Abstract
Background
The study describes the clinical manifestations and variant screening of two Chinese siblings with primary ciliary dyskinesia (PCD). They carry the same DNAAF2 genotype, which is an extremely rare PCD genotype in the Chinese population. In addition, the study illustrated an overview of published variants on DNAAF2 to date.
Methods
A two-child family was recruited for the study. Clinical manifestations, laboratory tests, bronchoscopic and otoscopic images, and radiographic data were collected. Whole blood was collected from siblings and their parents for whole-exome sequencing (WES) and Sanger sequencing to screen variants.
Results
The two siblings exhibited typical clinical manifestations of PCD. Two compound heterozygous variants in DNAAF2 were detected in both by WES. Nonsense variant c.156 C>A and frameshift variant c.177_178insA, which was a novel variant.
Conclusion
The study identified a novel variant of DNAAF2 in Chinese children with a typical phenotype of PCD, which may enrich our knowledge of the clinical, diagnostic and genetic information of DNAAF2-induced PCD in children.
Similar content being viewed by others
Background
According to European Respiratory Society (ERS) and American Thoracic Society (ATS), PCD is a rare inherited autosomal recessive disease caused by impaired function of the motile cilia, with an estimated incidence of one in 10,000–20,000 live births worldwide [1], resulting in recurrent respiratory inflammation, bronchiectasis, sinusitis, otitis media, and neonatal respiratory distress [2, 3]. Due to the random asymmetry of the left-right bodies, up to about 50% of PCD patients have situs inversus or ambiguous [4]. In addition to the clinical features, nasal nitric oxide (nNO) concentration, high speed video microscopy analysis (HSVMA) of cilia beat frequency and pattern, ciliary ultrastructure in cross section through transmission electron microscopy (TEM) are all helpful for the diagnosis of PCD, thereinto, genetic testing can help to confirm the diagnosis of PCD [2, 3].
The pathogenesis of PCD is cilia motility dysfunction. Movements of cilia can help to clear mucous from the airway, the flow of cerebrospinal fluid along the brain ventricular system, to support the transport of the oocyte to the uterus, and the movement of male germ cells along the female reproductive tract [5]. The normal structure of the cilia consists of the conserved basic 9 + 2 microtubule-based axonemes and several other functional modules [6]. Members of the multi-subunit motor protein complexes, outer dynein arms (ODAs) and the inner dynein arms (IDAs) are responsible for generating and regulating ciliary beating. Dynein arms are ATPase-based protein-complexes, hydrolyze ATP through dynein heavy chain, allowing the paired microtubules to slide relative to each other, leading to cilia curve over deformed microtubules [7, 8]. During the process, IDA causes the curvature of cilia, and ODA accelerates the active sliding of outer microtubules, leading to the cilia to generate effective propulsion, effectively paddling forward and backward. Appropriate rheological properties and functional structure of cilia are the basis for maintaining mucociliary clearance, which is essential for clearance defense of the respiratory tract [9].
There are two different types of respiratory cilia ODA, proximal A-tubule (type 1) and distal A-tubule (type 2). The components of ODA are gradually assembled into large multiprotein complexes by dynein axonemal assembly factor (DNAAFs) in the cytoplasm. DNAAF2, a facilitator of dynein pre-assembly, also known as Ktu/PF13, has been reported to encode the cytoplasmic proteins responsible for ODA type 2 and IDA light DNALI1 from ciliary axonemes. Variants in DNAAF2 lead to loss of cilia motility and PCD [5, 6].
In this paper, we reported the clinical manifestations, diagnosis and treatment processes of two siblings with PCD caused by the same compound heterozygous variants in DNAAF2, c.156 C>A /c.177_178insA, an extremely rare PCD genotype in Chinese population, which had never discovered in children. Furthermore, c.177_178insA gene variant is considered as a novel variant.
