.05, ****P < 0.0001. n = 3 for all samples
Similar to the cell proliferation results, ALP activity of cells treated with DPs increased with the incubation time and reached the peak on day 14, compared with the control group. Moreover, ALP activity of cells was the highest in the BD-DPs group, intermediate in the PD-DPs group, and lowest in the BP-DPs group (Fig. 4b).
To determine the effect of DPs on mineralization, the gene expressions of Runx2, OC, and Col 1, well-known osteoblast markers, were evaluated by real-time qPCR. As shown in Fig. 4c–e, after 7 days of treatment, when compared with the control group, there are no statistical differences in the OC, Runx2, and Col 1 expressions. However, when we prolonged the treatment to 14 days, all gene expressions noticeably increased. In addition, BD-DPs induced a higher level of Runx2, OC, and Col 1 gene expressions than PD-DPs and BP-DPs.
Analysis of the DPs of collagen membranes
Excluding common contaminating peptides and low abundance peptides, a total of 887 peptides were identified in the degradation products of the three collagen membranes by liquid chromatography-mass spectrometry (LC–MS). Among them, 334 peptides, derived from 24 proteins, were identified in the BD membrane. Fewer peptides were identified in the PD membrane, and the lowest number of peptides was identified in the BP membrane (Fig. 5a). The peptides ranged from 8 to 25 amino acids in length (Fig. 5b). Despite the differences in peptide sequences, the identified proteins, including collagen and elastin, were similar (Fig. 5c and Supplementary Table S1 and S2).
Discussion
The three kinds of collagen membranes display unique features in clinical applications. The PD membrane can resist degradation for a long time, thus achieving significant bone growth effect in clinical applications by virtue of its excellent histocompatibility and reliable biosafety [19, 20]. The potential of the BD membrane in promoting tissue healing has been widely explored, and it is often used to guide tissue regeneration in the clinic [21, 22]. The BP membrane is known for its excellent tensile strength and mechanical properties [23, 24]. These membranes with different characteristics have shown consistent osteogenic efficacy in animal models and clinical trials [21, 25, 26]. However, these data primarily arise from clinical observations and studies of a membrane in isolation of other membranes, not from a comparison between different membranes.
First, the tensile strength and Young’s modulus of the three collagen membranes, important indicators of the resistance of solid materials to deformation, were obtained. Elongation, a vital index to characterize the softness and elasticity of fibers, was also evaluated. The experimental results showed that the tensile strength and Young’s modulus of the BP membrane were significantly higher than those of the other membranes, reflecting its excellent spatial maintenance ability. Among the three membranes, the PD membrane had the highest elongation, indicating excellent plasticity. In addition, as common occurrences in clinical treatment, insufficient soft tissue at the bone defect and high wound suture tension are risk factors leading to partial leakage of the membrane, thus exposing the membrane to the oral cavity. Previous research showed that a moist environment has a significant impact on the mechanical properties of the membrane [27]. That’s why, to demonstrate the effect of oral saliva on the mechanical properties of collagen membranes, both the dry and wet groups were evaluated. The results showed that the tensile strength and Young’s modulus of the wet group were lower than those of the dry group, consistent with the results of previous studies [28]. It is well established that the mechanical properties of resorbable membranes are poorer than those of non-resorbable membranes [29]. Therefore, if collagen membranes are selected as the barrier membrane, clinicians should reduce the extra pressure, avoid overstretching the collagen membrane when fixing it with membrane tacks or sutures, and avoid unnecessary stretching when pre-trimming the collagen membrane before implantation. When additional stress is inevitable or more support space is needed, the BP membrane is the most suitable candidate among the three membranes because it exhibits high tensile strength, which minimally decreases after wetting, and it has the largest Young’s modulus, reflecting its greater stiffness. Furthermore, extra support, such as bone graft materials, membrane tacks, and suture retention, can be given to compensate for the mechanical strength of the membrane.
