Abstract
Background
A psychoneurological symptom cluster composed of cancer-related fatigue, emotional distress, sleep difficulties, and pain is very common among patients with cancer. Cognitive difficulties are also frequently associated with this cluster. Network analyses allow for an in-depth understanding of the relationships between symptoms in a cluster. This paper details the study protocol of a longitudinal assessment of the psychoneurological symptom cluster in two distinct cohorts: breast cancer and digestive cancer survivors, using network analyses.
Methods
Over two years, the symptoms involved in the psychoneurological symptom cluster, along with other common symptoms (e.g., digestive symptoms, financial difficulties) and variables (i.e., self-compassion, coping strategies) will be assessed in two cohorts: breast cancer survivors (N = 240) and digestive cancer survivors (N = 240). Online questionnaires will be completed at baseline, then 6, 12 and 24 months later. Network analyses will be used to assess the configuration of the symptom cluster at each measurement time and in each cohort. Comparison of networks between two measurement times or between the two cohorts will also be done with network comparison tests.
Discussion
This study will enable a better understanding of the relationships between common symptoms endured by patients with cancer. The results will be employed to develop more cost-effective interventions which, ultimately, will significantly improve the quality of life of patients with breast or digestive cancer.
Trial registration
ClinicalTrials.gov (NCT05867966). Registered on the 27th of April 2023. url: https://classic.clinicaltrials.gov/ct2/show/NCT05867966.
Similar content being viewed by others
Background
The presence of one psychoneurological symptom cluster (PNSC) composed of cancer-related fatigue (CRF), sleep difficulties, emotional distress, and pain is particularly well-documented among patients with cancer [1,2,3]. Cognitive impairments are also frequently associated with this symptom cluster [4, 5]. CRF is a distressing, persistent, and subjective feeling of physical, emotional and/or cognitive tiredness or exhaustion, which is related to cancer or its treatment. It is not proportional to the person’s recent activity and interferes with their routine functioning [6]. This multidimensional symptom is considered as the most severe one endured by patients with cancer, having a high impact on their quality of life (QOL) [7,8,9]. Yet, few attention has been received from the healthcare professionals [10]. Emotional distress (i.e., anxiety and depression), sleep difficulties (i.e., generally insomnia), pain, and cognitive difficulties (e.g., concentration or memory difficulties) are also very common among these patients and negatively influence their QOL [11]. In addition to their own negative consequences, these symptoms evolve together and reinforce each other, participating resulting in the high burden endured by patients with cancer [2, 12]. The mechanisms involved in the PNSC are uncertain [13, 14], but it is known that the more symptoms the patient presents, the lower their QOL [15]. Despite their severe impact and their persistence up to years after treatment completion, these symptoms remain underdiagnosed and undertreated [16]. The configuration of the PNSC (e.g., intensity of the relationships between different symptoms, core/central symptom of the cluster) seems to vary according to the cancer diagnosis [1, 2] and to evolve over time [17, 18]. Most studies in oncology focused on a single symptom, or on several symptoms considered independently from each other [3, 11, 19, 20]. However, the high prevalence of the PNSC underlines the relevance of studying multiple symptoms and their interactions.
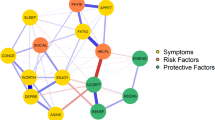
Network analysis is an innovative method that underlines a deeper understanding of the connections between symptoms. By assessing and visualizing symptom clusters as dynamic systems of interacting symptoms, these analyses allow to study symptoms in their full complexity [1, 2, 12, 18] and to compare patterns of clustering between distinct populations [2, 21] or measurement times [1, 18]. Core symptoms within a network are the ones with the strongest associations with the other symptoms, which may play a critical role in activating them [22]. Thus, targeting them could allow to design more cost-effective interventions to impact the whole cluster [1,2,3, 11, 17, 18, 20, 23]. As network analyses in symptom studies are scarce, there is no consensus regarding the core symptom of the PNSC yet, as it seems to vary according to the population studied, the phase of the cancer trajectory, and the methodology used. Depression [1, 20], CRF [2, 15, 18], distress [3, 17], or anxiety [23] have been recurrently shown to be the core symptoms in studies on patients with cancer.