Methods
Patients
The proband, a 7-year-old girl (II:1) and a 10-month-boy (II:2), were both born at full term, without remarkable family histories. Neither of the two probands had any history of fatty diarrhea, malnutrition, meconium intestinal obstruction. Both had normal cellular and humoral immune function, normal blood amylase and lipase levels, and negative pulmonary tuberculosis infection test. Clinical manifestations, laboratory tests, bronchoscopic and otoscopic images, and radiographic data were collected. Respiratory pathogens were identified from cultured bronchoalveolar lavage specimens. Whole blood was collected from siblings (II:1 & II:2) and their parents for whole-exome sequencing (WES) and Sanger sequencing. Literature of all DNAAF2 variants since the first reported were also searched. The clinical data of these patients were summarized. All methods were carried out in accordance with relevant guidelines and regulations. Informed consent was obtained from the parents. This study was conducted in accordance with the revised Declaration of Helsinki and approved by Ethics Committee of Henan Children’s Hospital (Approved No. 2022-k-049).
NNO measurement
Nasal NO was measured during quiet exhalation with EcoMedics CLD 88 ANALYZER (Dürnten, Switzerland). nNO measurement of proband II:1 was in line with ATS/ERS recommendations [2, 3]. For proband II:2, NO values were tested in both nostrils for 60s during tidal breathing and the larger value was recorded. nNO production (nL/min) was calculated by multiplying the nNO concentration (ppb) by the sampling flow rate (0.3 L/min).
Transmission electron microscopy
Obtained by bronchial biopsy, bronchial mucosa biopsy specimens were immediately immersed in glutaraldehyde at 4 °C, washed overnight, fixed in 1% osmium tetroxide, dehydrated, and then embedded in epoxy resin. After polymerization, sample sections were placed onto copper grids and stained with aqueous uranyl acetate and Reynold’s lead citrate. Ciliary ultrastructure was analyzed with a transmission electron microscope (JEM-1400; Jeol, Tokyo, Japan).
Genetic sequencing
Genomic DNA samples were isolated from EDTA-treated peripheral blood of probands and their parents using Chemagic 360 kit (CMG-536, PerkinElmer, USA). With standard protocols, whole exome sequencing was performed at WE-HEALTH Biomedical Technology Company (Shanghai, China) and raw data was assessed for quality and sequenced using the Illumina HiSeq X System sequencer after enriched. Sequence read was aligned to the NCBI human reference genome (hg19) and variants were evaluated according to American College of Medical Genetics and Genomics (ACMG) classification criteria. Sanger-sequencing was then used to confirm the genetic variation.
Results
The proband II:1 presented with wet cough and running nose for 2 months. She was diagnosed with pneumonia and treated in a local hospital for 15 days, however, the symptoms were not alleviated. Computed tomography (CT) (Fig. 1A, C) scanning of her lungs and sinuses showed atelectasis and potential bronchiectasis in the right middle lobe, and sinusitis with hypertrophy of tonsils and adenoids. Before being transferred to the respiratory medicine department of our hospital, she had suffered from recurrent respiratory tract infection for almost 5 years. Physical examination found pharyngeal congestion and tonsil swelling, with normal growth and development. Her nNO concentration (11.55nL/min) was well below the PCD-specific nNO cutoff value (77nL/min) [2, 3]. The pulmonary function tests were normal. Bronchoscopy revealed a large amount of mucilage secretions in lingula segments, left upper and right middle lobes (Fig. 1B). Bronchoalveolar lavage (BAL) culture showed that Haemophilus infuenzae (H. influenzae) was positive. Pure-tone audiometry confirmed conductive hearing loss. The air-bone gap of the right ear was10dB to 25dB, and that of the left ear was 15dB to 35dB. Otoscope and inner ear CT indicated otitis media in both of her ears, which was responsible for the hearing loss (Fig. 1D, E, F).
