Abstract
Background
Exploring the physiological and molecular mechanisms underlying goat sexual maturation can enhance breeding practices and optimize reproductive efficiency and is therefore substantially important for practical breeding purposes. As an essential neuroendocrine organ in animals, the hypothalamus is involved in sexual development and other reproductive processes in female animals. Although microRNAs (miRNAs) have been identified as significant regulators of goat reproduction, there is a lack of research on the molecular regulatory mechanisms of hypothalamic miRNAs that are involved in the sexual development of goats. Therefore, we examined the dynamic changes in serum hormone profiles and hypothalamic miRNA expression profiles at four developmental stages (1 day (neonatal, D1, n = 5), 2 months (prepubertal, M2, n = 5), 4 months (sexual maturity, M4, n = 5), and 6 months (breeding period, M6, n = 5)) during sexual development in Jining grey goats.
Results
Transcriptome analysis revealed 95 differentially expressed miRNAs (DEMs) in the hypothalamus of goats across the four developmental stages. The target genes of these miRNAs were significantly enriched in the GnRH signalling pathway, the PI3K-Akt signalling pathway, and the Ras signalling pathway (P < 0.05). Additionally, 16 DEMs are common among the M2 vs. D1, M4 vs. D1, and M6 vs. D1 comparisons, indicating that the transition from D1 to M2 represents a potentially critical period for sexual development in Jining grey goats. The bioinformatics analysis results indicate that miR-193a/miR-193b-3p-Annexin A7 (ANXA7), miR-324-5p-Adhesion G protein-coupled receptor A1 (ADGRA1), miR-324-3p-Erbb2 receptor tyrosine kinase 2 (ERBB2), and miR-324-3p-Rap guanine nucleotide exchange factor 3 (RAPGEF3) are potentially involved in biological processes such as hormone secretion, energy metabolism, and signal transduction. In addition, we further confirmed that miR-324-3p targets the regulatory gene RAPGEF3.
Conclusion
These results further enrich the expression profile of hypothalamic miRNAs in goats and provide important insights for studying the regulatory effects of hypothalamic miRNAs on the sexual development of goats after birth.
Similar content being viewed by others
Introduction
The goat is an important domestic animal worldwide, offering various products such as meat, milk, skin, and cashmere [1]. Reproductive capacity constrains the development of livestock husbandry, and the normal onset of puberty and sexual maturity are essential for the reproductive performance of goats [2]. Investigating the physiological and molecular mechanisms underlying sexual maturation in goats can enhance the breeding process and optimize reproductive efficiency, which is highly important for practical breeding purposes [3]. The Jining grey goat is an indigenous goat breed in China that is characterized by nonseasonal oestrous, high fertility and early sexual development [4]. Jining grey goats enter puberty at 2 months of age and achieve sexual maturity at 3–4 months of age [5]. These characteristics make Jining grey goats an ideal animal model for studying goat fecundity.
The sexual maturation of female mammals is controlled by the hypothalamic‒pituitary‒gonadal (HPG) axis. In mammals, the HPG axis remains stable after birth and is directly activated at the onset of puberty [6]. Following sexual maturity, follicles undergo development and maturation, resulting in a regular cycle of oestrous, ovulation, and fertility [7]. As a crucial neuroendocrine centre in animals, the hypothalamus significantly influences reproduction, energy metabolism, and circadian rhythms in animals [8,9,10]. In mammals, the initiation of puberty and the regulation of sexual development are coordinated by a complex neural network located within the hypothalamus [11]. The proper progression of sexual maturation holds paramount importance for the reproductive capacity of goats. The crucial determinant for triggering the onset of sexual maturation in animals involves increased pulsatile gonadotropin-releasing hormone (GnRH) secretion from the hypothalamus. This process is instigated and perpetuated by a combination of transsynaptic inputs and glial inputs, which influence a complex network of GnRH neurons [12, 13]. The hypothalamus produces and releases GnRH, which acts on the pituitary gland to promote the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) from the pituitary gland, which subsequently affects the production of ovarian sex steroid hormones [14]. Furthermore, the hypothalamus interacts with hormones from other tissues, such as adipose tissue and the liver, to establish a complex network of neuroendocrine regulation that ensures normal sexual development [15].
MicroRNAs (miRNAs), a cluster of small noncoding RNAs with an average length of approximately 22 nucleotides, are present in the genomes of different organisms [16]. These miRNAs regulate mRNA expression at the posttranscriptional level by forming complementary base pairs with the 3’ untranslated region (3’UTR) of the target mRNA [17, 18]. Recently, an increasing amount of research has indicated the potential involvement of miRNAs in the intricate signalling and genetic networks that control sexual development and maturation. In the hypothalamus of rats, Lin28/let-7 axis modifications are associated with postnatal sexual maturation [19]. Moreover, alterations in GnRH expression play crucial roles in pubertal and adult fertility [20]. miR-200 and miR-155 target zinc finger E-box binding homeobox 1 (ZEB1) and CCAAT enhancer binding protein beta (CEBPB), respectively, and participate in sexual development and reproduction through the regulation of GnRH expression [21]. MiR-7 inhibits the expression, synthesis, and secretion of gonadotropins. By regulating gonadotropins, miR-7 participates in the feedback regulation of the hypothalamic‒pituitary‒ovarian (HPO) axis [22]. These studies suggest that miRNAs in the hypothalamus play crucial roles in the regulation of reproduction and are also important components of the gene regulatory network involved in sexual maturation.
Although numerous studies have confirmed that hypothalamic miRNAs play important roles in reproductive regulation, most of the research on goat hypothalamic miRNAs has focused on high- and low-fertility goats [23, 24]. There is limited information on the hypothalamic miRNA expression profile of goats throughout their development from birth to sexual maturity. Given this, high-throughput sequencing technology was used to further investigate the dynamic changes in miRNA expression profiles during sexual development (1 day (neonatal, D1, n = 5), 2 months (prepubertal, M2, n = 5), 4 months (sexual maturity, M4, n = 5), and 6 months (breeding period, M6, n = 5)) in goats and to elucidate the potential molecular regulatory mechanisms involved. Our findings provide new perspectives for goat breeding by revealing the dynamic changes in hypothalamic miRNAs during sexual development and revealing the potential molecular regulatory mechanisms involved.
