Abstract
Background
Inactive physical behavior among the elderly is one risk factor for cardiovascular disease, immobility and increased all-cause mortality. We aimed to answer the question whether or not circulating and skeletal muscle biomarkers are differentially expressed depending on fitness status in a group of elderly individuals.
Methods
Twenty-eight elderly individuals (73.36 ± 5.46 years) participated in this exploratory study after participating as part of the multinational SITLESS-clinical trial (implementation of self-management and exercise programs over 16 weeks). A cardiopulmonary exercise test (CPX) and resting skeletal muscle biopsy were performed to determine individual physiological performance capacity. Participants were categorized into a high physical fitness group (HPF) and a low physical fitness group (LPF) depending on peak oxygen uptake (VO2peak). Serum blood samples were taken before (pre) and after (post) CPX and were examined regarding serum BDNF, HSP70, Kynurenine, Irisin and Il-6 concentrations. Skeletal muscle tissue was analyzed by silver staining to determine the myosin heavy chain (MyHC) composition and selected genes by qRT-PCR.
Results
HPF showed lower body weight and body fat, while skeletal muscle mass and oxygen uptake at the first ventilatory threshold (VO2T1) did not differ between groups. There were positive associations between VO2peak and VO2VT1 in HPF and LPF. MyHC isoform quantification revealed no differences between groups. qRT-PCR showed higher expression of BDNF and BRCA1 in LPF skeletal muscle while there were no differences in other examined genes regarding energy metabolism. Basal serum concentrations of Irisin were higher in HPF compared to LPF with a trend towards higher values in BDNF and HSP70 in HPF. Increases in Il-6 in both groups were observed post.
Conclusions
Although no association between muscle composition/VO2peak with fitness status in older people was detected, higher basal Irisin serum levels in HPF revealed slightly beneficial molecular serum and muscle adaptations.
Trial registration
ClinicalTrials.gov, NCT02629666. Registered 19 November 2015.
Similar content being viewed by others
Background
Sarcopenia is defined as the gradual loss of skeletal muscle mass, quality and strength during ageing and/or immobility [1]. Older people have a high prevalence of sarcopenia and altered skeletal muscle metabolism as a consequence of increased inactive lifestyle [2, 3]. Harvey et al. (2013) examined physical activity of older people in seven countries and found that approximately 67% of the age group > 60 years showed sedentary behavior of 8.5 h per day [4]. A systematic review of the amount of sedentary behavior in older people between 1981 and 2014 showed even higher values (average sedentary time of 9.4 h/day), equating to 65–80% of older people’s waking day [5]. Sarcopenia and increasing immobility is associated with a functional decline such as impaired gait speed, increased fall and hospitalization risk as well as a higher mortality rate [2, 6, 7]. Mechanistically, several pathways have been described. In addition to a downregulation of insulin sensitivity and cellular turnover, declining enzymatic activity in skeletal muscle [8, 9] has also been shown. Sarcopenic obesity, the combination of obesity and low muscle mass, is a serious health problem as it enhances the pro-inflammatory and adipokine response, promotes insulin resistance and is associated with increased strength loss and mortality [10].
This, however, can effectively be delayed by physical exercise [8, 11]. Several studies showed the beneficial effect of physical exercise in slowing down the loss of skeletal muscle mass and to increase life expectancy throughout different age groups [11, 12]. The beneficial effects of exercise training can be exerted at any time in life, regardless of the starting point [13, 14]. Exercise can also stimulate the expression of neurotrophic and angiogenic factors, reduce inflammation and thereby contributes to brain health and also reduce metabolic risk factors [15, 16].
The SITLESS project, a multicenter study conducted in four European countries (Spain, Denmark, Northern Ireland, Germany), addressed physical activity and sedentary behavior of community-dwelling elderly European citizens with a 16-week long training intervention [17]. In short, exercise referral schemes and self-management strategies were used to reduce sedentary behavior and improve health, quality of life and physical function, as well as psychosocial outcomes in the long term. In a sub-study, we aimed to test the hypothesis that fitter older individuals more likely have lower inflammation, improved cellular regulation processes involved in regeneration, metabolism and genomic health and a different muscle structure than unfit age-matched controls. Skeletal muscle biopsy samples at rest and blood samples before and after an acute exercise test were taken from fit and unfit study participants to analyze ageing and performance markers on the RNA and protein level for possible future diagnostic applications.
Materials and methods
A subgroup of the original SITLESS clinical trial [17] with residence in Germany was recruited to participate in this exploratory study and attended two additional visits at the Division of Sports- and Rehabilitation Medicine at Ulm University (Germany) after the end of the 16-week long SITLESS intervention study [18, 19]. Participants (16 female, 12 male) were divided into two groups depending on their peak oxygen consumption (VO2peak/kg): 1) Individuals with low physical fitness, (LPF, VO2peak/kg female ≤ 17 ml/min/kg, male ≤ 23 ml/min/kg); and 2) individuals with high physical fitness (HPF, VO2peak/kg female ≥ 18 ml/min/kg, male ≥ 24 ml/min/kg). As VO2peak represents the aerobic fitness status at higher intensities [20], we calculated the median for each woman and men and dichotomized the groups in HPF and LPF according to the higher and lower percentiles around these values. A graphical representation of the study design is provided in Fig. 1.