Clinical features of the probands, images A-F were of II:1 and G-L were of II:2. The chest high-resolution computed tomography (HRCT) scan showed bronchiectasis (red arrows) and atelectasis in the right middle lobe (A), sinusitis (C) and otitis media (D). Bronchoscopy revealed a large amount of mucilage secretions in the right middle lobe. Otoscope findings of otitis media in left ear (E) and the right one (F). Otoscope findings of otitis media of left and right ears (G&H). The CT scan of nasopharynx showed sinusitis (I&J). Chest HRCT scan illustrated atelectasis in lingular lobe (blue arrow, K&L)
Azithromycin plays an anti-inflammatory role in the treatment of H. influenzae. Budesonide and inhaled acetylcysteine were used for anti-inflammation and sputum dilution. In terms of pulmonary rehabilitation, mechanical vibration sputum expectoration, postural drainage and effective breathing techniques were performed to accelerate the clearance of lung mucus. During follow-up, she had improved symptoms but persistent atelectasis in the right middle lung (shown in supplemental Fig. 1) and recurrent sinusitis and otitis media (shown in supplemental Fig. 2).
The proband II:2 presented with cough and runny nose for one month. Actually, the runny nose initially started at the age of 2 months. He was diagnosed as otitis media (Fig. 1G&H), sinusitis (Fig. 1I&J), pneumonia in both lungs and atelectasis in lingular lobe (Fig. 1K&L), and hypertrophy of adenoids. Physical examination by stethoscope found crackles, and his growth and development were normal. Pure-tone audiometry confirmed conductive hearing loss. The air-bone gap of the right ear was 40dB to 60dB, and that of the left ear was 30dB to 55dB. Bilateral tympanotomy with tube placement and adenotomy were performed. His nNO was 9.84nL/min, even lower than that of proband II:1, which might be related to the recurrent respiratory infection. Pulmonary function tests showed obstructive ventilatory dysfunction. By transmission electron microscopy (TEM), ODAs and IDAs of bronchial cilia were not clearly displayed, indicating the possibility of the defect of ODAs and IDAs (Fig. 4). In addition, the echocardiography revealed residual left superior vena cava. Due to a history of emesis, he was diagnosed as intestine malrotation by abdominal ultrasound at 1.5 years old.
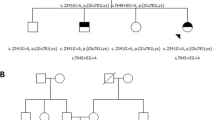
Sanger DNA sequencing chromatogram of probands and their parents. A novel variant DNAAF2, c.177_178insA (p.E60fs*3) was identified in the probands, their father had a heterozygous variant at the same location, whereas their mother did not. Another variant DNAAF2, c.156 C > A (p.Y52*,786) was also identified in both probands, their mother had a heterozygous variant at the same location, whereas their father did not
After treated with azithromycin, budesonide inhalation solution, and adjuvant mechanical vibration sputum expectoration, he was discharged soon. Although his symptoms improved during the 2-year follow-up, he still suffered from recurrent respiratory tract infection for 6–7 times, and his HRCT and otitis media became worse (shown in supplemental Figs. 3&4).
With the informed consent of the child’s parents, 2ml whole blood was separately collected from the siblings and their parents for WES and Sanger sequencing. Two compound heterozygous variants in DNAAF2 were found in both siblings, one nonsense variant of NM_018139: c.156 C > A (p.Y52*) was inherited from the mother (I:2) and one frameshift variant NM_018139: c.177_178insA (p.E60fs*3) was inherited from the father (I:1, Figs. 2 and 3). The variant of c.177_178insA (p.E60fs*3) has not been reported before.
Discussion
In this study, two compound heterozygous variants of DNAAF2 were identified in two siblings with similar PCD characteristics, including recurrent pneumonia dominated by the right middle and left lingular lobes with atelectasis, early bronchiectasis, sinusitis, otitis media, and significantly reduced nNO levels. One of the variants is the nonsense variant of NM_018139: c.156 C > A (p.Y52*), which has already been reported in a Chinese family and proved to be pathogenic [10]. Another variant is the frameshift variant (NM_018139: c.177_178insA (p.E60fs*3), which is a novel variant.
DNAAF2 is located on chromosome 14q21.3 and consists of three exons encoding cDNAs [10]. The variant p.E60fs*3 is located in exon1, near another variant p.Y52*. Exon1 is consists of 621 amino acids, and E60fs*3 variant leads to early transcription termination after 63 amino acids (Fig. 5). According to the standards and guidelines of American College of Medical Genetics and Genomics (ACMG), the frameshift variant p.E60fs*3 variant was pathogenic. In addition, none of cystic fibrosis (CF) or primary immune deficiency (PID) related genes were detected, which may also lead to recurrent pneumonia, atelectasis and bronchiectasis. Based on the clinical manifestations and hereditary mode of compound heterozygous variants from both parents, it is reasonable to consider that the novel p.E60fs*3 variant of DNAAF2 is responsible for the pathogenesis of PCD.
