Abstract
Background
Prediction of preoperative frailty risk in the emergency setting is a challenging issue because preoperative evaluation cannot be done sufficiently. In a previous study, the preoperative frailty risk prediction model used only diagnostic and operation codes for emergency surgery and found poor predictive performance. This study developed a preoperative frailty prediction model using machine learning techniques that can be used in various clinical settings with improved predictive performance.
Methods
This is a national cohort study including 22,448 patients who were older than 75 years and visited the hospital for emergency surgery from the cohort of older patients among the retrieved sample from the Korean National Health Insurance Service. The diagnostic and operation codes were one-hot encoded and entered into the predictive model using the extreme gradient boosting (XGBoost) as a machine learning technique. The predictive performance of the model for postoperative 90-day mortality was compared with those of previous frailty evaluation tools such as Operation Frailty Risk Score (OFRS) and Hospital Frailty Risk Score (HFRS) using the receiver operating characteristic curve analysis.
Results
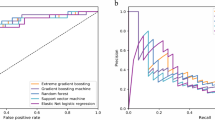
The predictive performance of the XGBoost, OFRS, and HFRS for postoperative 90-day mortality was 0.840, 0.607, and 0.588 on a c-statistics basis, respectively.
Conclusions
Using machine learning techniques, XGBoost to predict postoperative 90-day mortality, using diagnostic and operation codes, the prediction performance was improved significantly over the previous risk assessment models such as OFRS and HFRS.
Similar content being viewed by others
Introduction
As the proportion of older patients undergoing surgery increases worldwide, the evaluation of preoperative frailty is increasingly important [1, 2]. Frailty refers to a clinical condition in which physiological reserve is reduced and vulnerable to daily stressors [3]. Especially, an innocuous stress factor such as surgery in frail older patients is associated with poor clinical outcomes [4]. Predicting the frailty risk in older patients before surgery is an increasingly critical issue [1, 4]. Unfortunately, no established method or widely accepted model is available for the assessment of preoperative frailty in surgical patients. In previous frailty studies, the Fried model and Rockwood model were used [5, 6]. Most of the previous frailty measurement tools were time-consuming and required a lot of clinical evaluation and examination [7,8,9,10,11,12]. For example, frailty measurement methods such as gait speed, handgrip strength, or surveys of patients with many checklists [7,8,9,10,11,12,13,14]. Therefore, it had many limitations to be applied clinically, such as the condition in bed-ridden patients or emergency surgery, where there is not enough time for preoperative assessment. In a study by Gilbert et al., in 2018, the Hospital Frailty Risk Score (HFRS) was proposed as a frailty measurement tool using diagnostic code information from older patients [15] who were hospitalized in the emergency room. Therefore, there were many limitations to use in surgical patients. Similarly, another study predicted the preoperative frailty risk in surgical patients from only diagnostic code and operation code information that can be automatically extracted based on electronic medical records (EMR) data [16, 17] and Operation Frailty Risk Score (OFRS) using HFRS scores calculated from ICD-10 diagnostic codes and operation risk groups classified into eight categories were suggested [16, 17]. However, variation in subjective operation code classification, and difficulties to apply in their models based on specific hospital data, limit its usefulness [16, 17].
Therefore, in this study, we tried to develop a widely applicable predictive model based on the national health insurance database, which contains a wider range of multi-institutional data. Moreover, our study improved the predictive performance using artificial intelligence technologies such as machine learning, which are widely used in medical research recently.
Methods
This study was approved by our local institutional review board. Written informed consent was exempted due to the retrospectively collecting of the data. The study was conducted in accordance with relevant guidelines and regulations or declaration of Helsinki. Machine learning modeling in this study was conducted according to the guidelines entitled “Guidelines for Development and Reporting Machine Learning Predictive Models in Biomedical Research: A Multidisciplinary View” [18].