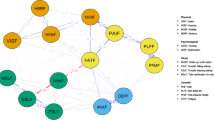
Cancer symptom management has often consisted in a “single symptom approach”, with the prescription of one specific intervention for each symptom reported by the patient, leading to complicated self-management of symptoms [11]. In the context of symptom clusters, interventions with benefits on one than more symptoms are thus highly relevant. Approaches such as mind-body techniques [11, 24,25,26,27], self-compassion learning [26,27,28,29,30], and cognitive-behavioural therapy [11] are relevant components to be included in such interventions. Different studies [10, 31,32,33,34] also suggested that the common-sense model of self-regulation of health and illness of Leventhal [31] (Fig. 1) could serve as a basis for developing interventions for cancer survivors, by targeting dysfunctional representations (top-down strategy) and enhancing adaptive coping (bottom-up strategy).
Leventhal’s common sense model of self-regulation of health, adapted from [31]
The studies discussed above however suffer from several limitations. First, literature on symptom clusters in oncology is heterogeneous, due to the different statistical procedures used, cancer diagnosis included, symptoms considered, and time since diagnosis. Most of them have been conducted during or shortly after treatment (i.e., generally up to one year), whereas symptoms can develop lately and persist for years after treatment [16]. Second, most of them were cross-sectional and lacked of a longitudinal evaluation of the symptom clusters [20]. Thus, there is a need for rigorous studies investigating the presence and evolution of the PNSC among distinct populations of cancer survivors [35]. There is also a critical need for cost-effective personalized interventions to alleviate these symptoms [18, 35]. Targeting a core symptom, determined for a specific population, to impact the whole PNSC seems to be of particular interest in oncology, and responds to the need for more personalized care.
Objectives
This study will consist of a longitudinal observational study that will assess the evolution of the PNSC (i.e., CRF, emotional distress, pain, sleep difficulties) in two cohorts of patients who had cancer (women with breast cancer and patients with digestive cancer), over two years, through network analyses. Evolution of other common symptoms (e.g., cognitive difficulties, nausea/vomiting, appetite loss), QOL, coping strategies, and self-compassion will be assessed in parallel. To note, the results from this study, especially the core symptom determined in each cohort, will be used in a future study to develop a new mind-body group intervention based on the common-sense model of self-regulation of health developed by Leventhal [31]. A randomized-controlled pilot trial will then be conducted to assess its feasibility and first benefits to improve the PNSC.
Methods
Design and procedures
We designed a longitudinal observational study to assess the evolution of the PNSC and the respective relevant variables in two distinct cohorts of cancer survivors (breast cancer survivors and digestive cancer survivors). Questionnaires (in French; see “Statistical analyses”for more details) will be completed online (on www.alchemer.eu) at four different times: at inclusion (T1), then 6 (T2), 12 (T3) and 24 (T4) months later. Written informed consent will be obtained from each participant at the beginning of the study (included at the beginning of the online questionnaires), and the inclusion criteria are checked at the beginning of the questionnaires, so that only participants who meet the criteria will be eligible to complete them. Participants will also have the possibility to contact the principal investigators by email or phone if needed (e.g., question, adverse event, or difficulty related to the study). At each measurement time, the risk of missing responses is very low, as the online questionnaires are designed in a way that does not allow for missing answers. The short duration of the questionnaires (approximately 20 min) will also increase completion and retention. All data will be anonymized: a code will be attributed to each participant and used during the entire study. Only the principal investigators involved in this study will have access to the complete datasets. Review of the trial process and difficulties encountered are discussed regularly between the researchers involved. As no standard procedure to determine the ideal sample size for network analysis is commonly used, the number of participants required in this study will be based on other studies in oncology using similar methodology [1,2,3, 17, 18, 20] and including between 172 and 342 patients per cohort, and set to 200. Accounting for a drop-out rate of 10% at one year in an observational study [36], and likely a higher drop-out rate at two years, we aim to include 240 participants in each cohort.
