Abstract
Background
Only five patients have previously been reported to harbor mutations in the MT-TT gene encoding mitochondrial tRNA threonine. The m.15923A > G mutation has been found in three severely affected children. One of these patients died within days after birth and two had a phenotype of myoclonic epilepsy with ragged red fibers (MERRF) in early childhood. We have now found the mutation in an adult patient with mild myopathy.
Case presentation
The patient is a 64-year-old Finnish man, who developed bilateral ptosis, diplopia and exercise intolerance in his fifties. Family history was unremarkable. Muscle histology showed cytochrome c-oxidase (COX) negative and ragged red fibres. The m.15923A > G mutation heteroplasmy was 33% in the skeletal muscle and 2% in buccal epithelial cells. The mutation was undetectable in the blood. Single-fibre analysis was performed and COX-negative fibres had a substantially higher heteroplasmy of 92%, than the normal fibres in which it was 43%.
Conclusions
We report the fourth patient with m. 15923A > G and with a remarkably milder phenotype than the previous three patients. Our findings and recent biochemical studies suggest that the mutation m.15923A > G is a definite disease-causing mutation. Our results also suggest that heteroplasmy of the m.15923A > G mutation correlates with the severity of the phenotype. This study expands the catalog of the phenotypes caused by mutations in mtDNA.
Similar content being viewed by others
Background
Mutations in mitochondrial DNA (mtDNA) were found to cause diseases for the first time in 1988 [1, 2], and since then, the number of new mutations and associated phenotypes has continuously increased. MtDNA contains 22 tRNAs, 2 rRNAs and 13 protein-coding genes and disease-causing mutations have been reported in all of them. Mitochondrial tRNA mutations are a well-established cause of mitochondrial disorder, and most of the patients carry the m.3243A > G mutation in MT-TL1. The prevalence of mt-tRNA mutations has been estimated to be 4.3 per 100,000 in U.K. [3]. Among these mutations, the prevalence of m.3243A > G solely has been reported to be 3.5 per 100,000. Interestingly, the prevalence is several fold, 16.3 per 100,000, in Finland [4].
While many tRNA genes seem to be mutational hotspots in patients with mitochondrial disorder, the mitochondrially encoded tRNA threonine (MT-TT) has been reported to be mutated in five patients only [5,6,7,8,9,10]. The m.15923A > G has previously been found in three patients; an infant dying from multisystem failure at the age of 2.5 days [5, 6], and two children with myoclonic epilepsy and ragged-red fibers (MERRF) syndrome [8, 10]. We report here the fourth patient with m.15923A > G and the first with an adult-onset phenotype.
Case presentation
Patient
The patient was a 64-year-old man, who had bilateral ptosis, diplopia and exercise intolerance. His early development had been normal and currently he had no regular medication. Ocular symptoms had started to develop at the age of 54 years, the right eye had been operated due to squint at the age of 58 years and ptosis surgery had been performed on the right at the age of 63 years. He had right clubfoot, which had been regarded as a complication of vaccination at the age of two years. There was muscle atrophy in the right leg and the leg movements were restricted. His parents, his seven siblings and his son were healthy. Patient’s maternal uncle had ocular symptoms and, interestingly, uncle’s granddaughter had ptosis and a 7.5 kb deletion in mtDNA.
On neurological examination, the patient limped slightly because of the right clubfoot. Ptosis was moderate on the right and mild on the left. Vertical gaze paresis and a slight restriction in horizontal movements was noted in both eyes. Otherwise, muscle examination was normal. Ankle reflexes were absent, while other tendon reflexes were normal.
Routine laboratory values including creatine kinase were normal. Blood lactate was 1.16 mmol/l (reference values 0.33–1.33 mmol/l) and pyruvate was 84 μmol/l (reference values 30–80 μmol/l). Brain MRI showed minimal nonspecific white matter lesions in the frontal lobe. Polyphasic units in frontal and nasal muscles were found in electromyography. Myasthenia gravis was first diagnosed at the age of 60 years and pyridostigmine was initiated. Because the treatment did not alleviate symptoms and all myasthenia studies were negative, treatment was discontinued. Lambert-Eaton myasthenic syndrome was excluded and PABP2 gene test for repeat expansion causing dominantly inherited oculopharyngeal muscle dystrophy was negative.
