Abstract
Background
Allergic asthma is a type I allergic reaction mediated by serum Immunoglobulin E (IgE). B cell-mediated humoral immune response to allergens in the pathophysiology of allergic asthma have not been thoroughly elucidated. Peripheral helper T cells (Tph) and follicular helper T cells (Tfh) promote B cell differentiation and antibody production in inflamed tissues.
Objective
To investigate the roles of B cell subsets, Tph cell subsets and Tfh cell subsets in allergic immune responses.
Methods
Circulating B cell subsets, Tph cell subsets and Tfh cell subsets in 33 children with allergic asthma and 17 healthy children were analyzed using multicolor flow cytometry. The level of serum total IgE was also assessed.
Results
Our study found that CD27+CD38+ plasmablasts and CD24hiCD38hi transitional B cells increased and were correlated with serum total IgE level, CD27− naive B cells and CD24hiCD27+ B cells decreased in children with allergic asthma. CXCR5− Tph, CXCR5−ICOS+ Tph, CXCR5−ICOS+PD-1+ Tph, CXCR5+ICOS+ Tfh and CXCR5+ICOS+PD-1+ Tfh increased in children with allergic asthma. Further analysis showed increased Tph2, Tph17, Tfh2 and Tfh17 subtypes while decreased Tph1 and Tfh1 subtypes in children with allergic asthma. Most interestingly, Tph2 or Tfh2 subtypes had a positive correlation with serum total IgE level.
Conclusion
Overall, these results provide insight into the allergens elicited B, Tph or Tfh cell response and identify heretofore unappreciated CD24hiCD38hi transitional B cells, CD24hiCD27+ B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 subtypes and Tfh2 subtypes response to allergens.
Similar content being viewed by others
Introduction
Hypersensitivity immune responses to allergens and other environmental factors, including helper T cell cytokine responses and the production of mmunoglobulin E (IgE) antibodies, are key immunologic features of allergic diseases [1]. Memory B cells, plasmablasts and antibody-producing cells arise during adaptive humoral immune responses in germinal center (GC) [2,3,4]. Regulatory CD24hiCD27+ B cells and CD24hiCD38hi transitional B cells have been identified to down-regulate allergic and auto-immune inflammation, suppress pro-inflammatory Th1/Th17 responses and induce regulatory T cells (Tregs) by producing IL-10 and/or TGF-β [5,6,7]. Sumimoto K et al. [8] found that circulating CD24hiCD27+ B cells decreased while CD24hiCD38hi B cells increased in patients with type I autoimmune pancreatitis. However, circulating CD24hiCD27+ B cells and CD24hiCD38hi B cells decreased and functionally impaired in psoriatic arthritis and systemic sclerosis [9, 10]. Van der Vlugt LE et al. [11] found that CD24hiCD27+ B cells from patients with allergic asthma have impaired regulatory function in response to lipopolysaccharide. Investigations have also revealed that the IgE level can be regulated by regulatory B cells by producing IL-10 [12]. However, the fundamental question of whether B cell response and IgE antibody production are regulated by the same immunological pathway in allergic asthma is still a matter of debate.
T follicular helper (Tfh) cells are distinguished from other CD4+ T cells by their selective role in orchestrating GC responses and in promoting the development of memory B cells and long-lived plasma cells. Previous studies demonstrated that Tfh cells orchestrated systemic IgE production in patients with allergic asthma [11, 13]. Yao Y et al. [14] also found a positive correction between Tfh2 subtypes and IgE in patients with allergic asthma. Achour A et al. [15] found human regulatory B cells control Tfh cell development and maturation, and further suppress the antibody secretion. However, understanding the role of Tfh cells in the regulation of IgE antibodies in allergic immune responses to allergens remains limited.
