Abstract
Background
It has been indicated that Angiotensin-Converting Enzyme Insertion/Deletion (ACE I/D) polymorphism (rs4646994) could be regarded as a genetic factor that raises the risk of CAD through its impact on the activity of Angiotensin-Converting Enzyme (ACE) and angiotensin II level. The present study seeks to examine the relationship between ACE I/D polymorphism with the risk of atherosclerosis. Moreover, its potential effects on ACE activity and oxLDL level are investigated.
Methods
In this study, 145 healthy individuals and 154 patients (143 males and 156 females) were selected among the subjects referred to Shahid Madani Hospital. Atherosclerosis was determined in all subjects with gold standard angiography. Blood samples were collected, used to isolate white blood cells (WBC) and serum separation. The DNA was extracted and the polymorphism was determined by polymerase chain reaction (PCR). The enzyme activity was measured using high-performance liquid chromatography (HPLC).
Results
This study indicated that patients with atherosclerosis had higher levels of oxidized Low-Density Lipoprotein (oxLDL) and ACE activity (P < 0.05) as compared to controls. Although we found a significant association between ACE I/D polymorphism genotype and the allele with atherosclerosis in the male group, there were no association when the entire patient group was compared to the entire control group.
Conclusion
Our study revealed the ACE I/D polymorphism of the ACE gene may not be an independent risk factor in the development of atherosclerosis and evaluation of ACE activity level is more important in evaluating the risk of disease. The researchers found no relation between ACE I/D polymorphism and atherosclerosis and also between types of genotype, ACE activity, and OxLDL level.
Similar content being viewed by others
Background
Even though therapeutic strategies concerning the deterrence of CAD have advanced, the rate of death induced by CAD is still rising. It is estimated by 2030 deaths due to CAD will be 23.4 million people. The most significant reason for CAD has been reported to be atherosclerotic coronary arteries [1, 2].
Atherosclerosis, as a fibroproliferative and inflammatory process, results from endothelial dysfunction induced chronic vascular damage [3]. A key step in initiating atherosclerosis is the LDL accumulation in the intima layer of the coronary artery [4]. LDL particles are converted to oxidized Low Density Lipoprotein (oxLDL) by reactive oxygen species (ROS) [5, 6]. Different risk factors have been identified in the occurrence of atherosclerosis including age, gender, high blood pressure, diabetes mellitus, hyperlipidemia, smoking, and obesity [7, 8].
Various published studies have emphasized that genetic factors could be involved in the initiation and progression of CAD [9]. For the first time, Rigat et al. discovered the ACE I/D polymorphism in their attempt to investigate the role of ACE gene in genetic control of plasma ACE level. They used this polymorphism as a genetic marker to examine the relationship between the type of polymorphism and the ACE concentration. Cambien et al. were the first to report the model for the genetic control of plasma ACE levels, based on the results of a family study. They showed 47% of the serum ACE variance was due to the allele effect of ACE I/D polymorphism. They also were the first to report the relationship of ACE I/D polymorphism with CAD.
Hence, we could consider insertion/ deletion (I/D) polymorphism of angiotensin-converting enzyme (ACE) gene as an effective genetic risk factors for CAD [10]. Many studies were conducted in different populations to investigate this association. We also decided to this for the first time in the Western Iran population. Maybe just say “The literature, however, is conflicting with some studies showing an association and others not.”
ACE is one of the components of the RAS and zinc-dependent metalloproteinase found widely in endothelial and epithelial cells. Moreover, the enzyme has been isolated from several sources including serum, lungs, seminal fluid, and plasma. ACE, as a key component in RAS, converts angiotensin I to angiotensin II Angiotensin II, increase the production of adhesion molecules and chemokines, stimulates LDL oxidation and foam cell formation in macrophages. Increased level of the ACE and subsequent ACE activity by raising the production of angiotensin II can lead to atherosclerosis [11,12,13].