The hydrophilicity of the collagen membranes was evaluated by measuring the water contact angles. Generally, surfaces with low contact angles exhibit strong biocompatibility and tissue integration. Conversely, surfaces with high contact angles can reduce cell adhesion and tissue regeneration [30], which is related to positioning the smooth surface to face the suture surface of the wound and the rough surface to face the bone defect area in clinical applications. The contact angle on both the smooth and rough surfaces of the PD membrane was the lowest, while that of the BP membrane was the highest. The hydrophilicity of the collagen membrane may be related to the arrangement of collagen fibers on the surface, specifically a looser arrangement tends to correlate with a lower contact angle and higher hydrophilicity [31]. This is consistent with the results of this study, specifically the hydrophilicity of the PD and BP membranes is higher on the rough surface than on the smooth surface, and the rough surface has a looser arrangement of the microstructure than the smooth surface.
In contrast to non-resorbable membranes, collagen membranes are biodegradable, which is a distinct advantage because a subsequent operation is unnecessary. In vivo studies of the collagen membranes show that these membranes significantly degrade within 8 to 12 weeks [32, 33]. In addition to cellular and enzymatic microenvironments under microbe-free conditions, spontaneous exposure to the oral enviroment is essential for the degradation of resorbable membranes [34,35,36,37]. Upon exposure to the oral environment, resorbable membranes degrade rapidly in the presence of saliva and collagenase derived from periodontal pathogens, such as Porphyromonas gingivalis and Bacteroides melaninogenicus [38]. Considering the requirements of clinical applications, degradation of membrane materials benefits the healing of damaged tissues. Even in the case of exposure to the oral environment, the exposed membrane should be kept in situ, thus continuing to function during the regenerative process [39, 40]. Therefore, artificial saliva and collagenase were introduced to evaluate the effects of the degradation of collagen membranes. In the present study, among the three membranes, the BD membrane degrades with the highest rate, while the PD membrane degrades with the lowest rate. Although in vitro studies have limitations in simulating the oral environment, the results of this experiment are consistent with those of Neto et al., that is, the PD membrane is less degraded than the BP membrane during bone regeneration in a rabbit bone defect model [41]. The PD membrane derived from porcine dermis is denser and degrades with a lower rate than the BD membrane, which is derived from bovine dermis, indicating that the degradation rate is related to the structure and source of the collagen [42].
It is well established that collagen peptides can promote osteogenesis by enhancing osteoblast (MC3T3-E1 cells) proliferation and differentiation [43, 44]. Although collagen membranes are known to provide a conducive environment for osteoblasts to adhere, survive, and grow, they are not considered a key player in promoting osteogenesis [45,46,47]. Because the three kinds of collagen membranes were found to have different degradation rates, this experiment explored the effect of DPs on bone defects during GBR by evaluating the proliferation of MC3T3-E1 cells, the activity of ALP (an osteoblast differentiation marker), and the messenger ribonucleic acid (mRNA) expression level of bone formation-related genes (Runx2, Col1, and OC). The results showed that the DPs of the three kinds of collagen membranes enhanced cell proliferation and ALP activity, in addition to up-regulating the mRNA expression level of osteoblast-related genes. This is the first demonstration, to our knowledge, showing that degradation products of collagen membrane can promote osteogenesis. Among the three membranes, the BD membrane is the most effective, followed by the PD membrane, in promoting osteogenesis. This high performance is likely due to the high total collagen in the BD membrane compared with that in the other membranes. Another reason is related to the source of the collagen.
Collagen membranes are composed of numerous proteins, including fibrillar collagens, non-fibrillar collagens, and leucine-rich repeat proteoglycans, as well as a small number of structural proteins, such as vimentin, actin-based microfilaments, annexins, tubulins, and histones [48]. Consistent with other studies, of the 887 peptides identified in the degradation products of the three collagen membranes, a large number is derived from different collagen chains and elastin. Interestingly, collagen peptides from different sources exhibit a wide range of biological functions, such as anti-inflammation, wound-healing, and anti-oxidative stress [49,50,51]. Two bioactive peptides, C2 and E1, derived from bovine tendon collagen can support cell adhesion and counter stress [52]. Liu et al. [53] also found that bovine collagen peptides with different molecular weights have different effects on the differentiation and mineralization of osteoblasts. In the present study, by identifying the DPs of the three collagen membranes, a pool of collagen peptides is available for the screening of peptides capable of promoting osteogenesis. A common strategy to extend the function of collagen membranes is to load or modify the membrane with bioactive components [46, 54]. Yu et al. modified a collagen membrane with stromal cell-derived factor-1 alpha (SDF-1α), a pro-osteogenic protein, by chemical conjugation, and reported that the modified membrane significantly promotes new bone and micro vessel formation [46]. Chen et al. used sonication to coat a collagen membrane with silver nanoparticles, thus conferring excellent anti-bacterial activity against Staphylococcus aureus and Pseudomonas aeruginosa [55]. These modified membranes not only require redesign of the bioactive component but also lack evaluation of biocompatibility. In contrast, collagen peptides derived from the degradation products of collagen membranes are potentially safer than the above bioactive components. Therefore, in future studies, identifying peptides with high bioactivity among the DPs of collagen membranes can be attempted to develop functional collagen membranes.
