Abstract
Background
Anemia and electrolyte disturbances are adverse outcomes of chronic kidney disease (CKD). This study explored the association between metabolic parameters with anemia and electrolyte and mineral disorders among CKD patients in Taiwan.
Methods
This cross-sectional study with a total of 2176 CKD stages 3–5 patients were collected from the Department of Nephrology at Shuang Ho Hospital, Taipei Medical University through the “Chronic Kidney Disease Common Care Network” database from December 2008 to April 2019. A multivariable-adjusted logistic regression expressed as odd ratios (OR) was performed to assess the association of metabolic parameters with anemia and electrolyte and mineral disorders.
Results
Elevated diastolic blood pressure, fasting blood glucose, and glycated hemoglobin A1c (HbA1c) were associated with presence of anemia. Similarly, elevated fasting blood glucose and HbA1c were associated with hyponatremia (OR = 1.59 and 1.58, P for both < 0.01) and hypercalcemia (OR = 1.38 and 1.33, P for both < 0.05). There was no significant association in serum lipid levels with presence of anemia. However, total triglycerides, total cholesterol and low-density lipoprotein-cholesterol were only associated with presence of hypercalcemia (OR = 1.43, 1.95 and 3.08, respectively, P for all < 0.05).
Conclusions
Elevated diastolic blood pressure, fasting blood glucose, HbA1c and blood lipids are associated with anemia or electrolyte and mineral disorders in CKD patients.
Similar content being viewed by others
Background
The United States Renal Data System reported in 2016 that Taiwan has the largest incidence and prevalence of end-stage renal disease (ESRD), the advanced stage of chronic kidney disease (CKD), and has continued to rank first in the world since 2002 [1]. Thus, CKD occupied the top list of medical expenditures of national health insurance, indicating that CKD threatens the health of people in Taiwan and becomes a significant financial burden on national medical resources. Anemia is an inevitable and common consequence of patients with CKD, which can develop in the early stages of CKD and is commonly observed in more advanced stages in CKD patients [2]. A prospective cohort study in Taiwan showed that 12.0% of CKD patients in stage 3 were anemic, and the prevalence increased to 58.8 and 92.5% in CKD patients in stages 4 and 5, respectively [3]. Moreover, CKD patients with anemia are more prone to CKD progression, cardiovascular comorbidities, poor quality of life, and higher mortality [2, 4, 5].
Additionally, the kidneys play a critical role in regulating body fluid, electrolytes, and acid-base balance, and CKD can lead to metabolic acidosis, hyperkalemia, hyponatremia, hypercalcemia, and hyperphosphatemia, resulting in serious adverse outcomes such as bone mineral disorders, vascular calcification, and even mortality [6, 7]. Hyperkalemia is increasingly common with the progression of CKD and is one of the life-threatening electrolyte disorders in CKD patients, with a nearly 10-fold risk of death in stages 4 and 5 [8]. CKD patients with hyperkalemia may develop certain clinical manifestations such as muscle weakness, cardiac arrhythmias, and cardiac arrest [6]. Meanwhile, hyponatremia is the most common electrolyte abnormality in CKD patients, which is likely due to fluid overload and positively correlates with mortality and morbidity [9,10,11]. Similarly, hypercalcemia and hyperphosphatemia are common bone mineral disorders in CKD patients and have been associated with vascular calcification, CKD progression, cardiovascular events, and mortality [12,13,14]. Overall, anemia and electrolyte and mineral disorders in CKD patients have major implications for cardiovascular-related comorbidities.
Hypertension, diabetes, and lipid abnormalities are the major causes of CKD [15, 16]. Previous studies have mentioned that high fasting glucose, CKD severity, body mass index, (BMI), and serum iron are independently associated with anemia among CKD patients [17, 18]. It is also well acknowledged that the use of angiotensin-converting enzyme inhibitors, diuretic treatment, impaired bone turnover, and use of calcium-based phosphate binders have been associated with electrolyte and mineral disorders in CKD patients [19, 20]. However, the existing studies scarcely focused on whether metabolic abnormalities are associated with anemia and electrolyte disturbances in CKD patients. Therefore, our study aimed to investigate the association of metabolic parameters such as elevated blood pressure, blood glucose, triglycerides, total cholesterol, low-density lipoprotein (LDL)-cholesterol, and high-density lipoprotein (HDL)-cholesterol with anemia and electrolyte and mineral disorders in CKD patients in Taiwan.