A variety of investigations have reported the impact of ACE I/D polymorphism in several cardiovascular diseases including endothelial dysfunction, atherosclerosis, and heart failure. It has been displayed that there is a relationship between ACE I/D polymorphism and the alteration of ACE activity [14, 15]. Various studies have revealed that the ACE level is higher in patients with DD than subjects with II genotype, and those with ID genotype have a medium level [16]. The literature, however, is conflicting with some studies showing an association and others not [17, 18]. We have summarized some previous ACE I/D polymorphism studies in a table below. (Table 1).
The purpose of the present study is the investigation of ACE I/D polymorphism distribution, measurement of ACE activity and oxLDL level and a number of biochemical factors. Furthermore, we examined the potential effect of ACE I/D polymorphism on ACE activity and the level of oxLDL in western Iran.
Methods
Study design and population
In this study, a total of 299 subjects (154 patients with atherosclerosis and 145 controls) who referred to Cardiology and Angiography Department of Shahid Madani Hospital, Khorramabad, Iran, between 2016 December and 2017 May were selected. Whether the participants had atherosclerosis or not was confirmed by the standard diagnostic angiography. In the case of plaque discernment inside arteries, participants divided into the patient’s group and, if the report showed normal coronary angiography, divided into the control group. The medical history of all subjects including age, sex, weight, the age of diagnosis, smoking, family history, hypertension, diabetes mellitus, drug abuse, and alcohol consumption were recorded. Patients with congenital heart disease, malignancy, chronic kidney disease, pulmonary obstruction, and steroid hormones consumption were excluded. Written informed consent was obtained from all participants. The study was approved by the Research Committee of Shahid Madani Hospital and the Ethics Committee of Lorestan University of Medical Sciences. Written informed consent was obtained from all subjects who participated in the study (code: LUMS.REC.1395.123). This study was administered in accordance with the Declaration of Helsinki and its following revisions.
Biochemical measurements
The blood samples were collected from all subjects following overnight fasting into tubes without anticoagulant. The lipid profile was evaluated applying an auto-analyzer (BT-1000, USA).
Small dense Low Density Lipoprotein (SdLDL) was evaluated using the method by Hirano et al. as follows: 1. Precipitating serum lipoproteins with 1.044 g/ml density using heparin sodium salt and MnCl2, 2. incubating for 10 min at 37 °C, 3. placing in the ice bath for 15 min. 4, Collecting the supernatant by centrifugation at 15,000 rpm for 15 min at 4 °C and 5. Measuring LDL-cholesterol in the supernatant by LDL assay kit (Pishtazteb, Iran) [30].
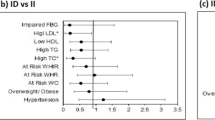
The measurement of oxLDL was performed using ELISA method according to the manufacturer protocol (Mercodia, Sweden). ACE activity was measured using the method by Horiuchi et al. In this measurement, 40 μl borate buffer containing 5.3 mM Hippuryl-L-Histidyl-Leucine (HHL) as a substrate, was added to 10 μl of serum, and then incubated for 30 min. The reaction was stopped by adding metaphosphoric acid, and was centrifuged for 5 min at 4000 rpm. Subsequently, 20 μl of supernatant was injected into the HPLC column. The amount of released hippuric acid was analyzed using HPLC (Shimadzu, Japan). The unit of enzyme activity is the amount of enzyme that can produce 1 μmol of hippuric acid at 37 °C for 1 min (Fig. 1) [31].