Schematic overview of the study design. After SITLESS, participants were asked to participate in this sub-study. A muscle biopsy was conducted at least one week before the CPX. Muscle samples were used for Myosin-Heavy chain (MyH) isoform composition and gene expression analysis. Blood was sampled before (pre) and directly after the CPX (post), and serum protein expression determined. Participants were classified according to the VO2peak measured during CPX by spiroergometry into HPF and LPF. (SITLESS: multinational 16-week long-intervention study to promote physical activity in the elderly population; CPX: cardiopulmonary exercise test; VO2peak: peak oxygen consumption; HPF: high physical fitness group; LPF: low physical fitness group)
Inclusion criteria were: aged ≥65 years; male or female; able to perform the physical exercise training and to walk a distance of at least 400 m by themselves; and previously physically inactive (< 150 min moderate exercise or < 75 min vigorous exercise per week). Dementia or other malignant progressive cardiovascular or oncologic diseases led to study exclusion. Detailed exclusion criteria are listed in Additional File 1.
This pilot study was approved by the Ethical Review Committee of Ulm University (No. 354/15). It conforms with the Declaration of Helsinki and all participants gave written informed consent to participate prior to taking part in this study.
Body composition and performance testing
Body composition was determined by body-impedance-analysis (BIA, InBody 770, Eschborn, Germany). Performance and aerobic capacity were determined by a spiroergometric cardiopulmonary exercise test (CPX). An incremental test on a bicycle ergometer (Lode Excalibur Sport, Groningen, The Netherlands) was performed to measure the first ventilatory threshold (VO2VT1) as well as peak oxygen consumption (VO2peak). After 2 min of unloaded pedaling, the incremental test started at 25 Watt (W) with a continual increase of 15 W/min. The test was aborted if an individual reached physical exhaustion or medical indications prevailed. A dynamic mixing chamber was used to determine breathing gases (Cortex MetaMax 3X, Leipzig, Germany). Calculation of the ventilatory thresholds were conducted by the V-slope method of Schneider et al. [21].
Blood analysis
Twenty milliliter of blood was drawn directly before (pre) and after (post) the CPX from the antecubital vein to determine adaptations of acute exercise on the molecular level. Blood was sampled in serum vacutainers, coagulated for at least 30 min in an upright position and centrifuged for 10 min at 3800 rpm. Serum was stored at − 80 °C until further analysis.
Concentrations of brain-derived neurotrophic factor (BDNF, R&D Systems, Minneapolis, USA), Interleukin 6 (Il-6, R&D Systems, Minneapolis, USA), Irisin (Phoenix Pharmaceuticals, California, USA) and Heat shock protein 70 (HSP70, Enzo Life Sciences GmbH, Lörrach, Germany) were analyzed by ELISAs according to the respective manufacturer’s instruction.
Kynurenine was determined by the following protocol: 1000 μM of L-kynurenine sulfate salt (Sigma-Aldrich, Munich, Germany) was diluted 1:20 with 15% Trichloric acid (TCA) to a final concentration of 50 μM. This stock solution was further diluted with TCA to 25 μM, 12.5 μM, 6.25 μM, 3.125 μM, 1.56 μM and 0.78 μM for standard curve determination. Aqua dest. Was used as blank solution. One fifty microliter serum sample was mixed with 100 μl of 30% TCA and centrifuged at 20000 g for 10 min and 4 °C. One fifty microliter of standard solutions and blank as well as 150 μl of the supernatant were pipetted into a 96 well plate in duplicates and incubated for 15 min at 65 °C. 0.15 g of diaminobenzoic acid (DABA) was diluted in 10 ml 100% acetic acid and 150 μl of this final solution was mixed with samples for 5 min at RT. Fluorescence was measured with a plate reader and the concentration of samples calculated by the slope of the linear regression line of the optical density from 492 to 620 nm.
Muscle biopsies
The skeletal muscle biopsy took place 1-7 days before the CPX using the Bergström technique according to published protocols [22]. In detail, participants were encouraged to perform their regular daily behavior after the end of SITLESS. The day before and on the day of the muscle biopsy, all participants were asked to refrain from strenuous physical exercise. A careful medical examination including medical history, medication and ECG was obtained and all examinations were performed between 1:30–3 pm to minimize inter-individual effects. When the participant was suitable for biopsy, skeletal muscle samples will be obtained under local anesthesia from the Musculus vastus lateralis (femur) of the dominant side of the subjects (or the healthy leg, respectively) approximately 20 cm above the knee. The Bergström-Needle with 4 mm outer diameter allows attaining of 100–200 mg muscle tissue.
For RNA isolation, muscle tissue was incubated for 24 h with RNAlater (QIAGEN GmbH, Hilden, Germany) at 4°C and then stored in cryotubes at − 80°C until further analysis. For protein examination, muscle tissue was immediately cryopreserved with liquid nitrogen and stored at − 80°C until further analysis.
The biopsy site was closed with suture tape and slight compression was applied using a flexible bandage. Participants could walk and train at the same day and were fully able to use their legs.