Data extraction
In this study, we retrieved the cohort data of older patients from the national sample cohorts (National Health Insurance Service – National Sample Cohort, version 2.0) provided by the Korean National Health Insurance Service (KNHIS). The cohort of older patients is a public health database of older patients (≥ 60 years old) from 2002 to 2013 [19]. The database covers insurance claim-related data and medical service-related data of approximately 550,000 older patients [19]. Among them, we extracted only 22,448 older patients (≥ 75 years old) who visited the hospital for emergency surgery (Fig. 1). Four variables such as age, sex, diagnosis code, and operation code, were used for analysis. Moreover, the national insurance claim codes were used for the operation code, and the ICD-10 code was used for the diagnostic code in which only the information recorded over the past year from the date of the surgery was extracted from the insurance database. The primary outcome of interest was 90-day postoperative mortality for the assessment tool of perioperative frailty. To obtain 90-day postoperative mortality, the data of all-cause deaths within 90 days after surgery were extracted from the death data in the KNHIS database. All data were downloaded only on the designated server computer according to KNHIS’ data policy, and all data analysis was conducted only on an allocated server.
Hospital frailty risk score
The HFRS can be applied quickly to evaluate the frailty risk in the clinical field using ICD-10 diagnostic codes extracted from EMR [15]. In HFRS, 109 ICD-10 diagnostic codes for frailty were scored according to the advice from geriatric medicine experts. The HFRS was developed using cluster analysis in which the scores were given to ICD-10 codes that were at least twice as prevalent in a frail group compared to a non-frail group. In our study, HFRS was calculated based on ICD-10 codes diagnosed within one year from the date of surgery. Moreover, the HFRS is the total corresponding score if the diagnostic code matches 109 frailty-related codes suggested in the previous study [15].
Operation frailty risk score
The OFRS was proposed in a recent study to evaluate preoperative frailty risk in surgical patients. Compared to HFRS, OFRS additionally used operation code information to reflect surgery-related risks. OFRS is the total points corresponding to the HFRS calculated from the diagnostic code, operation risk group for the operation code, and the patient’s age (Table S1) [16, 17]. Although the patient’s gender was originally considered in developing prediction model, it was excluded from the final scoring system because the effect of gender on outcome was not statistically significant (Table S1). According to the OFRS scoring system, age was divided into three categories, and HFRS as the total score calculated was divided by the three risk groups to give OFRS points to each group (Table S1) [16, 17]. Moreover, the operation-related insurance claim codes were classified into eight groups by clinical experts according to the risk of surgery (Table S2) [16, 17]. OFRS can obtain the final score by summing all the scores obtained in this way [16, 17]. The OFRS was classified as low risk if it was less than 2 points and high risk if it was greater than 4 points as described previously [16]. In this study, the total score was used in the analysis in the form of continuous variables instead of the risk groups.
Machine learning modeling
A total of 772 operation codes and 98 diagnostic codes were converted into dummy variables, and each code was used in the model as an independent input variable. The missing values of the continuous variable were filled with the median values of the corresponding variable, but if the values of the categorical variable were missing, the data in corresponding row was excluded from the entire research dataset. We used the extreme gradient boosting (XGBoost) as a machine learning technique [20]. XGBoost is a boosting tree-based ensemble model that regressively improves the performance of the model to minimize the residuals for each iteration [20]. For model training, the entire dataset was randomly allocated, and 80% was used for training and the remaining 20% for the test. We use a 10-fold cross-validation technique, a method of dividing the training dataset into 10-fold datasets and then cross-validating each other to prevent the model overfitting. Hyperparameters were tuned for the best predictive performance in which the root mean square error is minimized by using bayesian optimization. The final hyperparameters tuned are as follows: max_depth was 6, min_child_weight was 1, gamma was 0.1, eta was 0.2 and subsample was 0.8. The area under the receiver operating characteristics (AUROC) curve was used to evaluate the predictive performance of the machine learning based prediction model and other prediction tools in the test dataset. The explainability of the model was enhanced by showing the feature importance plot to know the important variables in predicting the model’s outcome. The feature importance of the model was extracted based on three metrics of XGBoost algorithms: weight, cover, and gain.