Genotyping of the ACE polymorphism
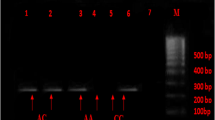
Genomic DNA was extracted from white blood cells with the DNA extraction kit (Yekta Tajhiz, Iran) based on the instruction of the manufacturer’s protocol. Genotyping for the ACE gene I/D polymorphism was performed using polymerase chain reaction (PCR) method and using two oligonucleotide primers, sense: 5′-CTGGAGACCACTCCCATCCTTTCT-3′ and antisense: 5′-GATGTGGCCATCACATTCGTCAGAT-3′ based on the flanking sequence of the insertion/deletion region on the ACE gene. The amplification was performed in a volume of 25 μl containing 50 ng template DNA, 10 μM of each primer, 2.5 μl 10X PCR buffer (Fermentas, Lithuania), 3 mM MgCl2, 200 μM each dNTP, and 1.5 units of Taq DNA polymerase (Fermentas, Lithuania). The PCR cycling conditions were as follows: initial denaturation at 94 °C for 5 min followed by 30 cycles of denaturation at 94 °C for 60 s, annealing at 59 °C for 60 s, extension at 72 °C for 2 min, and a final extension at 72 °C for 5 min. Amplification products were separated and sized by electrophoresis on a 2% agarose gel. The length of the D and I fragment alleles were 190 bp and 490 bp respectively (Fig. 2) [15].
To avoid DD mistyping, Second PCR was performed with a specific pair of primers that included sense: 5′-TGG GAC CAC AGC GCC CGC CAC TAC-3′ and antisense: 5′-TCG CCA GCC CTC CCA TGC CCA TAA-3′, in 25 μl reaction mixture volume on DD genotype samples. PCR conditions were identical except for the annealing temperature of 57 °C. The presence of I allele resulted in a 335 bp PCR product. All samples with DD genotype were re-genotyped, which I allele was detected in 8 samples.
Statistical analysis
The normality of data was tested by the use of the Kolmogorov-Smirnov test. The data of the numerical variables are presented as mean ± SD. Accordingly, t-test was used to compare continuous data, and Chi-square test was used to test the qualitative variables. Frequencies of genotype and allele was tested using Chi-square test and association with disease was tested using logistic regression. We used ANOVA and tukey’s test (for post-hoc analysis) for intra-group comparison of ACE activity and oxLDL level. Multivariate logistic regression analysis was used for testing the independent association of various variables. Data were analyzed with SPSS software version 16 (SPSS Inc., Chicago, IL, USA). P-values less than 0.05 were regarded to be statistically significant.
Results
Baseline characteristics of the study population
The baseline characteristics of the 299 participants have been presented in Table 2. The results of the present study revealed a significant difference between age (P < 0.001), weight (P = 0.045) and diagnosis age (Age of diagnosis the heart disease) (P < 0.001) of the patient and control groups. Remarkably, there was not any significant difference between height (P = 0.112), BMI (P = 0.390), systolic blood pressure (P = 0.140) or diastolic blood pressure (P = 0.147) in the two groups.
The biochemical characteristics of the study subjects have been indicated in Table 3. A significant difference was found between TC (P < 0.001), TG (P = 0.006), HDL (P = 0.035), LDL (P < 0.001), Sd-LDL (P < 0.001), Ox-LDL (P < 0.001), serum glucose (P < 0.001) and ACE activity (P = 0.026) between the patient and control groups. Furthermore, we found a significant difference between the family history of CAD, diabetes mellitus, cigarette smoking and hypertension between the two groups.
Genotype distribution and genotype frequencies
Genotype distribution did not deviate from Hardy-Weinberg equilibrium (patients (P = 0.2), controls (P = 0.08)). The results of the genotyping of the ACE polymorphism (Fig. 2) and its relationship with atherosclerosis have been presented in Table 4. In the present study, we did not find any significant difference between genotype and allele frequency in the patient and control groups. All the study groups and subgroups were separated by sex, and were subsequently analysed. We found a significant relation between ACE I/D polymorphism genotypes and the alleles with atherosclerosis in the male (81 patients, 62 control) group but there was no significant association in the female group (P-value = 0.02 for the male group, P value = 0.879 for the female group) (Table 5).
Patients with II and ID genotypes showed higher ACE activity as compared to controls with the same alleles (P < 0.05), but there was no significant association between ACE activity and DD genotype (P = 0.646) in the two groups. In contrast with the control group, the difference between the levels of ACE activity in different genotypes of the patient group was not statistically significant. The results indicated that in the control group, the level of enzyme activity of the DD genotype was significant (p < 0.05) compared to ID and II genotypes. No significant association was found between the other genotypes. The results also indicated that the relationship between genotype interaction and enzyme activity of ACE in increasing the chances of developing atherosclerosis was not significant.
