Abstract
Introduction
An acute Achilles tendon rupture represents a common tendon injury, and its operative methods have been developed over the years. This study aimed to quantify the learning curve for the minimally invasive acute Achilles tendon rupture repair.
Methods
From May 2020 to June 2022, sixty-seven patient cases who received minimally invasive tendon repair were reviewed. Baseline data and operative details were collected. The cumulative summation (CUSUM) control chart was used for the learning curve analyses. Achilles tendon rupture score (ATRS), American Orthopedic Foot and Ankle Society (AOFAS) ankle/hindfoot score, and visual analog scale (VAS) at 3/6/9/12 months were calculated to assess the clinical outcomes.
Results
Thirty-six cases underwent at least a year of follow up and were enrolled in this study. The gender ratio and average age were 80.5% and 32.5 years. The linear equation fitted well (R2 = 0.95), and CUSUM for operative time peaked in the 12th case, which was divided into the learning phase (n = 12) and master phase (n = 24). No significant difference was detected between the two groups in clinical variables, except for the operative time (71.1 ± 13.2 min vs 45.8 ± 7.2 min, p = 0.004). Moreover, we detected one case with a suture reaction and treated it properly.
Conclusion
Minimally invasive Achilles repair provides an opportunity for early rehabilitation. Notably, the learning curve showed that the “lumbar puncture needle and oval forceps” technique was accessible to surgeons.
Similar content being viewed by others
Background
The Achilles tendon is the strongest and largest tendon in the human body, and its rupture is a common sports-related injury [1, 2]. The incidence of acute Achilles tendon rupture keeps rising, and it has been reported to reach 18 per 100,000 people per year due to improper strength explosions in the ankle or the weakening of elasticity during frequent and high-intensity physical exercise [3,4,5]. Most acute Achilles tendon ruptures are observed in middle-aged men, predominantly during sports activities that require abrupt initiation and cessation of movement, including tennis, basketball, soccer, and badminton [6]. Notably, between 81 and 89% of rupture cases are attributed to the physical demands of athletic activities as the primary cause of injury [7,8,9]. Acute Achilles tendon rupture usually requires operative intervention. Otherwise, passive therapy may cause continuous pain and limited motion of the overall ankle joint and even leave a lifelong disability due to the extremity atrophy of the injured side.
Notably, this operative procedure has varied over the years. As such, the open repair requires a 10—15 cm incision to broadly separate the subcutaneous fascia and tendon sheath to search the broken ends for tendon anastomosis. This method was performed under direct supervision and could effectively protect the sural nerve, which was reported to significantly improve clinical outcomes compared with nonoperative options [10]. In 1977, Prof Ma and Griffith introduced the mini-open technique for acute Achilles tendon ruptures. They reported that this advanced attempt has many advantages, including a rapid recovery, a short hospitalization period, and a cosmetic appearance at the surgical site [11]. Recent literature also reported a series of surgical methods for minimally invasive approaches and described their clinical effects to minimize adverse events [12,13,14]. An Achilles tendon rupture guideline indicates that acute Achilles tendon rupture is a suitable indication for minimally invasive techniques, with clinical recovery rates of at least 85% [15]. Nevertheless, many institutions lack professional facilities to perform this surgery, and the importation of some equipment is limited for financial and administrative reasons.
In recent years, we have referred to previous reports that used the “lumbar puncture needle and oval forceps” technique to repair the ruptured Achilles tendon [16,17,18]. Generally, the surgical proficiency of orthopedic surgeons could be assessed using statistical analysis of surgical details, radiographic parameters, and postoperative clinical outcomes. However, this traditional evaluation has shown limited accuracy in demonstrating a surgeon’s ability. As such, the learning curve can be used to measure competency and proficiency in various surgical procedures [19, 20]. Notably, this progressive method has been widely used to estimate foot and ankle surgery [21, 22].
Based on this, our study used the cumulative sum (CUSUM) methodology to present the learning process of this minimally invasive operation in treating acute Achilles tendon rupture.
Methods
Study design
With the approval of the institutional review committee, a retrospective study was performed on patients who underwent operative treatments for acute Achilles tendon ruptures in 36 from May 2020 to June 2022.