Materials and methods
Animal sample collection
In this study, a total of 20 female Jining grey goats were selected from the Jining Grey Goat Breeding Farm (Jiaxiang County, Jining City, Shandong Province, China). The Jining grey goats in the experiment were divided into four groups according to age: the 1-day-old group (neonatal, D1, n = 5; body weight: 2.08 ± 0.11 kg), 2-month-old group (prepubertal, M2, n = 5; body weight: 4.42 ± 0.24 kg), 4-month-old group (sexual maturity, M4, n = 5; body weight: 7.62 ± 0.50 kg) and 6-month-old group (breeding period, M6, n = 5; body weight: 8.82 ± 0.53 kg). The selected goats demonstrated sound health and were free from any ailments. They were reared with consistent feeding and management practices. The goats had unrestricted access to food and water. The selected goats were not genetically related. All goats were slaughtered on the same day. Following stunning via electric shock, the goats were promptly slaughtered. Hypothalamic tissue samples were subsequently collected and frozen in liquid nitrogen for miRNA sequencing.
Serum sample collection and hormone content determination
All the goat serum samples were collected on the same day (8:00 to 10:00 a.m.). Blood was collected from the jugular vein of each experimental goat, placed in a 10-mL centrifuge tube for 30 min, centrifuged at 3 000 r/min at 4 °C for 10 min and stored at -20 °C for testing. In our study, the serum FSH, LH, P and E2 levels were detected using an ELISA kit (MDBio, Qingdao, China).
Total RNA extraction and library construction
Following the manufacturer’s instructions, total RNA was obtained from 20 hypothalamic tissue samples using TRIzol (Invitrogen, Carlsbad, CA, USA). The integrity of the sequenced RNA was evaluated using an RNA Nano 6000 assay kit and an Agilent Bioanalyzer 2100 system (Agilent Technologies, Santa Clara, CA, USA). The total RNA concentration was measured with a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). The quality of the total RNA was assessed using agarose gel electrophoresis.
The qualified RNA was subjected to PAGE for the purification and isolation of miRNAs ranging from 18 to 40 nucleotides in length. The 3’ and 5’ adaptors were subsequently ligated to the 3’ and 5’ ends of the small RNA molecules, respectively. Following reverse transcription primer hybridization, first-strand cDNA was synthesized, and a double-stranded cDNA library was generated through PCR enrichment. After purification and size selection, the library with lengths ranging from 18 to 40 base pairs was subjected to SE50 sequencing using the Illumina NovaSeq 6000 (Illumina, San Diego, CA, USA) platform.
Small RNA sequence data analysis
To ensure the quality of sequencing, a filtering process is performed to eliminate reads containing poly-N sequences, 5’ adapter contaminants, missing 3’ adapters or insertion tags, reads with poly A/T/G/C content, and those with low-quality readings. Simultaneously, the Q20, Q30, and GC content are calculated for the original data. A specific range of lengths was subsequently selected from the clean data for downstream analysis, and Bowtie2 was used to align the clean reads to the reference sequence of the goat. A comparison was subsequently performed between the quality-controlled clean tags and small RNAs in the GenBank database, Rfam database (http://rfam.xfam.org/), and the goat reference genome (GCF_001704415.2_ARS1.2) to remove other nonmiRNAs and tags present in exons, introns, and repetitive sequences. The filtered clean tags are subsequently used to search the miRBase database (http://www.mirbase.org) for the identification of known miRNAs, and both miREvo [25] and miRDeep2 [26] are employed to predict novel miRNAs.
Identification and analysis of DEMs
Differential expression analysis of miRNAs was performed using the DESeq2 package [27]. miRNAs with a |log2-fold change| > 1 and p.adj < 0.05 were identified as differentially expressed miRNAs (DEMs). The expression profiles for both known and novel miRNAs were calculated with normalization to transcripts per million (TPM), where TPM = actual miRNA read counts * 10^6 divided by the total number of clean labels. The ggplot2 and heatmap packages were used to conduct principal component analysis (PCA) and generate heatmaps for visualizing miRNAs across all samples. Expression pattern analysis was performed on the DEMs using Mfuzz [28].
Target gene prediction and functional enrichment of DEMs
We utilized RNAhybrid (version 2.1.2), TargetScan, and Miranda (version 3.3a) software to predict DEMs target genes in the hypothalamus of goats across four developmental stages. The predicted target genes were subjected to Gene Ontology (GO) enrichment analysis using the clusterProfiler software package (v3.10.1) [29] and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis using KOBAS [30]. Pathways with p values < 0.05 were identified as significantly enriched.
WGCNA and coexpression network construction
The miRNA expression in hypothalamic samples was analysed using weighted gene coexpression analysis (WGCNA) [31] with the WGCNA package of R software. The optimal soft-threshold value (β = 8) was determined using the pickSoftThreshold function. The minimum number of miRNAs within each module was set to 30, and the remaining parameters were set to the default settings to identify modules and construct a network. miRNAs with similar expression patterns were grouped into the same module. Phenotypic data, such as serum hormone levels, were imported to calculate the correlation coefficients and p values between the phenotypes and the modules for subsequent analysis. KEGG enrichment analysis was performed on the target genes of the miRNAs within the modules using KOBAS. miRNAs with a |KME| > 0.8 were identified as key miRNAs.
In this study, the target genes of key miRNAs were screened and identified, and a miRNA‒mRNA regulatory network map was constructed and visualized via Cytoscape [32].
qRT‒PCR validation
The SYBR PrimerScript miRNA RT‒PCR kit (Takara, Beijing, China) was used for miRNA reverse transcription (including the poly-A method) and fluorescence quantification in accordance with the product manual. For the design of the upstream-specific primers, the mature sequence of the miRNA was taken into consideration, and the downstream primers were selected from the universal miR qPCR primers provided in the kit. Six miRNAs were randomly selected for quantitative verification, and the screened miRNA‒mRNA regulatory relationships were verified via qPCR. The primer sequences can be found in Table S1.
For the miRNA RT‒PCR, a total volume of 20 µL was prepared, containing 2 × SYBR Premix ex Taq I (10 µL), 10 µM PCR upstream specific primers (0.8 µL), 10 µM downstream miR qPCR primers (0.8 µL), the cDNA template (2.0 µL), and RNase-free H2O. The reaction volume of mRNA was 20 µL, comprising 2 µL of cDNA, 0.5 µL each of upstream and downstream primers (200 nM), 10 µL of SYBR (Takara, Dalian, China), and 7 µL of RNA-free water. Quantitative detection was performed using a Roche 96 instrument with the following program: 95 °C for 30 s, predenaturation; 95 °C for 5 s, 60 °C for 20 s, 40 cycles. Five biological replicates were performed for each group of samples, and the relative expression levels of miRNAs and mRNAs were calculated using the 2−ΔΔCt method [33].