Patients with DD and ID genotype showed higher oxLDL level as compared to controls with DD and ID genotype (P < 0.05), but there was no important distinction between oxLDL level and II genotypes (P = 0.156) in the two groups.
No specific pattern was found for the relationship between type of genotype and oxLDL level (Table 6). The present study revealed that the interaction effect of ID (P = 0.713), DD (P = 0.142) and II (P = 0.125) genotypes and ACE activity on oxLDL levels were not statistically significant.
Using the multivariable logistic regression, the effects of ACE I/D polymorphism genotypes simultaneously with regard to age, sex, history of blood pressure, smoking and CAD, were simultaneously examined. The results indicated no significant relationship between genotype and the risk of developing atherosclerosis. In terms of age, the risk of developing a disease is increased by 0.8% per 1-year-old increase, which is statistically significant (P < 0.001) (CI = 1.05–1.11). As expected the risk of atherosclerosis in patients with a history of cardiovascular disease is 3.56 times more than those without a history of the same disease. (P < 0.001) (CI = 2–6.33) (Table 7).
Discussion
After reorganization of ACE I/D polymorphism as a genetic marker for cardiovascular disease, many investigations were done to find such a genetic risk relationship. This study was planned to investigate the association between ACE I/D polymorphism and the risk of atherosclerosis, and the effect of genotypes on ACE activity. We also interested in to study the difference of ACE activity, oxLDL level, and a number of biochemical characteristics between two groups. Distributions of the genotypes frequency of ACE I/D polymorphism in our study were found to be I/I (20%), I/D (48.3%), and DD (31.7%) in the control group and I/I (13%), I/D (50.5%), and DD (33.1%) in the patient group. In the present study, neither genotype nor allele frequency was remarkably different between the control and patient groups. This finding confirms the results of certain studies previously conducted that will be discussed below.
Many studies showed that genetic factors may play a role in the progression of cardiovascular diseases. Among these genetic factors, the ACE I/D gene polymorphism has been a recurrent subject in a variety of studies [32]. The ACE converts angiotensin into active octapeptide, called angiotensin II, which is the main active component in the RAS and has been known as an atherogenic factor [33]. Various studies have indicated that ACE I/D polymorphism could be as a risk factor for CAD, MI, and cardiomyopathy [34].
The relationship between DD genotype and CAD has been examined in several studies [35]. It has been found out that DD genotype is a risk factor for the development of atherosclerosis in carotid arteries in the Chinese, Australian, and Asian Indian populations [36, 37]. Moreover, the D allele has a role in the incidence of CAD by elevating the levels of ACE in several populations such as Turkish [38].
In accordance with the result obtained from our study, some studies previously carried out found no relationship between ACE I/D polymorphism and incidence of CAD. Studies carried out by Jeunemaitre et al. and Ferrieres et al. indicated that ACE I/D polymorphism could not play a role in the occurrence of CAD in the Caucasian and European populations [24, 39]. A meta-analysis study was conducted on 118 different studies, including 43.733 patients and 82.606 healthy individuals. Their results indicated that ACE I/D polymorphism is correlated with the increase in the risk of sever CAD [40].
The ACE enzyme plays an important role in the RAS by converting angiotensin I to angiotensin II. Angiotensin II has been shown to have atherogenic properties. Increased activity of the ACE enzyme can contribute to an increased risk of disease by raising angiotensin II production. Therefore, the level of ACE activity that may be affected by various factors, including genetic factors, can be considered as a risk for the disease [41]. Many studies have shown the relationship between ACE serum levels and ACE I/D polymorphism in different population [42,43,44]. In our study the level of ACE activity in patients with II + ID and DD genotypes was significantly higher than in control (II + ID and DD) that can consider as an important risk factor for disease in a study conducted by Sahin et al. (2015) on the population of Turkey, an increase was observed in ACE activity in the patient group compared to the control group. Furthermore, DD genotype was more prevalent among patients in their research [41]. It seems that evaluation of ACE activity level in comparison with ACEI/D polymorphism genotypes is more important in evaluating the risk of disease.