Inclusion criteria were as follows: (1) patients over 18 years old, (2) within two weeks after injury, (3) the definitive diagnosis of the Achilles tendon rupture confirmed by preoperative ultrasound and MRI, (4) positive results of Thompson test and the palpable gap at the rupture sites of Achilles tendon, (5) the Achilles tendon rupture occurring at 2–8 cm proximal to insertion (non-insertional type), (6) receiving a minimally invasive treatment of “lumbar puncture needle and oval forceps,” (7) follow-up more than twelve months. Exclusion criteria: (1) patients under 18 years old, (2) chronic injury (> 2 weeks), (3) open injury of Achilles tendons, (4) incomplete clinical data or follow-up of less than a year.
Two clinical investigators were responsible for individual data collection. Demographic data (gender and age), injury mechanism, and injury side were obtained from medical records. In addition, surgical details, including surgical time and postoperative length of stays (post-LOS), were also reviewed.
Operative technique
Minimally invasive repair (lumbar puncture needle and oval forceps) [16, 18]
The patient was under spinal anesthesia or general anesthesia, in a prone position with a pad under the ankle. (Fig. 1).
-
(i)
The surgeon palpated the tendon’s gap and observed the positive sign (“finger sign”). Then, a 2–3 cm longitudinal incision was immediately made over the ruptured Achilles tendon site.
-
(ii)
An Allis clamp was used to retrieve the proximal/distal broken end. An oval forceps was also inserted to penetrate both sides of the ruptured tendon. A lumber puncture needle (BD Medical System, NJ, USA) created a subcutaneous tunnel through the skin, subcutaneous fat, fascia, paratenon, tendon, and two eyelet rings of the oval forceps. Particularly in confirming whether the needle involved the ruptured tendon, the surgeon should attempt to withdraw the oval forceps.
-
(iii)
Then, a No. 0 polydioxanone (PDS) (Ethicon, Somerville, NJ, USA) was passed through the needle. After the needle and oval forceps were retrieved, the suture tails were concomitantly detached from the incision. Sutures were carefully separated and marked. The above procedure was repeated three times at the proximal/ distal site, and the needle pitch was about 0.8 cm. A No. 2 polyester suture (Ethicon, Somerville, NJ, USA) was used in each middle suture.
-
(iv)
Ruptured tendon strength was tested by gentle retraction of the sutures, which could minimize the risk of entrapping the sural nerve. The operated ankle was located at 30° of plantar flexion during repair. The most proximal suture from the proximal segment was tied to the most proximal from the distal, and so on. The loops were concealed severally beneath the paratenon.
-
(v)
The paratenon/subcutaneous tissue and skin were closed using 2/0 Vicryl (Ethicon, Somerville, NJ, USA) and 3/0 nonabsorbable suture (Ethicon, Somerville, NJ, USA), respectively.A sterile dressing and a plaster splint were applied to the ankle with 30 degrees of plantar flexion.
A series of interoperative photos illustrating the minimally invasive Achilles repair. A A pre-operative MRI showing a definite rupture of the Achilles tendon. B A 2-cm transverse incision was made over the central part of the Achilles tendon defect. C: An Allis clamp exposed and retracted the rupture site. D After the subcutaneous tunnel was built, the oval forceps was inserted and the Achilles tendon defect was grasped. Then, a lumbar puncture needle was passed through the tendon and the eyelet ring of the oval forceps. E Sutures went into the needle. The red circle denoted the nonabsorbable sutures. F It should be noted that the stitches were separated to avoid the concentration of suture knots. G The wound was sewed up in plantar flexion. H Postoperative radiographic data presented that the tendon healed well
Postoperative management
Stage 1: For the first two weeks postoperatively, patients were instructed to apply over-the-knee full casts with 30-degree knee flexion and 30-degree plantar flexion. Additionally, they were allowed to sustain 1/6 to 1/3 of the total weight. Heel cushions were available to adjust the plantar flexion angle and enhance the ankle proprioception.
Stage 2: A below-knee cast and walking brace were used for the next two weeks, and the goal was to gradually reach a full weight bearing. A full range of ankle motion attempts and concentric loading was instituted under the guidance of a physical therapist. Subsequent eccentric loading is gradually carried out according to the patient's recovery.