Recently, T peripheral helper (Tph) cells were found in the peripheral blood to possess B cell help functions like Tfh cells [16, 17]. Tph cells possess phenotypic characteristics similar to Tfh cells, such as inducible T-cell costimulator (ICOS), programmed cell death 1-positive (PD-1), the cytokines interleukin (IL)-21, chemokine (C-X-C motif) ligand 13, thus having the ability to regulate B-cell differentiation [18]. However, Tph cells are distinguished from Tfh cells by their lacking the expression of chemokine C-X-C receptor (CXCR) 5. Interestingly, CXCR5− Tph cells have hitherto been divided into three subtypes based on differential expression of CXCR3 and C–C receptor 6 (CCR6): CXCR3+CCR6− Tph1, CXCR3−CCR6− Tph2, and CXCR3−CCR6+ Tph17 [19]. Recently, studies found increased peripheral blood circulation Tph cells in patients with IgG4-related disease (IgG4-RD) [20, 21], rheumatoid arthritis (RA) [18, 22], systemic lupus erythematosus (SLE) [23, 24] and Sjögren's Syndrome (SS) [25]. Further, Tph cells in patients with IgG4-RD contribute to B cell reaction and plasma cell formation [21, 26, 27]. Moreover, the increased circulating Tph cells is positively related to the disease activity of SLE [23, 24, 28]. However, whether circulating Tph cell subsets contributes to the regulation of IgE antibodies in allergic immune responses deserves further investigation.
Accordingly, to better understand the contribution of B cell subsets, Tph cell subsets and Tfh cell subsets to the production of IgE antibodies in response to allergens and to elucidate their roles in allergic immune responses, we analyze circulating B cell subsets, Tph cell subsets and Tfh cell subsets in children with allergic asthma using multicolor flow cytometry in the current study. Our observations suggest that CD24hiCD38hi transitional B cells, CD24hiCD27+ B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 subtypes and Tfh2 subtypes could play a key role in the regulation of IgE antibody production. Our observations suggest CD24hiCD38hi transitional B cells, CD24hiCD27+ B cells, CXCR5−ICOS+PD-1+ Tph and Tph2 subtypes indreased in children with allergic asthma. Hence, allergic immune responses might be mediated by CD24hiCD38hi transitional B cells, CD24hiCD27+ B cells, CXCR5−ICOS+PD-1+ Tph and Tph2 subtypes.
Materials and methods
Subjects
In the current study, a total of 33 children with allergic asthma were recruited and met diagnostic criteria of “Guidelines for diagnosis, prevention and treatment of bronchial asthma in children” formulated by Chinese Medical Association. No children had a history of allergen-specific immunotherapy. Treatment with antihistamines and corticosteroids was stopped at least 4 weeks before children entered the study. Seventeen sex- and age-matched healthy children (HC) without allergic or respiratory diseases or experienced asthma-like syndromes were recruited for the present study (Table 1). EDTA-K2 anticoagulated peripheral blood were collected from all participants and were detected within 24 h of admission. Level of specific IgE to allergens and serum total IgE level was assessed using IMMAGE 800 specific protein analyzer (Beckman Coulter, USA). Written informed consents were obtained from parents of study participants. The experimental protocol followed the guidelines of the Declaration of Helsinki and was approved by the Human Ethics Committee of Zhejiang Provincial People’s Hospital.
Flow cytometry analysis
Peripheral blood mononuclear cells (PBMCs) were isolated from subjects by density-gradient centrifugation. Human PBMCs (106 cells/tube) were stained with following surface markers (Table 2). Fluorescence minus one (FMOs) were used for proper gate setting for all markers (Figure S1). After cells were incubated with these surface antibodies for 30 min at 4 °C in the dark, they were washed with Phosphate Buffered Saline (PBS) and then analyzed by BD FACSCantoTMflow cytometer (BD Biosciences, USA). Data were processed using BD FACSDivaTMsoftware (BD Biosciences, USA).
The B cell subsets were identified based on CD24, CD27 and CD38 after cells were gated on CD19+ B cells (Figure S2). Differential Tph and Tfh cell subsets were identified based on CXCR5, ICOS and PD-1 expression after cells were gated on CD3+CD4+ T cells. Tph or Tfh subtypes were determined according to CXCR3 and CCR6 expression after cells were gated on CXCR5− Tph or CXCR5+ Tfh (Figure S3).
Statistical analysis
Statistical analysis was performed with GraphPad Prism 5.01 software. The data are expressed by mean ± standard deviation. Statistical tests for data analysis included one-way ANOVA test and Spearman’s r correlation. P value < 0.05 were considered to be statistically significant.