Various pieces of evidence indicated that LDL peroxidation is one of the most important risk factors for atherosclerosis. In fact, changes in LDL oxidation seem to be the most important trigger for the development of atherosclerosis. Researchers have indicated that angiotensin 2, a product of the ACE, could play a role in increasing the absorption and oxidation of LDL [45,46,47].
For the first time we investigated the relationship between ACE I/D polymorphism and oxLDL level. In the present study a significant difference was observed in the level of oxLDL in DD + ID and DD genotypes in the patient group in contrast with the control group. No significant correlation was found between the simultaneous effect of ACE activity and ACE I/D polymorphism on oxLDL levels. Investigations by Shimada et al. showed high level of oxLDL in patient with coronary artery disease than controls [48]. In addition, our biochemical parameter’s results such as sdLDL, lipid profiles, blood pressure, FBS, and patient records were consistent with many previous findings [49].
Limitations
The limitations about this study include: first, in this study, we only collected samples from several western provinces in Iran, which, due to the genetic diversity and racial differences in different populations in Iran, this investigation could study a larger population in Iran, which may have different outcomes in different ethnic groups. Secondly, we did not find any relationship between different ACE I/D polymorphism genotypes and atherosclerosis in the control and patient groups, so other factors such as confirmation of genotyping results should be considered. Third, this study did not investigate the effects of oxidative stress, which, can increase the risk of heart disease. So, there are some other factors that can be very affected in this way such as environmental pollution factors.
Conclusion
In this study, we observed a considerable association between ACE I/D polymorphism and atherosclerosis in the male group, but there was no relationship between different ACE I/D polymorphism genotypes and atherosclerosis in the control and patient groups. This fact that the level of ACE activity and oxLDL were significantly higher in the patient group than the control group indicates the role of these two factors in increasing the risk of atherosclerosis. However, we did not find any evidence that ACE I/D polymorphism could effect on the level of enzyme activity and oxLDL. Despite the small sample size, this investigation showed the study of ACE I/D polymorphism did not proper for the prediction of atherosclerosis.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ACE I/D:
-
Angiotensin-converting enzyme insertion/deletion
- CAD:
-
Coronary artery disease
- HDL:
-
High-density lipoprotein
- LDL:
-
Low-density lipoprotein
- OxLDL:
-
Oxidized LDL
- RAS:
-
Renin-angiotensin system
- SdLDL:
-
Small dense LDL
References
Mack M, Gopal A. Epidemiology, traditional and novel risk factors in coronary artery disease. Heart Fail Clin. 2016;12(1):1–10.
Mathers C, Stevens G, Mascarenhas M. Global health risks: mortality and burden of disease attributable to selected major risks. World Health Organization; 2009.
Libby P, Okamoto Y, Rocha VZ, Folco E. Inflammation in atherosclerosis: transition from theory to practice. Circ J. 2010;74(2):213–20.
Conti P, Shaik-Dasthagirisaeb Y. Atherosclerosis: a chronic inflammatory disease mediated by mast cells. Cent Eur J Immunol. 2015;40(3):380–6.
Hansson GK, Hermansson A. The immune system in atherosclerosis. Nat Immunol. 2011;12(3):204–12.
Li H, Horke S, Förstermann U. Vascular oxidative stress, nitric oxide, and atherosclerosis. Atherosclerosis. 2014;237(1):208–19.
van Rooy MJ, Pretorius E. Obesity, hypertension and hypercholesterolemia as risk factors for atherosclerosis leading to ischemic events. Curr Med Chem. 2014;21(19):2121–9.