Stage 3: The walking braces were removed at 5 weeks postoperative, and they could walk with normal shoes. Plyometric exercises were permitted, and patients were encouraged to participate in athletic activities if they felt comfortable during the ninth week.
Clinical outcomes
To evaluate the operative outcome, we used the Achilles tendon rupture score (ATRS), the American Orthopedic Foot and Ankle Society (AOFAS) ankle/hindfoot score, and the visual analog scale (VAS). The ATRS scoring system is a validated Achilles tendon rupture instrument [23, 24]. The ATRS is a Patient-Reported Outcome Measure (PROM) presenting the difficulty level of various physical activities due to symptoms. The AOFAS scoring systems, including self-assessment and objective scoring, are broadly used to evaluate the ankle joint's clinical function, range of motion, and tolerance [25]. During the follow-up, we also recorded the time to return to work, full weight bearing, and return to previous activities. Furthermore, we collected patient complications during follow-up, including re-rupture, suture reaction, wound dehiscence, and deep infection.
Re-rupture was defined as a definite rupture after surgical repair. Suture reaction and wound dehiscence were counted when the redness/swelling of the incision and superficial wound breakdown, respectively [26]. Deep infection was referred to the previous definition of fracture-related infection, regarding as a postoperative infection presented a year after surgery and met at least one of the pathologic conditions: (1) clinical signs of a fistula, sinus, wound necrosis, or purulent exudation, (2) confirmed microorganisms by laboratory evidence from at least two separate deep tissues or implants during paracentesis or re-operation [27].
CUSUM analysis
Consecutive cases were enrolled in chronological order. The CUSUM method was applied to analyze the learning curve, focusing on surgical duration [28]. We also used the following formula for the CUSUM calculation: \(CUSUM={\sum }_{i=1}^{n}(Xi-U)\). 'Xi' was the operation time per case, 'U' was the average operation time across all cases, and 'n' was the consecutive numbering of each case. Python (version 3.9.6) was used for analysis and scatter plot generation was used to obtain the function formula through curve fitting. The efficacy of the curve fitting was assessed using the correlation coefficient R2, with values approaching 1 indicating more precise fitting. The first order of the curve was chosen and the curve apex was based on the slope. Notably, this apex contributed to segregating the patients into individual groups of learning and mastery, establishing a cut-off value representing the minimum number of cases in which a surgeon was required to obtain a certain level of proficiency [29].
Statistical analysis
According to the normal distribution results, continuous variables were analyzed using the independent samples t-test or Mann–Whitney u-test. The enumeration variables were analyzed using the chi-square or Fisher exact tests where appropriate. Descriptive data were presented as mean ± standard deviation (SD), median (interquartile range, IQR), and frequency (percentage) for normally and non-normally distributed continuous data and enumeration data. All statistical analyses were performed using SPSS software (IBM SPSS Statistics, Version 26.0). A p-value of < 0.05 was considered statistically significant in this study.
Results
Baseline data and learning curve quantification
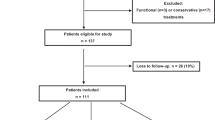
In general, a consecutive cohort of sixty-seven cases who received a “lumbar puncture needle and oval forceps” in treating acute Achilles rupture in our institution were reviewed. Finally, thirty-six patients were enrolled in this study (Fig. 2).
Since these results of surgical performance demonstrated that the learning curve peaked at the 12th case, we divided the cases into 2 groups: the learning phase (n = 12, case 1–12) and master phase (n = 24, case 13–36) (Fig. 3). The best-fit linear equation was: CUSUM = 1.68 × 10−2n3—1.30 × 100n2 + 2.42 × 101n + 4.14 × 101 (R2 = 0.95), where n represented the individual case.
The learning phase and master phase were statistically similar in gender ratio (the learning phase 83.3%, the master phase 79.2%, p = 0.766) and age (the learning phase 32.8 ± 6.8 years, the master phase 32.3 ± 5.2 years, p = 0.785) (Fig. 4). Other clinical data also demonstrated no statistical difference between these groups in injury mechanism, injury side, post-LOS, and blood loss (p > 0.05). However, the learning phase was significantly longer than the master phase for surgical time (71.1 ± 13.2 vs. 45.8 ± 7.2 min, p = 0.004).