Results
Increased CD27+CD38+ plasmablasts and CD24hiCD38hi transitional B cells while decreased CD24hiCD27+ B cells in children with allergic asthma
CD27+CD38+ plasmablasts and memory B cells are terminally differentiated B cells that arise during adaptive humoral immune responses. The frequency of CD19+ B cells (Fig. 1A, B), CD27+ memory B cells (Fig. 1C, E), CD24hiCD27− B cells (Fig. 1G, I), CD24+CD27− B cells (Fig. 1H, I), CD24hiCD38hi transitional B cells (Fig. 1J, M), CD24intCD38int B cells (Fig. 1K, M), CD27+CD38+ plasmablasts (Fig. 1N, Q) and CD27−CD38− B cells (Fig. 1P, Q) were significantly increased while the frequency of CD27− naïve B cells (Fig. 1D, E), CD24hiCD27+ B cells (Fig. 1F, I), CD24hiCD38− B cells (Fig. 1L, M) and CD27+CD38− B cells (Fig. 1O, Q) were significantly decreased in children with allergic asthma. Our data further suggest that B cell responses in children with allergic asthma were different compared to HC, which may be induced by allergens or other factors.
Increased CD27+CD38+ plasmablasts and CD24hiCD38hi transitional B cells while decreased CD24hiCD27+ B cells in children with allergic asthma. Frequency of CD19+ B cells in lymphocytes (A) and corresponding typical flow cytometry plot (B). Frequency of CD27+ memory B cells (C), CD27− naïve B cells (D) in CD19+ B cells and corresponding typical flow cytometry plot (E). Frequency of CD24hiCD27+ B cells (F), CD24hiCD27− B cells (G), CD24+CD27− B cells (H) in CD19+ B cells and corresponding typical flow cytometry plot (I). Frequency of CD24hiCD38hi transitional B cells (J), CD24intCD38int B cells (K), CD24hiCD38− B cells (L) in CD19+ B cells and corresponding typical flow cytometry plot (M). Frequency of CD27+CD38+ plasmablasts (N), CD27+CD38− B cells (O), CD27−CD38− B cells (P) in CD19+ B cells and corresponding typical flow cytometry plot (Q). All above cells are in CD19+ B cells of peripheral blood of children with allergic asthma (n = 33) and children with HC (n = 17). Symbols represent individual samples, the horizontal line represents the median of all data points, and error bars indicate the interquartile range
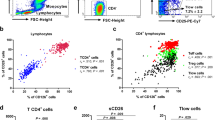
Expansion of CXCR5− Tph, CXCR5−ICOS+ Tph, CXCR5−ICOS+PD-1+ Tph, CXCR5+ICOS+ Tfh and CXCR+ICOS+PD-1+ Tfh cells and skewing to Tph2, Tph17, Tfh2 and Tfh17 subtypes in patients with allergic asthma
Since CD4+ T cells promotes B cells differentiation and antibody production in inflamed tissues, we further investigated different Tph and Tfh cell subsets. We first investigated the frequency of circulating Tph and Tfh from 33 children with allergic asthma, 26 of whom were followed up for Tph and Tfh subtypes. First, we found a higher frequency of CXCR5− Tph (Fig. 2A, C), CXCR5−ICOS+ Tph (Fig. 2D, F) and CXCR5−ICOS+PD-1+ Tph (Fig. 2G, J) in children with allergic asthma. Then, we further analyzed three Tph subtypes and found a lower frequency of Tph1 subtypes while a higher frequency of Tph2 and Tph17 subtypes in children with allergic asthma than those of HC (Fig. 2K-N).
Expansion of CXCR5− Tph, CXCR5−ICOS+ Tph, CXCR5−ICOS+PD-1+ Tph, CXCR5+ICOS+ Tfh and CXCR+ICOS+PD-1+ Tfh cells and skewing to Tph2, Tph17, Tfh2 and Tfh17 subtypes in patients with allergic asthma. Frequency of CXCR5− Tph (A), CXCR+ Tfh (B) in CD3+CD4+ T cells and corresponding typical flow cytometry plot (C). Frequency of CXCR5−ICOS+ Tph (D), CXCR5+ICOS + Tfh (E) in CD3+CD4+ T cells and corresponding typical flow cytometry plot (F). Frequency of CXCR5−ICOS+PD-1+ Tph (G) in CD3+CD4+ T cells and corresponding typical flow cytometry plot (H). Frequency of CXCR5+ICOS+PD-1+ Tfh (I) in CD3+CD4+ T cells and corresponding typical flow cytometry plot (J) in children with allergic asthma. Frequency of Tph1 subtypes (K), Tph2 subtypes (L) and Tph17 subtypes (M) in CXCR5− Tph cells and corresponding typical flow cytometry plot (N) in children with allergic asthma (n=26) and HC (n=17). Frequency of Tfh1 subtypes (O), Tfh2 subtypes (P) and Tfh17 subtypes (Q) in CXCR5+ Tfh cells and corresponding typical flow cytometry plot (R) in children with allergic asthma (n = 26) and HC (n = 17). The horizontal line represents the median of all data points, and error bars indicate the interquartile range
Similarly, we also found a lower frequency of CXCR5+ Tfh (Fig. 2B, C) while a higher frequency of CXCR5+ICOS+ Tfh (Fig. 2E, F) and CXCR+ICOS+PD-1+ Tfh (Fig. 2I, J) in children with allergic asthma. We then further analyzed three Tfh subtypes and found a lower frequency of Tfh1 subtypes while a higher frequency of Tfh2 and Tfh17 subtypes in children with allergic asthma than those of HC (Fig. 2O-R). In all, our findings indicate that skewed polarization of Tph2, Tph17, Tfh2 and Tfh17 subtypes may be a factor in the exaggerating immune response of allergic asthma.