Li D, Singh RM, Liu L, Chen H, Singh BM, Kazzaz N, et al. Oxidized-LDL through LOX-1 increases the expression of angiotensin-converting enzyme in human coronary artery endothelial cells. Cardiovasc Res. 2003;57(1):238–43.
Roy H, Bhardwaj S, Yla-Herttuala S. Molecular genetics of atherosclerosis. Hum Genet. 2009;125(5–6):467–91.
Hamelin BA, Zakrzewski-Jakubiak M, Robitaille NM, Bogaty P, Labbé L, Turgeon J. Increased Risk of Myocardial Infarction Associated With Angiotensin-Converting Enzyme Gene Polymorphism Is Age Dependent. J Clin Pharmacol. 2011;51(9):1286-92.
Nguyen Dinh Cat A, Touyz RM. A new look at the renin-angiotensin system--focusing on the vascular system. Peptides. 2011;32(10):2141–50.
Zhou JB, Yang JK, Lu JK, An YH. Angiotensin-converting enzyme gene polymorphism is associated with type 2 diabetes: a meta-analysis. Mol Biol Rep. 2010;37(1):67–73.
Kumar R, Thomas CM, Yong QC, Chen W, Baker KM. The intracrine renin-angiotensin system. Clin Sci (Lond). 2012;123(5):273–84.
Schmieder RE, Hilgers KF, Schlaich MP, Schmidt BM. Renin-angiotensin system and cardiovascular risk. Lancet. 2007;369(9568):1208–19.
Bai Y, Wang L, Hu S, Wei Y. Association of angiotensin-converting enzyme I/D polymorphism with heart failure: a meta-analysis. Mol Cell Biochem. 2012;361(1–2):297–304.
Seckin D, Ilhan N, Ilhan N, Ozbay Y. The relationship between ACE insertion/deletion polymorphism and coronary artery disease with or without myocardial infarction. Clin Biochem. 2006 Jan;39(1):50–4.
Acarturk E, Attila G, Bozkurt A, Akpinar O, Matyar S, Seydaoglu G. Insertion/deletion polymorphism of the angiotensin converting enzyme gene in coronary artery disease in southern Turkey. J Biochem Mol Biol. 2005;38(4):486–90.
Amara A, Mrad M, Sayeh A, Lahideb D, Layouni S, Haggui A, et al. The effect of ACE I/D polymorphisms alone and with concomitant risk factors on coronary artery disease. Clin Appl Thromb Hemost. 2018;24(1):157–63.
Nakai K, Itoh C, Miura Y, Hotta K, Musha T, Itoh T, Miyakawa T, Iwasaki R, Hiramori K. Deletion polymorphism of the angiotensin I-converting enzyme gene is associated with serum ACE concentration and increased risk for CAD in the Japanese. Circulation. 1994;90(5):2199–202.
Moradzadegan A, Vaisi-Raygani A, Nikzamir A, Rahimi Z. Angiotensin converting enzyme insertion/deletion (I/D) (rs4646994) and Vegf polymorphism (+ 405G/C; rs2010963) in type II diabetic patients: association with the risk of coronary artery disease. J Renin-Angiotensin-Aldosterone Syst. 2015;16(3):672–80.
Nagi DK, Foy CA, Mohamed-Ali V, Yudkin JS, Grant PJ, Knowler WC. Angiotensin-1—converting enzyme (ACE) gene polymorphism, plasma ACE levels, and their association with the metabolic syndrome and electrocardiographic coronary artery disease in Pima Indians. Metabolism. 1998;47(5):622–6.
Saha N, Talmud PJ, Tay JS, Humphries SE, Basair J. Lack of association of angiotensin-converting enzyme (ACE). Gene insertion/deletion polymorphism with CAD in two Asian populations. Clin Genet. 1996;50(3):121–5.
Arca M, Pannitteri G, Campagna F, Candeloro A, Montali A, Cantini R, Seccareccia F, Campa PP, Marino B, Ricci G. Angiotensin-converting enzyme gene polymorphism is not associated with coronary atherosclerosis and myocardial infarction in a sample of Italian patients. Eur J Clin Investig. 1998;28(6):485–90.