Eligibility
Inclusion criteria will be: ≥18-years-old, fluent in French, diagnosis of non-metastatic breast cancer or digestive cancer (i.e., anal, colorectal, gastric, oesophageal, liver, pancreatic cancers), no history of cancer and not currently in relapse, time since end of active treatments (i.e., surgery, chemotherapy, radiation therapy) < 5 years (based on the methodology and recommendations of previous studies on symptom clusters in oncology [2, 3, 35, 37]). Breast and digestive cancer were chosen because they are among the most common cancers [38]. Thus, they would be easier to recruit, and our results could be of interest for more patients, as well as their relatives and healthcare professionals. Metastatic cancers will not be included, as done in one of our previous studies [26], in order to minimize the baseline differences in the sample, as the changing nature and complexity of metastatic cancers could make it difficult to compare the results with non-metastatic cases [39].
Recruitment
Participants will be recruited over a 2-year period. Online questionnaires will be promoted and diffused through different means: websites of the involved departments and institutions, social networks, waiting rooms of the hospital, patients’ associations, and direct contact with health professionals working with these patients, in French-speaking Belgium but also in France or other French-speaking areas.
Assessments
Approximate duration of questionnaires completion is 20 min at each measurement time. First, socio-demographic and medical data will be collected (i.e., sex, age, marital situation, education level, professional activity, cancer diagnosis, cancer stage, personal history of cancer and treatments, possible recurrence during the study, comorbidities, neurological and psychiatric history, history of drug or alcohol abuse, previous participation in a mind-body intervention, psychotherapy, major life events). Symptoms involved in the PNSC, as well as other common symptoms and QOL, will be assessed as follows:
-
European Organization for Research and Treatment of Cancer - Quality of Life Core Questionnaire-30 (EORTC QLQ-C30) [40]: This questionnaire (30 items) assesses the QOL of patients with cancer through 5 functional scales (physical, role, emotional, cognitive, and social functioning), three symptom scales (fatigue, nausea and vomiting, pain), a global health status/QOL scale and several single items for additional difficulties (dyspnoea, insomnia, appetite loss, constipation, diarrhoea, financial difficulties). Each item is scored on a 4-point Likert scale (‘not at all’ to ‘very much’), excepted for the global health status/QOL that is a 7-point scale (‘very bad’ to ‘excellent’). For functional scales and QOL scale, higher scores indicate better functionality and QOL. For symptom scales and single items, higher scores indicate higher symptomatology.
-
Multidimensional Fatigue Inventory (MFI-20) [41]: This 20-item scale is designed to measure fatigue on 5 dimensions: general fatigue, physical fatigue, mental fatigue, reduced motivation and reduced activity. Suggested cut-off scores for significant general fatigue have been proposed for the general population: ≥ 11 (for men between 40 and 59 years-old), ≥ 12 (for women between 40 and 59 years-old), and ≥ 14 (for men and women older than 60 years-old) [42, 43].
-
Hospital Anxiety and Depression Scale (HADS) [44]: This 14-item scale measures anxiety and depression, and has been validated for people with somatic illnesses. Scores between 8 and 10 on a dimension suggest the presence of anxious and/or depressive disorders, while scores between, 11 and 21 indicate the presence of such disorders.
Other variables of interest (i.e., coping strategies and self-compassion) will be assessed as follows:
-
Ways of Coping Checklist [45, 46]: This 27-item scale assesses coping through 3 dimensions: problem-oriented coping (score range: 10–40), emotion-oriented coping (score range: 9–36), and seeking social support (score range: 8–32). Higher scores indicate higher use of specific coping strategies.
-
Self-compassion scale [47]: This 26-item questionnaire measures 6 dimensions related to self-compassion: self-kindness, self-judgment, common humanity, isolation, mindfulness, and over-identification, with higher scores indicating higher self-compassion. A total mean score is also generated and indicates low (i.e., score between 1 and 2.49), moderate (i.e., score between 2.5 and 3.49), or high (i.e., score between 3.5 and 5) self-compassion.