Protein preparation and silver staining
To determine the respective protein myosin heavy chain (MHC) isoform abundance, muscle samples were incubated with 250 μl Pierce™ RIPA buffer (Thermo Fisher Scientific, MA, USA), mixed with cOmplete™ Mini EDTA-free Protease Inhibitor Cocktail (Roche, Basel, Switzerland), homogenized and incubated for 10 min on ice in Pierce™ RIPA buffer to a final concentration of 5 μg/μl muscle protein. After centrifugation (4°C, 14000 rpm, 20 min), the supernatant was used for SDS-PAGE according to the manufacturer’s instructions (Mini-Protean TGX Gels 4–20%, BIO-RAD, Berkeley, USA). Afterwards, silver staining was performed with the Silver Stain kit (BIO-RAD, #161–0443, Berkeley, USA) according to the manufacturer’s instructions. The relative protein abundance was analyzed with Image J 1.51 (ImageJ, NIH, Maryland, USA).
RT-PCR
Muscle biopsy samples of HPF and LPF were examined regarding gene expression of genes involved in aerobic metabolism, anti-oxidative response, aging and tumor development. Targets were PGC1a, G6PD, UCP3, VEGFa, NR4A1, NR4A2, SCO2, SOD2, IL16, BDNF, BRCA1 and TP53. RNA was quantified with spectrophotometry (NanoDrop 2000c, Thermo Scientific, Massachusetts, USA) and transcribed to cDNA with the QantiTect® Reverse Transcription Kit (QIAGEN GmbH, Hilden, Germany) according to the manufacturer’s instructions. cDNA was used to determine the expression with Real Time qPCR (RT-qPCR) analogous to established protocols [23] with GAPDH as established reference gene for endurance exercise [23, 24].
Statistical analysis
Statistical analysis was performed using GraphPad PRISM (Version 9, La Jolla, USA). Data were tested for Gaussian distribution. Normally distributed data of blood variables were analyzed for inter- and intra-group differences with a 2-way ANOVA followed by Tukey Post-hoc test. Otherwise, a mixed model analysis was used followed by Dunn’s Post-hoc test. To determine associations between skeletal muscle mass (SMM), age, VO2peak and VO2T1, Spearman correlation analyses followed by two-tailored t-test were performed to determine statistical significance. Descriptive statistics of the data are presented as mean ± standard deviation. Statistical differences were considered to be significant for values of p ≤ 0.05.
Results
Physical performance measures and anthropometry
The study population consisted of 28 participants (14 HPF, 14 LPF). A detailed anthropometric description is provided in Table 1.
HPF participants had lower body mass (p = 0.0119), BMI (p = 0.0026) and body fat mass (p < 0.001) (Table 1) as well as lower percentage body fat (p < 0.001) and higher VO2peak (p < 0.001) compared to LPF (Fig. 2). Examination of the respiratory quotient (RQ) at maximal exertion showed a mean of 1.10 +/− 0.09, which is clear sign of objective exhaustion in all subjects. Gender analysis revealed no significant differences in the fat parameters or VO2peak between female and male participants. Only VO2T1 was significantly higher in males vs. females (p = 0.0003) (Additional File 1). As the results were based on the fitness level determined by VO2peak, no gender effect is assumed to bias the data.
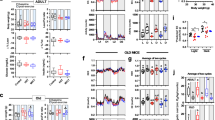
Differences in aerobic capacity and anthropometry in HPF and LPF. A HPF showed significantly higher VO2peak/kg values compared with LPF (p < 0.001) with no difference in first ventilatory threshold VO2VT1. B Skeletal muscle mass (SSM) was statistically not different between the groups while body fat was higher in LPF (p < 0.001)
Significant positive correlation between VO2peak and VO2VT1 in HPF and LPF
Although there was no correlation between SMM, age, VO2peak and VO2T1 (Fig. 3), a positive correlation between VO2peak and VO2VT1 in HPF (p = 0.009) and LPF (p = 0.004) was observed (Additional File 1). A separate analysis showed no effect of sex on the tested parameters. In female participants, there were negative correlations seen between body fat percentage and VO2peak (p = 0.003) and body fat mass and age (0.022) (Additional File 1).
RT-PCR revealed no differences except for BDNF and BRCA1
Analysis of selected genes involved in aerobic metabolism, anti-oxidative response, ageing, and tumor development showed no difference in the expression between HPF and LPF, except for BDNF and BRCA1 (Additional File 2). Except for a limited number of PCR-results for BDNF (HPF n = 5, LPF n = 6), all targets could be reliable detected.
Myosin heavy chain (MyHC) composition did not differ between HPF (n = 13) and LPF (n = 12)
Both groups showed highest fiber type percentage in MyHC IId/x fibers, followed by MyHC IIa and MyHC I fibers (Fig. 4).