Postoperative assessments
The clinical outcome evaluation was assessed using the ATRS and AOFAS. The VAS scoring system determined the pain. We found no statistical correlation between these ATRS, AOFAS, and VAS groups in 3/6/9/12 months (Table 1, Figs. 5 and 6). Similarly, neither time to return to work/ full-weight bearing/ previous activities showed a significant difference (p > 0.05).
Regarding the complication within a year, one patient (case 4) was detected with a suture reaction, which presented in the fourth month postoperatively. After removing the nonabsorbable sutures, the redness and swelling of his injured Achilles tendon gradually improved in the following seven days. Major complications, including re-rupture, wound dehiscence, and deep infection, were not found in these thirty-six patients.
Discussion
Achilles tendon rupture is a common injury in daily life, accounting for 35% of the rate of human tendon damage. This injury always occurs during motor activity, of which men aged 30 – 50 are the most susceptible [30, 31]. A clinical study found that the male proportion and the mean age of the entire study were 88.9% and 36.9 years, consistent with our research [32]. We also detected that patients were keen on exercise and usually in good physical condition before the tendon got hurt. For such patients, they were enthusiastic about rehabilitation.
Despite it usually suddenly occurring, however, there is still a fair amount of chronic Achilles tendon rupture that is ascribed to misdiagnosis or disregarding this injury. Chronic Achilles tendon rupture mainly presents with cicatricial tissues irregularly buttressing the rupture gap and causing broken ends retraction, gastrocnemius atrophy, and gait incoordination [33, 34]. Consequently, these uncertainties and diversity produce different therapeutic regimens compared to acute injuries. Therefore, we excluded patients with chronic Achilles tendon rupture from this study.
Many different techniques have been introduced for operative interventions. Previous research pointed out that open surgery had a deficiency in wound healing and deep infection [35,36,37]. Although the percutaneous method minimized the wound complications, it increased the risk of the sural nerve injury and re-rupture [38]. A cadaveric study also observed that a minimally invasive method could be safe for sural nerves if the technique were used correctly [11, 39]. Campillo-Recio et al. disagreed that the percutaneous method was superior to the conservative treatment because the latter allowed the earlier weight-bearing and controlled rehabilitation protocol at a young age [40]. Furthermore, Attia et al. reported that the incidence of deep infection for open surgery was over 20% [41]. However, our results did not detect deep infection, possibly due to the small number of patients we included and the short follow-up period.
In 2014, a technique was described that included a mini-open surgery combined with a knotless percutaneous instrument (the Achilles Midsubstance SpeedBridge) to repair the injured site [42]. Hoskins et al. also reported that patients treated by an Arthrex PARS Achilles Jig System and their postoperative AOFAS and ATRS reached 90.3 and 88.0 points [9]. Concerning complications, they observed cases of re-rupture (1.2%) and suture irritation (1.2%). Notably, these follow-up data were similar to ours. Thus, patients could obtain a full weight bearing in the ninth week after surgery, conforming to the principle of rehabilitation.
For economic reasons, our institution did not import these instruments. Therefore, we referred to previous studies and used the simplified instrument, “the oral forceps and lumbar puncture needles.” This method was first reported by Ngai et al. and significantly reduced the surgery cost, which was widely used worldwide [18]. Liu et al. inserted an additional two anchors into the calcaneus from this previous study. However, we thought this also caused unnecessary waste [16]. Thus, our study used only three sutures and ensured these knots were staggered to avoid tissue cutting and centralization. Follow-up results also showed a relatively clinical outcome. Biz et al. used Tenolig technology to repair the Achilles tendon rupture, which cost nearly 1,000 euros [43].
In this study, the average operative time was 45.8 min after reaching mastery. Attia et al. included ten randomized controlled studies on open versus minimal invasive acute Achilles tendon rupture repair. The results showed that the average time was 51.0 and 29.7 min [41]. We took a longer surgical time for each procedure, which might be attributed to different calculations, defined as the time interval from surgical area disinfection to wound closing in our institution.