Conclusion
In this study, the physical, mechanical, and in vitro biological properties of three kinds of collagen membranes were determined. The findings indicate that the PD membrane is the best choice for shaping and maintenance time, the BD membrane is good for osteogenesis, and the BP membrane is suitable for spatial maintenance. More importantly, two kinds of collagen membranes can be used concurrently to exert their respective advantages for clinical needs during GBR, for example, placing the PD membrane as the outer layer to prolong maintenance time and using the BD membrane as the inner layer for osteogenesis. Further studies are necessary to determine the osteogenic effect of single and combined membranes on GBR. Because of the limitations of in vitro experiments, in vivo experimental studies are needed to provide clinicians with reliable indicators.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
French D, Grandin HM, Ofec R. Retrospective cohort study of 4,591 dental implants: Analysis of risk indicators for bone loss and prevalence of peri-implant mucositis and peri-implantitis. J Periodontol. 2019;90(7):691–700.
Abe GL, Sasaki JI, Katata C, Kohno T, Tsuboi R, Kitagawa H, Imazato S. Fabrication of novel poly(lactic acid/caprolactone) bilayer membrane for GBR application. Dent Mater. 2020;36(5):626–34.
Zhao D, Dong H, Niu Y, Fan W, Jiang M, Li K, Wei Q, Palin WM, Zhang Z. Electrophoretic deposition of novel semi-permeable coatings on 3D-printed Ti-Nb alloy meshes for guided alveolar bone regeneration. Dent Mater. 2022;38(2):431–43.
Sbricoli L, Guazzo R, Annunziata M, Gobbato L, Bressan E, Nastri L. Selection of Collagen Membranes for Bone Regeneration: A Literature Review. Materials. 2020;13(3):786.
Calciolari E, Ravanetti F, Strange A, Mardas N, Bozec L, Cacchioli A, Kostomitsopoulos N, Donos N. Degradation pattern of a porcine collagen membrane in an in vivo model of guided bone regeneration. J Periodontal Res. 2018;53(3):430–9.
Jung RE, Brugger LV, Bienz SP, Husler J, Hammerle CHF, Zitzmann NU. Clinical and radiographical performance of implants placed with simultaneous guided bone regeneration using resorbable and nonresorbable membranes after 22–24 years, a prospective, controlled clinical trial. Clin Oral Implants Res. 2021;32(12):1455–65.
Thoma DS, Bienz SP, Figuero E, Jung RE, Sanz-Martin I. Efficacy of lateral bone augmentation performed simultaneously with dental implant placement: A systematic review and meta-analysis. J Clin Periodontol. 2019;46:257–76.
Friedl P, Wolf K. Plasticity of cell migration: a multiscale tuning model. J Cell Biol. 2010;188(1):11–9.
Dimitriou R, Mataliotakis GI, Calori GM, Giannoudis PV. The role of barrier membranes for guided bone regeneration and restoration of large bone defects: current experimental and clinical evidence. BMC Med. 2012;10:81.
Benic GI, Hämmerle CH. Horizontal bone augmentation by means of guided bone regeneration. Periodontol 2000. 2014;66(1):13-40.
Wu Z, Wu J, Peng T, Li Y, Lin D, Xing B, Li C, Yang Y, Yang L, Zhang L et al. Preparation and Application of Starch/Polyvinyl Alcohol/Citric Acid Ternary Blend Antimicrobial Functional Food Packaging Films. Polymers. 2017;9(3):102.
