Abstract
Background
In the general population, the epidemiological relationships between delirium and adverse outcomes are not well defined. The aims of this study were to: (1) construct an algorithm for the diagnosis of delirium using the Geriatric Mental State (GMS) examination; (2) test the criterion validity of this algorithm against mortality and dementia risk; (3) report the age-specific prevalence of delirium as determined by this algorithm.
Methods
Participant and informant data in a randomly weighted subsample of the Cognitive Function and Ageing Study were taken from a standardized assessment battery. The algorithmic definition of delirium was based on the DSM-IV classification. Outcomes were: proportional hazard ratios for death; odds ratios of dementia at 2-year follow-up.
Results
Data from 2197 persons (representative of 13,004) were used, median age 77 years, 64% women. Study-defined delirium was associated with a new dementia diagnosis at two years (OR 8.82, 95% CI 2.76 to 28.2) and death (HR 1.28, 95% CI 1.03 to 1.60), even after adjustment for acute illness severity. Similar associations were seen for study-defined subsyndromal delirium. Age-specific prevalence as determined by the algorithm increased with age from 1.8% in the 65-69 year age group to 10.1% in the ≥85 age group (p < 0.01 for trend). For study-defined subsyndromal delirium, age-specific period prevalence ranged from 8.2% (65-69 years) to 36.1% (≥85 years).
Conclusions
These results demonstrate the possibility of constructing an algorithmic diagnosis for study-defined delirium using data from the GMS schedule, with predictive criterion validity for mortality and dementia risk. These are the first population-based analyses able to account prospectively for both illness severity and an earlier study diagnosis of dementia.
Similar content being viewed by others
Background
Delirium is a serious neuropsychiatric syndrome presenting with inattention and global changes in cognition [1–3]. Delirium arises as a consequence of a neurological or systemic illness, medications and psychological stress. It is well-recognized that there is a relationship between predisposing (ageing, cognitive impairment) and precipitating (illness severity) factors such that in the setting of multiple (or severe) predisposing factors, fewer (or less severe) precipitating factors are required [4]. Delirium is therefore a sensitive marker of acute illness in vulnerable older people. This association with acute illness has resulted in the vast majority of delirium studies being undertaken in hospital cohorts [5]. However, this introduces selection biases as not all persons with delirium may reach medical attention. In addition, comparisons to pre-morbid cognitive functions are difficult.
In hospital samples, a common finding is that delirium contributes to persistent cognitive deficits, independently of predisposing and precipitating factors [6]. This has also been reported for subsyndromal delirium, where individuals have one or more of the diagnostic features of delirium [7]. Indeed, any examination of the utility of a delirium definition should incorporate criterion validity tests for future dementia. In prospective community cohort studies, hospitalization predicts adverse cognitive outcomes [8–10], though none has been able to specify if delirium is a key determinant. Delirium is also associated with increased mortality [11], and this should be another criterion by which any definition of delirium should be validated.
The point-prevalence of delirium in the community is thought to be low (0.7%, 95% CI 0.5 to 1.0 in the population aged ≥60 years), though this understanding is based on a systematic review identifying only three prevalence estimates in population samples [12]. Furthermore, epidemiological studies may under-estimate acute illness and/or prevalent delirium because people who are unwell are less likely to be interviewed. However, the period-prevalence may be higher. The Gerontological Regional Database (GERDA) study reported that 27% of persons aged 85 and older in the general population with delirium in the previous month [13]. This suggests that whole population samples could potentially investigate delirium more efficiently if stratified subsamples at higher risk for cognitive impairment are more intensively studied.
Delirium is clinically defined by application of a psychiatric reference standard such as the Diagnostic and Statistical Manual (DSM), where the core features are inattention, altered consciousness, cognitive and/or perceptual disturbance, acute and fluctuating change, in relation to a general medical condition. Based on this, there is an opportunity to construct an algorithmic diagnosis for delirium in population-based cohort studies collecting psychiatric symptoms. Such an approach is well-established in dementia, but yet to be systematically applied in delirium, and particularly not in population studies. Accordingly, using data from the population-based Medical Research Council (MRC) Cognitive Function and Ageing Study (CFAS) the aims of this study were to: (1) construct an algorithm for the diagnosis of delirium in population-based studies using the Geriatric Mental State (GMS) examination based on clinical principles; (2) test the predictive criterion validity of this algorithm against mortality and dementia risk; (3) report the age-specific prevalence of delirium as determined by this algorithm.