Dual-luciferase reporter assay
RNAhybrid was used to predict miRNA binding site information for target genes. Wild-type plasmids (miR-324-3p-RAPGEF3-WT, miR-331-3p-RAPGEF3-WT) and mutant plasmids (miR-324-3p-RAPGEF3-MUT, miR-331-3p-RAPGEF3-MUT) were synthesized using the pmirGLO vector (Table S2). The wild-type and mutant plasmids were cotransfected with the miR-324-3p mimics/NC or the miR-331-3p mimics/NC into HEK-293T cells, and dual luciferase activity was measured 48 h later. The sequences of the miRNA mimics and miRNA NC are shown in Table S3. Three biological replicates were established for each group.
Statistical analysis
All the statistical analyses were performed using SPSS 17.0 software (IBM Corporation, Armonk, NY). All experimental data are expressed as the mean ± standard error (mean ± SEM). The analysis between the two sets of data is conducted using a t test. One-way ANOVA was used for data analysis for more than two groups of data. P < 0.05 was considered statistically significant.
Results
Overview of the miRNA sequencing data
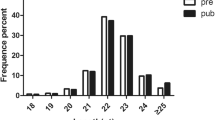
We performed high-throughput sequencing of 20 small RNA libraries from goats at 4 different developmental stages via the NovaSeq 6000 platform. Twenty libraries generated a total of 227,480,000 raw reads, with a Q30 > 95.56%, indicating that the sequencing data exhibited high quality and could be used for subsequent data analysis (Table S4). Statistics on the length distribution of sRNAs (Fig. 1a) revealed that the number of clean reads with a length of 22 nt was the highest, and the percentage of clean reads with a length between 21 nt and 23 nt ranged from 64.1 to 79.9%.
Overview of the miRNA sequencing data. (a). Distribution of the lengths of clean reads in the goat hypothalamus. (b). Distribution characteristics of small RNAs in the hypothalamus of goats at different developmental stages. (c). The distribution range of identified miRNA TPM. (d). The expression of the top 20 miRNAs in the hypothalamus at four developmental stages (D1, 1 day; M2, 2 months; M4, 4 months; M6, 6 months)
To identify known miRNAs in the hypothalamus of goats at different developmental stages, we compared the sRNA reads with sequences in the specified range in miRBase. The results (Fig. 1b) revealed that 69.25% of the clean reads were mapped to known miRNAs and that 0.55% of the clean reads were mapped to novel miRNAs. We determined the TPM values of the miRNAs, and the results revealed that the expression of the miRNAs varied across the goat hypothalamus tissues (Fig. 1c).
Identification and characterization of miRNAs
A total of 412 known mature miRNAs were identified, and these miRNAs belong to 193 miRNA families. In addition, we assessed the number of members of different miRNA families (Figure S1). Thirty-two of these families contained three or more miRNA members, and the family with the greatest number of members was the let-7 family, which consisted of 16 family members, followed by the miR-30 and miR-376 families, which included 11 and 6 family members, respectively.
In addition, a total of 148 novel miRNAs were identified by predicting hairpin structures in unannotated reads. Among the identified miRNAs, the top 20 highly expressed miRNAs are shown in Fig. 1d. Interestingly, over half of the identified miRNAs belong to the let-7 family.
Differential expression miRNA analysis
To identify miRNAs associated with sexual development in goats and understand their potential molecular regulatory mechanisms, we first performed PCA on the sequence data of 20 tissue samples. The results showed (Fig. 2a) that the D1 group was more clearly differentiated from the other groups, whereas M2, M4, and M6 were closer together, suggesting that there were fewer differences between the groups.
Differential expression analysis of miRNAs. (a). PCA of 20 hypothalamus samples from goats at different developmental stages. (b). The number of DEMs in different comparison groups. (c). Upset plots of DEMs in the hypothalamus of goats at different developmental stages. (d). Heatmap of the top 20 DEMs associated with hypothalamus expression in goats during sexual development. (e). Analysis of DEMs expression patterns during sexual development
A total of 95 DEMs were identified via differential analysis of miRNAs in the hypothalamus of goats at different developmental stages (Fig. 2b, Figure S2; Tables S5-S8). Among the DEMs, 72 were known, and 23 were novel. A total of 52 DEMs (14 upregulated and 38 downregulated) were identified between M2 and D1. Similarly, in the comparison between M4 and D1, we detected 61 DEMs (18 upregulated and 43 downregulated). In the comparison between M6 and D1, we identified 31 DEMs (10 upregulated and 21 downregulated). Finally, in the comparison between M4 and M2, we identified 6 DEMs (1 upregulated and 5 downregulated). No differential miRNAs were identified in the M6 vs. M4 and M6 vs. M2 comparison groups, which is consistent with the results of PCA and DEMs clustering analyses. These results revealed relatively small differences among the M2, M4, and M6 groups. To determine key miRNAs involved in hypothalamic development, an UpSet plot (Fig. 2c) was generated to visualize the miRNAs across different developmental stages. Interestingly, the M4 vs. D1 group presented the greatest number of independently differentially expressed miRNAs, with 16 miRNAs coexpressed among the M2, M4, and M6 periods compared with the D1 period. Figure 2d shows the expression patterns of the DEMs with the top 20 TPM values, with let-7c-5p exhibiting the highest expression level.
Furthermore, we assessed the expression profiles of the DEMs using Mfuzz software. The analysis delineated three distinct clusters of DEMs on the basis of their expression patterns (Fig. 2e; Table S9). Cluster 1 comprised 17 DEMs with peak expression at M4 and characterized by a progressive increase from D1 to M4 followed by a decline towards M6. Cluster 2 included 12 DEMs that were predominantly expressed at M2, exhibiting a marked increase from D1 to M2 and then a sustained elevation through M6. Finally, Cluster 3 had the largest number of DEMs at 65; the expression of these DEMs was highest in D1, decreased significantly in M2, and then plateaued in M6.
We selected 6 key miRNAs for qRT‒PCR validation. The qRT‒PCR results showed consistent expression trends with the sRNA sequencing results in each group (D1, M2, M4, and M6), indicating high reliability of the sRNA sequencing results (Fig. 3). This was also supported by the high correlation between the two sets of data.
DEMs target gene prediction and functional annotation
To explore the roles of DEMs with varying expression patterns, we predicted that 95 distinct miRNA targets would regulate 7,393 genes. Specifically, 51 genes in Cluster 1, 402 genes in Cluster 2, and 7234 genes in Cluster 3 were identified as targets of DEMs (Table S10).
We subsequently analysed the GO and KEGG enrichment of the miRNA target genes in each cluster (Fig. 4, Table S11-S14). The results revealed that the target genes of the DEMs in Cluster 1 were not significantly enriched in the GO terms or KEGG pathways.