Statistical analysis
All data were analyzed only in the dedicated server space provided by KNHIS according to the policy on preventing the leakage of public data. Categorical variables are expressed as numbers and percentages, while continuous variables are expressed as means and standard deviations. The receiver operating characteristic curve analysis was used to compare the predictive performances of the prediction model using machine learning techniques and other risk scores. The significance level of statistical analysis was considered p < 0.05. All statistical analyses and machine learning modeling were performed with the R statistical language (R version 3·5·1, R Foundation for Statistical Computing, Vienna, Austria).
Results
Patients
The baseline characteristics of the studied patients according to the postoperative 90-day mortality rate are shown in Table 1. Of the total 22,448 patients, 3225 (14.4%) died within 90 days. The mean age of the patient group who died within 90-day post-operatively (death group) was higher than the no-death group (82.2 years vs. 81.0). Moreover, a significantly lower proportion of females was observed in the death group (50.2% vs. 62.2%, p-value < 0.001). Both HFRS and OFRS showed that the death group has higher points than the no-death group (Table 1). Subgroup analysis revealed a higher proportion of the death group in all high risk operation groups except in low risk operation groups 1 and 2 (p-value < 0.001).
Prediction performance of the models
The predictive performance of each prediction model for the postoperative 90-day mortality rate was compared with each other by the AUROC graph (Fig. 2). The prediction model using the XGBOOST algorithm showed the highest prediction performance at 0.840 on AUROC, while OFRS and HFRS showed relatively low prediction performance (AUROC of OFRS: 0.607, AUROC of HFRS: 0.588).
Feature importance
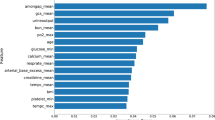
Figure 3 shows the feature importance of the XGBoost model. In addition to age, ICD-10 diagnostic codes A09 (Infectious gastroenteritis and colitis, unspecified), I95 (Hypotension), and L08 (Other local effects of skin and subcutaneous tissues) and operation code (O1502, Irrigation of empyema cavity) were shown as major factors affecting the postoperative 90-day mortality rate.
Feature of importance in the predictive model using machine learning method (XGBoost). XGBoost: Extreme Gradient Boosting, DX: Diagnostic code, OP: Operation code, E83: Disorders of mineral metabolism, L08: Other local infections of skin and subcutaneous tissue, R11: Nausea, and vomiting, O1502: Irrigation of empyema cavity, M81: Osteoporosis without current pathological fracture, M6730: Percutaneous gastrostomy, M15: Polyosteoarthritis, A04: Other bacterial intestinal infections, L89: Pressure ulcer, O1264: Operation of vocal cord paralysis, UX044: Temporomandibular joint arthrocentesis, W19: Unspecified fall, M6650: Percutaneous installation of inferior vena cava filter, O2004: Implantation of internal pulse generator by thoracotomy, G30: Alzheimer’s disease, M41L: Scoliosis, S32: Fracture of lumbar spine and pelvis, E16: Other disorders of pancreatic internal secretion
Discussion
In this study, the predictive model made by learning operation code and diagnostic code using a machine learning technique called XGBoost had a better predictive performance for postoperative 90-day mortality, one of the indicators for preoperative frailty, compared to risk scoring systems such as OFRS and HFRS developed through conventional regression analysis.
Despite many attempts to evaluate and predict frailty in older patients in the last decades, a few frailty evaluation tools could be applied to older patients undergoing emergency surgery [16, 21]. Since most older patients undergoing emergency surgery are vulnerable to surgical stress and are likely to develop postoperative complications, predicting or evaluating the preoperative frailty of these patients clinically is very important [22]. To evaluate the frailty risk, most previous studies required interviews with patients or specific measurements such as grip strength or gait speed [7,8,9,10,11,12,13,14]. However, older patients who come to the hospital for emergency surgery are often restricted in communication, and many of them are bed-ridden conditions. Therefore, previous methods of frailty evaluation cannot be applied in clinical practice.