Variants of DNAAF2 gene identified in the probands and all published variants on protein level. The newly identified c.177_178insA (p.E60fs*3) variant structure (A) and its wide type (B), and the protein in bold (C) was another identified variant in this study and other published variants on protein level were all displayed (C)
Over 50 genes have been reported to be associated with PCD, including DNAH5, DNAH11, DNAI1, DNAI2, DNAL1, TXNDC3, DNAAF1, DNAAF2, etc. [1, 6, 11]. In 2008, two heterozygous variants c.C23A [pS8X] and c.1214-1215insACGATACCTGCGTGGC [p.G406Rfs89X] variants of DNAAF2 were first identified in two consanguineous families with PCD presented with recurrent respiratory tract infections, laterality defects and impaired fertility [6]. DNAAF2 functions as a facilitator of dynein pre-assembly. The dysfunction of DNAAF2 is associated with the specific defects of the interaction between intermediate and heavy chains in the cytoplasm, leading to complete or partial loss of ODAs and IDAs and loss of motility [6]. Since then, only 14 PCD cases have been reported to be caused by DNAAF2 with 17 variants. The cases of DNAAF2 variant are summarized in Table 1 and variants on protein level are illustrated in Fig. 5 [6, 10, 12,13,14,15,16,17,18,19,20,21,22,23,24]. DNAAF2 gene is rare for Chinese Children. As reported by Guan et al. in 81 Chinese children, the genes causing the highest variant rate of PCD was DNAH11, followed by HYDIN, DNAH5, CCDC39, DNAH1 and CCNO, no variant of DNAAF2 gene was detected [25]. In Chinese adult patients, Sun et.at have identified two heterozygous variants, c.C156A [p.Y52X] and c.C26A [p.S9X], in the DNAAF2 gene, leading to defects in the outer dynein arms and inner dynein arms, thereby resulting in PCD with the manifestation of male infertility [10]. Lu C et al. detected another two heterozygous variants, c.491T > C [p.L164P] and c.822del [p.A275Profs*10], in two females with bronchiectasis, sinusitis, situs inversus, and infertility. In addition, one woman with c.491T > C variant had scoliosis [24].
Consequently, we made an early diagnosis of PCD in two siblings with a rare novel variant of DNAAF2 gene. Early PCD may be easily misdiagnosed as common pneumonia or sinusitis. PCD should be considered when a child presented with recurrent pneumonia or atelectasis accompanied with recurrent sinusitis or otitis media. The characteristic HRCT manifestations include right middle and left lingular atelectasis, thickened bronchial wall, and mild bronchiectasis, which can provide a diagnostic clue for PCD. Moreover, the decrease of nNO is conducive to the further diagnosis, and genetic tests is conducive to confirming the diagnosis. TEM of bronchial cilia is also helpful for the diagnosis of PCD.
Conclusion
Our study suggested that, c.156 C>A/c.177_178insA variant of DNAAF2 is a novel pathogenic genotype in Chinese children with typical clinical features of PCD, which may broaden the gene spectrum, and enrich our knowledge of the clinical, diagnostic and genetic information of DNAAF2-induced PCD in children.
Data Availability
The datasets generated and/or analyzed during the current study are available in the Genome Sequence Archive (Genomics, Proteomics & Bioinformatics 2021) in National Genomics Data Center (Nucleic Acids Res 2022), China National Center for Bioinformation / Beijing Institute of Genomics, Chinese Academy of Sciences (accession number: HRA002729) that are publicly accessible at https://ngdc.cncb.ac.cn/gsa-human.