Serum protein quantification of selected factors revealed significant basal differences in Irisin and adaptations in Il-6 response in both groups
Serum samples were taken before (pre) and after (post) an acute exercise test on a bicycle ergometer for analysis of HSP70, BDNF, Irisin, Il-6 and kynurenine protein concentration (Fig. 5). Subsequent ELISA analysis showed significant differences between HPF and LPF in basal values of irisin (p = 0.0195) (Fig. 5C). There was an increase in HPF (p = 0.0124) and LPF (p = 0.0146) after exercise in Il-6 (Fig. 5D). Kynurenine (p = 0.0927) and HSP70 (p = 0.069) only showed post a tendency for inter-group changes (Fig. 5E).
Changes in serum concentrations of molecules involved in ageing, muscle damage and neurotrophic adaptation. HPF and LPF were examined after an acute exercise test (pre vs. post). A HSP70 showed non-significant higher basal values in HPF (p = 0.069) compared to LPF. B Plasma levels of BDNF were non-significantly higher in HPF pre and post. C Irisin concentrations were pre higher in HPF compared to LPF, but this difference was blunted post. D Serum concentration of inflammation marker Il-6 increased post in HPF and LPF. E Kynurenine did not differ in both groups, but due to an increase in LPF and a decrease in HPF post a tendency for different adaptations (p = 0.0927) was observed
Discussion
In this pilot study we found that although maximal oxygen uptake and body composition of fitter older adults were higher compared to unfit peers, most of the basal gene expression related to ageing, performance, and associated metabolism as well as fiber type composition did not significantly differ between HPF and LPF. On the other hand, slight differences were observed regarding protein concentrations before and after an acute exercise test.
Performance and body/muscle composition
Several studies have assessed sedentary behavior and physical inactivity in older adults taking into account differing nationalities, social/educational level and sex [4, 25,26,27], and uniformly observed a tendency for increasing inactive physical behavior among the elderly population. Although the popular allusion that “sitting is the new smoking” is scientifically not sound [28, 29], an inactive lifestyle, especially in older people, increases the risk for cardiovascular diseases, loss of skeletal muscle mass and malfunction as well as all-cause mortality [7, 13, 28].
The combination of increasing age and loss of skeletal muscle mass and/or functionality is associated with decreased aerobic capacity, shown by decreased VO2max [30]. The examination of our fitter HPF and unfit LPF showed distinct differences in performance and body composition. Especially the workload at higher intensities could be better maintained by HPF, seen by higher VO2peak/kg, whereas there seemed no difference between groups during moderate intensities at VO2VT1. Maintaining a higher VO2peak during life can reduce the risk of multiple comorbidities, all-cause mortality, and loss of independence during aging [31].
The aging-associated capillarization and aerobic enzyme activity decline is equally slowed down by lifelong exercise, which can have a direct beneficial influence on cardiovascular health in women and men [32]. In our mostly inactive LPF participants, the body fat percentage was significantly higher. It is well known that a high body fat percentage contributes to increased risk of developing cardiovascular diseases, diabetes and immobility [33]. On the other hand, several studies have shown that weight loss intervention with a combination of diet and exercise improves muscle strength and muscle quality in addition to fat loss among older obese people and consequently attenuates sarcopenic symptoms and pro-inflammatory responses [10]. On the muscular level, increasing age and immobility lead to alterations in the MyHC isoform distribution, fiber type composition and fiber activation [2, 12, 34]. Interestingly, we did not detect any correlation between skeletal muscle mass, VO2peak and age in both groups and, what is more, skeletal muscle mass as well as MyHC composition were not different between HPF and LPF.
Fiber size was not assessed, which may have given further insights into muscle adaptations and decline as muscle strength is a strong predictor of severe mobility limitation, slow gait speed, increased fall risk, risk of hospitalization, and high mortality rate [2, 34,35,36]. For example, Miljkovic et al. showed that elderly adults with low muscle strength have a 2.6-fold greater risk of severe mobility limitation, 4.3-fold greater risk for slow gait speed, and 2.1-fold greater risk of mortality compared to elderly adults with high muscle strength [2].
Skeletal muscle gene expression
Beside adaptations in fiber type morphology, regular exercise can slow down age-related alterations on the molecular skeletal muscle level with beneficial changes in oxidative enzyme activity, DNA repair and skeletal neurotrophic factors like BDNF [11, 12, 37]. Skeletal muscular expression analysis of genes involved in gene stability, energy metabolism and mitochondrial activity (PGC1a, G6PD, UCP3, VEGFa, NR4A1, NR4A2, SCO2, SOD2, IL16, and TP53) revealed no difference in HPF and LPF regarding energy metabolism or mitochondrial activity. Significant differences could only be observed in BDNF and BRCA1 levels with higher basal expression in LPF. The latter contrasts with previous studies, which showed increased expression in BRCA1, responsible for genomic stability, with regular exercise [38, 39]. We observed a high intra-group variability in the expression profiles which may contribute to this result. A further possible explanation may be the known blunted response on the protein level in older people [9].
Serum protein adaptations
Although a higher basal BDNF skeletal muscle mRNA expression was seen for LPF, serum protein concentrations showed non-significant elevated levels in HPF pre and post exercise compared to LPF. Until now, skeletal muscle expression of BDNF in older people after exercise is incompletely understood and has been rarely examined: accumulating evidence on circulating protein levels seem to hint to transiently up-regulation of BDNF concentration after acute exercise, but with no long-term changes with regular training [40,41,42]. Therefore, BDNF may not be differently regulated depending on physical fitness status in older individuals and similar levels might be expected.