Serum total IgE level positively correlate with CD27+CD38+ plasmablasts, CD24hiCD38hi transitional B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 and Tfh2 subtypes in children with allergic asthma
To further explore the potential contribution to dysregulated B cell subsets, Tph or Tfh subsets in the development of allergic asthma, we analyzed correlation between frequency of B cell subsets, Tph or Tfh subsets and serum total IgE level. As expected, the frequency of CD27+CD38+ plasmablasts, CD24hiCD38hi transitional B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 subtypes and Tfh2 subtypes were weak positively correlated with serum total IgE level in children with allergic asthma (Fig. 3A-F). However, no correlation was found between other B cell subsets, Tph or Tfh subsets and serum total IgE level (Figure S4).
Serum total IgE level positively correlate with CD27+CD38+ plasmablasts, CD24hiCD38hi transitional B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 subtypes and Tfh2 subtypes in children with allergic asthma. The positive correlation between CD27+CD38+ plasmablasts (A), CD24hiCD38hi transitional B cells (B), CXCR5− Tph (C), CXCR5−ICOS+PD-1+ Tph (D), Tph2 subtypes (E) or Tph2 subtypes (F) and serum total IgE level
Discussion
Allergic asthma is IgE-mediated type I hypersensitivity reaction to allergens [29]. Efforts are being made to understand the dysregulation of IgE production in patients with allergic asthma. In the current study, we analyzed changes in circulating B cell subsets, Tfh cell subsets, and Tph cell subsets in children with allergic asthma. A novel concept derived from our study was IgE antibody production was closely related to CD27+CD38+ plasmablasts, CD24hiCD38hi transitional B cells, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 subtypes and Tfh2 subtypes. In the current study, we sought to obtain a better understanding of B cell response in children with allergic asthma.
CD27 is a recognized surface marker of memory B cells [30]. We found increased CD19+ B cells, CD27+ memory B cells and CD27+CD38+ plasmablasts while decreased CD27− naïve B cells in children with allergic asthma. Moreover, we found a weak positive correlation between CD27+CD38+ plasmablasts and serum total IgE level. These results demonstrated that the allergen induced B cell activation and a high plasmablasts response, consistent with previous studies [4, 31, 32]. Currently, Flores-Borja Fet al. [33] and Noble A et al. [34] reported that CD24hiCD38hi B cells in asthmatic mice induced CD4+CD25− T effector cells transform into Tregs, so as to inhibit allergic airway inflammation. Our observations, for the first time, demonstrated that increased CD24hiCD38hi transitional B cells in children with allergic asthma were weakly positively correlated with serum total IgE level, indicating that CD24hiCD38hi transitional B cells may emerged after exposure to allergens.
We also found significantly decreased CD24hiCD27+ B cells in children with allergic asthma, consistent with previous study [1, 2, 4, 6, 11]. Therefore, together with our findings, indicate that CD27+CD38+ plasmablasts and CD24hiCD38hi transitional B cells could play an important role in IgE antibody production.
