Abstract
Objectives
The early detection and identification of stroke are essential to the prognosis of patients with suspected stroke symptoms out-of-hospital. We aimed to develop a risk prediction model based on the FAST score to identify the different types of strokes early for emergency medical services (EMS).
Methods
This retrospective observational study enrolled 394 stroke patients at a single center from January 2020 to December 2021. Demographic data, clinical characteristics, and stroke risk factors with patients were collected from the EMS record database. Univariate and multivariate logistic regression analysis was used to identify the independent risk predictors. The nomogram was developed based on the independent predictors, in which the discriminative value and calibration of the nomogram were verified by the receiver operator characteristic (ROC) curve and calibration plots.
Results
A total of 31.90% (88/276) of patients were diagnosed with hemorrhagic stroke in the training set, while 36.40% (43/118) in the validation set. The nomogram was developed based on the multivariate analysis, including age, systolic blood pressure, hypertension, vomiting, arm weakness, and slurred speech. The area under the curve (AUC) of the ROC with nomogram was 0.796 (95% CI: 0.740–0.852, P < 0.001) and 0.808 (95% CI:0.728–0.887, P < 0.001) in the training set and validation set, respectively. In addition, the AUC with the nomogram was superior to the FAST score in both two sets. The calibration curve showed a good agreement with the nomogram and the decision curves analysis also demonstrated that the nomogram had a wider range of threshold probabilities than the FAST score in the prediction risk of hemorrhagic stroke.
Conclusions
This novel noninvasive clinical nomogram shows a good performance in differentiating hemorrhagic and ischemic stroke for EMS staff prehospital. Moreover, all of the variables of nomogram are acquired in clinical practice easily and inexpensively out-of-hospital.
Similar content being viewed by others
Background
Stroke is a severe manifestation of cardiovascular disease, which leads to the second cause of death in the world [1]. Nearly 20 million people experience stroke annually, and the incidence is increasing because of the aging of the population [1, 2]. The ischemic stroke makes up about 88% of all strokes, and the remainder is hemorrhagic stroke [3, 4]. At present, endovascular therapy and intravenous tissue-type plasminogen activators are the most effective therapies for acute ischemic stroke in the therapeutic time window [5]. In addition, receiving operation and intervention treatment in time for hemorrhagic stroke is crucial [6]. Thus, it is critical to improve the prognosis of stroke patients for establish the stroke center network and stroke “Green Channel”, which shortens the treatment time and integrates the medical resources.
Almost 50% of stroke occurred out-of-hospital. Emergency medical services (EMS) are the first point to contract patients who appear suspected stroke symptoms [7]. Early identification of ischemic or hemorrhagic stroke from stroke patients could provide earlier diagnosis, referral to the appropriate emergency department, and given a better treatment decision [8]. Therefore, distinguishing hemorrhagic stroke from ischemia stroke has an important implication for EMS.
Some prehospital stroke scales were used for EMS to identify stroke in recent studies, such as the Face Arm Speech Test (FAST), Los Angeles Prehospital Stroke Screen (LAPSS), and Recognition of Stroke in the Emergency Room (ROSIER), parts of which had moderate-to-good sensitivity and the lower levels of specificity [9,10,11,12]. In addition, some studies have proved that serum markers had a certain advantage in distinguishing hemorrhagic stroke and ischemic stroke, including glial fibrillary acidic protein (GFAP), N-terminal proB-type natriuretic peptide (NT-proBNP), and retinol-binding protein 4 (RBP-4) [13, 14]. However, the difficulty to detect prehospital limited their application.
Therefore, this study aims to establish a simple and reliable clinical tool to identify ischemic and hemorrhagic stroke prehospital based on the easy-to-obtain prehospital clinical data and FAST scale. Moreover, the use of the tool would be convenient for EMS staff to identify stroke types early and improve emergency efficiency in future clinical applications.
Methods
Study design and participants
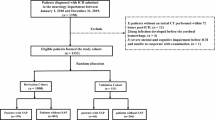
This retrospective observational study enrolled patients with suspected stroke symptoms onset from January 1, 2020 to December 1, 2021 in the second affiliated hospital of Wannan Medical College. All patients were admitted to the emergency department by the EMS. The study was approved by the Ethics Committee of the Second Affiliated Hospital of Wannan Medical College (Number: wyefyls202205) and followed the principles of the Declaration of Helsinki. The informed consent of patients was waived due to the retrospective observational design.