We recognize that there are several limitations to this study. Firstly, the relatively small series size, including its retrospective design, limits the reliability of our research. A power analysis was not conducted, and our results must be interpreted cautiously. Secondly, this minimally invasion surgery was performed by an individual surgeon. Discrepancies in the learning curves exist among surgeons, so its generalization remains unknown. For all this, our study has proved surgeons could be proficient with this technique over time. However, additional studies are needed to support these findings.
Conclusions
Our learning curve analysis demonstrated that the surgical competence in treating an acute Achilles tendon rupture with the “lumbar puncture needle and oval forceps” was obtained after the primary learning period of 12 cases. Furthermore, this technique benefited early rehabilitation and rapid return to normal life.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Hosny H, Salama W, Marzouk A, Kenawey M. Repair of traumatic avulsed Achilles tendon using double cerclage trans-calcaneal stainless steel sutures: prospective study and review of literature. Injury. 2020;51:1887–92. https://doi.org/10.1016/j.injury.2020.05.010.
Gould HP, Bano JM, Akman JL, Fillar AL. Postoperative rehabilitation following Achilles tendon repair: a systematic review. Sports Med Arthrosc Rev. 2021;29:130–45. https://doi.org/10.1097/JSA.0000000000000309.
Houshian S, Tscherning T, Riegels-Nielsen P. The epidemiology of Achilles tendon rupture in a Danish county. Injury. 1998;29:651–4. https://doi.org/10.1016/S0020-1383(98)00147-8.
Leppilahti J, Puranen J, Orava S. Incidence of Achilles tendon rupture. Acta Orthop Scand. 1996;67:277–9. https://doi.org/10.3109/17453679608994688.
Maffulli N, Waterston SW, Squair J, et al. Changing incidence of Achilles tendon rupture in Scotland: a 15-year study. Clin J Sport Med. 1999;9:157–60. https://doi.org/10.1097/00042752-199907000-00007.
Jozsa L, Kvist M, Balint BJ, et al. The role of recreational sport activity in Achilles tendon rupture. Am J Sports Med. 1989;17:338–43. https://doi.org/10.1177/036354658901700305.
Leppilahti J, Forsman K, Puranen J, Orava S. Outcome and prognostic factors of Achilles rupture repair using a new scoring method. Clin Orthop Relat Res. 1998;346:152 ???161. https://doi.org/10.1097/00003086-199801000-00022.
Möller M, Movin T, Granhed H, et al. Acute rupture of tendon Achillis. A prospective randomised study of comparison between surgical and non-surgical treatment. J Bone Joint Surg Br. 2001;83:843–8. https://doi.org/10.1302/0301-620X.83B6.0830843.
Pajala A, Kangas J, Ohtonen P, Leppilahti J. Rerupture and deep infection following treatment of total Achilles tendon rupture. ThJ Bone Joint Surg Am. 2002;84:2016–21. https://doi.org/10.2106/00004623-200211000-00017.
Wilkins R, Bisson LJ. Operative versus nonoperative management of acute Achilles tendon ruptures: a quantitative systematic review of randomized controlled trials. Am J Sports Med. 2012;40:2154–60. https://doi.org/10.1177/0363546512453293.
Ma GW, Griffith TG. Percutaneous repair of acute closed ruptured Achilles tendon: a new technique. Clin Orthop Relat Res. 1977;128:247–55.
Maffulli N, Sammaria G, Ziello S, et al. Percutaneous cruciate repair of ruptured Achilles tendon. J Orthop Surg Res. 2023;18:677. https://doi.org/10.1186/s13018-023-04167-9.
Hoskins T, Patel J, Choi JH, et al. Mini-open Achilles tendon repair: improving outcomes while decreasing complications. Foot Ankle Spec. 2023;16:363–9. https://doi.org/10.1177/19386400221078671.
Attia AK, Mahmoud K, d’Hooghe P, et al. Outcomes and complications of open versus minimally invasive repair of acute achilles tendon ruptures: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2023;51. https://doi.org/10.1177/03635465211053619.
Oliva F, Rugiero C, Giai Via A, et al. I.S.Mu.L.T. Achilles tendon ruptures guidelines. Muscle Ligaments and Tendons J. 2019;08:310. https://doi.org/10.32098/mltj.03.2018.03.