The previous study predicted the clinical outcome by measuring the frailty of hospitalized patients using ICD10 diagnostic codes related to frailty [15]. This was called HFRS, and the patient’s frailty was measured using an ICD10 diagnostic code that can be automatically extracted from EMR for inpatients who visited the emergency room [15]. However, since HFRS is a model designed for inpatients, there were many limitations to apply to the patients undergoing surgery. Therefore, recently, a study suggested a frailty prediction model, which was called OFRS that applied to surgical patients by additionally applying surgical code information [16, 17]. OFRS is a model that evaluates preoperative frailty using only the patient’s age, diagnostic code, and surgical code information [16, 17]. Therefore, OFRS has the advantage of being able to predict preoperative frailty without specific measurements or patient interviews in older patients undergoing emergency surgery. In the previous study, OFRS had poor predictive performance for clinical outcomes [16]. Previous work developed predictive models based on regression models, that is difficult to put a lot of operation code as input [16]. To overcome these, surgical codes were grouped according to similar risks, divided into the eight risk groups, and modeled for analysis[16] by the three clinical experts as they reviewed each other [16]. This type of grouping is not based on data, but the subjective views and experiences of clinicians [16]. Therefore, these subjective parts may lead to poor predictive performance, and these problems should be solved in a data-based or reliable objective manner [16]. In this study, to improve this low predictive power, we tried to overcome these limitations by utilizing an artificial intelligence model that has recently been applied in various medical fields.
We used an artificial intelligence technique, a tree-based ensemble model called XGBoost [20]. Existing tree-based machine learning methods are more useful in the medical field because they are similar to the traditional regression models that are commonly known and easier to find the cause for the results [23, 24]. The ensemble model refers to a technique that improves the final prediction performance by generating multiple trees and combining prediction results [20]. These machine learning techniques can be very useful for many input variables or the low incidences of the response variables [25]. To use statistical methods of existing traditional methods for modeling, the input variables must be grouped or the data must be artificially altered. However, the machine learning method works using the input without grouping the variables or data deterioration. Therefore, the prediction model of the machine learning method based on data is more accurate [25].
Moreover, our study was not based on the data retrieved from a single institution, but from a national public dataset. Therefore, this study presented a more robust predictive model compared to previous models based on specific institutions’ data. Furthermore, our work was based on a more generalized dataset and will serve as a cornerstone for creating predictive models applicable in many healthcare environments.
The prediction model using XGBoost showed higher prediction performance than OFRS or HFRS. This confirms that the predictive performance of the machine learning method’s modeling, which learns data as it is, is superior to the statistical modeling of the existing traditional method when there are many input variables as in this study. Using a machine learning technique, modeling is easy because there is no need to group surgical codes or process data. It may also be useful for application in changing clinical environments to update the predictive models using additional data. However, “black boxes” of the artificial intelligence are existed as to how artificial intelligence techniques work and why these results come out, are unable to be explained. Therefore, many techniques have recently been developed to overcome these limitations [26,27,28] identifying the main factors and causes of the predictive model with the model’s feature importance [27, 28].
Our study has many limitations. First, since it is a model using the sample cohort dataset provided by KNHIS, it is expected to be a more robust predictive model unlike previous studies, but it could not be confirmed without external validation with other institutional data. In future studies, external validation is needed. The second is that no comparative assessment was included on the diverse algorithms using other machine learning techniques such as random forest and support vector machine or deep learning methods other than the XGBoost. Moreover, according to the public data policy, data analysis could only be performed within the designated server provided by KNHIS, and it was difficult to apply the latest updates and algorithms of the designated server. Consequently, we were unable to conduct additional application and comparative research on various algorithms. Additionally, due to these policy-related limitations, it is currently difficult to publicly share and apply the developed prediction model in clinical settings. In the near future, it is expected that changes in data-related policies will enable the application of prediction models using public data in actual clinical fields. Another limitation of our study was that the predictive power of various clinical outcomes such as long hospital stay, readmission, extended intensive care unit stay, and reoperation other than postoperative mortality were not analyzed. Future studies were necessary to compare the performance of the prediction model presented in this study with other risk scores for more diverse clinical outcomes.