Methods
Population
Data from the MRC Cognitive Function and Ageing Study (CFAS) were used in this secondary analysis. The principal methods for CFAS have previously been reported [14]. In brief, CFAS was a multi-center study, with sampling from four urban (Newcastle, Nottingham, Oxford and Liverpool), and two rural areas (Cambridgeshire and Gwynedd) in the UK. The present report only concerns the five identical sites (excluding Liverpool). Each center had ethical approval from the Local Research Ethics Committee (Cambridge: North West Anglia Health Authority Local Research Ethics Committee (Peterborough); Huntingdon Local Research Ethics Committee; Cambridge Local Research Ethics Committee. Gwynedd: Gwynedd Hospitals NHS Trust – NWHA Research Ethics Committee (West). Newcastle: Newcastle & North Tyneside Health Authority – Joint Ethics Committee; Northumberland and Tyne & Wear Health Authority – Local Research Ethics Committee. Nottingham: QMC NHS Trust Ethics Committee; Nottingham University Medical School Ethical Committee; City Hospital Ethics Committee. Oxford: Oxfordshire Health Authority: Central Oxford Research Ethics Committee). Family Health Service Authority lists were used as the sampling frame within a defined geographical area, and this specifically included people resident in institutions. Each individual gave written consent to participate in the study.
Figure 1 shows the two stage sampling process for case ascertainment. A screening examination was started in 1991 (“Screen”, n = 13004)a. Then, a stratified sample consisted of approximately 20% was selected using center, age (equal numbers aged 65–74 and ≥75), and cognitive ability (weighted toward the more cognitively impaired, based on the screening assessment), and a random subsample from the remaining 80% (“Ascertain”, n = 2640) (mean interval between “Screen” and “Ascertain” was 3 months). Interviews of participants’ nearest informants were also undertaken (“Informant”, n = 2159) (mean interval between “Ascertain” and “Informant” was 3 weeks). Participants were followed at two years, with further subsets examined more frequently, and the whole cohort was re-examined at 6 and 10 years. The number of participants at baseline and at the first two-year follow-up is shown in Figure 1. Mortality outcomes were notified through reports linked to the UK Office of National Statistics.
Interviews
Interviews were carried out in participants’ usual residence (including if in a care home) by trained interviewers. At screening, information on socio-demographic, physical and behavioral status was collected in addition to health (including self-reported chronic conditions) and cognitive function, assessed using the MMSE. The assessment interview was based on the GMS [15], which was designed to be a structured schedule amenable to administration by non-clinicians. The GMS comprises measures assessing psychiatric symptoms of organicity, depression, anxiety and psychosis, with ratings for each derived using the AGECAT algorithm. The study diagnosis of dementia was based on the GMS B3 AGECAT algorithmic differential diagnosis, where the dementia component is organicity at case level 03 and above. This approach has been validated against formal clinical diagnoses based on DSM-III-R [16]. All information was based on self-report. Informant proxies were also interviewed in a standardized manner using a set of questions complementary to the GMS known as the History and Aetiology Schedule (HAS). Informants were asked questions covering psychiatric symptom clusters occurring in ‘recent weeks and months’. Each interviewer for “Follow-up” undertook assessments blinded to data acquired in the baseline phase.
Questions from “Ascertain” and “Informant” (Figure 1) pertaining to delirium symptoms are shown in Table 1. These were used to define an algorithm for a study definition of delirium based on DSM-IV, where participants were required to demonstrate all three of: (i) acute change; (ii) fluctuation; (iii) inattention and/or drowsiness (Table 2). Subsyndromal delirium was defined as having at least one of these features. In addition, interviewers were asked to judge if responses were affected by their subjective rating of any current acute illness in the participants (categorized as: none, mild, moderate, or severe).
Statistical analyses
The criterion validity of the study-defined delirium algorithm was tested in two ways: (i) hazard for mortality and (ii) odds of a new diagnosis of dementia at two year follow-up. The association between study-defined delirium and mortality was evaluated using Cox proportional hazards models, adjusted by age, sex and prevalent dementia. The association between study-defined delirium and dementia was assessed using logistic regression where the outcome was new dementia at two-year follow-up (“Follow-up”, Figure 1) in the sample known to be dementia-free at baseline, adjusted by age and sex (“Ascertain”, Figure 1). Each delirium symptom was tested for both outcomes, as well as the overall algorithmic diagnosis. Testing the criterion validity of the algorithmic diagnosis also adjusted for interviewers’ rating of current acute illness severity. Post-estimation tests for underlying assumptions included Hosmer-Lemershow goodness-of-fit and Schoenfeld residuals for logistic and proportional hazards models respectively.