Rezvanian M, Ahmad N, Mohd Amin MC, Ng SF. Optimization, characterization, and in vitro assessment of alginate-pectin ionic cross-linked hydrogel film for wound dressing applications. Int J Biol Macromol. 2017;97:131–40.
Tamburaci S, Tihminlioglu F. Development of Si doped nano hydroxyapatite reinforced bilayer chitosan nanocomposite barrier membranes for guided bone regeneration. Mater Sci Eng C Mater Biol Appl. 2021;128: 112298.
Schägger H. Tricine-SDS-PAGE. Nature protocols. 2006;1(1):16–22.
Cheon SY, Kim J, Lee KY, Lee CM. Poly (l-lactic acid) membrane crosslinked with Genipin for guided bone regeneration. Int J Biol macromol. 2021;191:1228–39.
Gao X, Al-Baadani MA, Wu M, Tong N, Shen X, Ding X, Liu J. Study on the Local Anti-Osteoporosis Effect of Polaprezinc-Loaded Antioxidant Electrospun Membrane. Int J Nanomedicine. 2022;17:17–29.
Yang M, Vesterlund M, Siavelis I, Moura-Castro LH, Castor A, Fioretos T, Jafari R, Lilljebjorn H, Odom DT, Olsson L, et al. Proteogenomics and Hi-C reveal transcriptional dysregulation in high hyperdiploid childhood acute lymphoblastic leukemia. Nat Commun. 2019;10(1):1519.
Balestra AC, Koussis K, Klages N, Howell SA, Flynn HR, Bantscheff M, Pasquarello C, Perrin AJ, Brusini L, Arboit P, et al. Ca2+ signals critical for egress and gametogenesis in malaria parasites depend on a multipass membrane protein that interacts with PKG. Sci Adv. 2021;7(13):eabe5396.
Urban IA, Lozada JL, Wessing B, del Amo FSL, Wang HL. Vertical Bone Grafting and Periosteal Vertical Mattress Suture for the Fixation of Resorbable Membranes and Stabilization of Particulate Grafts in Horizontal Guided Bone Regeneration to Achieve More Predictable Results: A Technical Report. Int J Periodontics Restorative Dent. 2016;36(2):153–9.
Jung RE, Fenner N, Hammerle CHF, Zitzmann NU. Long-term outcome of implants placed with guided bone regeneration (GBR) using resorbable and non-resorbable membranes after 12–14years. Clin Oral Implants Res. 2013;24(10):1065–73.
Tang J, Han Y, Zhang F, Ge Z, Liu X, Lu Q. Buccal mucosa repair with electrospun silk fibroin matrix in a rat model. Int J Artif Organs. 2015;38(2):105–12.
de Andrade PF, de Souza SLS, Macedo GD, Novaes AB, Grisi MFD, Taba M, Palioto DB. Acellular dermal matrix as a membrane for guided tissue regeneration in the treatment of class II furcation lesions: A histometric and clinical study in dogs. J Periodontol. 2007;78(7):1288–99.
Pakzaban P. Technique for Mini-open Decompression of Chiari Type I Malformation in Adults. Oper Neurosurg. 2017;13(4):465–70.
Kyu KK. Treatment of Peri-implantitis: Cases Report. Maxillofacial Plastic Reconstruct Surg. 2013;35(2):112-123.
Chen L, Cheng G, Meng S, Ding Y. Collagen Membrane Derived from Fish Scales for Application in Bone Tissue Engineering. Polymers. 2022;14(13):2532.
Doan TL, Le LD. Efficacy of the tent-pole technique in horizontal ridge augmentation. Pesquisa Brasileira em Odontopediatria e Clinica Integrada. 2020;20:1–8.
Coïc M, Placet V, Jacquet E, Meyer C. Mechanical properties of collagen membranes used in guided bone regeneration: a comparative study of three models. Revue de stomatologie et de chirurgie maxillo-faciale. 2010;111(5–6):286–90.
Raz P, Brosh T, Ronen G, Tal H. Tensile Properties of Three Selected Collagen Membranes. BioMed Res Int. 2019;2019:5163603.
Zhang M, Zhou Z, Yun J, Liu R, Li J, Chen Y, Cai H, Jiang HB, Lee ES, Han J, et al. Effect of Different Membranes on Vertical Bone Regeneration: A Systematic Review and Network Meta-Analysis. BioMed Res Int. 2022;2022:7742687.