Liu Y, Lin L, Lin C, et al. The improved oval forceps suture-guiding method for minimally invasive Achilles tendon repair. Injury. 2018;49:1228–32. https://doi.org/10.1016/j.injury.2018.05.002.
Dai G, Zheng Y, Lu X, et al. The percutaneous oval forceps suture-guiding method with anchor nails for Achilles tendon repair. Injury. 2022;53:1552–6. https://doi.org/10.1016/j.injury.2021.09.007.
Ngai WYH, Chan SCF. An uncomplicated method for minimally invasive achilles tendon repair. J Foot Ankle Surg. 2010;49:208–11. https://doi.org/10.1053/j.jfas.2009.12.011.
Lee DK, Kim KK, Ham CU, et al. The learning curve for biplane medial open wedge high tibial osteotomy in 100 consecutive cases assessed using the cumulative summation method. Knee Surg Relat Res. 2018;30:303–10. https://doi.org/10.5792/ksrr.17.064.
Delman AM, Turner KM, Ammann AM, et al. A method for identifying the learning curve for the surgical stabilization of rib fractures. J Trauma Acute Care Surg. 2022;93:743–9. https://doi.org/10.1097/TA.0000000000003788.
Park YH, Kim W, Jang KS, et al. Learning curve of the Krackow suture technique for the repair of Achilles tendon rupture. Arch Orthop Trauma Surg. 2022;142:3747–54. https://doi.org/10.1007/s00402-021-04213-3.
Maccario C, Tan EW, Di Silvestri CA, et al. Learning curve assessment for total ankle replacement using the transfibular approach. Foot Ankle Surg. 2021;27:129–37. https://doi.org/10.1016/j.fas.2020.03.005.
Nilsson-Helander K, Thomeé R, Silbernagel KG, et al. The Achilles tendon Total Rupture Score (ATRS): development and validation. Am J Sports Med. 2007;35:421–6. https://doi.org/10.1177/0363546506294856.
Park YH, Cho HW, Choi JW, Kim HJ. Validation and cross-cultural adaptation of the Korean translation of the Achilles tendon total rupture score. BMC Musculoskelet Disord. 2021;22:876. https://doi.org/10.1186/s12891-021-04765-w.
Kitaoka HB, Alexander IJ, Adelaar RS, et al. Clinical rating systems for the ankle-hindfoot, midfoot, hallux, and lesser toes. Foot Ankle Int. 1994;15:349–53. https://doi.org/10.1177/107110079401500701.
Molloy A, Wood EV. Complications of the treatment of Achilles tendon ruptures. Foot Ankle Clin. 2009;14:745–59. https://doi.org/10.1016/j.fcl.2009.07.004.
Wj M, Morgenstern M, McNally MA, et al. Fracture-related infection: a consensus on definition from an international expert group. Injury. 2018;49:505–10. https://doi.org/10.1016/j.injury.2017.08.040.
Kim CW, Kim WR, Kim HY, et al. Learning curve for single-incision laparoscopic anterior resection for sigmoid colon cancer. J Am Coll Surg. 2015;221:397–403. https://doi.org/10.1016/j.jamcollsurg.2015.02.016.
Vakharia VN, Rodionov R, McEvoy AW, et al. Improving patient safety during introduction of novel medical devices through cumulative summation analysis. J Neurosurg. 2018;130:213–9. https://doi.org/10.3171/2017.8.JNS17936.
Eliasson P, Agergaard A-S, Couppé C, et al. The ruptured Achilles tendon elongates for 6 months after surgical repair regardless of early or late weightbearing in combination with ankle mobilization: a randomized clinical trial. Am J Sports Med. 2018;46:2492–502. https://doi.org/10.1177/0363546518781826.
Nilsson-Helander K, Grävare Silbernagel K, Thomeé R, et al. Acute Achilles tendon rupture: a randomized, controlled study comparing surgical and nonsurgical treatments using validated outcome measures. Am J Sports Med. 2010;38:2186–93. https://doi.org/10.1177/0363546510376052.
Okoroha KR, Ussef N, Jildeh TR, et al. Comparison of tendon lengthening with traditional versus accelerated rehabilitation after Achilles tendon repair: a prospective randomized controlled trial. Am J Sports Med. 2020;48:1720–6. https://doi.org/10.1177/0363546520909389.