Results
The subsample selected for this analysis included 2197 individuals assessed by both the GMS (participant) and HAS (informant) schedules at the assessment interview (“Ascertain”). Median age was 77 (interquartile range 71-84), and 1403 (64%) were women. In this weighted subsample of the whole baseline cohort, 511 (23%) had prevalent dementia. Table 1 lists the delirium symptom clusters and questions used to ascertain these, along with the prevalence of individual symptoms. Table 3 reports the age and sex characteristics of the sample, number of persons with study-defined delirium and dementia, and incident dementia and mortality at two years.
Table 4 shows the results of the Cox proportional hazards survival analyses, adjusted by age, sex and prevalent dementia. In this weighted subsample, each delirium symptom was independently associated with higher mortality. This was also the case for the algorithmic diagnosis, even after adjustment for acute illness severity (HR 1.28, 95% CI 1.03 to 1.60). A similar risk for study-defined subsyndromal delirium was apparent (HR 1.41, 95% CI 1.23 to 1.62).
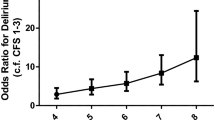
Table 5 gives the results of the logistic regression analyses assessing the odds of a dementia diagnosis at two year follow-up, adjusted by age and sex. In this weighted subsample, all delirium symptoms were associated with odds ratios greater than 1.0, but this was only statistically significant for acute change, fluctuation and drowsiness. The algorithmic diagnosis was significantly associated with a two year dementia diagnosis (OR 8.82, 95% CI 2.76 to 28.2). The estimate for study-defined subsyndromal delirium was half that of full syndromal delirium (OR 4.31, 95% CI 2.41 to 7.73).The estimated age-specific period prevalence of the algorithmic diagnosis of delirium is given in Figure 2. The overall period prevalence in this subsample of enriched for cognitive impairment is estimated at 5.6% (95% CI 4.6 to 6.5). Age-specific prevalence increases with age from 1.8% in the 65-69 year age group to 10.1% in the ≥85 age group (p < 0.01 for trend). For study-defined subsyndromal delirium, age-specific period prevalence ranged from 8.2% (65-69 years) to 36.1% (≥85 years). In persons with prevalent dementia, 16.8% (95% CI 13.6 to 20.1%) had superimposed delirium.
Prevalence of study-defined delirium and subsyndromal delirium, by age group. Bar chart showing estimated age-specific prevalence of the algorithm diagnosis of delirium (grey) and subsyndromal delirium (dark grey) as a proportion of the assessed subsample. Upper and lower bars show 95% confidence intervals.
Discussion
These results demonstrate the possibility of constructing an algorithmic diagnosis for delirium within a population-based framework using data from the GMS schedule including self and informant reported responses. This algorithm has criterion validity for mortality and dementia risk. These are the first population-based analyses able to account for both illness severity and prevalent dementia, suggesting that our study-defined delirium has a deleterious effect on mortality and dementia risk beyond that expected from precipitating and predisposing factors alone. These findings also highlight the importance of age in the prevalence of delirium with the highest prevalence in the oldest-old group (i.e. ≥85 years).
The strengths of this study derive from its large population-based sample size and availability of serial cognitive assessments in relation to incident dementia. The major limitation is that the algorithm was not validated with concurrent clinical diagnosis of delirium. Furthermore, the period over which informants were asked to comment on delirium symptoms was not strictly defined (‘in recent weeks and months’) and may be overstated due to recall bias. The lack of assessments by clinicians also limits the precision of the data. Though dementia as a binary predisposing factor in delirium is a well-established construct, delirium could be associated with mild or minimal dementia-type states not captured by the AGECAT diagnosis. Analyses taking the continuum of cognition into account would address this concern to some extent, though there is real difficulty in disentangling such closely associated phenomena, both in clinical and epidemiological studies. Nonetheless, this does not detract from the prognostic significance of delirium symptoms for future dementia. As with any observational study, both over-adjustment and residual confounding remain considerations. It is recognized that the effect of age on total dementia risk is so large that it overshadows many of other associations under investigation (particularly modifiable risk factors) [17, 18]. This has been specifically observed in previous CFAS analyses [19, 20], and therefore it is not clear which co-morbidities, if any, should also be included in dementia risk models. Finally, though the CFAS sample was population-representative in 1991, the age-specific prevalence of dementia is lower in 2011 [21] and so secular trends may constrain the accuracy of the delirium prevalence estimates.