Conclusion
In conclusion, by using machine learning techniques such as XGBoost to predict postoperative 90-day mortality, one of the indicators of preoperative frailty, using diagnostic and operation codes, the prediction performance improved over previous risks assessment models such as OFRS and HFRS. In the future, various artificial intelligence algorithms and external validation studies should be conducted.
Data Availability
All data regarding this study is available upon reasonable request to the corresponding author.
References
Chan SP, Ip KY, Irwin MG. Peri-operative optimisation of elderly and frail patients: a narrative review. Anaesthesia. 2019;74(Suppl 1):80–9.
Lim BG, Lee IO. Anesthetic management of geriatric patients. Korean J Anesthesiol. 2020;73(1):8–29.
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381(9868):752–62.
Partridge JS, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41(2):142–7.
Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146–156.
Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, Mitnitski A. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173(5):489–95.
Arjunan A, Peel NM, Hubbard RE. Gait Speed and Frailty Status in relation to adverse outcomes in Geriatric Rehabilitation. Arch Phys Med Rehabil. 2019;100(5):859–64.
Castell MV, Sanchez M, Julian R, Queipo R, Martin S, Otero A. Frailty prevalence and slow walking speed in persons age 65 and older: implications for primary care. BMC Fam Pract. 2013;14:86.
Choi JY, Kim KI, Choi Y, Ahn SH, Kang E, Oh HK, Kim DW, Kim EK, Yoon YS, Kang SB, et al. Comparison of multidimensional frailty score, grip strength, and gait speed in older surgical patients. J Cachexia Sarcopenia Muscle. 2020;11(2):432–40.
Dudzinska-Griszek J, Szuster K, Szewieczek J. Grip strength as a frailty diagnostic component in geriatric inpatients. Clin Interv Aging. 2017;12:1151–7.
Jung HW, Jang IY, Lee CK, Yu SS, Hwang JK, Jeon C, Lee YS, Lee E. Usual gait speed is associated with frailty status, institutionalization, and mortality in community-dwelling rural older adults: a longitudinal analysis of the Aging Study of Pyeongchang Rural Area. Clin Interv Aging. 2018;13:1079–89.
Reeve TEt, Ur R, Craven TE, Kaan JH, Goldman MP, Edwards MS, Hurie JB, Velazquez-Ramirez G, Corriere MA. Grip strength measurement for frailty assessment in patients with vascular disease and associations with comorbidity, cardiac risk, and sarcopenia. J Vasc Surg. 2018;67(5):1512–20.
Mitnitski AB, Mogilner AJ, Rockwood K. Accumulation of deficits as a proxy measure of aging. ScientificWorldJournal. 2001;1:323–36.
Rockwood K, Mitnitski A. Frailty defined by deficit accumulation and geriatric medicine defined by frailty. Clin Geriatr Med. 2011;27(1):17–26.
Gilbert T, Neuburger J, Kraindler J, Keeble E, Smith P, Ariti C, Arora S, Street A, Parker S, Roberts HC, et al. Development and validation of a hospital frailty risk score focusing on older people in acute care settings using electronic hospital records: an observational study. Lancet. 2018;391(10132):1775–82.
Lee SW, Nam JS, Kim YJ, Kim MJ, Choi JH, Lee EH, Joung KW, Choi IC. Predictive Model for the Assessment of Preoperative Frailty Risk in the Elderly.J Clin Med2021, 10(19).
Lee SW, Kim KS, Park SW, Kim J, Choi JH, Lee S, Joung KW, Choi IC. Application of the New Preoperative Frailty Risk Score in Elderly Patients Undergoing Emergency Surgery.Gerontology2022:1–9.