Abbreviations
- PCD:
-
Primary ciliary dyskinesia
- WES:
-
Whole-exome sequencing
- ERS:
-
European Respiratory Society
- ATS:
-
American Thoracic Society
- HSVMA:
-
High speed video microscopy analysis
- TEM:
-
Transmission electron microscopy
- ODAs:
-
Outer dynein arms
- IDAs:
-
Inner dynein arms
- DNAAFs:
-
Dynein axonemal assembly factor
- nNO:
-
Nasal nitric oxide
- CT:
-
Computed tomography
- BAL:
-
Bronchoalveolar lavage
- HRCT:
-
High-resolution computed tomography
- ACMG:
-
American College of Medical Genetics and Genomics
- CF:
-
Cystic fibrosis
- PID:
-
Primary immune deficiency
References
Mirra V, Werner C, Santamaria F. Primary ciliary dyskinesia: an update on clinical aspects, Genetics, diagnosis, and future treatment strategies. Front Pediatr. 2017 Jun;9:5:135.
Lucas JS, Barbato A, Collins SA, Goutaki M, Behan L, Caudri D, et al. European respiratory society guidelines for the diagnosis of primary ciliary dyskinesia. Eur Respir J. 2017;49(1):1601090.
Shapiro AJ, Davis SD, Polineni D, Manion M, Rosenfeld M, Dell SD, et al. Diagnosis of primary ciliary Dyskinesia. An official american thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2018;197(12):e24–e39.
Nöthe-Menchen T, Wallmeier J, Pennekamp P, Höben IM, Olbrich H, Loges NT, et al. Randomization of Left-right asymmetry and congenital heart defects: the role of DNAH5 in humans and mice. Circ Genom Precis Med. 2019 Oct;22. https://doi.org/10.1161/CIRCGEN.119.002686.
Wallmeier J, Nielsen KG, Kuehni CE, Lucas JS, Leigh MW, Zariwala MA, et al. Motile ciliopathies. Nat Rev Dis Primers. 2020 Sep;17(1):77.
Omran H, Kobayashi D, Olbrich H, Tsukahara T, Loges NT, Hagiwara H et al. Ktu/PF13 is required for cytoplasmic pre-assembly of axonemal dyneins. Nat 2008 Dec 4;456(7222):611–6.
Ibañez-Tallon I, Heintz N, Omran H. To beat or not to beat: roles of cilia in development and disease. Hum Mol Genet 2003 Apr 1;12 Spec No 1:R27–35.
Takeuchi K, Kitano M, Ishinaga H, Kobayashi M, Ogawa S, Nakatani K, et al. Recent advances in primary ciliary dyskinesia. Auris Nasus Larynx. 2016 Jun;43(3):229–36.
Lucas JS, Davis SD, Omran H, Shoemark A. Primary ciliary dyskinesia in the genomics age. Lancet Respir Med. 2020 Feb;8(2):202–16.
Sun M, Zhang Y, JiyunYang, Wang Y, Tan H, Wang H, et al. Novel compound heterozygous DNAAF2 mutations cause primary ciliary dyskinesia in a Han Chinese family. J Assist Reprod Genet. 2020 Sep;37(9):2159–70.
Niziolek M, Bicka M, Osinka A, Samsel Z, Sekretarska J, Poprzeczko M et al. PCD Genes-From Patients to Model Organisms and Back to Humans. Int J Mol Sci. 2022 Feb 3;23(3):1749.
Paff T, Kooi IE, Moutaouakil Y, Riesebos E, Sistermans EA, Daniels HJMA, et al. Diagnostic yield of a targeted gene panel in primary ciliary dyskinesia patients. Hum Mutat. 2018 May;39(5):653–65.
Emiralioğlu N, Taşkıran EZ, Koşukcu C, Bilgiç E, Atilla P, Kaya B, et al. Genotype and phenotype evaluation of patients with primary ciliary dyskinesia: first results from Turkey. Pediatr Pulmonol. 2020 Feb;55(2):383–93.
Zhu N, Welch CL, Wang J, Allen PM, Gonzaga-Jauregui C, Ma L, et al. Rare variants in SOX17 are associated with pulmonary arterial hypertension with congenital heart disease. Genome Med. 2018 Jul;20(1):56.