Functional enrichment analysis of differential miRNA target genes in different expression clusters. (a). GO analysis of DEMs target genes in Cluster 2. Red, green and blue represent biological process (BP), molecular function (MF) and molecular function (CC), respectively. (b). KEGG enrichment analysis of DEMs target genes in Cluster 2. (c). GO analysis of DEMs target genes in Cluster 3. (d). KEGG enrichment analysis of DEMs target genes in Cluster 3. P < 0.05 indicates a significantly enriched KEGG pathway
GO enrichment analysis revealed that the target genes in Cluster 2 were significantly enriched in biological processes such as cell surface receptor signalling pathways, protein phosphorylation, the Wnt signalling pathway, and cellular signal transduction (p < 0.05). The GO terms significantly enriched for target genes of DEM in Cluster 3 included phosphorylation, protein phosphorylation, regulation of small GTPase-mediated signal transduction, intracellular signal transduction, regulation of intracellular signal transduction, small GTPase-mediated signal transduction, regulation of signalling, DNA repair, and regulation of signal transduction (P < 0.05).
Figure 4b and d list the top 20 pathways for the KEGG enrichment results for Cluster 2 and Cluster 3 target genes. The target genes in Cluster 2 were significantly enriched in the regulation of the actin cytoskeleton, GnRH signalling pathway, PI3K-Akt signalling pathway, Ras signalling pathway and JAK-STAT signalling pathway (P < 0.05). The target genes in Cluster 3 were significantly enriched in the MAPK signalling pathway, human papillomavirus infection, axon guidance, endocrine resistance, insulin signalling pathway, glycerophospholipid metabolism, the mTOR signalling pathway, the neurotrophin signalling pathway, the Ras signalling pathway, the GnRH signalling pathway, the ECM‒receptor interaction, GnRH secretion, the PI3K‒Akt signalling pathway, the Wnt signalling pathway, and the ErbB signalling pathway (P < 0.05).
WGCNA analysis
To identify key miRNAs involved in sexual development in goats, we performed WGCNA using miRNA expression data. With a selected threshold of β = 8, we built a coexpression module (Figure S3a). Next, we analysed miRNA expression data in combination with phenotypic indicators such as serum hormones and developmental stages to identify key miRNAs. A significant negative correlation was noted between the miRNAs characterized in the turquoise module and the serum FSH (R=-0.49, P = 0.03) and E2 (R=-0.47, P = 0.04) levels. These results suggest that miRNAs in this module may be involved in reproductive hormone secretion during sexual development and may play a key role in the D1 stage (R=-0.47, P = 0.04) (Fig. 5a and b). A total of 129 miRNAs were identified in the module (Figure S3), including 100 known miRNAs and 29 novel miRNAs (Table S15).
Weighted gene network analysis of DEMs during sexual development in goats. (a). Hierarchical clustering results of the miRNA signature modules. miRNAs of the same colour are classified according to the same module. (b). Correlation analysis between feature modules and phenotypic indices such as hormones in goats. The x-axis represents the phenotypic information of goats at different developmental stages: FSH (follicle-stimulating hormone); LH (luteinizing hormone); P (progesterone); E2 (Estradiol); D1 (1 day old); M2 (2 months old); M4 (4 months old); and M6 (4 months old). (c). GO analysis of the DEMs target genes that appear in turquoise. (d). KEGG analysis of the DEMs target genes that appear in turquoise
GO and KEGG enrichment analyses were conducted using the miRNAs within the turquoise module (Fig. 5c and d; Table S16, S17). A total of 856 GO terms were enriched for target genes of miRNAs in the turquoise module. Biological processes, including phosphorylation, small GTPase-mediated signalling, and intracellular signalling (P < 0.05), were significantly enriched. The KEGG enrichment analysis revealed that the miRNA target genes in the module were significantly enriched in the focal adhesion, axon guidance, endocrine resistance, cholinergic synapse, MAPK signalling, insulin signalling, Rap1 signalling, mTOR signalling, GnRH signalling, and calcium signalling pathways (P < 0.05). These results suggest that miRNAs in the turquoise module may be involved in energy metabolism, hypothalamic signalling, hormone secretion and other processes during the sexual maturation of goats.
The hub miRNA is the miRNA with the highest connectivity in the module. We screened the top 20 candidate miRNAs according to the screening criterion |KME|>0.8 and then identified the DEMs with an average TPM > 100 as hub miRNAs (Table S18). We identified 12 hub miRNAs in the turquoise module: let-7c-5p, miR-140-3p, novel_73, novel_90, miR-331-3p, miR-363-3p, miR-193b-3p, miR-193a, miR-532-3p, miR-324-5p, miR-497-5p, and miR-326-3p.
Identification of key miRNAs and their correlation with serum hormones
A total of 18 key DEMs were obtained by intersecting miRNAs and hub DEMs identified using WGCNA of all three periods (M2 vs. D1, M4 vs. D1, and M6 vs. D1). The results of the detection of serum hormone levels revealed that the FSH, LH, P and E2 levels in the D1 stage were significantly different from those in the M2 and M4 stages (P < 0.05) (Figure S4). We further screened key miRNAs associated with hormone secretion during goat sexual development by calculating the correlation of these DEMs with serum hormone levels (Fig. 6). The results revealed that miR-335-3p and miR-3955-3p levels were significantly positively correlated with the serum FSH, LH, and E2 hormone levels (p < 0.05), and miR-324-5p, miR-877-5p, miR-331-3p, miR-542-5p, and miR-324-3p levels were negatively correlated with FSH hormone levels (P < 0.05).
Correlation heatmap of key miRNAs in the hypothalamus and hormone levels in the serum during the sexual development of Jining grey goats. Red represents a positive correlation, and blue represents a negative correlation. *, P < 0.05; **, P < 0.01. Follicle-stimulating hormone, FSH; luteinizing hormone, LH; estradiol, E2; progesterone, P
miRNA‒mRNA interaction network analysis
KEGG enrichment analysis was performed on the target genes of the 18 key DEMs identified, and genes that were significantly enriched in biological processes such as reproduction, energy metabolism, and signal transduction were subsequently screened. A regulatory network of miRNA‒mRNA interactions during sexual development in goats was constructed. This interaction network included 7 DEMs, 54 target genes, and a total of 70 pairs of target regulatory relationships (Fig. 7). In the constructed regulatory network.
In the constructed regulatory network, a single miRNA can regulate multiple target genes, and multiple miRNAs can also regulate a single target gene, such as miR-193a/miR-193b-3p-Annexin A7 (ANXA7), miR-324-5p-Adhesion G protein-coupled receptor A1 (ADGRA1), miR-324-3p- Erbb2 receptor tyrosine kinase 2 (ERBB2), and miR-324-3p-Rap guanine nucleotide exchange factor 3 (RAPGEF3).