Methods
Research design and patients
In this cross-sectional study, pre-ESRD patients (CKD stages 3 to 5 with proteinuria) were enrolled in the Department of Nephrology at Shuang Ho Hospital, Taipei Medical University, Taiwan from December 2008 to April 2019. The data were retrieved from the “Chronic Kidney Disease Common Care Network” that has been developed by the physician of the hospital for more than 10 years. We retrieved the patients’ data including sociodemographic information, lifestyle, medical records, anthropometry, and blood biochemical data. Before visiting the hospital for CKD treatment and participating in the pre-ESRD nutrition education program, all patients signed an informed consent allowing their data to be used without personal identification for research only. The Taipei Medical University Joint Institutional Review Board approved this study (N202001055).
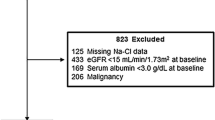
The data of 4094 patients who had participated in the pre-ESRD program were retrieved from the platform. Among the 4094 CKD patients, 3885 were stages 3–5 CKD patients, and a total of 1709 patients were excluded due to a history of cardiovascular disease (CVD) (n = 1072), a history of chronic liver disease and cancer (n = 232), tuberculosis and autoimmune disease (n = 48), current erythropoietin treatment (n = 353) and a lack of estimated glomerular filtration rate (eGFR) measurement (n = 4). The reason for excluding patients with presence of CVD and other comorbidities was because the predictor variables were highly associated with CVD, thus presence of CVD may become a confounding factor in our study. Finally, 2176 CKD patients were analyzed in this study.
Clinical and blood biochemical parameters
Anthropometric data, including body weight and height, were measured using high-accuracy ultrasonic sensors (AHS 700, Kaohsiung, Taiwan). BMI was determined using body weight (kg) divided by height squared (m2). Systolic and diastolic blood pressure (BP) was determined by oscillometry (OMRON HBP-9020, Taipei, Taiwan). For blood biochemical measurements, all patients were required to fast at least 8 h prior to blood tests. Blood biochemical data including hemoglobin (Hb), fasting blood glucose, albumin, triglycerides, total cholesterol, LDL-cholesterol, HDL-cholesterol, potassium (K), sodium (Na), calcium (Ca), and phosphorus (P) were determined using an auto chemical analyzer (Beckman DxC 800, California, USA). Glycated hemoglobin A1c (HbA1c) was determined by capillary electrophoresis (Sebia II, Lisses, France).
Anemia was defined as Hb < 130 g/Lfor men and < 120 g/L for women, or current iron supplementation according to the Kidney Disease Improving Global Outcomes (KDIGO) Anemia Work Group [21]. Serum calcium levels were corrected for serum albumin by using Payne’s formula: corrected calcium (C-Ca) (mmol/L = calcium (mmol/L) + 0.02 × [40 – serum albumin (g/L)]) [22]. Hyperkalemia was defined as serum potassium > 5.0 mmol/L and hyponatremia was defined as serum sodium < 135 mmol/L [6]. Hypercalcemia was defined as serum levels of corrected calcium ≥2.37 mmol/L (9.5 mg/dL), while hyperphosphatemia was defined as serum levels of phosphorus ≥1.49 mmol/L (4.6 mg/dL) based on guidelines from National Kidney Foundation [23]. Metabolic parameters were defined as: high systolic BP if ≥130 mmHg with medication, high diastolic BP if ≥85 mmHg with medication [24], high fasting blood glucose if ≥7.0 mmol/L (126 mg/dL) with treatment and high HbA1C if ≥6.5% with treatment [25]. Moreover, high triglycerides if ≥2.3 mmol/L (200 mg/dL) or with treatment, high total cholesterol if ≥6.2 mmol/L (240 mg/dL) or with treatment, high LDL-cholesterol if ≥4.1 mmol/L (160 mg/dL) or with treatment and low HDL-cholesterol if < 1.04 mmol/L (40 mg/dL) or with treatment [26]. The value of eGFR was calculated using the equation of the Modification of Diet in Renal Disease study [27]. Moreover, CKD stages were classified based on eGFR values into: CKD stages 3a (45–59 mL/min/1.73 m2), 3b (30–44 mL/min/1.73 m2), 4 (15–29 mL/min/1.73 m2) and 5 (< 15 mL/min/1.73 m2).