Schieber R, Lasserre F, Hans M, Fernández-Yagüe M, Díaz-Ricart M, Escolar G, Ginebra MP, Mücklich F, Pegueroles M. Direct Laser Interference Patterning of CoCr Alloy Surfaces to Control Endothelial Cell and Platelet Response for Cardiovascular Applications. Adv Healthcare Mater. 2017;6(19).
Strong SE, Eaves JD. The Dynamics of Water in Porous Two-Dimensional Crystals. J Phys Chem B. 2017;121(1):189–207.
Rothamel D, Schwarz F, Fienitz T, Smeets R, Dreiseidler T, Ritter L, Happe A, Zöller J. Biocompatibility and biodegradation of a native porcine pericardium membrane: results of in vitro and in vivo examinations. Int J Oral Maxillofac Implants. 2012;27(1):146–54.
Bornert F, Herber V, Sandgren R, Witek L, Coelho PG, Pippenger BE, Shahdad S. Comparative barrier membrane degradation over time: Pericardium versus dermal membranes. Clin Exp Dental Res. 2021;7(5):711–8.
Tal H, Kozlovsky A, Artzi Z, Nemcovsky CE, Moses O. Long-term bio-degradation of cross-linked and non-cross-linked collagen barriers in human guided bone regeneration. Clin Oral Implants Res. 2008;19(3):295–302.
Brunel G, Piantoni P, Elharar F, Benqué E, Marin P, Zahedi S. Regeneration of rat calvarial defects using a bioabsorbable membrane technique: influence of collagen cross-linking. J Periodontol. 1996;67(12):1342–8.
Almutairi AS: Case Report: Managing the postoperative exposure of a non-resorbable membrane surgically. F1000Research. 2018;7:685.
Hartmann A, Seiler M. Minimizing risk of customized titanium mesh exposures - a retrospective analysis. BMC Oral Health. 2020;20(1):36.
Mayrand D, Grenier D. Detection of collagenase activity in oral bacteria. Can J Microbiol. 1985;31(2):134–8.
Chen K, Zhou G, Li Q, Tang H, Wang S, Li P, Gu X, Fan Y. In vitro degradation, biocompatibility and antibacterial properties of pure zinc: assessing the potential of Zn as a guided bone regeneration membrane. J Mater Chem B. 2021;9(25):5114–27.
Sanz M, Dahlin C, Apatzidou D, Artzi Z, Bozic D, Calciolari E, De Bruyn H, Dommisch H, Donos N, Eickholz P, et al. Biomaterials and regenerative technologies used in bone regeneration in the craniomaxillofacial region: Consensus report of group 2 of the 15th European Workshop on Periodontology on Bone Regeneration. J Clin Periodontol. 2019;46(Suppl 21):82–91.
Neto AMD, Sartoretto SC, Duarte IM, Resende RFB, Neves Novellino Alves AT, Mourão C, Calasans-Maia J, Montemezzi P, Tristão GC, Calasans-Maia MD. In Vivo Comparative Evaluation of Biocompatibility and Biodegradation of Bovine and Porcine Collagen Membranes. Membranes. 2020;10(12):423.
Vallecillo-Rivas M, Toledano-Osorio M, Vallecillo C, Toledano M, Osorio R. The Collagen Origin Influences the Degradation Kinetics of Guided Bone Regeneration Membranes. Polymers. 2021;13(17):3007.
Liu J, Zhang B, Song S, Ma M, Si S, Wang Y, Xu B, Feng K, Wu J, Guo Y. Bovine collagen peptides compounds promote the proliferation and differentiation of MC3T3-E1 pre-osteoblasts. PloS one. 2014;9(6): e99920.
Rousseau M, Boulzaguet H, Biagianti J, Duplat D, Milet C, Lopez E, Bédouet L. Low molecular weight molecules of oyster nacre induce mineralization of the MC3T3-E1 cells. J Biomed Mater Res Part A. 2008;85(2):487–97.
El-Jawhari JJ, Moisley K, Jones E, Giannoudis PV. A crosslinked collagen membrane versus a non-crosslinked bilayer collagen membrane for supporting osteogenic functions of human bone marrow-multipotent stromal cells. Eur Cells Mater. 2019;37:292–309.