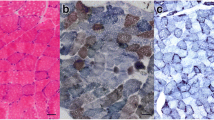
Muscle biopsy from vastus lateralis was compatible with mitochondrial myopathy (Fig. 1). Ten percent of the muscle fibers were COX-negative and few RRFs were found as well. Ultrastructural examination revealed an increased number of mitochondria and changes in the internal structure of mitochondria.
Mutation m.15923A > G is situated in a conservative position and causes typical mitochondria myopathy. a Sequence chromatograms showing variable heteroplasmy. P, patient; COX+, biochemically normal fibers; COX−, cytochrome c-oxidase negative fibers. b Histology stainings. Left panel: Hematoxylin & eosin staining showing ragged red fibers. Right panel: Cytochrome c-oxidase staining showing COX-negative (blue) fibers. Arrows denote biochemically abnormal fibers. c Clustal Omega [15] alingnment for multiple sequences showing complete conservation between species
Molecular methods and muscle histology
DNA of blood leucocytes and buccal epithelial cells was extracted by using QIAamp DNA Blood Mini Kit (QIAGEN, Hilden, Germany) and that of muscle by using Wizard® Genomic DNA purification kit (Promega Corporation, Madison, WI). Mitochondrial DNA was amplified and sequenced in twelve overlapping fragments. The amplification reactions were done by using Phire Hot Start II DNA polymerase (Thermo Fisher Scientific, Waltham, MA, U.S.A.) according to the provided protocol. Sequencing was carried out at Biocenter Oulu sequencing core facility.
The muscle sample for histological staining was fresh-frozen and cryostat sections (5 μm) were stained with routine histochemical techniques [11]. The stainings included hematoxylin and eosin and combined cytochrome c-oxidase and succinate dehydrogenase (COX-SDH). Laser-capture microdissection of COX-SDH stained frozen sections was done using Carl Zeiss P.A.L.M. microscope (Microlaser Technologies GmbH, Bernried, Germany) in Turku Centre for Biotechnology, University of Turku and Åbo Akademi University. Ten COX-negative and ten COX-positive fibers were collected into Carl Zeiss AdhesiveCap tubes (Carl Zeiss Gmbh, Göttingen, Germany) and DNA was released incubating fibers 30 min in 65 °C in lysis buffer containing 200 mM potassium hydroxide and 50 mM dithiothreitol followed by neutralization step with 900 mM Tris-HCl, pH 8.3. Amplification was carried out using Phusion High-Fidelity DNA polymerase (Thermo Fisher Scientific). Heteroplasmy was determined by cloning using CloneJET PCR Cloning Kit with blunt-end cloning protocol and DH5α competent cells (Thermo Fisher Scientific). Colony screening was done by using FastDigest XmiI (Thermo Fisher Scientific). XL-PCR for whole mtDNA amplification was carried out using Phusion High-Fidelity DNA polymerase with GC Buffer according to the original protocol (Thermo Fisher Scientific).
We found the m.15923A > G mutation in MT-TT in the skeletal muscle of the patient. The heteroplasmy was determined and, interestingly, the mutation was undetectable in the blood of the patient, while it was present with a 33% heteroplasmy in the skeletal muscle and with 2% heteroplasmy in the buccal mucosa. In pooled COX-negative fibers, the heteroplasmy was 92% while it was 43% in biochemically normal fibers. The investigation of mtDNA deletions remained negative.
Discussion and conclusions
We found the m.15923A > G mutation in an adult patient with ptosis and exercise intolerance. The pathogenicity of the mutation has been unclear, because the mutation has been found only in three patients before our study. Recent studies have shown, however, that m.15923A > G affects post-translational modification of tRNA threonine [10, 12]. The nucleotide in position 38 (m.15923A) has been shown to be crucial in N6-threonylcarbamoyladenosine (t6A) modification that occurs in position 37, and decreased level of t6A-modified tRNA has been detected in patient cell lines [10]. In addition, according to the Yarham scoring [13], the m.15923A > G is classified as definitely pathogenic. The score includes evaluation of nucleotide site conservation, heteroplasmy and mutation segregation with disease, and evidence of biochemical defect from single-fiber studies and mitochondrial complex activity measurements [13].