Other covariates
We also retrieved sociodemographic and lifestyle data such as age, sex, marital status, educational level, occupation (unemployment and professional), cigarette smoking, alcohol consumption, and physical activity from the database. Marital status was dichotomized as no (divorced, widowed, and separated) and yes (currently married). Educational level was categorized as low (high school or below) and high (above high school). Cigarette smoking and alcohol use were categorized as no, former (quit smoking or drinking), and current. Data on type of physical activity (e.g., regular walking, fast walking, jogging, dancing, gymnastics, biking or hiking), frequency (5 response options: from never to ≥7 times/week), and duration (5 response options: from never to > 90 min) were collected and retrieved from the database. Physical activity was defined as no (< 30 min/week) and yes (≥30 min/week). Current medication use, including angiotensin II receptor blockers, angiotensin-converting enzyme inhibitors, calcium channel blockers, calcium phosphate binders, iron supplements, antihypertensives, hypolipidemic agents, hypoglycemic agents, or insulin injections were also queried. Patients were categorized as no (never) and yes (≥once) for participation in the nutrition education program.
Data analysis
Data are presented as numbers and percentages for categorical variables or mean ± standard deviation (SD) for continuous variables. For categorical variables, a chi-square test was performed to examine differences in characteristics of CKD patients with or without anemia, whereas the general linear model was used to examine differences in means for continuous variables. The association between metabolic parameters and anemia or electrolyte and mineral disorders was analyzed using a multivariable-adjusted logistic regression model, and the data are reported as odds ratio (OR) and 95% confidence interval (CI). Considering different Hb cut-off values for anemia in men and women, the association between metabolic parameters and anemia was stratified by sex. The regression models were adjusted for age, marital status, educational level, occupation, smoking status, drinking status, physical activity, drug use, nutritional education, and BMI in the association between metabolic parameters and anemia. Besides the confounders adjusted in the regression model, sex was also adjusted in the association between metabolic parameters and electrolyte and mineral disorders. Patients with ‘normal’ status in all dependent variables were selected as the reference group. A P-value of < 0.05 was considered statistically significant, and STATA software version 13 (STATA Corp LLC, Texas, USA) was used to perform the statistical analysis.
Results
Characteristics of the patients
Table 1 shows the characteristics of patients with and without anemia. Of 2176 CKD patients, 67% were anemic, and 56.1% of anemic patients were men. Patients with anemia were more likely to be older (72.1 ± 14.0 vs. 68.7 ± 13.3 years) and had higher proportions of advanced stages (stages 4 and 5: 35.5 and 29.3% vs. 20.6 and 4.6%), lower educational level (69.9% vs. 60.4%), inactive physical activity (72.6% vs. 65.4%) and drug use (61.2% vs. 56.8%) compared to those without anemia. Moreover, patients with anemia had lower BMI (25.2 ± 4.4 vs. 26.3 ± 4.2 kg/m2), diastolic BP (72 ± 14 vs. 75 ± 13 mmHg), albumin (39.5 ± 5.9 vs. 43.1 ± 4.5 g/L) and triglycerides (1.8 ± 1.9 vs. 1.9 ± 1.3 mmol/L) compared to those without anemia. The characteristics of CKD patients stratified by electrolyte and mineral disorders are shown in Additional file 1: Table S1. Among all CKD patients, 18.3% of patients were hyperkalemic, 10.2% of patients were hyponatremic, 12.4% of patients were hypercalcemic and 25.7% of patients were hyperphosphatemic.