Beside BDNF, Küster et al. evaluated Irisin and the kynurenine pathway as novel blood-based biomarkers for physical and cognitive performance [42]. Ten weeks of cognitive and physical exercise training in older people were associated with potentially beneficial decreased kynurenine metabolites, whereas no significant changes in Irisin levels could be observed, which is in contrast to other studies [43]. The main function of the myokine Irisin is thermogenesis regulation by hormonal inducement of increasing energy expenditure, promoting weight loss, and decreasing insulin resistance [44]. Irisin is also a potential circulating mediator of exercise which benefits brain health and neuroprotection, as it was shown that it is secreted upon exercise in mice and humans, where it apparently promotes BDNF release [16]. Furthermore, the PGC-1α-FNDC5/Irisin-BDNF-signaling pathway has been proposed for resistance exercise that increases the expression of FNDC5/Irisin and in turn induces BNDF with subsequently beneficial adaptations in the transcription and transport of mRNA along dendrites, growth, differentiation, and survival of neurons [44].
In our study, significantly higher serum baseline values of circulating Irisin in HPF compared to LPF were found. Circulating Irisin levels were shown to be negatively correlated with cardiometabolic risk in sedentary participants, with an active lifestyle positively increasing Irisin levels [15]. This is in line with higher VO2peak data of HPF, which implies a higher general fitness of this sub-group. For serum kynurenine, only a trend for decreased concentrations in HPF and increased concentration in LPF was observed post. This might suggest better coping capacity with the physical exercise test, as elevated kynurenine levels were associated during and after stressful cognitive and physical periods [42].
Serum HSP70 concentration did not significantly differ between HPF and LPF. The versatile heat shock protein HSP70 serves as a molecular chaperone involved in many cellular processes [45]. It has a profound impact on protein turnover, energy metabolism, muscle function/regeneration, hypertrophy and adaptation, while sarcopenia leads to decreased HSP70 content due to muscle loss [46]. Furthermore, a blunted expression of HSP70 in skeletal muscle in response to chronic exercise during aging was observed [9, 46], which may support our observation of unchanged HSP70 response in both HPF and LPF.
The proinflammatory cytokine Il-6 is induced after stressful events like intensive exercise, but has the additional function as an anti-inflammatory myokine [47]. It is one of the key molecules which controls the catabolic effect in myoblasts through inhibition of myogenesis and protein synthesis [48]. Higher plasma levels of Il-6 are predominately found in older people and were associated with lower muscle mass and muscle strength [48], which may be driven by the chronic low-grade inflammatory status and contributes to muscular functional impairment [8]. Il-6 serum basal levels of HPF and LPF were not significantly different and both groups showed increased values after the exercise test. This observation indicates a similar inflammation process regardless of physical fitness status, which may modulate the so called “inflammageing” in older people [8]. A third sample time point after the exercise test, e.g. 1 or 2 h later, may have given further insights in the long-term process of the Il-6 regulation. Short-term increases of inflammatory cytokines are common responses to exercise, whereas prolonged increases have harmful consequences.
Limitations
In this pilot-study, physically fit and unfit older men and women performed an acute exercise test with pre and post blood sampling, but a muscle biopsy sample was only taken at rest and so restricted to one time point. The relatively small participant number and muscle sample amount limits to a certain extent the genetic profiling, muscle protein analysis possibility and thus definitive conclusions thereof due to high interindividual expression. Nevertheless, the examined rare muscle biopsy samples in this susceptible older population give valuable insights into structural and adaptive processes in the ageing musculature.
Conclusions
Valenzuela et al. stated that “The sooner the better, but never too late” in their seminal review regarding exercise adaptations and health effects in the older population [14]. Serum molecular adaptations in older individuals to an acute exercise test seem to depend on fitness level. Although we did not detect a significant association between basal muscle parameters like SSM or fiber type composition and VO2peak/kg with fitness status, higher basal levels of Irisin in HPF revealed slightly beneficial long-term adaptations due to higher fitness level but most adaptations may be blunted during ageing. A significant lower inflammation after the acute test or a different muscle composition of fit- compared to unfit age-matched subjects could not be detected, suggesting that probably a still higher fitness status than our previously unactive participants has to be maintained to gain beneficial long-term adaptations in the older population.
Availability of data and materials
All pseudonymized data which are not included in the additional files or the manuscript are available on reasonable request from the corresponding author.
Abbreviations
- BDNF:
-
Brain-derived neurotrophic factor
- BRCA1:
-
Breast cancer gene 1
- BMI:
-
Body mass index
- CPX:
-
Cardiopulmonary exercise test
- HPF:
-
High physical fitness group
- HSP70:
-
Heat-shock protein 70
- Il-6:
-
Interleukin 6
- LPF:
-
Low physical fitness group
- MyHC:
-
Myosin heavy chain
- RQ:
-
Respiratory quotient
- SMM:
-
Skeletal muscle mass
- VO2peak:
-
Peak oxygen uptake
- VO2T1 :
-
Oxygen uptake at the first ventilatory threshold
References
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16–31. https://doi.org/10.1093/ageing/afy169.