Validation of the miRNA‒mRNA target regulatory relationship
We selected miR-324-3p, miR-331-3p, and RAPGEF3 for targeted regulation for verification. The qRT‒PCR results revealed significant differences in the expression of RAPGEF3 in hypothalamic tissue during the sexual development of Jining grey goats (P < 0.05), and RAPGEF3 expression levels in M2 and M4 were significantly greater than those in D1 (P < 0.05) (Fig. 8a). In addition, miR-324-3p and miR-331-3p were negatively correlated with RAPGEF3 expression during sexual development in Jining grey goats (Fig. 8b and c). Furthermore, we predicted the potential targets of miR-331-3p, miR-324-3p, and RAPGEF3 and performed a dual luciferase reporter vector experiment (Fig. 8d). The results revealed no significant difference in the relative expression of firefly luciferase from the RAPGEF3 wild-type and mutant vectors after transfection of miR-331-3p mimics compared with the NC group (P > 0.05) (Fig. 8e). Compared with that in the NC group, the relative expression of firefly luciferase in the RAPGEF3-WT group was significantly inhibited after transfection with the miR-324-3p mimics (P < 0.05) (Fig. 8f), whereas there was no significant difference in the relative expression of luciferase in the RAPGEF3-MUT group (P > 0.05). These results suggest that miR-324-3p has a targeted regulatory relationship with RAPGEF3, whereas miR-331-3p does not have a targeted regulatory relationship with RAPGEF3.
Verification of the regulatory relationships among miR-324-3p, miR-331-3p and RAPGEF3. (a). Relative RAPGEF3 expression in the hypothalamus at four developmental stages in Jining grey goats (n = 5). Different lowercase letters represent significant differences (P < 0.05). (b). Validation of the correlation between miR-331-3p and RAPGEF3. (c). Validation of the correlation between miR-324-3p and RAPGEF3. (d). Schematic diagram of the predicted binding sites of miR-331-3p, miR-324-3p, and RAPGEF3. (e). miR-331-3p with RAPGEF3 dual-luciferase activity assay (n = 3). (f). miR-331-3p with RAPGEF3 dual-luciferase activity assay (n = 3). WT represents the luciferase reporter vector for the wild-type RAPGEF3 3’-UTR, and MUT represents the luciferase reporter vector for the mutant RAPGEF3 3’-UTR. The data are presented as the means ± SEMs. n = 5, ns, P > 0.05; **P < 0.001
Discussion
In this study, we identified 95 DEMs in the hypothalamic tissues of Jining grey goats during various sexual development stages (D1, M2, M4, and M6), comprising 72 known miRNAs and 23 newly discovered miRNAs. Among these miRNAs, let-7c presented the highest expression level. Research has indicated that let-7 family members, such as let-7c, are prominently expressed in the hypothalamus and play significant roles in reproductive processes, puberty initiation, and sexual maturation [34, 35]. In addition, miR-30b-5p is another highly expressed DEMs. Recent studies in rats have shown that miR-30b expression is low in the neonatal hypothalamus and increases progressively during postnatal maturation. Interference with the interaction between miR-30 and MkRN3 in the hypothalamus results in delayed sexual development in rats [36]. In this study, miR-30b-5p expression mirrors its lowest level postbirth, and a gradual increase occurs during the M4 period, indicating a potential key role of miR-30b-5p in sexual development in goats.
MiRNAs with the same expression pattern may have similar regulatory functions [37]. In this study, 95 DEMs presented 3 different expression patterns. The target genes of the DEMs in Cluster 2 were significantly enriched in the JAK-STAT signalling pathway, the PI3K-Akt signalling pathway, the T-cell receptor signalling pathway, and other pathways related to immunity and energy metabolism. The miRNA content of this cluster was lowest after birth and significantly increased at M2, which may be related to the transition from passive to active immunity in goats from birth to M2 [38, 39]. The expression of miRNAs in Cluster 3 was high at the D1 stage and then significantly decreased at 2 months of age. Interestingly, 14 of the 16 DEMs identified in M2 vs. D1, M4 vs. D1, and M6 vs. D1 exhibited Cluster 3 expression patterns. The target genes of the DEMs in Cluster 3 were significantly enriched in energy metabolism, signalling pathways and hormone secretion-related pathways, such as the GnRH signalling pathway and ECM receptor interaction. These pathways are involved in the regulation of reproductive ability and sexual development of hypothalamic tissue in goats and other domestic animals [23, 40, 41]. These results suggest that DEMs with different expression patterns may be involved in corresponding biological processes and play important roles in the sexual development of goats.
Moreover, we identified key DEMs associated with hormone secretion and sexual development at different stages in Jining grey goats using WGCNA, including miR-193a, miR-193b-3p, miR-324-3p, and miR-331-3p. Among them, miR-193a and miR-193b-3p are both members of the miR-193 family and regulate cell proliferation by regulating the expression of cell cycle-related genes [42, 43]. In addition, miR-193b-3p is involved in the regulation of adiponectin production in white adipose tissue [44,45,46] and the regulation of glucose metabolism by regulating Forkhead Box O1 (FOXO1) [47]. In this study, ANXA7 was predicted to be a target gene for both miR-193a and miR-193b-3p. ANXA7, a member of the annexin family, is involved in biological processes such as neuronal development, calcium homeostasis, and hormone secretion [48,49,50]. ANXA7 is also involved in glucose metabolism by regulating insulin secretion [49, 51], and energy reserve levels and metabolic status plays important roles in the sexual maturation of female animals [52]. In this study, miR-193a and miR-193b-3p levels were significantly different between M2 and D1 and between M4 and D1. These results further suggest that miR-193a and miR-193b-3p may participate in the energy metabolism process of goats by targeting ANXA7, thus affecting the sexual maturation of goats.
It has been suggested that miR-324-3p may be involved in biological processes such as apoptosis, cAMP and Ca2+ signalling, cellular stress, and metabolism [53]. KISS1/kisspeptins expression was significantly downregulated by inhibiting the interaction with miR-324-3p in early ectopic pregnancy. In addition, miR-324-3p regulates ovarian development and ovarian granulosa cell proliferation in goats [54]. In our study, ERBB2 was predicted to be a target gene of miR-324-3p. ERBB2 is required for the onset of normal puberty, and inhibition of its expression delays puberty in females and impairs reproductive capacity [55]. In addition, ERBB2 is involved in the regulation of GnRH secretion during sexual maturation [56]. In this study, miR-324-5p was significantly differentially expressed between M2 and D1, M4 and D1, and M6 and D1. Additionally, miR-324-3p was negatively correlated with FSH hormone secretion. These results suggest that miR-324-3p may regulate the secretion of the FSH hormone by targeting ERBB2 to participate in hypothalamic ERBB receptor-mediated neuronal glial cell signalling and regulate the release of GnRH during sexual maturation in Jining grey goats [57].