Statistical analyses
Normality of data will be checked using Shapiro-Wilk test. Sociodemographic and medical data will be described using frequencies and percentages, means and standard deviations, or medians and interquartile ranges, depending on the distribution. To estimate the networks’ structure, a standardized score (z score [1]) will be calculated for each variable or dimension. Three centrality indices will be used: strength (number and strength of the direct connections to a node/symptom, i.e., sum of absolute weights; a high strength means that the symptom is more likely to occur in conjunction with other symptoms), closeness (node’s relationship to all other nodes; closer nodes mean stronger correlation), and betweenness (importance of a node in the average pathway between other pairs of nodes; a node with a higher betweenness has greater influence in the network) [1, 2]. Symptom networks will be created for both cohorts, at each of the four measurement times. To assess the differences between two networks (i.e., between breast and digestive cancers), and over time for each cohort (i.e., between T1, T2, T3 and T4), network comparison tests will be used [1, 17]. The evolution of the other variables over time (i.e., self-compassion, self-care agency, self-care strategies already implemented to manage CRF, and the representations about CRF) will also be measured and compared through repeated measures MANOVA and post-hoc comparisons.
Data coding and storage
The questionnaires will be completed electronically (on www.alchemer.eu) by the participants. Data encoding will be done automatically by Alchemer and will be regularly checked by one of the principal investigators. Final databases will be stored on a protected server from the University of Liège, protected by a password, only accessible to the principal investigators. Data coding and storage comply with the European General Data Protection Regulation (GDPR).
Discussion
Recent studies highlighted the prevalence and negative consequences of the PNSC in oncology [1,2,3]. The core symptom of this cluster could be of major importance in the management of the cluster of symptoms as a whole [22] but it is still unclear which symptom from the cluster is the most central among patients with breast or digestive cancer. Determining this core symptom could allow to develop more cost-effective interventions, as addressing this symptom will likely decrease the other symptoms from the cluster [1,2,3, 11, 17, 18, 20, 23]. The development of multi-component interventions, especially those aiming to manage the whole PNSC, is then urgently needed in oncology [11]. We thus designed a longitudinal study assessing the PNSC and other symptoms in breast cancer and digestive cancer survivors, over 2 years. We hypothesize that: (1) one dimension of CRF or of emotional distress (i.e., depression or anxiety) will be the core symptom in all the obtained networks (i.e., for both cohorts and at each of the four measurement times), (2) the physical and mental dimensions of CRF will have different relationships with the other symptoms of the networks (e.g., physical fatigue could have stronger relationships with other physical symptoms such as loss of appetite or nausea/vomiting), (3) the configuration of the networks will differ according to the cohort considered (e.g., nausea/vomiting and diarrhoea/constipation could have stronger relationships with CRF, anxiety or depression in the digestive cancer cohort), and the measurement time (e.g., some correlations could weaken over time, as the intensity of some symptoms will decrease and became less important in the networks), and (4) self-compassion and coping strategies will not significantly evolve over time, even if a trend in this sense is possible due to a better adaptation to cancer.
The present study will suffer from several limitations. First, as no standard procedure to determine the ideal sample size for network analyses exist, we based our calculation on existing studies with similar methodology. It is then possible that the sample size will remain too small to allow robust results. Second, we decided to include patients who completed their active treatment within the last 5 years. One could argue that 5 years is a rather long period of time, that the psychological state and quality of life of patients are very likely to evolve during that time, and that the participants would be very different from each other, leading to an unexpected heterogeneity of our sample. However, the threshold of 5 years is commonly used in oncology studies, as it is considered to distinguish the “short-term” and “long-term” cancer survivors [2, 35, 48]. Our decision is thus in line with existing practices in the field. Finally, we will focus on two populations of cancer patients only (i.e., survivors of breast or digestive cancer). These results will then be relevant to these patients only. However, breast and digestive cancers are among the most frequent cancers [38], making our results applicable for many patients and health professionals.