Miljkovic N, Lim J-Y, Miljkovic I, Frontera WR. Aging of skeletal muscle fibers. Ann Rehabil Med. 2015;39:155–62. https://doi.org/10.5535/arm.2015.39.2.155.
Smith L, Tully M, Jacob L, Blackburn N, Adlakha D, Caserotti P, et al. The association between sedentary behavior and sarcopenia among adults aged ≥65 years in low- and middle-income countries. Int J Environ Res Public Health. 2020;17:1708. https://doi.org/10.3390/ijerph17051708.
Harvey JA, Chastin SFM, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645–61. https://doi.org/10.3390/ijerph10126645.
Harvey JA, Chastin SFM, Skelton DA. How sedentary are older people? A systematic review of the amount of sedentary behavior. J Aging Phys Act. 2015;23:471–87. https://doi.org/10.1123/japa.2014-0164.
Nilwik R, Snijders T, Leenders M, Groen BBL, van Kranenburg J, Verdijk LB, et al. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp Gerontol. 2013;48:492–8. https://doi.org/10.1016/j.exger.2013.02.012.
Wilmot EG, Edwardson CL, Achana FA, Davies MJ, Gorely T, Gray LJ, et al. Sedentary time in adults and the association with diabetes, cardiovascular disease and death: systematic review and meta-analysis. Diabetologia. 2012;55:2895–905. https://doi.org/10.1007/s00125-012-2677-z.
Crescioli C. Targeting age-dependent functional and metabolic decline of human skeletal muscle: the Geroprotective role of exercise, Myokine IL-6, and vitamin D. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21031010.
Kayani AC, Morton JP, McArdle A. The exercise-induced stress response in skeletal muscle: failure during aging. Appl Physiol Nutr Metab. 2008;33:1033–41. https://doi.org/10.1139/H08-089.
Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. Sarcopenic obesity: definition, cause and consequences. Curr Opin Clin Nutr Metab Care. 2008;11:693–700. https://doi.org/10.1097/MCO.0b013e328312c37d.
Tarpenning KM, Hamilton-Wessler M, Wiswell RA, Hawkins SA. Endurance training delays age of decline in leg strength and muscle morphology. Med Sci Sports Exerc. 2004;36:74–8. https://doi.org/10.1249/01.MSS.0000106179.73735.A6.
Grimby G. Muscle performance and structure in the elderly as studied cross-sectionally and longitudinally. J Gerontol A Biol Sci Med Sci. 1995;50 Spec No:17–22. https://doi.org/10.1093/gerona/50a.special_issue.17.
Blair SN, Kohl HW, Paffenbarger RS, Clark DG, Cooper KH, Gibbons LW. Physical fitness and all-cause mortality. A prospective study of healthy men and women. JAMA. 1989;262:2395–401. https://doi.org/10.1001/jama.262.17.2395.
Valenzuela PL, Castillo-García A, Morales JS, Izquierdo M, Serra-Rexach JA, Santos-Lozano A, et al. Physical exercise in the oldest old. Compr Physiol. 2019;9:1281–304. https://doi.org/10.1002/cphy.c190002.
Moreno M, Moreno-Navarrete JM, Serrano M, Ortega F, Delgado E, Sanchez-Ragnarsson C, et al. Circulating irisin levels are positively associated with metabolic risk factors in sedentary subjects. PLoS One. 2015;10:e0124100. https://doi.org/10.1371/journal.pone.0124100.
Tari AR, Norevik CS, Scrimgeour NR, Kobro-Flatmoen A, Storm-Mathisen J, Bergersen LH, et al. Are the neuroprotective effects of exercise training systemically mediated? Prog Cardiovasc Dis. 2019;62:94–101. https://doi.org/10.1016/j.pcad.2019.02.003.
Giné-Garriga M, Coll-Planas L, Guerra M, Domingo À, Roqué M, Caserotti P, et al. The SITLESS project: exercise referral schemes enhanced by self-management strategies to battle sedentary behaviour in older adults: study protocol for a randomised controlled trial. Trials. 2017;18:221. https://doi.org/10.1186/s13063-017-1956-x.
Coll-Planas L, Blancafort Alias S, Tully M, Caserotti P, Giné-Garriga M, Blackburn N, et al. Exercise referral schemes enhanced by self-management strategies to reduce sedentary behaviour and increase physical activity among community-dwelling older adults from four European countries: protocol for the process evaluation of the SITLESS randomised controlled trial. BMJ Open. 2019;9:e027073. https://doi.org/10.1136/bmjopen-2018-027073.
McMullan II, Bunting BP, Blackburn NE, Wilson JJ, Deidda M, Caserotti P, et al. The mediating role of self-regulation and self-efficacy on physical activity change in community-dwelling older adults (≥65 years): an experimental cross-lagged analysis using data from SITLESS. J Aging Phys Act. 2021:1–10. https://doi.org/10.1123/japa.2020-0322.