Participants of the following criteria were included in this study: 1) age > 18 years; 2) admission to the emergency department with a suspected diagnosis of stroke; 3) computed tomography (CT) or magnetic resonance imaging (MRI) scan during hospitalization. The exclusion criteria were:1) patients using EMS as a secondary transport; 2) data missed with EMS records; 3) patients died before CT or MRI was performed; 4) stroke caused by trauma.
A total of 670 patients were included in this study. Finally, 394 patients were enrolled in the final analysis according to the inclusion and exclusion criteria. All patients were divided into a training set (n = 276) and a validation set (n = 118) in a ratio of 7:3 (Fig. 1).
Data collection
Baseline demographic data, prehospital clinical characteristics, and stroke risk factors were collected from the EMS record database in the Wuhu Emergency Medical Center, including age, gender, blood pressure, clinical features, diabetes, hypertension, duration of symptom, etc. The prehospital vital signs (blood pressure and heart rate) were measured using an electrocardiogram blood pressure monitor (Mindray, China) by EMS personnel out-of-hospital.
Definition
Hemorrhagic stroke was defined as a new symptomatic neurologic deterioration accompanied by neuroimaging evidence of extravasation of blood into the brain parenchyma spontaneous and non-traumatic, including intraparenchymal hemorrhage and subarachnoid hemorrhage [15, 16]. Ischemic stroke was defined as a sudden neurologic dysfunction with imaging evidence of acute infarction by MRI or CT scan [17]. Transient cerebral ischemic attack (TIA) was defined as new neurologic symptoms lasting less than 24 h with no new infarction or hemorrhage on neuroimaging [18]. The Face Arm Speech Test (FAST) contained items that unilateral facial droop (F) or arm weakness (A) and Slurred speech (S) [19]. Duration of symptom was defined as the duration since suspected stroke symptoms onset until the EMS arrived.
Statistical analysis
All statistical analyses were performed using R software (version 3.6.2, http://www.r-project.org) and SPSS software (version 22.0, IBM, New York, USA). Kolmogorov–Smirnov(K-S) test was used to discriminate normal distribution for all continuous variables. Continuous variables were expressed as mean ± standard deviation (SD) or interquartile range (IQR), which depended on the normal distribution. Categorical variables were expressed as numbers (percentages). Differences between the two groups for continuous variables were analyzed by the student’s test or Mann–Whitney U test, and the chi-squared test was used for categorical variables. Multivariate logistic regression models with the entering method were accomplished to determine the independent risk predictors of hemorrhagic stroke in the nomogram. The variables included factors with significance at P < 0.1 in the univariate analysis. Moreover, the final total score of the nomogram was constructed by a preliminary score of each predictor with a point ranging from 0–100, which was converted to the probability of hemorrhagic stroke (0–100%). Calibration was implemented to assess the fitting degree between the actual and nomogram-predicted hemorrhagic stroke by using a calibration plot with bootstraps of 1000 resamples.
The area under the curve (AUC) of receiver operating characteristic (ROC) was used to compare the discrimination ability of nomogram and traditional FAST score. Decision curve analysis (DCA) was further performed to estimate the clinical value of the nomogram and traditional FAST score with the net benefit for multiple threshold probabilities. All accuracy estimates and regression coefficients reported 95% confidence intervals (CI) and a two-tailed P < 0.05 was considered statistically significant.
Results
Demographic and prehospital clinical characteristics of the study
A total of 394 patients who met the eligibility criteria were retrospectively enrolled during the period between Jan 2020 and Dec 2021. The incidence of hemorrhagic stroke in the training set was 31.90% (88/276), while 36.40% (43/118) in the validation set. There were no statistical differences between the two sets in all clinical characteristics (all P > 0.05) (Table 1).
In the training set, the frequency of hypertension (P = 0.003) was higher in hemorrhagic stroke patients, compared to ischemic stroke patients. The incidence of headache (P = 0.029) and vomiting (P = 0.007) was significantly increased in hemorrhagic stroke. Meanwhile, a higher incidence of the FAST Scale (arm weakness, and slurred speech) was observed in hemorrhagic stroke patients (P = 0.004, and P = 0.002). In addition, patients with hemorrhagic stroke tended to have the lower age (P < 0.001) and higher systolic blood pressure (P = 0.006) (Table 2).