MiR-331-3p is associated with the cell cycle and plays important roles in tumorigenesis, cancer cell proliferation, and differentiation [58]. Hosako et al. (2009) reported that miR-331-3p expression was significantly altered in mice with p53 deletion, resulting in embryonic craniocephalic malformations [59]. miR-331-3p also inhibits the proliferation of preadipocytes, promotes adipocyte differentiation and fatty acid metabolism, and promotes fatty acid synthesis by regulating the target gene dihydrolipoamide S-succinyltransferase (DLST) [60]. Interestingly, we observed that miR-331-3p and miR-324-3p cotargeted regulatory RAPGEF3. RAPGEF3, also known as EPAC1, is a gene encoding a small GTPase-activating protein of Rap1 [61]. RAPGEF3 is involved in a variety of biological processes, such as cell proliferation, inflammation regulation, energy metabolism, and neuronal signalling through the regulation of intracellular cAMP levels by mediating the cAMP signalling pathway [62,63,64,65,66]. GnRH secretion in hypothalamic GnRH neurons is highly dependent on the calcium concentration and is regulated by cAMP [67, 68]. In our study, miR-331-3p was differentially expressed in the M2 vs. D1 and M4 vs. D1 groups, and the expression level in the D1 stage was significantly greater than that in the M2 and M4 stages (P < 0.05). miR-331-3p and miR-324-3p were significantly negatively correlated with FSH hormone secretion. We hypothesize that miR-331-3p and miR-324-3p cotarget regulatory RAPGEF3 and participate in neuronal development and GnRH hormone secretion during goat sexual development, subsequently affecting FSH serum levels.
We verified the targeted regulatory relationship between miR-324-3p and RAPGEF3 via qRT‒PCR and dual-luciferase reporter assays. In follow-up studies, we will further verify the role of miR-324-3p and its target gene RAPGEF3 in the regulation of primary cell proliferation and hormone secretion in the goat hypothalamus through molecular biology experiments such as qRT‒PCR, the Cell Counting Kit-8 (CCK8) assay, Western blotting, and ELISA.
Conclusion
In summary, this study described the dynamic changes in the hypothalamic miRNA expression profile during postnatal sexual development in Jining grey goats. We identified 95 DEMs at four developmental stages. Among them, key miRNAs such as miR-193a, miR-193b-3p, miR-324-3p and miR-331-3p may be involved in biological processes such as hormone secretion and energy metabolism during goat sexual development by regulating target genes. Notably, miR-324-3p has been shown to target RAPGEF3. This research provides a foundation for understanding the role of hypothalamic miRNAs in goat sexual development; however, further investigations into the biological functions and molecular mechanisms of these miRNAs are warranted.
Data availability
The data generated from this study has been submitted to the Gene Expression Omnibus (GEO) database and is publicly available. It can be accessed under the accession number GSE246961.
Abbreviations
- MiRNA:
-
MicroRNA
- GnRH:
-
Gonadotropin-releasing hormone
- FSH:
-
Follicle stimulating Hormone
- LH:
-
Luteinizing hormone
- P:
-
Progesterone
- E2:
-
Estradiol
- GO:
-
Gene Ontology
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
References
Miller BA, Lu CD. Current status of global dairy goat production: an overview. Asian-Australas J Anim Sci. 2019;32(8):1219–32.
Taberlet P, Coissac E, Pansu J, Pompanon F. Conservation genetics of cattle, sheep, and goats. C R Biol. 2011;334(3):247–54.
Shelton M. Reproduction and breeding of goats. J Dairy Sci. 1978;61(7):994–1010.
National Committee for Livestock and Poultry Genetic Resources. Animal Genetic resources in China: Sheep and Goats. China National Commission of Animal Genetic Resources. Beijing; 2011. pp. 283–6.
Shi Y, Wang S, Bai S, Huang L, Hou Y. Postnatal ovarian development and its relationship with steroid hormone receptors in JiNing Grey goats. Anim Reprod Sci. 2015;154:39–47.
Collu R, Savoie S, Hamel R, Gibb W, Ducharme JR. Maturation of the hypothalamic–pituitary–gonadal axis in the male lamb: a review. Psychoneuroendocrinology. 1983;8(2):213–24.
Sliwowska JH, Ziarniak K, Dudek M, Matuszewska J, Tena-Sempere M. Dangerous liaisons for pubertal maturation: the impact of alcohol consumption and obesity on the timing of puberty. Biol Reprod. 2019;100(1):25–40.
Seeburg PH, Mason AJ, Stewart TA, Nikolics K. The mammalian GnRH gene and its pivotal role in reproduction. Recent Prog Horm Res. 1987;43:69–98.
Stevenson JA, THE HYPOTHALAMUS IN THE REGULATION OF ENERGY AND WATER BALANCE. Physiologist. 1964;99:305–18.
Gao Y, Sun T. Molecular regulation of hypothalamic development and physiological functions. Mol Neurobiol. 2016;53(7):4275–85.
Spaziani M, Tarantino C, Tahani N, Gianfrilli D, Sbardella E, Lenzi A, Radicioni AF. Hypothalamo-pituitary axis and puberty. Mol Cell Endocrinol. 2021;520:111094.
Pinilla L, Aguilar E, Dieguez C, Millar RP, Tena-Sempere M. Kisspeptins and reproduction: physiological roles and regulatory mechanisms. Physiol Rev. 2012;92(3):1235–316.
Nebesio TD, Eugster EA. Current concepts in normal and abnormal puberty. Curr Probl Pediatr Adolesc Health Care. 2007;37(2):50–72.
Mancini A, Magnotto JC, Abreu AP. Genetics of pubertal timing. Best Pract Res Clin Endocrinol Metab. 2022;36(1):101618.
Abreu AP, Kaiser UB. Pubertal development and regulation. Lancet Diabetes Endocrinol. 2016;4(3):254–64.
Cai Y, Yu X, Hu S, Yu J. A brief review on the mechanisms of miRNA regulation. Genomics Proteom Bioinf. 2009;7(4):147–54.
He L, Hannon GJ. MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5(7):522–31.
Brodersen P, Voinnet O. Revisiting the principles of microRNA target recognition and mode of action. Nat Rev Mol Cell Biol. 2009;10(2):141–8.
Sangiao-Alvarellos S, Manfredi-Lozano M, Ruiz-Pino F, Navarro VM, Sánchez-Garrido MA, Leon S, Dieguez C, Cordido F, Matagne V, Dissen GA, et al. Changes in hypothalamic expression of the Lin28/let-7 system and related microRNAs during postnatal maturation and after experimental manipulations of puberty. Endocrinology. 2013;154(2):942–55.