Ferrières J, Ruidavets JB, Fauvel J, Perret B, Taraszkiewicz D, Fourcade J, et al. Angiotensin I-converting enzyme gene polymorphism in a low-risk European population for coronary artery disease. Atherosclerosis. 1999;142(1):211–6.
Akar N, Aras Ö, Ömürlü K, Cin Ş. Deletion polymorphism at the angiotensin-converting enzyme gene in Turkish patients with coronary artery disease. Scand J Clin Lab Invest. 1998;58(6):491–6.
Acarturk E, Attila G, Bozkurt A, Akpinar O, Matyar S, Seydaoglu G. Insertion/deletion polymorphism of the angiotensin converting enzyme gene in coronary artery disease in southern Turkey. BMB Rep. 2005;38(4):486–90.
Van Bockxmeer FM, Mamotte CD, Burke V, Taylor RR. Angiotensin-converting enzyme gene polymorphism and premature coronary heart disease. Clin Sci. 2000;99(3):247–51.
Chiang FT, Lai ZP, Chern TH, Tseng CD, Hsu KL, Lo HM, Tseng YZ. Lack of association between angiotensin-converting enzyme gene polymorphism and coronary heart disease in a Chinese population. Jpn Heart J. 1997;38(2):227–36.
Pandey U, Kumari R, Nath B, Ganesh S, Banerjee I, Hasan OM, Midha T, Pandey S. Association of angiotensin-converting enzyme, methylene tetrahydrofolate reductase and paraoxonase gene polymorphism and coronary artery disease in an Indian population. Cardiol J. 2011;18(4):385–94.
Hirano T, Ito Y, Saegusa H, Yoshino G. A novel and simple method for quantification of small, dense LDL. J Lipid Res. 2003;44(11):2193–201.
Horiuchi M, Fujimura K, Terashima T, Iso T. Method for determination of angiotensin-converting enzyme activity in blood and tissue by high-performance liquid chromatography. J Chromatogr. 1982;233:123–30.
Jia EZ, Xu ZX, Guo CY, Li L, Gu Y, Zhu TB, et al. Renin-angiotensin-aldosterone system gene polymorphisms and coronary artery disease: detection of gene-gene and gene-environment interactions. Cell Physiol Biochem. 2012;29(3–4):443–52.
Ikonomidis I, Tzortzis S, Tsantes A, Ntai K, Triantafyllidi H, Trivilou P, et al. The interplay between renin-angiotensin system activation, abnormal myocardial deformation and neurohumoral activation in hypertensive heart disease: a speckle tracking echocardiography study. Int J Cardiovasc Imaging. 2017;33(3):323–9.
Guney AI, Ergec D, Kirac D, Ozturhan H, Caner M, Koc G, et al. Effects of ACE polymorphisms and other risk factors on the severity of coronary artery disease. Genet Mol Res. 2013 Dec 19;12(4):6895–906.
Cambien F, Poirier O, Lecerf L, Evans A, Cambou JP, Arveiler D, et al. Deletion polymorphism in the gene for angiotensin-converting enzyme is a potent risk factor for myocardial infarction. Nature. 1992;359(6396):641–4.
Jeng JR. Carotid thickening, cardiac hypertrophy, and angiotensin converting enzyme gene polymorphism in patients with hypertension. Am J Hypertens. 2000;13(1 Pt 1):111–9.
Wang XL, McCredie RM, Wilcken DE. Genotype distribution of angiotensin-converting enzyme polymorphism in Australian healthy and coronary populations and relevance to myocardial infarction and coronary artery disease. Arterioscler Thromb Vasc Biol. 1996;16(1):115–9.
Akar N, Aras O, Omürlü K, Cin S. Deletion polymorphism at the angiotensin-converting enzyme gene in Turkish patients with coronary artery disease. Scand J Clin Lab Invest. 1998;58(6):491–5.