Multivariate logistic regression analyses of independent risk factors for hemorrhagic stroke
Multivariate logistic regression analysis was used to calculate the adjusted odds ratio (aOR) value of each independent risk factor in the training set (Table 3). After multivariable adjustment, systolic blood pressure (aOR:1.014, 95% CI: 1.002–1.025, P = 0.023), hypertension (aOR:2.440, 95% CI: 1.291–4.613, P = 0.006), age (aOR:0.942, 95% CI: 0.921–0.964, P < 0.001), and vomiting (aOR:2.741, 95% CI: 1.465–5.129, P = 0.002) remained significant after adjusting for confounders. Besides, arm weakness (aOR:2.559, 95% CI: 1.397–4.687, P = 0.002) and slurred speech (aOR:2.072, 95% CI: 1.142–3.760, P = 0.017) were independently associated with hemorrhagic stroke.
Establishment of nomogram in predicting hemorrhagic stroke
A clinical nomogram to predict hemorrhagic stroke was developed based on multivariate logistic regression analysis using the 6 independent risk factors. Each of the major significant predictors was assigned with points ranging from 0 to 100, and the preliminary scores were summarized as the total predictive score. Based on the total score, a visualized percentage to predict the risk of hemorrhagic stroke was shown in the nomogram (Fig. 2). The calibration plots with 1000 Bootstrap resamples were described, which demonstrated a good-predictive performance between the predicted probability of hemorrhagic stroke and the actual observations, with data points on the plots close to the ideal curve (Fig. 3).
Calibration curves of the nomogram in the training set and the validation set. Notes: A the nomogram in the training set (n = 276); B the nomogram in the validation set (n = 118). The y-axis represents the observed rate of hemorrhagic stroke, and the x-axis represents the nomogram-predicted probability of hemorrhagic stroke. The dotted lines represented by the nomogram are closer to the diagonal grey lines representing a better prediction
Validation of nomogram with receiver operating characteristic (ROC) curve
The AUC of ROC was analyzed to investigate the discrimination of the nomogram, which was 0.796 (95% CI:0.740–0.852, P < 0.001) in the training set and 0.808 (95% CI:0.728–0.887, P < 0.001) in the validation set.
Furthermore, we compared the discrimination between the nomogram and the FAST score, and the results indicated the AUC of the nomogram was superior to that of the FAST score in both the training set (AUC = 0.796 vs 0.660, P < 0.001) and validation set (AUC = 0.808 vs 0.664, P = 0.004), indicating the novel nomogram had higher predictive efficiency (Fig. 4).
Clinical use compared nomogram with the FAST score
Decision curve analysis (DCA) curves were applied to compare the clinical validity of the nomogram and the FAST score, suggesting that the nomogram could augment net benefits and demonstrate a wider range of threshold probabilities than the FAST score in the prediction of hemorrhagic stroke (Fig. 5).
Discussion
Stroke is a major cause of death and long-term cognitive impairment in China [20]. Timely treatment is critical to the prognosis of stroke patients, which reduces mortality and improves neurological prognosis [21]. Efficient pre-hospital assessment is essential for EMS to differentiate between hemorrhagic and ischemic stroke [22]. Traditionally, ischemic and hemorrhagic stroke had the common risk factors. However, the risk factors for identifying the different subtypes of stroke are unclear.
This study established a practical and convenient tool based on the FAST score and combined with age, systolic blood pressure, hypertension, and vomiting to predict the risk of hemorrhagic stroke in patients with suspected stroke symptoms for EMS staff. All independent predictors were acquired in clinical practice easily and inexpensively out-of-hospital. This nomogram has proven clinical utility and is useful for risk decision-making in patients with hemorrhagic stroke during pre-hospital first aid.
We found that age was an important independent factor to distinguish hemorrhagic stroke from ischemic stroke. The incidence of stroke among young adults has increased in the past two decades [23]. The patients with hemorrhagic stroke were younger than ischemic stroke [24, 25]. One study of 1,880 non-fatal stroke patients in Japan found that the mean age was 74.1 years for ischemic stroke, and 68.2 years for hemorrhagic stroke [26]. Further, a recent study also confirmed that the median age of patients was 74 (66–82) years for ischemic stroke, 70 (59–79) years for intracerebral hemorrhage, and 64 (53–75) years for subarachnoid hemorrhage among the 183,080 stroke patients [27]. Thus, younger patients who suspected stroke may have an increased risk of hemorrhagic stroke prehospital, which was associated with the poorer blood pressure control and an increased proportion of subarachnoid hemorrhage [28].