Yu X, Sun H, Yang J, Liu Y, Zhang Z, Wang J, Deng F. Evaluation of bone-regeneration effects and ectopic osteogenesis of collagen membrane chemically conjugated with stromal cell-derived factor-1 in vivo. Biomed Mater. 2019;15(1): 015009.
Caballé-Serrano J, Munar-Frau A, Delgado L, Pérez R, Hernández-Alfaro F. Physicochemical characterization of barrier membranes for bone regeneration. J Mech Behav Biomed Mater. 2019;97:13–20.
Lee JS, Mitulović G, Panahipour L, Gruber R. Proteomic Analysis of Porcine-Derived Collagen Membrane and Matrix. Materials (Basel). 2020;13(22):5187.
Woo M, Seol BG, Kang KH, Choi YH, Cho EJ, Noh JS. Effects of collagen peptides from skate (Raja kenojei) skin on improvements of the insulin signaling pathway via attenuation of oxidative stress and inflammation. Food Funct. 2020;11(3):2017–25.
Lin H, Zheng Z, Yuan J, Zhang C, Cao W, Qin X. Collagen Peptides Derived from Sipunculus nudus Accelerate Wound Healing. Molecules. 2021;26(5):1385.
Hakuta A, Yamaguchi Y, Okawa T, Yamamoto S, Sakai Y, Aihara M. Anti-inflammatory effect of collagen tripeptide in atopic dermatitis. J Dermatol Sci. 2017;88(3):357–64.
Banerjee P, Mehta A, Shanthi C. Investigation into the cyto-protective and wound healing properties of cryptic peptides from bovine Achilles tendon collagen. Chemico-Biol Interact. 2014;211:1–10.
Liu J, Si S, Qin Y, Zhang B, Song S, Guo Y. The effect of different molecular weight collagen peptides on MC3T3-E1 cells differentiation. Biomed Mater Eng. 2015;26(Suppl 1):S2041–2047.
Khorsand B, Elangovan S, Hong L, Kormann MSD, Salem AK. A bioactive collagen membrane that enhances bone regeneration. J Biomed Mater Res Part B Appl Biomater. 2019;107(6):1824–32.
Chen P, Wu Z, Leung A, Chen X, Landao-Bassonga E, Gao J, Chen L, Zheng M, Yao F, Yang H, et al. Fabrication of a silver nanoparticle-coated collagen membrane with anti-bacterial and anti-inflammatory activities for guided bone regeneration. Biomed Mater. 2018;13(6): 065014.
Acknowledgments
We thank Liwen Bianji (Edanz) (http://www.liwenbianji.cn) for editing the language of a draft of this manuscript.
Funding
This work was supported by the Scientific Research Project of Jilin Provincial Science and Technology Department (No. 20230203065SF), Scientific Research Project of Jilin Provincial Department of Education (No. JJKH20231291KJ), Scientific Research Project of Jilin Provincial Department of Finance, China (Grant No. JCSZ2019378-23), the Science and Technology Development Project of Jilin Province (Grant No. 20210204190YY), and Traditional Chinese Medicine Science and Technology Project of Jilin Province (Grant No. 2021042).
Author information
Authors and Affiliations
Contributions
X.S. led the writing. X.L. did most of the experiments. Y.T., Y.C., X.Q., and S.C. critically revised the manuscript. S.Z. and Y.L. analyzed the experimental data. Y.C. and W.J. contributed to charting. Every author gave final approval and agreed to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations. This study did not involve human or animal experiments, and the cell lines used were artificially cultured and did not require ethical approval or moral explanation.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Supplementary Table S1. Identified proteins.
Additional file 2:
Supplementary Table S2. Identified peptides.
Additional file 3:
Figure 3. Original figure of Figure 3d in the manuscript. (A) Original figure. (B) The picture after exposure. The red boxes represent the regions of the original blots used in main figures. The label (M,1,2,3,4,5,7,6) matched to the cropped versions in the manuscript figures.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Shi, X., Li, X., Tian, Y. et al. Physical, mechanical, and biological properties of collagen membranes for guided bone regeneration: a comparative in vitro study. BMC Oral Health 23, 510 (2023). https://doi.org/10.1186/s12903-023-03223-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12903-023-03223-4