Jeunemaitre X, Ledru F, Battaglia S, Guillanneuf MT, Courbon D, Dumont C, et al. Genetic polymorphisms of the renin-angiotensin system and angiographic extent and severity of coronary artery disease: the CORGENE study. Hum Genet. 1997;99(1):66–73.
Zintzaras E, Raman G, Kitsios G, Lau J. Angiotensin-converting enzyme insertion/deletion gene polymorphic variant as a marker of coronary artery disease: a meta-analysis. Arch Intern Med. 2008;168(10):1077–89.
Sahin S, Ceyhan K, Benli I, Ozyurt H, Naseri E, Tumuklu MM, et al. Traditional risk factors and angiotensin-converting enzyme insertion/deletion gene polymorphism in coronary artery disease. Genet Mol Res. 2015;14(1):2063–8.
Lee EJ. Population genetics of the angiotensin-converting enzyme in Chinese. Br J Clin Pharmacol. 1994;37(2):212–4.
Tsutaya S, Kitaya H, Saito Y, Nakata S, Takamatsu H, Yasujima M. Angiotensin converting enzyme gene polymorphism and its enzyme activity in serum in young Japanese females. Tohoku J Exp Med. 1997;182(2):151–5.
Yamamoto K, Kataoka S, Hashimoto N, Kakihara T, Tanaka A, Kawasaki T, et al. Serum level and gene polymorphism of angiotensin I converting enzyme in Japanese children. Acta Paediatr Jpn. 1997;39(1):1–5 30.
Sato A, Ueda C, Kimura R, Kobayashi C, Yamazaki Y, Ebina K. Angiotensin II induces the aggregation of native and oxidized low-density lipoprotein. Eur Biophys J. 2018;47(1):1–9.
Shao B, Heinecke JW. HDL, lipid peroxidation, and atherosclerosis. J Lipid Res. 2009;50(4):599–601.
Salvayre R, Negre-Salvayre A, Camaré C. Oxidative theory of atherosclerosis and antioxidants. Biochimie. 2016;125:281–96.
Shimada K, Mokuno H, Matsunaga E, Miyazaki T, Sumiyoshi K, Miyauchi K, Daida H. Circulating oxidized low-density lipoprotein is an independent predictor for cardiac event in patients with coronary artery disease. Atherosclerosis. 2004;174(2):343–7.
Dahlen GH, Guyton JR, Attar M, Farmer JA, Kautz JA, Gotto AM Jr. Association of levels of lipoprotein Lp (a), plasma lipids, and other lipoproteins with coronary artery disease documented by angiography. Circulation. 1986;74(4):758–65.
Acknowledgments
The authors would like to thank all the people involved in the investigation and with their support, we were encouraged to do this project. We also gratefully acknowledge all staff of Shahid Madani Hospital and all the people participating in this study.
Funding
The work described in this manuscript was part of the dissertation of Negar Nouryazdan, submitted to the Lorestan University of Medical Sciences for the MSc in clinical biochemistry. This work was supported by a grant from the Vice Chancellor for Research, Lorestan University of Medical Sciences (grant number A-10-1665-1).
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to the study presented in this paper. GS was responsible for the design and management of the project and also the final edition of the paper. NN played a practical role in collect samples, performed the experiments and wrote the paper. RH performed some experiments. MB analyzed the data. GA and BY contributed to the writing and editing of the article. All authors have read and approved the final manuscript submitted.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee at Shahid Madani Hospital, Khorramabad, Iran and Lorestan University of Medical Science. Written informed consent was obtained from all subjects who participated in the study (code: LUMS.REC.1395.123). This study was administered in accordance with the Declaration of Helsinki and its following revisions.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Nouryazdan, N., Adibhesami, G., Birjandi, M. et al. Study of angiotensin-converting enzyme insertion/deletion polymorphism, enzyme activity and oxidized low density lipoprotein in Western Iranians with atherosclerosis: a case-control study. BMC Cardiovasc Disord 19, 184 (2019). https://doi.org/10.1186/s12872-019-1158-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12872-019-1158-4