Despite these limitations, this study will be one of the first to assess over a long period of time the evolution of the PNSC on two distinct populations. As described, the conduction of the present study will allow us to design a new multi-component intervention specifically targeting the core symptom determined in each cohort, and to assess its feasibility and preliminary benefits. Taken as a whole, this project is thus particularly original, timely relevant and innovative. It will bring new knowledge about the PNSC, its evolution, and its specificities according to the diagnosis, and its results will open a lot of scientific and clinical perspectives to improve symptom (self-)management of patients with cancer.
Data availability
The datasets used during the current study will be available from the corresponding author on reasonable request (ch.gregoire@uliege.be).
References
Lin Y, Bruner DW, Paul S, Miller AH, Saba NF, Higgins KA, et al. A network analysis of self-reported psychoneurological symptoms in patients with head and neck cancer undergoing intensity-modulated radiotherapy. Cancer. 2022;128(20):3734–43.
de Rooij BH, Oerlemans S, van Deun K, Mols F, de Ligt KM, Husson O, et al. Symptom clusters in 1330 survivors of 7 cancer types from the PROFILES registry: a network analysis. Cancer. 2021;127(24):4665–74.
Zhu Z, Sun Y, Kuang Y, Yuan X, Gu H, Zhu J et al. Contemporaneous symptom networks of multidimensional symptom experiences in cancer survivors: a network analysis. Cancer Med. 2022.
Sanford SD, Beaumont JL, Butt Z, Sweet JJ, Cella D, Wagner LI. Prospective longitudinal evaluation of a symptom cluster in breast cancer. J Pain Symptom Manag. 2014;47(4):721–30.
Pullens MJJ, De Vries J, Roukema JA. Subjective cognitive dysfunction in breast cancer patients: a systematic review. Psychooncology. 2010;19(11):1127–38.
Koh WJ, Abu-Rustum NR, Bean S, Bradley K, Campos SM, Cho KR, et al. Cervical Cancer, Version 3.2019, NCCN Clinical Practice guidelines in Oncology. J Natl Compr Canc Netw. 2019;17(1):64–84.
Ma Y, He B, Jiang M, Yang Y, Wang C, Huang C, et al. Prevalence and risk factors of cancer-related fatigue: a systematic review and meta-analysis. Int J Nurs Stud. 2020;111:103707.
Abrahams H, Gielissen M, Verhagen C, Knoop H. The relationship of fatigue in breast cancer survivors with quality of life and factors to address in psychological interventions: a systematic review. Clin Psychol Rev. 2018;63:1–11.
Wang XS, Zhao F, Fisch MJ, O’Mara AM, Cella D, Mendoza TR, et al. Prevalence and characteristics of moderate to severe fatigue: a multicenter study in cancer patients and survivors. Cancer. 2014;120(3):425–32.
Corbett T, Groarke A, Walsh JC, McGuire BE. Cancer-related fatigue in post-treatment cancer survivors: application of the common sense model of illness representations. BMC Cancer. 2016;16(1):919.
Kwekkeboom KL. Cancer Symptom Cluster Management. Semin Oncol Nurs. 2016;32(4):373–82.
Borsboom D, Cramer AOJ. Network analysis: an integrative approach to the structure of psychopathology. Annu Rev Clin Psychol. 2013;9:91–121.
Kim HJ, Barsevick AM, Fang CY, Miaskowski C. Common biological pathways underlying the psychoneurological symptom cluster in cancer patients. Cancer Nurs. 2012;35(6):E1–20.
Miaskowski C, Aouizerat BE. Is there a biological basis for the clustering of symptoms? Semin Oncol Nurs. 2007;23(2):99–105.
Mathew A, Tirkey AJ, Li H, Steffen A, Lockwood MB, Patil CL, et al. Symptom clusters in Head and Neck Cancer: a systematic review and conceptual model. Semin Oncol Nurs. 2021;37(5):151215.
Wu HS, Harden JK. Symptom burden and quality of life in survivorship: a review of the literature. Cancer Nurs. 2015;38(1):E29–54.
Shim EJ, Ha H, Suh YS, Kong SH, Lee HJ, Yang HK, et al. Network analyses of associations between cancer-related physical and psychological symptoms and quality of life in gastric cancer patients. Psychooncology. 2021;30(6):946–53.