Association of metabolic parameters with anemia and electrolyte and mineral disorders
The results of multivariable-adjusted logistic regression showed that CKD patients with increased values of diastolic BP (OR = 1.54, 95% CI 1.09–2.19, P = 0.015), fasting blood glucose (OR = 1.63, 95% CI 1.27–2.09, P < 0.001) or HbA1c (OR = 1.48, 95% CI 1.15–1.89, P = 0.002) were associated with presence of anemia compared to those with normal values (Table 2). However, no significant association was found between elevated blood lipid parameters and anemia in either sex. The results of multivariable-adjusted logistic regression also showed that CKD patients with high diastolic BP were associated with presence of hyperkalemia (OR = 1.54, 95% CI 1.08–2.21, P = 0.017) compared to those with normal diastolic BP. Patients with elevated fasting blood glucose were associated with presence of hyponatremia (OR = 1.59, 95% CI 1.16–2.16, P = 0.004), hypercalcemia (OR = 1.38, 95% CI 1.05–1.83, P = 0.025) or hyperphosphatemia (OR = 1.32, 95% CI 1.06–1.65, P = 0.014). Additionally, CKD patients with elevated HbA1c were significantly associated with presence of hyperkalemia (OR = 1.26, 95% CI 1.00–1.59, P = 0.049), hyponatremia (OR = 1.58, 95% CI 1.16–2.16, P = 0.004) or hypercalcemia (OR = 1.33, 95% CI 1.00–1.76, P = 0.049). Meanwhile, CKD patients with high levels of triglycerides (OR = 1.43, 95% CI 1.03–1.99, P = 0.034), total cholesterol (OR = 1.95, 95% CI 1.28–2.96, P = 0.002) and LDL-cholesterol (OR = 3.08, 95% CI 1.77–5.34, P < 0.001) were significantly associated with hypercalcemia only. Moreover, anemic CKD patients had a significant association with presence of hyperkalemia (OR = 3.13, 95% CI 2.32–4.22, P < 0.001), hyponatremia (OR = 2.28, 95% CI 1.54–3.38, P < 0.001) and hyperphosphatemia (OR = 3.91, 95% CI 2.93–5.20, P < 0.001) (Table 3).
Discussion
In our study, anemic CKD patients were more likely to be older and male, and 67% of the subjects in stages 3–5 had anemia. Additionally, high fasting blood glucose and HbA1c levels were associated with presence of anemia and electrolyte and mineral imbalance including hyponatremia, hypercalcemia, and hyperphosphatemia. Similar results were also found in Japanese CKD patients, in whom the prevalence of anemia in stages 4 and 5 was 40.1 and 60.3%, respectively [28]. Age, gender, and presence of complications could be the factors affecting the development and/or severity of anemia in CKD patients [28, 29]. A previous study also showed that CKD patients with a diastolic BP between 80 and 89 mmHg had a lower risk of anemia (OR = 0.38, 95% CI 0.16–0.92) compared to those with a diastolic BP between 60 and 79 mmHg, but systolic BP was not significantly associated with anemia risk [30]. The prevalence of anemia was higher in uncontrolled hypertensives than in well-controlled hypertensives, suggesting that hypertension is independently associated with increased anemia risk [31]. Moreover, presence of anemia was independently associated with renal events even in overall hypertensive patients with well-controlled blood pressure [32]. The progression of anemia in CKD patients may be related to the left ventricular hypertrophy that exists in persistent renal failure in combination with decreased Hb levels and increased BP [33].
Similar findings were reported in diabetic patients that the incidence of anemia (33.5% vs. 27.9%) was increased in diabetic patients with uncontrolled HbA1c levels (> 7.5%) compared to those with controlled HbA1c levels (≤7.5%) [34]. Chronic hyperglycemia could lead to a cellular hypoxic state in the renal interstitium, which contributes to impaired production of erythropoietin in renal peritubular fibroblasts [35, 36]. Low erythropoietin levels are a major cause of early anemia in patients with impaired glucose homeostasis [36]. Moreover, anemia per se could be a risk factor for hyperglycemia. Elevated HbA1c levels were associated with decreased serum transferrin saturation, ferritin and mean corpuscular hemoglobin levels in Japanese diabetic women in late pregnancy, and decreased anemia parameters (24–35 weeks of gestation) occurred before elevated HbA1c levels (32–35 weeks of gestation) [37], indicating that anemia might be one of the risk factors for hyperglycemia. Thus, these results suggest that anemia could be a risk factor and consequence of hyperglycemia [35,36,37].
Electrolyte and mineral imbalances are common in patients with impaired renal function because the kidney plays an important role in regulating body fluid, electrolytes, acid-base balance, and iron metabolism [6]. Additionally, anemia in patients with impaired renal function may be associated with disturbance in the metabolism of calcium, phosphate, and hydrogen ions [38]. An increased risk of developing hyperkalemia has also been found in hypertensive patients on antihypertensive therapy [39]. Likewise, the present study showed that CKD patients with high diastolic BP were associated with presence of hyperkalemia. Imbalance of electrolytes potassium, sodium, and calcium were observed more frequently in patients with type 2 diabetes (> 70%) than in patients with hypoglycemia or normal fasting blood glucose, and changes in electrolyte distribution could be due to osmotic fluid shift by hyperglycemia or loss of electrolytes by osmotic diuresis [40]. Our study showed that high HbA1c was significantly associated with presence of hyperkalemia in CKD patients (P = 0.049). Similarly, a study in Benin found that mean serum potassium levels were significantly higher in diabetic patients compared to non-diabetic controls of the same age and sex [41].