Blanchon S, Legendre M, Bottier M, Tamalet A, Montantin G, Collot N, et al. Deep phenotyping, including quantitative ciliary beating parameters, and extensive genotyping in primary ciliary dyskinesia. J Med Genet. 2020 Apr;57(4):237–44.
Turner TN, Wilfert AB, Bakken TE, Bernier RA, Pepper MR, Zhang Z, et al. Sex-based analysis of De Novo Variants in Neurodevelopmental Disorders. Am J Hum Genet. 2019 Dec;5(6):1274–85.
Stranneheim H, Lagerstedt-Robinson K, Magnusson M, Kvarnung M, Nilsson D, Lesko N, et al. Integration of whole genome sequencing into a healthcare setting: high diagnostic rates across multiple clinical entities in 3219 rare disease patients. Genome Med. 2021 Mar;17(1):40.
LaDuca H, Farwell KD, Vuong H, Lu HM, Mu W, Shahmirzadi L et al. Exome sequencing covers > 98% of mutations identified on targeted next generation sequencing panels. PLoS One. 2017 Feb 2;12(2):e0170843.
Yiallouros PK, Kouis P, Kyriacou K, Evriviadou A, Anagnostopoulou P, Matthaiou A, et al. Implementation of multigene panel NGS diagnosis in the national primary ciliary dyskinesia cohort of Cyprus: an island with a high disease prevalence. Hum Mutat. 2021 Jun;42(6):e62–e77.
Zhao X, Bian C, Liu K, Xu W, Liu Y, Tian X et al. Clinical characteristics and genetic spectrum of 26 individuals of Chinese origin with primary ciliary dyskinesia. Orphanet J Rare Dis. 2021 Jul 1;16(1):293.
Capalbo A, Valero RA, Jimenez-Almazan J, Pardo PM, Fabiani M, Jiménez D et al. Optimizing clinical exome design and parallel gene-testing for recessive genetic conditions in preconception carrier screening: Translational research genomic data from 14,125 exomes. PLoS Genet. 2019 Oct 7;15(10):e1008409.
Alhathal N, Maddirevula S, Coskun S, Alali H, Assoum M, Morris T, et al. A genomics approach to male infertility. Genet Med. 2020 Dec;22(12):1967–75.
Kim RH, Hall A, Cutz D, Knowles E, Nelligan MR, Nykamp KA. The role of molecular genetic analysis in the diagnosis of primary ciliary dyskinesia. Ann Am Thorac Soc. 2014 Mar;11(3):351–9.
Lu C, Yang D, Lei C, Wang R, Guo T, Luo H. Identification of two Novel DNAAF2 variants in two Consanguineous families with primary ciliary Dyskinesia. Pharmgenomics Pers Med. 2021 Nov;10:14:1415–23.
Guan Y, Yang H, Yao X, Xu H, Liu H, Tang X, et al. Clinical and genetic spectrum of children with primary ciliary Dyskinesia in China. Chest. 2021 May;159(5):1768–81.
Acknowledgements
We are grateful to the patients and their family for their collaboration.
Funding
Medical Science and Technology Project of Henan Province (2018020614, 2018020621); Demonstration application of remote collaborative service network for complex genetic diseases (SJPT-03-01).
Author information
Authors and Affiliations
Contributions
YT, SZ and XT were principal investigators of the study. They participated in the design of the study. YT and XT also participated in the drafting of the manuscript. LD participated in the data collection, data analysis and took the lead on drafting the manuscript. LZ, XL, SM, YS, LF participated in the data collection and data analysis. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Ethical approval was obtained from Henan Children’s Hospital Ethics Committee with ethical approval number 2022-k-049. Informed consent was obtained from the parents. All procedures performed were carried out in accordance with the ethical standards in the 1964 Declaration of Helsinki and its later amendments and the institutional ethics guidelines.
Consent for publication
Not Applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.




Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Dong, L., Zhang, L., Li, X. et al. Clinical and genetic analysis of two patients with primary ciliary dyskinesia caused by a novel variant of DNAAF2. BMC Pediatr 23, 616 (2023). https://doi.org/10.1186/s12887-023-04185-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12887-023-04185-w