Tph cells hardly express CXCR5 [24, 35], and can promote B cell differentiation and the production of autoantibodies [28, 36, 37]. Previous studies have shown that Tph cells regulate B cell response and plasma cell differentiation in rheumatoid arthritis [18]. Ekman I et al. found increased circulating Tph cells in children with newly diagnosed type I diabetes, which has the potential to further become a biomarker of disease progress, and monitor the effect of immunotherapy [38]. Recent study have shown that Tph cells are associated with serum IgG level and may be a biomarker for monitoring disease activity [26]. In the current study, we observed increased frequency of CXCR5− Tph, CXCR5−ICOS+ Tph, CXCR5−ICOS+PD-1+ Tph, Tph2 and Tph17 subtypes in children with allergic asthma. Most interestingly, CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph and Tph2 subtypes were weakly positively correlated with serum total IgE level, suggesting that CXCR5− Tph, CXCR5−ICOS+PD-1+ Tph and Tph2 subtypes may play an important role in the excessive accumulation of serum total IgE in allergic asthma.
Previous reports have demonstated that a skewed distribution of circulating Tfh2 subtypes contributes to the pathogenesis of inflammatory airway diseases such as allergic rhinitis and asthma, and that Tfh2 subtypes promote the polarization of IgE production in patients with allergic asthma [14, 39,40,41,42,43]. We found a lower frequency of CXCR5+ Tfh or Tfh1 subtypes and a higher frequency of CXCR5+ICOS+ Tfh, CXCR+ICOS+PD-1+ Tfh, Tfh2 or Tfh17 subtypes in children with allergic asthma. Our data also showed that Tfh2 subtypes were positively correlated with serum total IgE level in children with allergic asthma. Achour A et al. [15] found that humans regulate B cells to control Tfh maturation and inhibit Tfh cell-mediated antibody secretion. Thus, decreased CD24hiCD27+ B cells could bring about excessive Tfh cell-dependent humoral responses and might lead to aberrant immune responses.
In summary, it has become clear that the increased CD24hiCD38hi transitional B cell, CXCR5−ICOS+PD-1+ Tph, Tph2 and Tfh2 subtypes may contribute to the development of aberrant immune responses in children with allergic asthma. Although this study was conducted in a small number of subjects, a deeper understanding of human Tph or Tfh and B cell subsets interrelations is worthy of pursuit to elaborate new therapeutic strategies in allergic asthma. Further research is required to elucidate the role of various B cell subsets, Tph and Tfh in allergic asthma.
Availability of data and materials
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Naja AS, Permaul P, Phipatanakul W. Taming asthma in school-aged children: a comprehensive review. J Allergy Clin Immunol Pract. 2018;6(3):726–35. https://doi.org/10.1016/j.jaip.2018.01.023.
Bousquet J, et al. Factors responsible for differences between asymptomatic subjects and patients presenting an IgE sensitization to allergens. A GA2LEN project. Allergy. 2006;61(6):671–80. https://doi.org/10.1111/j.1398-9995.2006.01048.x.
Douwes J, et al. Importance of allergy in asthma: an epidemiologic perspective. Curr Allergy Asthma Rep. 2011;11(5):434–44. https://doi.org/10.1007/s11882-011-0215-6.
Cyster JG, Allen CDC. B cell responses: cell interaction dynamics and decisions. Cell. 2019;177(3):524–40. https://doi.org/10.1016/j.cell.2019.03.016.
Carter NA, et al. Mice lacking endogenous IL-10-producing regulatory B cells develop exacerbated disease and present with an increased frequency of Th1/Th17 but a decrease in regulatory T cells. J Immunol. 2011;186(10):5569–79. https://doi.org/10.4049/jimmunol.1100284.
Mauri C, Bosma A. Immune regulatory function of B cells. Annu Rev Immunol. 2012;30:221–41. https://doi.org/10.1146/annurev-immunol-020711-074934.
Siewe B, et al. Regulatory B cell frequency correlates with markers of HIV disease progression and attenuates anti-HIV CD8+ T cell function in vitro. J Leukoc Biol. 2013;93(5):811–8. https://doi.org/10.1189/jlb.0912436.
Sumimoto K, et al. The role of CD19+ CD24high CD38high and CD19+ CD24high CD27+ regulatory B cells in patients with type 1 autoimmune pancreatitis. Pancreatology. 2014;14(3):193–200. https://doi.org/10.1016/j.pan.2014.02.004.
Mavropoulos A, et al. IL-10 producing Bregs are impaired in psoriatic arthritis and psoriasis and inversely correlate with IL-17- and IFNγ-producing T cells. Clin Immunol. 2017;184:33–41. https://doi.org/10.1016/j.clim.2017.04.010.