Rha SY, Lee J. Stable Symptom clusters and evolving Symptom networks in relation to chemotherapy cycles. J Pain Symptom Manag. 2021;61(3):544–54.
Miaskowski C, Barsevick A, Berger A, Casagrande R, Grady PA, Jacobsen P et al. Advancing Symptom Science Through Symptom Cluster Research: Expert Panel Proceedings and Recommendations. J Natl Cancer Inst. 2017;109(4).
Schellekens MPJ, Wolvers MDJ, Schroevers MJ, Bootsma TI, Cramer AOJ, van der Lee ML. Exploring the interconnectedness of fatigue, depression, anxiety and potential risk and protective factors in cancer patients: a network approach. J Behav Med. 2020;43(4):553–63.
van Borkulo CD, van Bork R, Boschloo L, Kossakowski JJ, Tio P, Schoevers RA et al. Comparing network structures on three aspects: a permutation test. Psychol Methods. 2022.
Hevey D. Network analysis: a brief overview and tutorial. Health Psychol Behav Med. 2018;6(1):301–28.
Kim HJ, Malone PS. Roles of biological and psychosocial factors in experiencing a psychoneurological symptom cluster in cancer patients. Eur J Oncol Nurs. 2019;42:97–102.
Deleemans J, Mather H, Spiropoulos A, Toivonen K, Baydoun M, Carlson L. Recent progress in mind–body therapies in Cancer Care. Curr Oncol Rep. 2023;1–15.
Grégoire C, Bragard I, Jerusalem G, Etienne AM, Coucke P, Dupuis G, et al. Group interventions to reduce emotional distress and fatigue in breast cancer patients: a 9-month follow-up pragmatic trial. Br J Cancer. 2017;117(10):1442–9.
Grégoire C, Faymonville ME, Vanhaudenhuyse A, Charland-Verville V, Jerusalem G, Willems S, et al. Effects of an intervention combining self-care and self-hypnosis on fatigue and associated symptoms in post-treatment cancer patients: a randomized-controlled trial. Psycho-oncology. 2020;29(7):1165–73.
Grégoire C, Faymonville ME, Vanhaudenhuyse A, Jerusalem G, Monseur J, Bragard I. A group intervention combining self-hypnosis and self-care in oncology: implementation in daily life and perceived usefulness. Int J Clin Exp Hypn. 2023;71(3).
O’Regan P, McCarthy G, O’Reilly S, Power D, Bird BH, Murphy CG, et al. Cancer-related fatigue and self-care agency: a multicentre survey of patients receiving chemotherapy. J Clin Nurs. 2019;28(23–24):4424–33.
O’Regan P, Hegarty J. The importance of self-care for fatigue amongst patients undergoing chemotherapy for primary cancer. J Oncol Nurs. 2017;47–55.
Zessin U, Dickhäuser O, Garbade S. The Relationship between Self-Compassion and Well-Being: a Meta-analysis. Appl Psychol Health Well Being. 2015;7(3):340–64.
Leventhal H, Brissette I, Leventhal EA. The common-sense model of self-regulation of health and illness. The self-regulation of health and illness behaviour. D. Cameron&H. Leventhal. New York, NY, US: Routledge; 2003. pp. 42–65.
Cannon M, Credé M, Kimber JM, Brunkow A, Nelson R, McAndrew LM. The common-sense model and mental illness outcomes: a meta-analysis. Clin Psychol Psychother. 2022;29(4):1186–202.
Hagger MS, Koch S, Chatzisarantis NLD, Orbell S. The common sense model of self-regulation: Meta-analysis and test of a process model. Psychol Bull. 2017;143:1117–54.
McAndrew LM, Musumeci-Szabó TJ, Mora PA, Vileikyte L, Burns E, Halm EA, et al. Using the common sense model to design interventions for the prevention and management of chronic illness threats: from description to process. Br J Health Psychol. 2008;13(Pt 2):195–204.
de Ligt KM, de Rooij BH, Walraven I, Heins MJ, Verloop J, Siesling S et al. Varying severities of symptoms underline the relevance of personalized follow-up care in breast cancer survivors: latent class cluster analyses in a cross-sectional cohort. Support Care Cancer. 2022.