Three children harboring m.15923A > G have been reported to suffer from severe mitochondrial disorder (Table 1) [5, 6, 10]. Patient 1 was a girl who died 2.5 after birth of a fatal cardiopulmonary arrest [5, 6]. The mother had a history of five miscarriages and a delivery of a boy who died within two days. The two pregnancies and deliveries and the first 24 h of the infants had been normal. The mutation was heteroplasmic in the girl, but samples were not available from her deceased brother. Patient 2 was a girl with symptoms from early childhood [8]. The patient had exercise intolerance, vomiting and generalized seizures during her early childhood. She developed deafness, migraine, retinitis pigmentosa, and cognitive delay. Her symptoms were progressive. Mutation heteroplasmy was 78% in the muscle, 10% in the blood and 18% in buccal mucosa. Patient 3 was a 15-year-old girl with bilateral hearing impairment as the first manifestation at the age of 6 years [10]. At the time of the latest clinical examination, she had retinitis pigmentosa, lactic acidosis, myoclonic epilepsy, proteinuria and migraine suggesting a MERRF syndrome.
Decreased activity of OXPHOS complexes is a common finding in patients with mitochondrial disorder. The activity of complexes III and IV have been reported to be decreased in the muscle, liver and kidney of patient 1 [5, 6], whereas the activity of complex IV was normal in the muscle of patient 2 with mutation heteroplasmy of 78% [8]. Unfortunately, lack of remaining muscle prevented us from carrying out these analyses. However, previous studies and our single-fibre analysis suggest that the heteroplasmy required to cause an OXPHOS defect is very high and, therefore, the heteroplasmy of 33% in our patient may not be sufficient to cause the defect.
Pathogenic mutations in MT-TT are rare. In addition to m.15923A > G in the three patients, two other mutations have been found previously in single patients [5,6,7,8,9,10]. The m.15915A > G mutation has been found in a 16-year-old boy, whose symptoms started at the age of 8 years [7]. He had muscle weakness, hearing impairment, seizures, ptosis, intellectual disability, growth failure, and mitochondrial myopathy in muscle histology. The m.15933G > A mutation has been found in an adult patient, who had exercise-induced muscle weakness, myalgia, dysphagia and ptosis with mitochondrial myopathy in the skeletal muscle [9].
We conclude that the pathogenicity of the m.15923A > G mutation is now confirmed on the basis of the following facts: The mutation has been found in three severely affected children [5, 6, 8] and we describe the first adult patient harboring the mutation and with a mild mitochondrial disorder. The mutation has not been reported in population controls or listed among polymorphisms in MITOMAP database [14]. Mutation heteroplasmy has been shown to be high in the children with fatal mitochondrial disorder, while it was lower in our patient with a milder phenotype. COX-negative and ragged red fibers have been found in the skeletal muscle in all the four patients and decreased OXPHOS enzyme activities have been reported in the most severely affected infant. Differences in mutation load between biochemically deficient and normal fibers in our study further confirm the functional consequences of the mutation.
Abbreviations
- COX:
-
Cytochrome c-oxidase
- mtDNA:
-
Mitochondrial DNA
- MT-TT :
-
Mitochondrialally encoded transferRNA threonine
- OXPHOS:
-
Oxidative phosphorylation
References
Zeviani MI, Moraes CT, DiMauro S, Nakase H, Bonilla E, Schon EA, et al. Deletions of mitochondrial DNA in Kearns-Sayre syndrome. Neurology. 1988;38:1339–46.
Wallace DC, Singh G, Lott MT, Hodge JA, Schurr TG, Lezza AM, et al. Mitochondrial DNA mutation associated with Leber's hereditary optic neuropathy. Science. 1988;242:1427–30.
Gorman GS, Schaefer AM, Ng Y, Gomez N, Blakely EL, Alston CL, et al. Prevalence of nuclear and mitochondrial DNA mutations related to adult mitochondrial disease. Ann Neurol. 2015;77:753–9.