At present, hypertension has been recognized as the most important risk factor affecting the occurrence of stroke [29, 30]. This may be related to cerebral vascular remodeling caused by the decrease in the diameter of the cerebrovascular lumen and the increase in the thickness of the vascular wall when hypertension occurs [31]. The elevated blood pressure that occurred in the hyperacute phase of stroke was often associated with sympathetic overactivity [32, 33]. Rawshani et al.’s study found that systolic blood pressure was a risk factor that affected cerebrovascular accidents [34]. Importantly, Katsanos confirmed that the lower the systolic blood pressure, the lower risk of hemorrhagic stroke happened, which was consistent with our results [35]. Therefore, we should pay more attention to uncontrolled systolic blood pressure in hypertensive patients, which induced the increased risk of hemorrhagic stroke [36]. Furthermore, we should focus on individual blood pressure treatment goals to reduce the risk of hemorrhagic stroke in hypertensive patients.
The FAST score is a traditional tool for identifying strokes with large vessel occlusion [37]. For patients suspected of acute stroke, questions including facial drooping, arm weakness, and slurred speech should be evaluated according to the FAST score [38, 39]. It was worth noting that stroke was the leading cause of adult-acquired disability [40]. In our study, we found that a higher incidence of slurred speech and arm weakness was observed in hemorrhagic stroke patients. Slurred speech was a manifestation of progressive central nervous system damage [19], always manifested as dysarthria, and was caused by weak, slow, or uncoordinated muscle control [41,42,43]. The appearance of arm weakness might be related to the regulation of hand function by the corticoreticulospinal tract [44]. Therefore, slurred speech and arm weakness played the important roles in the diagnosis of stroke, especially in hemorrhagic stroke.
Intracranial pressure could be increased after ischemic or hemorrhagic stroke [45, 46]. The typical clinical manifestations of elevated intracranial pressure were headache, vomiting, and even loss of consciousness [47]. Especially in hemorrhagic stroke, blood could extravasate into surrounding brain tissue due to blood vessel ruptures [48]. Our study also demonstrated that vomiting was an important clinical manifestation to distinguish hemorrhagic stroke. More importantly, in contrast to other symptoms, vomiting was a typical symptom that can be assessed even in patients with unconsciousness [49]. In addition, vomiting was the most common manifestation in children with hemorrhagic stroke [50], and it was rarely presented in children with ischemic stroke [51].
The AUC with nomogram was 0.790 in the training set and 0.808 in the validation set, which has moderate prediction efficiency. In addition, the AUC of the nomogram was superior to that of the FAST score in both sets. DCA curves showed that the nomogram demonstrated a wider range of threshold probabilities than the FAST score in the prediction of hemorrhagic stroke. Given the discriminative ability of the model, it was useful to identify the type of stroke early and optimize the nursing procedure.
Our study has several limitations. First, it was a retrospective single-center study, in which potential selection bias and recall bias were inevitable. Second, this study was not external validation because of the smaller sample size, and multicenter studies should be conducted future in verifying the clinical usefulness of the model. Furthermore, other stroke-related risk factors, such as alcohol consumption, smoking, and exercise habits were not included in our research, which were important factors affecting stroke and may have influenced our results.
Conclusions
In summary, we identified several associated risk factors that could differentiate hemorrhagic and ischemic stroke prehospital. In addition, we developed a clinical nomogram based on the FAST score for differentiating hemorrhagic and ischemic stroke.
Availability of data and materials
The data and R codes are available from the corresponding author on reasonable request.
References
Katan M, Luft A. Global burden of stroke. Semin Neurol. 2018;38(2):208–11. https://doi.org/10.1055/s-0038-1649503.
Jones SP, Bray JE, Gibson JM, et al. Characteristics of patients who had a stroke not initially identified during emergency prehospital assessment: a systematic review. Emerg Med J. 2021;38(5):387–93. https://doi.org/10.1136/emermed-2020-209607.
Wu S, Wu B, Liu M, et al. Stroke in China: advances and challenges in epidemiology, prevention, and management. The Lancet Neurology. 2019;18(4):394–405. https://doi.org/10.1016/s1474-4422(18)30500-3.