Our results showed that high fasting blood glucose and high HbA1c were associated with presence of hyponatremia in CKD patients. Similarly, a previous study found that hyperglycemia in healthy adults with acute insulin deficiency rapidly decreased serum sodium levels, and hyponatremia was reversed by normoglycemia [42]. The plausible mechanism is that hyperglycemia could increase serum osmolality, cause efflux of water from cells, and lead to further hyponatremia due to dilution [42]. Additionally, our results showed that high fasting blood glucose and HbA1c were associated with presence of hypercalcemia and hyperphosphatemia in CKD patients. Consistent with our findings, serum calcium levels were positively associated with fasting blood glucose and insulin resistance, but negatively associated with pancreatic β-cell function in healthy adults [43], suggesting that abnormal regulation of calcium homeostasis may be related to impaired β-cell function and elevated glucose levels. According to the results of previous and current studies, the relationship between hyperglycemia and hypercalcemia could be cyclic. Hyperphosphatemia frequently occurred in late-stage CKD patients because the excretory capacity of phosphate by the kidney is exhausted [44, 45]. Moreover, hyperphosphatemia has been associated with vascular calcification and abnormal bone mineralization and turnover [46], which was frequently and positively associated with CVD morbidity and mortality in CKD or diabetic patients [47]. Combined hyperphosphatemia with hyperglycemia promoted vascular calcification in human aortic smooth muscle cells compared with hyperphosphatemia or hyperglycemia alone [47]. Similar to the previous studies [48, 49], we also found a strong association between hyperphosphatemia and presence of anemia. High serum phosphorus may lead to increases in the production of uremic toxins as higher polyamines and the secretion of parathyroid hormone which has been shown to inhibit erythropoiesis [48].
The present study showed an association between blood lipids and hypercalcemia in CKD patients. Similarly, previous studies also found a positive association between serum calcium and triglycerides, total cholesterol or HDL-cholesterol [50, 51]. Elevated calcium levels contributed to a decrease in hepatic cholesterol catabolism via a reduction in 7α-hydroxylase, and to an increase in de novo lipid synthesis via an increase in sterol regulatory element-binding protein-1c, which may serve as a plausible mechanism for this association [50]. However, in the present study, no significant relationship was found between lipid profile and anemia. Previous studies also found no significant association between Hb levels and serum lipid concentrations [52, 53], and the results have been inconsistent [54]. Therefore, it should be further investigated whether and how iron status affects serum lipids in CKD patients.
This study had several limitations. First, this study was a cross-sectional design, making it difficult to draw a causal conclusion using our model. A longitudinal study is needed to clarify the relationship. Second, we did not collect data on patients’ dietary habits in relation to mineral or iron sources. Third, the definition of anemia in the present study lacks other parameters such as serum iron, ferritin, transferrin saturation, and total iron-binding capacity. For clinical diagnosis of anemia in CKD patients, measurement of hemoglobin along with body iron status is recommended. The strength of our study is that it includes a large sample population of CKD patients, which provides better evidence for general public interpretation.
Conclusions
In summary, fasting blood glucose and HbA1c are associated with presence of anemia, hyperkalemia, hyponatremia, hypercalcemia, and hyperphosphatemia in patients with stage 3–5 CKD. Moreover, the results of the present study suggest that there is a discordant association of blood pressure or serum lipid concentrations with anemia or electrolyte and mineral imbalances. A longitudinal study with prospective measurements is needed to further investigate this association.
Availability of data and materials
The data that support the findings of this study are available from the Department of Nephrology at Shuang Ho Hospital, Taipei Medical University, but restricted for research use only. The data are not publicly available. Data are available from the authors upon reasonable request and with permission of Shuang Ho Hospital, Taipei Medical University.