Luo W, Phung D, Tran T, Gupta S, Rana S, Karmakar C, Shilton A, Yearwood J, Dimitrova N, Ho TB, et al. Guidelines for developing and reporting machine learning predictive models in Biomedical Research: a multidisciplinary view. J Med Internet Res. 2016;18(12):e323.
Kim YI, Kim YY, Yoon JL, Won CW, Ha S, Cho KD, Park BR, Bae S, Lee EJ, Park SY, et al. Cohort Profile: National health insurance service-senior (NHIS-senior) cohort in Korea. BMJ Open. 2019;9(7):e024344.
Chen TQ, Guestrin C. XGBoost: A Scalable Tree Boosting System. Kdd’16: Proceedings of the 22nd Acm Sigkdd International Conference on Knowledge Discovery and Data Mining. 2016:785–794.
Costa G, Bersigotti L, Massa G, Lepre L, Fransvea P, Lucarini A, Mercantini P, Balducci G, Sganga G, Crucitti A, et al. The emergency surgery Frailty Index (EmSFI): development and internal validation of a novel simple bedside risk score for elderly patients undergoing emergency surgery. Aging Clin Exp Res. 2021;33(8):2191–201.
Joseph B, Zangbar B, Pandit V, Fain M, Mohler MJ, Kulvatunyou N, Jokar TO, O’Keeffe T, Friese RS, Rhee P. Emergency general surgery in the Elderly: Too Old or too frail? J Am Coll Surg. 2016;222(5):805–13.
Parikh RB, Manz C, Chivers C, Regli SH, Braun J, Draugelis ME, Schuchter LM, Shulman LN, Navathe AS, Patel MS et al. Machine Learning Approaches to Predict 6-Month Mortality Among Patients With Cancer.Jama Netw Open2019, 2(10).
Wong A, Young AT, Liang AS, Gonzales R, Douglas VC, Hadley D. Development and validation of an Electronic Health Record-Based machine learning model to Estimate Delirium Risk in newly hospitalized patients without known cognitive impairment. Jama Netw Open. 2018;1(4):e181018.
Zhang ZH, Zhao YM, Canes A, Steinberg D, Lyashevska O. Collab AB-DCT: Predictive analytics with gradient boosting in clinical medicine.Ann Transl Med2019, 7(7).
Watson DS, Krutzinna J, Bruce IN, Griffiths CEM, McInnes IB, Barnes MR, Floridi L. Clinical applications of machine learning algorithms: beyond the black box.Bmj-Brit Med J2019,364.
Stojic A, Stanic N, Vukovic G, Stanisic S, Perisic M, Sostaric A, Lazic L. Explainable extreme gradient boosting tree-based prediction of toluene, ethylbenzene and xylene wet deposition. Sci Total Environ. 2019;653:140–7.
Lundberg SM, Lee SI. A Unified Approach to Interpreting Model Predictions.Adv Neur In2017,30.
Acknowledgements
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
Sang-Wook Lee participated in the design of the study, data statistical analysis, data interpretation, and manuscript drafting. Eun-Ho Lee and In-Cheol Choi participated in the design of the study and manuscript revision. All authors read and approved the final manuscript. The corresponding author had full access to data used in the study and had final responsibility to submit for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Asan Medical Center, Seoul, Korea (approval number 2021 − 0282, approval date April 08, 2021). Written informed consent was exempted by the Institutional Review Board of Asan Medical Center, because of the retrospective nature of study.
Consent for publication
Not applicable.
Competing interests
The authors have no competing interests to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12877_2023_3969_MOESM1_ESM.docx
Supplementary Material 1. Table S1. Scoring system for prediction of 90 day mortality. Table S2. Operation Groups according to surgical risk.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Lee, SW., Lee, EH. & Choi, IC. An ensemble machine learning approach to predict postoperative mortality in older patients undergoing emergency surgery. BMC Geriatr 23, 262 (2023). https://doi.org/10.1186/s12877-023-03969-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12877-023-03969-0