Posner JD, Gorman KM, Klein HS, Cline CJ. Ventilatory threshold: measurement and variation with age. J Appl Physiol. 1985;1987(63):1519–25. https://doi.org/10.1152/jappl.1987.63.4.1519.
Schneider DA, Phillips SE, Stoffolano S. The simplified V-slope method of detecting the gas exchange threshold. Med Sci Sports Exerc. 1993;25:1180–4.
Shanely RA, Zwetsloot KA, Triplett NT, Meaney MP, Farris GE, Nieman DC. Human skeletal muscle biopsy procedures using the modified Bergström technique. J Vis Exp. 2014;51812. https://doi.org/10.3791/51812.
Mahoney D, Carey K, Fu H-H, Snow R, Cameron-Smith D, Parise G, et al. Real-time RT-PCR analysis of housekeeping genes in human skeletal muscle following acute exercise. Physiol Genomics. 2004;18:226–31. https://doi.org/10.1152/physiolgenomics.00067.2004.
Jemiolo B, Trappe S. Single muscle fiber gene expression in human skeletal muscle: validation of internal control with exercise. Biochem Biophys Res Commun. 2004;320:1043–50. https://doi.org/10.1016/j.bbrc.2004.05.223.
Loyen A, Clarke-Cornwell AM, Anderssen SA, Hagströmer M, Sardinha LB, Sundquist K, et al. Sedentary time and physical activity surveillance through accelerometer pooling in four European countries. Sports Med. 2017;47:1421–35. https://doi.org/10.1007/s40279-016-0658-y.
Loyen A, van der Ploeg HP, Bauman A, Brug J, Lakerveld J. European sitting championship: prevalence and correlates of self-reported sitting time in the 28 European Union member states. PLoS One. 2016;11:e0149320. https://doi.org/10.1371/journal.pone.0149320.
Jelsma JGM, Gale J, Loyen A, van Nassau F, Bauman A, van der Ploeg HP. Time trends between 2002 and 2017 in correlates of self-reported sitting time in European adults. PLoS One. 2019;14:e0225228. https://doi.org/10.1371/journal.pone.0225228.
Baddeley B, Sornalingam S, Cooper M. Sitting is the new smoking: where do we stand? Br J Gen Pract. 2016;66:258. https://doi.org/10.3399/bjgp16X685009.
Vallance JK, Gardiner PA, Lynch BM, D'Silva A, Boyle T, Taylor LM, et al. Evaluating the evidence on sitting, smoking, and health: is sitting really the new smoking? Am J Public Health. 2018;108:1478–82. https://doi.org/10.2105/AJPH.2018.304649.
Fleg JL, Lakatta EG. Role of muscle loss in the age-associated reduction in VO2 max. J Appl Physiol. 1985;1988(65):1147–51. https://doi.org/10.1152/jappl.1988.65.3.1147.
Ross R, Blair SN, Arena R, Church TS, Després J-P, Franklin BA, et al. Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653–99. https://doi.org/10.1161/CIR.0000000000000461.
Gries KJ, Raue U, Perkins RK, Lavin KM, Overstreet BS, D'Acquisto LJ, et al. Cardiovascular and skeletal muscle health with lifelong exercise. J Appl Physiol. 1985;2018(125):1636–45. https://doi.org/10.1152/japplphysiol.00174.2018.
Wilson PWF, Kannel WB. Obesity, diabetes, and risk of cardiovascular disease in the elderly. Am J Geriatr Cardiol. 2002;11(119–23):125. https://doi.org/10.1111/j.1076-7460.2002.00998.x.
Andersen JL. Muscle fibre type adaptation in the elderly human muscle. Scand J Med Sci Sports. 2003;13:40–7. https://doi.org/10.1034/j.1600-0838.2003.00299.x.
Gollnick PD, Armstrong RB, Saubert CW, Piehl K, Saltin B. Enzyme activity and fiber composition in skeletal muscle of untrained and trained men. J Appl Physiol. 1972;33:312–9. https://doi.org/10.1152/jappl.1972.33.3.312.
Coggan AR, Spina RJ, King DS, Rogers MA, Brown M, Nemeth PM, et al. Skeletal muscle adaptations to endurance training in 60- to 70-yr-old men and women. J Appl Physiol. 1985;1992(72):1780–6. https://doi.org/10.1152/jappl.1992.72.5.1780.
Máderová D, Krumpolec P, Slobodová L, Schön M, Tirpáková V, Kovaničová Z, et al. Acute and regular exercise distinctly modulate serum, plasma and skeletal muscle BDNF in the elderly. Neuropeptides. 2019;78:101961. https://doi.org/10.1016/j.npep.2019.101961.
Jackson KC, Gidlund E-K, Norrbom J, Valencia AP, Thomson DM, Schuh RA, et al. BRCA1 is a novel regulator of metabolic function in skeletal muscle. J Lipid Res. 2014;55:668–80. https://doi.org/10.1194/jlr.M043851.
Bizjak DA, Schulz SVW, Schumann U, Otto S, Kirsten J, Ebner F, et al. Beneficial molecular adaptations in BRCA-mutation carriers by combined HIT/HIRT intervention: results from a pilot study. Cancers (Basel). 2020. https://doi.org/10.3390/cancers12061526.