Mavropoulos A, et al. Breg cells are numerically decreased and functionally impaired in patients with systemic sclerosis. Arthritis Rheumatol. 2016;68(2):494–504. https://doi.org/10.1002/art.39437.
van der Vlugt LE, et al. CD24(hi)CD27(+) B cells from patients with allergic asthma have impaired regulatory activity in response to lipopolysaccharide. Clin Exp Allergy. 2014;44(4):517–28. https://doi.org/10.1111/cea.12238.
Palomares O, et al. Regulatory T cells and immune regulation of allergic diseases: roles of IL-10 and TGF-β. Genes Immun. 2014;15(8):511–20. https://doi.org/10.1038/gene.2014.45.
Kamekura R, et al. Alteration of circulating type 2 follicular helper T cells and regulatory B cells underlies the comorbid association of allergic rhinitis with bronchial asthma. Clin Immunol. 2015;158(2):204–11. https://doi.org/10.1016/j.clim.2015.02.016.
Yao Y, et al. Role of allergen-specific T-follicular helper cells in immunotherapy. Curr Opin Allergy Clin Immunol. 2018;18(6):495–501. https://doi.org/10.1097/aci.0000000000000480.
Achour A, et al. Human regulatory B cells control the T(FH) cell response. J Allergy Clin Immunol. 2017;140(1):215–22. https://doi.org/10.1016/j.jaci.2016.09.042.
Wacleche VS, Wang R, Rao DA. Identification of T Peripheral Helper (Tph) Cells. Methods Mol Biol. 2022;2380:59–76. https://doi.org/10.1007/978-1-0716-1736-6_6.
Yoshitomi H, Ueno H. Shared and distinct roles of T peripheral helper and T follicular helper cells in human diseases. Cell Mol Immunol. 2021;18(3):523–7. https://doi.org/10.1038/s41423-020-00529-z.
Rao DA, et al. Pathologically expanded peripheral T helper cell subset drives B cells in rheumatoid arthritis. Nature. 2017;542(7639):110–4. https://doi.org/10.1038/nature20810.
Liu W, et al. Increased peripheral helper T cells type 17 subset correlates with the severity of psoriasis vulgaris. Immunol Lett. 2021;229:48–54. https://doi.org/10.1016/j.imlet.2020.11.005.
Cargill T, Barnes E, Culver EL. Expansion of a Novel Subset of PD1+CXCR5-CD4+ T Peripheral Helper Cells in IgG4-Related Disease. Clin Transl Gastroenterol. 2020;11(1):e00111. https://doi.org/10.14309/ctg.0000000000000111.
Yabe H, et al. Cytotoxic Tph-like cells are involved in persistent tissue damage in IgG4-related disease. Mod Rheumatol. 2021;31(1):249–60. https://doi.org/10.1080/14397595.2020.1719576.
Fortea-Gordo P, et al. Two populations of circulating PD-1hiCD4 T cells with distinct B cell helping capacity are elevated in early rheumatoid arthritis. Rheumatology (Oxford). 2019;58(9):1662–73. https://doi.org/10.1093/rheumatology/kez169.
Makiyama A, et al. Expanded circulating peripheral helper T cells in systemic lupus erythematosus: association with disease activity and B cell differentiation. Rheumatology (Oxford). 2019;58(10):1861–9. https://doi.org/10.1093/rheumatology/kez077.
Lin J, et al. PD-1+CXCR5-CD4+T cells are correlated with the severity of systemic lupus erythematosus. Rheumatology (Oxford). 2019;58(12):2188–92. https://doi.org/10.1093/rheumatology/kez228.
Verstappen GM, et al. Attenuation of Follicular Helper T Cell-Dependent B Cell Hyperactivity by Abatacept Treatment in Primary Sjögren’s Syndrome. Arthritis Rheumatol. 2017;69(9):1850–61. https://doi.org/10.1002/art.40165.
Zhang P, et al. Expanded CD4+CXCR5-PD-1+ peripheral T helper like cells and clinical significance in IgG4-related disease. Clin Immunol. 2022;237:108975. https://doi.org/10.1016/j.clim.2022.108975.
Kamekura R, et al. Circulating PD-1(+)CXCR5(-)CD4(+) T cells underlying the immunological mechanisms of IgG4-related disease. Rheumatol Adv Pract. 2018;2(2):rky043. https://doi.org/10.1093/rap/rky043.