Jansen F, Brakenhoff RH, Baatenburg de Jong RJ, Langendijk JA, Leemans CR, Takes RP, et al. Study retention and attrition in a longitudinal cohort study including patient-reported outcomes, fieldwork and biobank samples: results of the Netherlands quality of life and Biomedical cohort study (NET-QUBIC) among 739 head and neck cancer patients and 262 informal caregivers. BMC Med Res Methodol. 2022;22(1):27.
Bickel EA, Schellekens MPJ, Smink JG, Mul VEM, Ranchor AV, Fleer J et al. Looking at individual symptoms: the dynamic network structure of depressive symptoms in cancer survivors and their preferences for psychological care. J Cancer Surviv. 2022.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49.
Cardoso F, Costa A, Norton L, Cameron D, Cufer T, Fallowfield L, et al. 1st International consensus guidelines for advanced breast cancer (ABC 1). Breast. 2012;21(3):242–52.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–76.
Smets EM, Garssen B, Bonke B, De Haes JC. The multidimensional fatigue inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res. 1995;39(3):315–25.
Singer S, Kuhnt S, Zwerenz R, Eckert K, Hofmeister D, Dietz A, et al. Age- and sex-standardised prevalence rates of fatigue in a large hospital-based sample of cancer patients. Br J Cancer. 2011;105(3):445–51.
Schwarz R, Krauss O, Hinz A. Fatigue in the general population. Onkologie. 2003;26(2):140–4.
Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–70.
Cousson F, Bruchon-Schweitzer M, Quintard B, Nuissier J, Rascle N. Analyse Multidimensionnelle D’une échelle de coping: validation française de la W.C.C. (ways of coping checklist). Psychologie française. 1996;41(2):155–64.
Folkman S, Lazarus RS. An analysis of coping in a middle-aged community sample. J Health Soc Behav. 1980;21(3):219–39.
Kotsou I, Leys C. Self-Compassion Scale (SCS): Psychometric properties of the French translation and its relations with Psychological Well-Being, affect and Depression. PLoS ONE. 2016;11(4):e0152880.
Victoria Puzo L. What Are the Needs of Cancer Survivors 5 Years or More After Treatment? 2019 Oct 5 [cited 2023 Sep 11];13. Available from: https://www.oncnursingnews.com/view/what-are-the-needs-of-cancer-survivors-5-years-or-more-after-treatment.
Acknowledgements
We are grateful to all the patients who will participate in the study. We also thank all the health professionals working at the CHU of Liège or elsewhere who will help us to recruit the participants needed for our study.
Funding
This study is supported by the University of Liège and University Hospital of Liège, the Belgian National Funds for Scientific Research (FRS-FNRS)– Télévie, the Belgium Foundation Against Cancer (Grants Number: 2017064, C/2020/1357 and Social Grant 2023), and the Sector Council for research and valorization in Health Sciences (University of Liège). These funds finance the different researchers involved in this study. The FRS-FNRS Télévie peer-reviewed and approved this study protocol. The other funding bodies have no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript. CG is a postdoctoral researcher at FRS-FNRS.
Author information
Authors and Affiliations
Contributions
All authors participated in the conception and design of the study. CG drafted the manuscript, and all the other authors revised it critically for important intellectual content. All authors read and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in this study will be in accordance with the ethical standards of the institutional and national research committee, with the GDPR, and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study is approved by the Institutional Ethics Board ‘Hospital-Faculty Ethics Committee of Liège’ (N°B7072023000016), with each participant providing written informed consent. All modifications in the protocol will be transmitted to the Ethics Board.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Grégoire, C., Baussard, L., Ernst, M. et al. Evaluation of a psychoneurological symptom cluster in patients with breast or digestive cancer: a longitudinal observational study. BMC Cancer 24, 51 (2024). https://doi.org/10.1186/s12885-023-11799-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-023-11799-x