Abbreviations
- BMI:
-
Body mass index
- BP:
-
Blood pressure
- Ca:
-
Calcium
- C-Ca:
-
Corrected calcium
- CKD:
-
Chronic kidney disease
- eGFR:
-
Estimated glomerular filtration rate
- ESRD:
-
End stage renal disease
- Hb:
-
Hemoglobin
- HbA1c:
-
Glycated hemoglobin A1c
- HDL:
-
High-density lipoprotein
- K:
-
Potassium
- LDL:
-
Low-density lipoprotein
- Na:
-
Sodium
- P:
-
Phosphorus
References
Saran R, Robinson B, Abbott K. United States renal data system 2016 USRDS annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2017;69(Suppl. 1):A7–8.
Stauffer ME, Fan T. Prevalence of anemia in chronic kidney disease in the United States. PLoS One. 2014;9:e84943.
Wen CP, Cheng TYD, Tsai MK, Chang YC, Chan HT, Tsai SP, et al. All-cause mortality attributable to chronic kidney disease: a prospective cohort study based on 462293 adults in Taiwan. Lancet. 2008;371:2173–82.
Mehdi U, Toto RD. Anemia, diabetes, and chronic kidney disease. Diabetes Care. 2009;32:1320–6.
Astor BC, Coresh J, Heiss G, Pettitt D, Sarnak MJ. Kidney function and anemia as risk factors for coronary heart disease and mortality: the atherosclerosis risk in communities (ARIC) study. Am Heart J. 2006;151:492–500.
Dhondup T, Qian Q. Electrolyte and acid-base disorders in chronic kidney disease and end-stage kidney failure. Blood Purif. 2017;43:179–88.
Dhingra R, Sullivan LM, Fox CS, Wang TJ, D’Agostino RB, Gaziano JM, et al. Relations of serum phosphorus and calcium levels to the incidence of cardiovascular disease in the community. Arch Intern Med. 2007;167:879–85.
Einhorn LM, Zhan M, Hsu V, Walker LD, Moen MF, Seliger SL, et al. The frequency of hyperkalemia and its significance in chronic kidney disease. Arch Intern Med. 2009;169:1156–62.
Kovesdy CP, Lott EH, Lu JL, Malakauskas SM, Ma JZ, Molnar MZ, et al. Hyponatremia, hypernatremia, and mortality in patients with chronic kidney disease with and without congestive heart failure. Circulation. 2012;125:677–84.
Zhang R, Wang SY, Zhang M, Cui LJ. Hyponatremia in patients with chronic kidney disease. Hemodial Int. 2017;21:3–10.
Lim LM, Tsai NC, Lin MY, Hwang DY, Lin HYH, Lee JJ, et al. Hyponatremia is associated with fluid imbalance and adverse renal outcome in chronic kidney disease patients treated with diuretics. Sci Rep. 2016;6:36817.
Shanahan CM, Crouthamel MH, Kapustin A, Giachelli CM. Arterial calcification in chronic kidney disease: key roles for calcium and phosphate. Circ Res. 2011;109:697–711.
Da JJ, Xie XF, Wolf M, Disthabanchong S, Wang JW, Zha Y, et al. Serum phosphorus and progression of CKD and mortality: a meta-analysis of cohort studies. Am J Kidney Dis. 2015;66:258–65.
O'Seaghdha CM, Hwang SJ, Muntner P, Melamed ML, Fox CS. Serum phosphorus predicts incident chronic kidney disease and end-stage renal disease. Nephrol Dial Transplant. 2011;26:2885–90.
Jha V, Garcia-Garcia G, Iseki K, Li Z, Naicker S, Plattner B, et al. Chronic kidney disease: global dimension and perspectives. Lancet. 2013;382:260–72.
Lee PH, Chang HY, Tung CW, Hsu YC, Lei CC, Chang HH, et al. Hypertriglyceridemia: an independent risk factor of chronic kidney disease in Taiwanese adults. Am J Med Sci. 2009;338:185–9.
Loutradis C, Skodra A, Georgianos P, Tolika P, Alexandrou D, Avdelidou A, et al. Diabetes mellitus increases the prevalence of anemia in patients with chronic kidney disease: a nested case-control study. World J Nephrol. 2016;5:358–66.
Alagoz S, Dincer MT, Eren N, Bakir A, Pekpak M, Trabulus S, et al. Prevalence of anemia in predialysis chronic kidney disease: is the study center a significant factor? PLoS One. 2020;15:e0230980.
Loutradis C, Tolika P, Skodra A, Avdelidou A, Sarafidis PA. Prevalence of hyperkalemia in diabetic and non-diabetic patients with chronic kidney disease: a nested case-control study. Am J Nephrol. 2015;42:351–60.