Bocharnikov AV, et al. PD-1hiCXCR5- T peripheral helper cells promote B cell responses in lupus via MAF and IL-21. JCI Insight. 2019;4(20):e130062. https://doi.org/10.1172/jci.insight.130062.
Gans MD, Gavrilova T. Understanding the immunology of asthma: Pathophysiology, biomarkers, and treatments for asthma endotypes. Paediatr Respir Rev. 2020;36:118–27. https://doi.org/10.1016/j.prrv.2019.08.002.
Cancro MP, Tomayko MM. Memory B cells and plasma cells: The differentiative continuum of humoral immunity. Immunol Rev. 2021;303(1):72–82. https://doi.org/10.1111/imr.13016.
Nutt SL, et al. The generation of antibody-secreting plasma cells. Nat Rev Immunol. 2015;15(3):160–71. https://doi.org/10.1038/nri3795.
van de Veen W, et al. Role of regulatory B cells in immune tolerance to allergens and beyond. J Allergy Clin Immunol. 2016;138(3):654–65. https://doi.org/10.1016/j.jaci.2016.07.006.
Flores-Borja F, et al. CD19+CD24hiCD38hi B cells maintain regulatory T cells while limiting TH1 and TH17 differentiation. Sci Transl Med. 2013;5(173):173ra23.
Noble A, Zhao J. Follicular helper T cells are responsible for IgE responses to Der p 1 following house dust mite sensitization in mice. Clin Exp Allergy. 2016;46(8):1075–1082.
Chen W, Yang F, Lin J. Tph cells expanded in primary sjögren’s syndrome. Front Med (Lausanne). 2022;9:900349. https://doi.org/10.3389/fmed.2022.900349.
Rao DA. T cells that help B cells in chronically inflamed tissues. Front Immunol. 2018;9:1924. https://doi.org/10.3389/fimmu.2018.01924.
Yoshitomi H. Peripheral helper T cell responses in human diseases. Front Immunol. 2022;13:946786. https://doi.org/10.3389/fimmu.2022.946786.
Ekman I, et al. Circulating CXCR5(-)PD-1(hi) peripheral T helper cells are associated with progression to type 1 diabetes. Diabetologia. 2019;62(9):1681–8. https://doi.org/10.1007/s00125-019-4936-8.
Olatunde AC, Hale JS, Lamb TJ. Cytokine-skewed Tfh cells: functional consequences for B cell help. Trends Immunol. 2021;42(6):536–50. https://doi.org/10.1016/j.it.2021.04.006.
Zhou M, et al. Circulating TFH cells is correlated with disease activity in anti-MDA5 antibody positive idiopathic inflammatory myopathies. Clin Exp Rheumatol. 2021;39(4):804–10. https://doi.org/10.55563/clinexprheumatol/gfqlcz.
Chen W, et al. Follicular helper T cells and follicular regulatory T cells in the immunopathology of primary Sjögren’s syndrome. J Leukoc Biol. 2021;109(2):437–47. https://doi.org/10.1002/jlb.5mr1020-057rr.
Varricchi G, et al. T follicular helper (Tfh ) cells in normal immune responses and in allergic disorders. Allergy. 2016;71(8):1086–94. https://doi.org/10.1111/all.12878.
Yao Y, et al. Roles of follicular helper and regulatory T cells in allergic diseases and allergen immunotherapy. Allergy. 2021;76(2):456–70. https://doi.org/10.1111/all.14639.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural Science Foundation of Zhejiang Province (grant no. Y18H200014).
Author information
Authors and Affiliations
Contributions
LQ designed the experiment. QY performed flow cytometry. LQ and QJ confirm the authenticity of all original data. QJ, CL and YZ conducted experiments and data analysis and prepared the first manuscript. YZ revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Written informed consents were obtained from parents of study participants. The experimental protocol followed the guidelines of the Declaration of Helsinki and was approved by the Human Ethics Committee of Zhejiang Provincial People’s Hospital.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhu, Y., Jiang, Q., Lei, C. et al. The response of CD27+CD38+ plasmablasts, CD24hiCD38hi transitional B cells, CXCR5−ICOS+PD-1+ Tph, Tph2 and Tfh2 subtypes to allergens in children with allergic asthma. BMC Pediatr 24, 154 (2024). https://doi.org/10.1186/s12887-024-04622-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12887-024-04622-4