Ojeda SR, Lomniczi A. Puberty in 2013: unravelling the mystery of puberty. Nat Rev Endocrinol. 2014;10(2):67–9.
Messina A, Langlet F, Chachlaki K, Roa J, Rasika S, Jouy N, Gallet S, Gaytan F, Parkash J, Tena-Sempere M, et al. A microRNA switch regulates the rise in hypothalamic GnRH production before puberty. Nat Neurosci. 2016;19(6):835–44.
He J, Xu S, Ji Z, Sun Y, Cai B, Zhang S, Wang P. The role of miR-7 as a potential switch in the mouse hypothalamus-pituitary-ovary axis through regulation of gonadotropins. Mol Cell Endocrinol. 2020;518:110969.
Liang C, Han M, Zhou Z, Liu Y, He X, Jiang Y, Ouyang Y, Hong Q, Chu M. Hypothalamic transcriptome analysis reveals the crucial MicroRNAs and mRNAs affecting litter size in goats. Front Vet Sci. 2021;8:747100.
Han M, Liang C, Liu Y, He X, Chu M. Integrated Transcriptome Analysis Reveals the Crucial mRNAs and miRNAs Related to Fecundity in the Hypothalamus of Yunshang Black Goats during the Luteal Phase. Animals (Basel) 2022, 12(23).
Wen M, Shen Y, Shi S, Tang T. miREvo: an integrative microRNA evolutionary analysis platform for next-generation sequencing experiments. BMC Bioinformatics. 2012;13:140.
Friedländer MR, Mackowiak SD, Li N, Chen W, Rajewsky N. miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucleic Acids Res. 2012;40(1):37–52.
Love MI, Huber W, Anders S. Moderated estimation of Fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15(12):550.
Kumar L. Mfuzz: a software package for soft clustering of microarray data. Bioinformation. 2007;2(1):5–7.
Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics. 2012;16(5):284–7.
Bu D, Luo H, Huo P, Wang Z, Zhang S, He Z, Wu Y, Zhao L, Liu J, Guo J, et al. KOBAS-i: intelligent prioritization and exploratory visualization of biological functions for gene enrichment analysis. Nucleic Acids Res. 2021;49(W1):W317–25.
Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008;9:559.
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13(11):2498–504.
Rao X, Huang X, Zhou Z, Lin X. An improvement of the 2ˆ(-delta delta CT) method for quantitative real-time polymerase chain reaction data analysis. Biostat Bioinforma Biomath. 2013;3(3):71–85.
Zhang XD, Zhang YH, Ling YH, Liu Y, Cao HG, Yin ZJ, Ding JP, Zhang XR. Characterization and differential expression of microRNAs in the ovaries of pregnant and non-pregnant goats (Capra hircus). BMC Genomics. 2013;14:157.
Sangiao-Alvarellos S, Manfredi-Lozano M, Ruiz-Pino F, León S, Morales C, Cordido F, Gaytán F, Pinilla L, Tena-Sempere M. Testicular expression of the Lin28/let-7 system: hormonal regulation and changes during postnatal maturation and after manipulations of puberty. Sci Rep. 2015;5:15683.
Heras V, Sangiao-Alvarellos S, Manfredi-Lozano M, Sanchez-Tapia MJ, Ruiz-Pino F, Roa J, Lara-Chica M, Morrugares-Carmona R, Jouy N, Abreu AP, et al. Hypothalamic miR-30 regulates puberty onset via repression of the puberty-suppressing factor, Mkrn3. PLoS Biol. 2019;17(11):e3000532.
Lu M, Lu F, Liao C, Guo Y, Mao C, Lai Y, Chen X, Chen W. High throughput miRNA sequencing and bioinformatics analysis identify the mesenchymal cell proliferation and apoptosis related miRNAs during fetal mice palate development. J Gene Med. 2023;25(9):e3531.
Struff WG, Sprotte G. Bovine colostrum as a biologic in clinical medicine: a review. Part I: biotechnological standards, pharmacodynamic and pharmacokinetic characteristics and principles of treatment. Int J Clin Pharmacol Ther. 2007;45(4):193–202.
Guedes MT, Zacharias F, Couto RD, Portela RW, Santos LC, Santos SC, Pedroza KC, Peixoto AP, López JA, Mendonça-Lima FW. Maternal transference of passive humoral immunity to Haemonchus contortus in goats. Vet Immunol Immunopathol. 2010;136(1–2):138–43.
Pan X, Li Q, Chen D, Gong W, Li N, Jiang Y, Zhang H, Chen Y, Yuan X. Alternative Splicing dynamics of the Hypothalamus-Pituitary-Ovary Axis during Pubertal Transition in Gilts. Front Genet. 2021;12:592669.
Gao X, Ren C, Zhang W, Fang F. An integrated analysis of mRNAs and lncRNAs in goat’s hypothalamus to explore the onset of puberty. Reprod Domest Anim. 2023;58(8):1046–54.
Nakano H, Yamada Y, Miyazawa T, Yoshida T. Gain-of-function microRNA screens identify miR-193a regulating proliferation and apoptosis in epithelial ovarian cancer cells. Int J Oncol. 2013;42(6):1875–82.
Lin SR, Yeh HC, Wang WJ, Ke HL, Lin HH, Hsu WC, Chao SY, Hour TC, Wu WJ, Pu YS, et al. MiR-193b mediates CEBPD-Induced Cisplatin Sensitization through Targeting ETS1 and cyclin D1 in human urothelial carcinoma cells. J Cell Biochem. 2017;118(6):1563–73.
Mazzu YZ, Hu Y, Soni RK, Mojica KM, Qin LX, Agius P, Waxman ZM, Mihailovic A, Socci ND, Hendrickson RC, et al. miR-193b-Regulated signaling networks serve as Tumor suppressors in Liposarcoma and promote adipogenesis in adipose-derived stem cells. Cancer Res. 2017;77(21):5728–40.
Mazzu YZ, Hu Y, Shen Y, Tuschl T, Singer S. miR-193b regulates tumorigenesis in liposarcoma cells via PDGFR, TGFβ, and wnt signaling. Sci Rep. 2019;9(1):3197.
Belarbi Y, Mejhert N, Lorente-Cebrián S, Dahlman I, Arner P, Rydén M, Kulyté A. MicroRNA-193b controls Adiponectin Production in Human White Adipose tissue. J Clin Endocrinol Metab. 2015;100(8):E1084–1088.
Hu H, Zhao M, Li Z, Nie H, He J, Chen Z, Yuan J, Guo H, Zhang X, Yang H, et al. Plasma miR-193b-3p is elevated in type 2 diabetes and could impair glucose metabolism. Front Endocrinol (Lausanne). 2022;13:814347.