Seng JJB, Tan YLC, Lim RW, Ng HTS, Lee PH, Wong J. Prevalence and risk factors for hypercalcemia among non-dialysis patients with chronic kidney disease-mineral and bone disorder. Int Urol Nephrol. 2018;50:1871–7.
Kidney Disease Improving Global Outcomes (KDIGO). KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279–35.
Payne RB, Little AJ, Williams RB, Milner JR. Interpretation of serum calcium in patients with abnormal serum proteins. Br Med J. 1973;4:643–6.
Bailie GR, Massry SG. Clinical practice guidelines for bone metabolism and disease in chronic kidney disease: an overview. Pharmacotherapy. 2005;25:1687–707.
Syauqy A, Hsu CY, Rau HH, Chao JC. Association of dietary patterns, anthropometric measurements, and metabolic parameters with C-reactive protein and neutrophil-to-lymphocyte ratio in middle-aged and older adults with metabolic syndrome in Taiwan: a cross-sectional study. Nutr J. 2018;17:106.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37(Suppl. 1):S81–90.
Kurniawan AL, Hsu CY, Rau HH, Lin LY, Chao JC. Association of kidney function-related dietary pattern, weight status, and cardiovascular risk factors with severity of impaired kidney function in middle-aged and older adults with chronic kidney disease: a cross-sectional population study. Nutr J. 2019;18:27.
Levey AS, Coresh J. Chronic kidney disease. Lancet. 2012;379:165–80.
Sofue T, Nakagawa N, Kanda E, Nagasu H, Matsushita K, Nangaku M, et al. Prevalence of anemia in patients with chronic kidney disease in Japan: a nationwide, cross-sectional cohort study using data from the Japan chronic kidney disease database (J-CKD-DB). PLoS One. 2020;15:e0236132.
Liu H, Ye Y, Chen Y, Zhang Y, Li S, Hu W, et al. Therapeutic targets for the anemia of predialysis chronic kidney disease: a meta-analysis of randomized, controlled trials. J Investig Med. 2019;67:1002–8.
Adera H, Hailu W, Adane A, Tadesse A. Prevalence of anemia and its associated factors among chronic kidney disease patients at University of Gondar Hospital, Northwest Ethiopia: a hospital-based cross sectional study. Int J Nephrol Renov Dis. 2019;12:219–28.
Paul B, Wilfred NC, Woodman R, Depasquale C. Prevalence and correlates of anaemia in essential hypertension. Clin Exp Pharmacol Physiol. 2008;35:1461–4.
Kim-Mitsuyama S, Soejima H, Yasuda O, Node K, Jinnouchi H, Yamamoto E, et al. Anemia is an independent risk factor for cardiovascular and renal events in hypertensive outpatients with well-controlled blood pressure: a subgroup analysis of the ATTEMPT-CVD randomized trial. Hypertens Res. 2019;42:883–91.
Levin A, Thompson CR, Ethier J, Carlisle EJ, Tobe S, Mendelssohn D, et al. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999;34:125–34.
AlDallal SM, Jena N. Prevalence of anemia in type 2 diabetic patients. J Hematol. 2018;7:57–61.
Singh DK, Winocour P, Farrington K. Mechanisms of disease: the hypoxic tubular hypothesis of diabetic nephropathy. Nat Clin Pract Nephrol. 2008;4:216–26.
Bosman DR, Winkler AS, Marsden JT, Macdougall IC, Watkins PJ. Anemia with erythropoietin deficiency occurs early in diabetic nephropathy. Diabetes Care. 2001;24:495–9.
Hashimoto K, Osugi T, Noguchi S, Morimoto Y, Wasada K, Imai S, et al. A1C but not serum glycated albumin is elevated because of iron deficiency in late pregnancy in diabetic women. Diabetes Care. 2010;33:509–11.
Zarzecki M, Chudek J, Kukla M, Kopeć P, Mamcarz E, Wnuk Z, et al. Prevalence of anemia, calcium-phosphorus abnormalities and metabolic acidosis in different stages of chronic renal failure. Pol Arch Med Wewn. 2004;112:1211–9.
Kovesdy CP. Epidemiology of hyperkalemia: an update. Kidney Int Suppl. 2016;6:3–6.
Sayouba JPD, Asanghanwa M, Njouendou AJ, Kamga HL, Assob JCN. Electrolyte imbalance in type 2 diabetes: a case study from the west region of Cameroon. Int J Res Med Sci. 2020;8:2017–22.