Barthels D, Das H. Current advances in ischemic stroke research and therapies. Biochim Biophys Acta Mol Basis Dis. 2020;1866(4). https://doi.org/10.1016/j.bbadis.2018.09.012
Uchida K, Yoshimura S, Hiyama N, et al. Clinical prediction rules to classify types of stroke at prehospital stage. Stroke. 2018;49(8):1820–7. https://doi.org/10.1161/STROKEAHA.118.021794.
Kase CS, Hanley DF. Intracerebral Hemorrhage: advances in emergency care. Neurol Clin. 2021;39(2):405–18. https://doi.org/10.1016/j.ncl.2021.02.002.
Boulanger JM, Lindsay MP, Gubitz G, et al. Canadian Stroke Best Practice Recommendations for Acute Stroke Management: Prehospital, Emergency Department, and Acute Inpatient Stroke Care, 6th Edition, Update 2018. Int J Stroke. 2018;13(9):949–84. https://doi.org/10.1177/1747493018786616.
Millin MG, Gullett T, Daya MR. EMS management of acute stroke–out-of-hospital treatment and stroke system development (resource document to NAEMSP position statement). Prehosp Emerg Care. 2007;11(3):318–25. https://doi.org/10.1080/10903120701347885.
Zhelev Z, Walker G, Henschke N, et al. Prehospital stroke scales as screening tools for early identification of stroke and transient ischemic attack. Cochrane Database Syst Rev. 2019;4:CD011427. http://doi.org/10.1002/14651858.CD011427.pub2.
Han F, Zuo C, Zheng G. A systematic review and meta-analysis to evaluate the diagnostic accuracy of recognition of stroke in the emergency department (ROSIER) scale. BMC Neurol. 2020;20(1):304. https://doi.org/10.1186/s12883-020-01841-x.
Bergs J, Sabbe M, Moons P. Prehospital stroke scales in a Belgian prehospital setting: a pilot study. Eur J Emerg Med. 2010;17(1):2–6. https://doi.org/10.1097/MEJ.0b013e32831040ec.
Harbison J, Hossain O, Jenkinson D, et al. Diagnostic accuracy of stroke referrals from primary care, emergency room physicians, and ambulance staff using the face arm speech test. Stroke. 2003;34(1):71–6. https://doi.org/10.1161/01.str.0000044170.46643.5e.
Bustamante A, Penalba A, Orset C, et al. Blood Biomarkers to Differentiate Ischemic and Hemorrhagic Strokes. Neurology. 2021;96(15):e1928–39. https://doi.org/10.1212/WNL.0000000000011742.
Luger S, Jaeger HS, Dixon J, et al. Diagnostic accuracy of glial fibrillary acidic protein and ubiquitin Carboxy-terminal Hydrolase-L1 serum concentrations for differentiating acute intracerebral hemorrhage from ischemic stroke. Neurocrit Care. 2020;33(1):39–48. https://doi.org/10.1007/s12028-020-00931-5.
Morotti A, Goldstein JN. Diagnosis and management of acute intracerebral hemorrhage. Emerg Med Clin North Am. 2016;34(4):883–99. https://doi.org/10.1016/j.emc.2016.06.010.
Dastur CK, Yu W. Current management of spontaneous intracerebral haemorrhage. Stroke Vasc Neurol. 2017;2(1):21–9. https://doi.org/10.1136/svn-2016-000047.
Mendelson SJ, Prabhakaran S. Diagnosis and management of transient ischemic attack and acute ischemic stroke: a review. JAMA. 2021;325(11):1088–98. https://doi.org/10.1001/jama.2020.26867.
Amarenco P, Lavallee PC, Monteiro Tavares L, et al. Five-year risk of stroke after TIA or minor ischemic stroke. N Engl J Med. 2018;378(23):2182–90. https://doi.org/10.1056/NEJMoa1802712.
Purrucker JC, Hametner C, Engelbrecht A, et al. Comparison of stroke recognition and stroke severity scores for stroke detection in a single cohort. J Neurol Neurosurg Psychiatry. 2015;86(9):1021–8. https://doi.org/10.1136/jnnp-2014-309260.
Chao BH, Yan F, Hua Y, et al. Stroke prevention and control system in China: CSPPC-stroke program. Int J Stroke. 2021;16(3):265–72. https://doi.org/10.1177/1747493020913557.
Karlinski M, Kozera-Strzelinska D, Sienkiewicz-Jarosz H, et al. Reliability of prehospital diagnosis of acute cerebrovascular accident. Neurol Neurochir Pol. 2022;56(1):89–95. https://doi.org/10.5603/PJNNS.a2022.0011.