Majamaa K, Moilanen JS, Uimonen S, Remes AM, Salmela PI, Kärppä M, et al. Epidemiology of A3243G, the mutation for mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes: prevalence of the mutation in an adult population. Am J Hum Genet. 1998;63:447–54.
Yoon KL, Aprille JR, Ernst SG. Mitochondrial tRNAThr mutation in fatal infantile respiratory enzyme deficiency. Biochem Biophys Res Commun. 1991;176:1112–5.
Yoon KL, Ernst SG, Rasmussen C, Dooling EC, Aprille JR. Mitochondrial disorder associated with newborn cardiopulmonary arrest. Pediatr Res. 1993;33:433–40.
Nishino I, Seki A, Maegaki Y, Takeshita K, Horai S, Nonaka I, et al. A novel mutation in the mitochondrial tRNAThr gene associated with a mitochondrial encephalomyopathy. Biochem Biophys Res Commun. 1996;225:180–5.
Del Mar O'Callaghan M, Emperador S, Lopez-Gallardo E, Jou C, Bujan N, Montero R, et al. New mitochondrial DNA mutations in tRNA associated with three severe encephalopamyopathic phenotypes: neonatal, infantile, and childhood onset. Neurogenetics. 2012;13:245–50.
Soini HK, Vaisanen A, Karppa M, Hinttala R, Kytovuori L, Moilanen JS, et al. A novel MTTT mutation m.15933G>a revealed in analysis of mitochondrial DNA in patients with suspected mitochondrial disease. BMC Med Genet. 2017;18:14.
Lin H, Miyauchi K, Harada T, Okita R, Takeshita E, Komaki H, et al. CO2-sensitive tRNA modification associated with human mitochondrial disease. Nat Commun. 1875;2018:9.
Dubowitz V, Sewry C, Oldfors A. Muscle biopsy: a practical approach. 4th edition. London: Saunders, Elsevier; 2006.
Wang Y, Zeng QY, Zheng WQ, Ji QQ, Zhou XL, Wang ED. A natural non-Watson-crick base pair in human mitochondrial tRNAThr causes structural and functional susceptibility to local mutations. Nucleic Acids Res. 2018;46:4662–76.
Yarham JW, Al-Dosary M, Blakely EL, Alston CL, Taylor RW, Elson JL, et al. A comparative analysis approach to determining the pathogenicity of mitochondrial tRNA mutations. Hum Mutat. 2011;32:1319–25.
MITOMAP: A human mitochondrial genome database. http://www.mitomap.org, 2018.
Sievers F, Wilm A, Dineen D, Gibson TJ, Karplus K, Li W, et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal omega. Mol Syst Biol. 2011;7:539.
Acknowledgements
The authors gratefully acknowledge the expert technical assistance of Ms. Anja Heikkinen. Laser capture microdissection was performed at the Cell Imaging Core, Turku Centre for Biotechnology, University of Turku and Åbo Akademi University. The Cell Imaging Core is supported by Biocenter Finland infrastructure.
Funding
The study was supported by grants from the Sigrid Juselius Foundation, Medical Research Center, University of Oulu and Oulu University Hospital, and State research funding from Oulu University Hospital.
Availability of data and materials
All data generated or analyzed during this study is available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
MK performed clinical examinations. LK carried out molecular analyses and wrote the first draft of the manuscript. MS designed and conducted the LCM protocol. KM participated in designing the project, and all investigations occurred under his supervision. All authors have revised the manuscript and approved the manuscript for submission.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
High standard of ethics according to the WMA Declaration of Helsinki was applied in all investigations and clinical work described in this manuscript. The study protocol was approved by the Oulu University Hospital ethics committee. Written informed consent of participation was obtained from the patient. A copy of the written consent is available for review by the Editor of this journal.
Consent for publication
Written informed consent to publish this case report was given by the patient.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Kärppä, M., Kytövuori, L., Saari, M. et al. Mutation m.15923A>G in the MT-TT gene causes mild myopathy – case report of an adult-onset phenotype. BMC Neurol 18, 149 (2018). https://doi.org/10.1186/s12883-018-1159-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12883-018-1159-4