Anago E, Medehouenou T, Akpovi CD, Tchehouenou H. Electrolyte disturbances in diabetic patients in Cotonou, Benin. Int J Res Med Sci. 2016;4:5430–5.
Hillier TA, Abbott RD, Barrett EJ. Hyponatremia: evaluating the correction factor for hyperglycemia. Am J Med. 1999;106:399–403.
Sun G, Vasdev S, Martin GR, Gadag V, Zhang H. Altered calcium homeostasis is correlated with abnormalities of fasting serum glucose, insulin resistance, and β-cell function in the Newfoundland population. Diabetes. 2005;54:3336–9.
Tejwani V, Qian Q. Calcium regulation and bone mineral metabolism in elderly patients with chronic kidney disease. Nutrients. 2013;5:1913–36.
Levin A, Bakris GL, Molitch M, Smulders M, Tian J, Williams LA, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007;71:31–8.
Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, et al. Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med. 2000;342:1478–83.
Wang P, Zhou P, Chen W, Peng D. Combined effects of hyperphosphatemia and hyperglycemia on the calcification of cultured human aortic smooth muscle cells. Exp Ther Med. 2019;17:863–8.
Tran L, Batech M, Rhee CM, Streja E, Kalantar-Zadeh K, Jacobsen SJ, et al. Serum phosphorus and association with anemia among a large diverse population with and without chronic kidney disease. Nephrol Dial Transplant. 2016;31:636–45.
Ryu SR, Park SK, Jung JY, Kim YH, Oh YK, Yoo TH, et al. The prevalence and management of anemia in chronic kidney disease patients: result from the KoreaN cohort study for outcomes in patients with chronic kidney disease (KNOW-CKD). J Korean Med Sci. 2017;32:249–56.
Gallo L, Faniello MC, Canino G, Tripolono C, Gnasso A, Cuda G, et al. Serum calcium increase correlates with worsening of lipid profile: an observational study on a large cohort from South Italy. Medicine. 2016;95:e2774.
Jorde R, Sundsfjord J, Fitzgerald P, Bønaa KH. Serum calcium and cardiovascular risk factors and diseases: the Tromsø study. Hypertension. 1999;34:484–90.
Choi JW, Kim SK, Pai SH. Changes in serum lipid concentrations during iron depletion and after iron supplementation. Ann Clin Lab Sci. 2001;31:151–6.
Zaribaf F, Entezari MH, Hassanzadeh A, Mirzaian S. Association between dietary iron, iron stores, and serum lipid profile in reproductive age women. J Educ Health Promot. 2014;3:15.
Zorca S, Freeman L, Hildesheim M, Allen D, Remaley AT, Taylor JG, et al. Lipid levels in sickle-cell disease associated with hemolytic severity, vascular dysfunction and pulmonary hypertension. Br J Haematol. 2010;149:436–45.
Acknowledgements
The authors thank the Department of Nephrology at Shuang Ho Hospital, Taipei Medical University, Taiwan for collecting and providing the database available for this study.
Funding
This research was funded by Shuang Ho Hospital, Taipei Medical University, Taiwan, grant number 110TMU-SHH-31.
Author information
Authors and Affiliations
Contributions
A.L.K. and J.C.J.C. conceived and designed the study. Y.L.Y., C.Y.H., H.A.L., P.Y.N. and M.Y.C. managed and retrieved the data. A.L.K. analyzed and performed the statistical analysis. C.Y.H. and J.C.J.C. supervised the study. A.L.K., R.P. and J.C.J.C. wrote the manuscript. M.Y.C. and J.C.J.C. provided funding acquisition. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by The Taipei Medical University Joint Institutional Review Board approved this study (N202001055). All patients signed an informed consent allowing their data to be used without personal identification for research only. All methods for the human study were conducted in accordance with ethical quidelines and regulations.
Consent for publication
The data in this study did not include any personal information, and all patients were adults. Not applicable.
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1
. Characteristics of CKD patients stratified by electrolyte and mineral disorders.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Kurniawan, A.L., Yang, YL., Hsu, CY. et al. Association between metabolic parameters and risks of anemia and electrolyte disturbances among stages 3–5 chronic kidney disease patients in Taiwan. BMC Nephrol 22, 385 (2021). https://doi.org/10.1186/s12882-021-02590-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12882-021-02590-w