Ebinger M, Audebert HJ. Shifting acute stroke management to the prehospital setting. Curr Opin Neurol. 2022;35(1):4–9. https://doi.org/10.1097/WCO.0000000000001012.
Hussain A, Lee M, Rana J, et al. Epidemiology and risk factors for stroke in young individuals: implications for prevention. Curr Opin Cardiol. 2021;36(5):565–71. https://doi.org/10.1097/HCO.0000000000000894.
Henriksson KM, Farahmand B, Asberg S, et al. Comparison of cardiovascular risk factors and survival in patients with ischemic or hemorrhagic stroke. Int J Stroke. 2012;7(4):276–81. https://doi.org/10.1111/j.1747-4949.2011.00706.x.
Tsai CF, Jeng JS, Anderson N, et al. Comparisons of Risk Factors for Intracerebral Hemorrhage versus Ischemic Stroke in Chinese Patients. Neuroepidemiology. 2017;48(1–2):72–8. https://doi.org/10.1159/000475667.
Takashima N, Arima H, Kita Y, et al. Long-term survival after stroke in 1.4 Million Japanese population: Shiga stroke and heart attack registry. J Stroke. 2020;22(3):336–44. https://doi.org/10.5853/jos.2020.00325.
Toyoda K, Yoshimura S, Nakai M, et al. Twenty-year change in severity and outcome of ischemic and hemorrhagic strokes. JAMA Neurol. 2022;79(1):61–9. https://doi.org/10.1001/jamaneurol.2021.4346.
Minhas JS, Moullaali TJ, Rinkel GJE, et al. Blood pressure management after intracerebral and subarachnoid hemorrhage: the knowns and known unknowns. Stroke. 2022;53(4):1065–73. https://doi.org/10.1161/STROKEAHA.121.036139.
Feigin VL, Krishnamurthi RV, Parmar P, et al. Update on the global burden of ischemic and hemorrhagic stroke in 1990–2013: the GBD 2013 study. Neuroepidemiology. 2015;45(3):161–76. https://doi.org/10.1159/000441085.
Castilla-Guerra L, Fernandez-Moreno MC. Chronic management of hypertension after stroke: the role of ambulatory blood pressure monitoring. J Stroke. 2016;18(1):31–7. https://doi.org/10.5853/jos.2015.01102.
Pires PW, Rogers CT, McClain JL, et al. Doxycycline, a matrix metalloprotease inhibitor, reduces vascular remodeling and damage after cerebral ischemia in stroke-prone spontaneously hypertensive rats. Am J Physiol Heart Circ Physiol. 2011;301(1):H87-97. https://doi.org/10.1152/ajpheart.01206.2010.
Ko SB, Yoon BW. Blood pressure management for acute ischemic and hemorrhagic stroke: the evidence. Semin Respir Crit Care Med. 2017;38(6):718–25. https://doi.org/10.1055/s-0037-1608777.
Montano A, Hanley DF, Hemphill JC 3rd. Hemorrhagic stroke. Handb Clin Neurol. 2021;176:229–48. https://doi.org/10.1016/B978-0-444-64034-5.00019-5.
Rawshani A, Rawshani A, Sattar N, et al. Relative prognostic importance and optimal levels of risk factors for mortality and cardiovascular outcomes in type 1 diabetes mellitus. Circulation. 2019;139(16):1900–12. https://doi.org/10.1161/CIRCULATIONAHA.118.037454.
Katsanos AH, Filippatou A, Manios E, et al. Blood pressure reduction and secondary stroke prevention: a systematic review and Metaregression analysis of randomized clinical trials. Hypertension. 2017;69(1):171–9. https://doi.org/10.1161/HYPERTENSIONAHA.116.08485.
Kernan WN, Ovbiagele B, Black HR, et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45(7):2160–236. https://doi.org/10.1161/STR.0000000000000024.
Frank B, Fabian F, Brune B, et al. Validation of a shortened FAST-ED algorithm for smartphone app guided stroke triage. Ther Adv Neurol Disord. 2021;14:17562864211057640. https://doi.org/10.1177/17562864211057639.
Puolakka T, Virtanen P, Kinnunen J, et al. Prehospital identification of large vessel occlusion using the FAST-ED score. Acta Neurol Scand. 2021;144(4):400–7. https://doi.org/10.1111/ane.13474.