Knaepen K, Goekint M, Heyman EM, Meeusen R. Neuroplasticity - exercise-induced response of peripheral brain-derived neurotrophic factor: a systematic review of experimental studies in human subjects. Sports Med. 2010;40:765–801. https://doi.org/10.2165/11534530-000000000-00000.
Tarassova O, Ekblom MM, Moberg M, Lövdén M, Nilsson J. Peripheral BDNF response to physical and cognitive exercise and its association with cardiorespiratory fitness in healthy older adults. Front Physiol. 2020;11:1080. https://doi.org/10.3389/fphys.2020.01080.
Küster OC, Laptinskaya D, Fissler P, Schnack C, Zügel M, Nold V, et al. Novel blood-based biomarkers of cognition, stress, and physical or cognitive training in older adults at risk of dementia: preliminary evidence for a role of BDNF, Irisin, and the Kynurenine pathway. J Alzheimers Dis. 2017;59:1097–111. https://doi.org/10.3233/JAD-170447.
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8. https://doi.org/10.1038/nature10777.
Martinez Munoz IY, Del Camarillo Romero ES, Garduno Garcia JJ. Irisin a novel metabolic biomarker: present knowledge and future directions. Int J Endocrinol. 2018;2018:7816806. https://doi.org/10.1155/2018/7816806.
Kilgore JL, Musch TI, Ross CR. Physical activity, muscle, and the HSP70 response. Can J Appl Physiol. 1998;23:245–60. https://doi.org/10.1139/h98-013.
Liu Y, Gampert L, Nething K, Steinacker JM. Response and function of skeletal muscle heat shock protein 70. Front Biosci. 2006;11:2802–27. https://doi.org/10.2741/2011.
Petersen AMW, Pedersen BK. The anti-inflammatory effect of exercise. J Appl Physiol. 2005;98:1154–62. https://doi.org/10.1152/japplphysiol.00164.2004.
Visser M, Pahor M, Taaffe DR, Goodpaster BH, Simonsick EM, Newman AB, et al. Relationship of Interleukin-6 and tumor necrosis factor-α with muscle mass and muscle strength in elderly men and women: the health ABC study. J Gerontol A Biol Sci Med Sci. 2002;57:M326–32. https://doi.org/10.1093/gerona/57.5.M326.
Acknowledgements
The authors thank Pia Bürger, Jasmine-Leonike Buhl, Nadine Rolser and the SITLESS-Study-group (Dietrich Rothenbacher, Nicole Blackburn, Paolo Caserotti, Maria Giné Garriga, Mathias Skjødt, Laura Coll Planas, Jason Wilson) for invaluable technical assistance and study support.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was part of the EU Horizon 2020 research and innovation program under grant agreement No 634270. The funding body had no role in the collection, analysis, interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
JMS, MD, DD and US designed the work and recruited participants. US and MZ sample material. DAB, JS, MZ and US analyzed and interpreted the participant data and wrote the manuscript. JS, MZ, MD and DD, MAT substantively revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This pilot study was approved by the Ethical Review Committee of Ulm University (No. 354/15). It conforms with the Declaration of Helsinki and all participants gave written informed consent to participate prior to taking part in this study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no financial or non-financial competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Detailed inclusion and exclusion criteria of study participants. Table a. Descriptive statistics of body fat mass (kg), % body fat, relative VO2 peak, VO2VT1 for males and females. Table b. Values subdivided to the respective male and female participants. Table c: P-values after Spearman correlation analysis for aerobic parameters VO2peak and VO2VT1, age and skeletal muscle mass SMM in the respective HPF and LPF. Figure a. Spearman correlation matrix with respective r-value after analysis of age, maximal oxygen consumption VO2peak/kg, first ventilatory threshold VO2VT1 and skeletal muscle mass SMM in HPF and LPF. Figure b. Spearman correlation matrix with respective r-value after analysis of age, maximal oxygen consumption VO2peak/kg, first ventilatory threshold VO2VT1 and skeletal muscle mass SMM in female and male participants.
Additional file 2: Figure c.
Violin plot of Relative Quantification (RQ) of selected gene expression related to ageing, performance and associated metabolism. Except for BDNF (p = 0.0303) and BRCA1 (p = 0.0214), no difference was observed between HPF and LPF. Figure d: Correlation matrix and respective Spearman’s r for VO2peak of HPF vs gene expression. Table d. Relative Quantification (RQ) of selected gene expression related to ageing, performance and associated metabolism. Except for BDNF (p = 0.0303) and BRCA1 (p = 0.0214), no difference was observed between HPF and LPF. * p ≤ 0.05 indicate inter-group differences. Table e. Mean MyHC composition as well as MyHC composition for HPF and LPF with regard to MyHC I, MyHC IIa and MyHC IId/x fiber type.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Bizjak, D.A., Zügel, M., Schumann, U. et al. Do skeletal muscle composition and gene expression as well as acute exercise-induced serum adaptations in older adults depend on fitness status?. BMC Geriatr 21, 697 (2021). https://doi.org/10.1186/s12877-021-02666-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12877-021-02666-0