Rick M, Ramos Garrido SI, Herr C, Thal DR, Noegel AA, Clemen CS. Nuclear localization of Annexin A7 during murine brain development. BMC Neurosci. 2005;6:25.
Mears D, Zimliki CL, Atwater I, Rojas E, Glassman M, Leighton X, Pollard HB, Srivastava M. The Anx7(+/-) knockout mutation alters electrical and secretory responses to ca(2+)-mobilizing agents in pancreatic β-cells. Cell Physiol Biochem. 2012;29(5–6):697–704.
Kuijpers GA, Lee G, Pollard HB. Immunolocalization of synexin (annexin VII) in adrenal chromaffin granules and chromaffin cells: evidence for a dynamic role in the secretory process. Cell Tissue Res. 1992;269(2):323–30.
Luo D, Fajol A, Umbach AT, Noegel AA, Laufer S, Lang F, Föller M. Influence of annexin A7 on insulin sensitivity of cellular glucose uptake. Pflugers Arch. 2015;467(4):641–9.
Roa J, Tena-Sempere M. Energy balance and puberty onset: emerging role of central mTOR signaling. Trends Endocrinol Metab. 2010;21(9):519–28.
Díaz I, Calderón-Sánchez E, Toro RD, Ávila-Médina J, de Rojas-de Pedro ES, Domínguez-Rodríguez A, Rosado JA, Hmadcha A, Ordóñez A, Smani T. miR-125a, miR-139 and miR-324 contribute to Urocortin protection against myocardial ischemia-reperfusion injury. Sci Rep. 2017;7(1):8898.
Liu Y, Chen Y, Zhou Z, He X, Tao L, Jiang Y, Lan R, Hong Q, Chu M. Chi-mir-324-3p regulates Goat Granulosa Cell Proliferation by Targeting DENND1A. Front Vet Sci. 2021;8:732440.
Moeller-Gnangra H, Ernst J, Pfeifer M, Heger S. ErbB4 point mutation in CU3 inbred rats affects gonadotropin-releasing-hormone neuronal function via compromised neuregulin-stimulated prostaglandin E2 release from astrocytes. Glia. 2019;67(2):309–20.
Hou J, Li B, Yang Z, Fager N, Ma MY. Functional integrity of ErbB-4/-2 tyrosine kinase receptor complex in the hypothalamus is required for maintaining normal reproduction in young adult female rats. Endocrinology. 2002;143(5):1901–12.
Prevot V, Rio C, Cho GJ, Lomniczi A, Heger S, Neville CM, Rosenthal NA, Ojeda SR, Corfas G. Normal female sexual development requires neuregulin-erbb receptor signaling in hypothalamic astrocytes. J Neurosci. 2003;23(1):230–9.
Chang RM, Yang H, Fang F, Xu JF, Yang LY. MicroRNA-331-3p promotes proliferation and metastasis of hepatocellular carcinoma by targeting PH domain and leucine-rich repeat protein phosphatase. Hepatology. 2014;60(4):1251–63.
Hosako H, Martin GS, Barrier M, Chen YA, Ivanov IV, Mirkes PE. Gene and microRNA expression in p53-deficient day 8.5 mouse embryos. Birth Defects Res Clin Mol Teratol. 2009;85(6):546–55.
Chen T, Cui J, Ma L, Zeng Y, Chen W. The Effect of MicroRNA-331-3p on Preadipocytes Proliferation and Differentiation and Fatty Acid Accumulation in Laiwu Pigs. Biomed Res Int 2019, 2019:9287804.
Laurent AC, Breckler M, Berthouze M, Lezoualc’h F. Role of Epac in brain and heart. Biochem Soc Trans. 2012;40(1):51–7.
Banerjee U, Cheng X. Exchange protein directly activated by cAMP encoded by the mammalian rapgef3 gene: structure, function and therapeutics. Gene. 2015;570(2):157–67.
Liu X, Song L, Ma X, Liu Y, Huang H, Xu Y, Yan W. Overexpression of RAPGEF3 enhances the therapeutic effect of dezocine in treatment of neuropathic pain. Genet Mol Biol. 2021;44(4):e20200463.
Gao ZZ, Li YC, Shao CY, Xiao J, Shen Y, Zhou L. EPAC negatively regulates myelination via Controlling Proliferation of Oligodendrocyte Precursor cells. Neurosci Bull. 2020;36(6):639–48.
Lee K. Epac: new emerging cAMP-binding protein. BMB Rep. 2021;54(3):149–56.
Reverte-Salisa L, Siddig S, Hildebrand S, Yao X, Zurkovic J, Jaeckstein MY, Heeren J, Lezoualc’h F, Krahmer N, Pfeifer A. EPAC1 enhances brown fat growth and beige adipogenesis. Nat Cell Biol. 2024;26(1):113–23.
Frattarelli JL, Krsmanovic LZ, Catt KJ. The relationship between pulsatile GnRH secretion and cAMP production in immortalized GnRH neurons. Am J Physiol Endocrinol Metab. 2011;300(6):E1022–1030.
Krsmanovic LZ, Hu L, Leung PK, Feng H, Catt KJ. Pulsatile GnRH secretion: roles of G protein-coupled receptors, second messengers and ion channels. Mol Cell Endocrinol. 2010;314(2):158–63.
Funding
This project was financed by the Project of Improved Agricultural Varieties in Shandong Province (2021LZGC010), The National key R & D plan (Integration and Demonstration of Key Technologies for Large-scale, Efficient and Safe Breeding of Goats) (2018YFD0501906), Natural Science Foundation project of Shandong Province (ZR2023QC044), Shandong Provincial Sheep and Goat Industry Technology System (SDAIT-10-01), Shandong “Double Tops” Program (SYL2017YSTD12), and The Project of Improved Agricultural Varieties in Shandong Province (2019LZGC012).
Author information
Authors and Affiliations
Contributions
Q.L.: Conceptualization; Data curation; Formal analysis; Supervision; Validation; Visualization; Writing - original draft; and Writing - review & editing. T.C.: Supervision; Validation; Visualization, and Writing - review & editing. Y.W.: Software. R.X.: Validation; Visualization. YG: Validation. P.H.: Software. L.Z.: Visualization. J.W.: Funding acquisition; Investigation; Methodology; Project administration; and Writing - review & editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Animal Care and Use Committee of Shandong Agricultural University ethics committee (SDAUA-2023-157), and all methods are reported in accordance with ARRIVE guidelines (https://arriveguidelines.org).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Q., Chao, T., Wang, Y. et al. Transcriptome analysis reveals miRNA expression profiles in hypothalamus tissues during the sexual development of Jining grey goats. BMC Genomics 25, 832 (2024). https://doi.org/10.1186/s12864-024-10735-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12864-024-10735-y