Lima FO, Silva GS, Furie KL, et al. Field assessment stroke triage for emergency destination: a simple and accurate prehospital scale to detect large vessel occlusion strokes. Stroke. 2016;47(8):1997–2002. https://doi.org/10.1161/STROKEAHA.116.013301.
Vafadar AK, Cote JN, Archambault PS. Effectiveness of functional electrical stimulation in improving clinical outcomes in the upper arm following stroke: a systematic review and meta-analysis. Biomed Res Int. 2015;2015:729768. https://doi.org/10.1155/2015/729768.
Gordon C, Ellis-Hill C, Ashburn A. The use of conversational analysis: nurse-patient interaction in communication disability after stroke. J Adv Nurs. 2009;65(3):544–53. https://doi.org/10.1111/j.1365-2648.2008.04917.x.
Kent RD, Vorperian HK, Kent JF, et al. Voice dysfunction in dysarthria: application of the multi-dimensional voice program™. J Commun Disord. 2003;36(4):281–306. https://doi.org/10.1016/s0021-9924(03)00016-9.
Mitchell C, Bowen A, Tyson S, et al. Interventions for dysarthria due to stroke and other adult-acquired, non-progressive brain injury. Cochrane Database Syst Rev. 2017;1:CD002088. https://doi.org/10.1002/14651858.CD002088.pub3.
Taga M, Charalambous CC, Raju S, et al. Corticoreticulospinal tract neurophysiology in an arm and hand muscle in healthy and stroke subjects. J Physiol. 2021;599(16):3955–71. https://doi.org/10.1113/JP281681.
Freeman WD. Management of Intracranial Pressure. Continuum (Minneap Minn). 2015;21(5 Neurocritical Care):1299–323. https://doi.org/10.1212/CON.0000000000000235.
Holbrook J, Saindane AM. Imaging of intracranial pressure disorders. Neurosurgery. 2017;80(3):341–54. https://doi.org/10.1227/NEU.0000000000001362.
Leinonen V, Vanninen R, Rauramaa T. Raised intracranial pressure and brain edema. Handb Clin Neurol. 2017;145:25–37. https://doi.org/10.1016/B978-0-12-802395-2.00004-3.
Syahrul S, Maliga HA, Ilmawan M, et al. Hemorrhagic and ischemic stroke in patients with coronavirus disease 2019: incidence, risk factors, and pathogenesis - a systematic review and meta-analysis. F1000Res. 2021;10:34. https://doi.org/10.12688/f1000research.42308.1.
Shigematsu K, Shimamura O, Nakano H, et al. Vomiting should be a prompt predictor of stroke outcome. Emerg Med J. 2013;30(9):728–31. https://doi.org/10.1136/emermed-2012-201586.
Gerstl L, Badura K, Heinen F, et al. Childhood haemorrhagic stroke: a 7-year single-centre experience. Arch Dis Child. 2019;104(12):1198–202. https://doi.org/10.1136/archdischild-2018-316749.
Yock-Corrales A, Mackay MT, Mosley I, et al. Acute childhood arterial ischemic and hemorrhagic stroke in the emergency department. Ann Emerg Med. 2011;58(2):156–63. https://doi.org/10.1016/j.annemergmed.2010.10.013.
Acknowledgements
Not applicable.
Funding
This work was supported by the Key Research Fund Project of Wannan Medical College (grant number: WK2021ZF24, WK2021ZF25), the Provincial Quality Project of the Education Department of Anhui Province (number:2021jyxm1618), and “Summit Plan” of the Second Affiliated Hospital of Wannan Medical College (grant number: DFJH202207).
Author information
Authors and Affiliations
Contributions
Hailong Zhang and Sheng Ye designed the research. Sheng Ye and Huiqing Pan drafted the manuscript. Weijia Li and Jinqiang Wang contributed to data collection. Sheng Ye, Huiqing Pan analyzed the data and discussed the results. Hailong Zhang revised the final manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The research was approved by the Medical Ethics Committee of the Second Affiliated Hospital of Wannan Medical College (Number: wyefyls202205). The informed consent of patients was waived by the Medical Ethics Committee of the Second Affiliated Hospital of Wannan Medical College due to the retrospective observational design.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Ye, S., Pan, H., Li, W. et al. Development and validation of a clinical nomogram for differentiating hemorrhagic and ischemic stroke prehospital. BMC Neurol 23, 95 (2023). https://doi.org/10.1186/s12883-023-03138-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12883-023-03138-1