Adukia V, Jha S. Management of chronic Achilles tendon ruptures. Orthopaedics and Trauma. 2024;38:11–7. https://doi.org/10.1016/j.mporth.2023.11.003.
Tarczyńska M, Szubstarski M, Gawęda K, et al. Outcomes of open repair treatment for acute versus chronic Achilles tendon ruptures: long-term retrospective follow-up of a minimum 10 years—a pilot study. Medical Sciences. 2023;11:25. https://doi.org/10.3390/medsci11020025.
Del Buono A, Volpin A, Maffulli N. Minimally invasive versus open surgery for acute Achilles tendon rupture: a systematic review. Br Med Bull. 2014;109:45–54. https://doi.org/10.1093/bmb/ldt029.
Bruggeman NB, Turner NS, Dahm DL, et al. Wound complications after open Achilles tendon repair: an analysis of risk factors. Clin Orthop Relat Res. 2004;427:63–6. https://doi.org/10.1097/01.blo.0000144475.05543.e7.
Saxena A, Maffulli N, Nguyen A, Li A. Wound complications from surgeries pertaining to the Achilles tendon: an analysis of 219 surgeries. JAPMA. 2008;98:95–101. https://doi.org/10.7547/0980095.
Yang B, Liu Y, Kan S, et al. Outcomes and complications of percutaneous versus open repair of acute Achilles tendon rupture: a meta-analysis. Int J Surg. 2017;40:178–86. https://doi.org/10.1016/j.ijsu.2017.03.021.
Wong J, Barrass V, Maffulli N. Quantitative review of operative and nonoperative management of Achilles tendon ruptures. Am J Sports Med. 2002;30:565. https://doi.org/10.1177/03635465020300041701.
Campillo-Recio D, Comas-Aguilar M, Ibáñez M, et al. Percutaneous Achilles tendon repair with absorbable suture: outcomes and complications. Revista Española de Cirugía Ortopédica y Traumatología. 2023;67:56–61. https://doi.org/10.1016/j.recot.2022.06.008.
Attia AK, Mahmoud K, d’Hooghe P, et al. Outcomes and complications of open versus minimally invasive repair of acute Achilles tendon ruptures: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2023;51:825–36. https://doi.org/10.1177/03635465211053619.
Demetracopoulos CA, Gilbert SL, Young E, et al. Limited-open Achilles tendon repair using locking sutures versus nonlocking sutures: an in vitro model. Foot Ankle Int. 2014;35:612–8. https://doi.org/10.1177/1071100714524550.
Biz C, Cerchiaro M, Belluzzi E, et al. Long term clinical–functional and ultrasound outcomes in recreational athletes after achilles tendon rupture: ma and griffith versus tenolig. 2021. https://doi.org/10.3390/medicina57101073.
Acknowledgements
We appreciate all co-authors for their contributions to this study and the writing of this manuscript. The authors thank AiMi Academic Services (www.aimieditor.com) for English language editing and review services.
Funding
This work is Supported by National Natural Science Foundation of China (82272469) and Beijing Key Clinical Specialty Project.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. Material preparation, data collection, and analysis were performed by ZYR, WHZ, and ZBZ. WHZ and ZBZ wrote the first draft of the manuscript. SD and YLG were responsible for the production of the study illustrations. Throughout the process, YL and JLZ provided guidance and insight. All authors have read and approved the final manuscript for submission.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Beijing Chaoyang Hospital. The Ethics Committee of Beijing Chaoyang Hospital waived the requirement for written informed consent because the study was retrospective, it had no adverse effect on patients’ health, and it reported anonymized patient data. The authors announce that all methods were performed in accordance with the relevant guidelines and regulations. This study is a retrospective study and not a clinical trial; therefore, trial registration is not applicable.
Consent for publications
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhao, Y., Wang, H., Zhao, B. et al. The learning curve for minimally invasive Achilles repair using the “lumbar puncture needle and oval forceps” technique. BMC Musculoskelet Disord 25, 373 (2024). https://doi.org/10.1186/s12891-024-07489-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12891-024-07489-9