The estimated age-specific population prevalence of study-defined delirium is lower than the only other estimate available (from GERDA), even though diagnoses included information from community medical records [13]. Previously, the only population-based cohort to have assessed a delirium measure in relation to adverse outcomes is the Vantaa 85+ study [22, 23]. In Vantaa 85+, delirium history was assessed at each interview by a neurologist with access to an informant and medical records, amounting to an estimate of period prevalence for the intervening 2-3 years between waves. The present analysis is four times bigger (CFAS = 2197 versus Vantaa = 553). Though medical records were not available here, the advantage in CFAS is the possibility of accounting for illness severity, even though this assessment was subjective. The point estimates for mortality (CFAS HR 1.55 (when unadjusted by illness severity) versus Vantaa HR 1.61) and two-year dementia risk (CFAS OR 8.82 versus Vantaa OR 8.65) are effectively the same. Though delirium diagnoses were derived through different approaches, this suggests the core features of inattention, altered arousal (here characterized as ‘drowsiness’) and acute fluctuations in cognitive function represent an adverse state for future outcomes regardless of the exact methods for operationalizing the syndrome.
Conclusions
These data add to the small literature on the population-based epidemiology of delirium. Delirium, as defined in this study, is associated with increased dementia, strengthening the argument that interventions for delirium may have an impact on the burden of cognitive impairment. Nonetheless, the core elements of the delirium-dementia relationship still require further exploration (e.g. if the key factors responsible for this association relate to etiology, duration, severity or some combination), particularly in the general population and in other cohort studies [18]. At the least, these findings indicate that it is possible to identify population samples with delirium and subsyndromal delirium at higher risk for dementia.
Endnote
aIn other CFAS publications, the stages: S0; A0; H0; C2 are described here as “Screen”; “Ascertainment”; “Informant”; “Follow-up” respectively.
References
MacLullich AMJ, Hall RJ: Who understands delirium?. Age Ageing. 2011, 40 (4): 412-414.
Maclullich AM, Anand A, Davis DH, Jackson T, Barugh AJ, Hall RJ, Ferguson KJ, Meagher DJ, Cunningham C: New horizons in the pathogenesis, assessment and management of delirium. Age Ageing. 2013, 42 (6): 667-674.
Inouye SK, Westendorp RG, Saczynski JS: Delirium in elderly people. Lancet. 2014, 383 (9920): 911-922.
O'Hanlon S, O'Regan N, Maclullich AM, Cullen W, Dunne C, Exton C, Meagher D: J Neurol Neurosurg Psychiatry. 2014, 85 (2): 207-213.
Khan BA, Zawahiri M, Campbell NL, Fox GC, Weinstein EJ, Nazir A, Farber MO, Buckley JD, Maclullich A, Boustani MA: Delirium in hospitalized patients: implications of current evidence on clinical practice and future avenues for research-A systematic evidence review. J Hosp Med. 2012, 7 (7): 580-589.
Witlox J, Eurelings LSM, De Jonghe JFM, Kalisvaart KJ, Eikelenboom P, Van Gool WA: Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: A meta-analysis. JAMA. 2010, 304 (4): 443-451.
Cole M, McCusker J, Dendukuri N, Han L: The prognostic significance of subsyndromal delirium in elderly medical inpatients. J Am Geriatr Soc. 2003, 51 (6): 754-760.
Iwashyna TJ, Ely EW, Smith DM, Langa KM: Long-term cognitive impairment and functional disability among survivors of severe sepsis. JAMA. 2010, 304 (16): 1787-1794.
Ehlenbach WJ, Hough CL, Crane PK, Haneuse SJ, Carson SS, Curtis JR, Larson EB: Association between acute care and critical illness hospitalization and cognitive function in older adults. JAMA. 2010, 303 (8): 763-770.
Wilson RS, Hebert LE, Scherr PA, Dong X, Leurgens SE, Evans DA: Cognitive decline after hospitalization in a community population of older persons. Neurology. 2012, 78 (13): 950-956.
Siddiqi N, House AO, Holmes JD: Occurrence and outcome of delirium in medical in-patients: A systematic literature review. Age Ageing. 2006, 35 (4): 350-364.
Davis DH, Kreisel SH, Muniz Terrera G, Hall AJ, Morandi A, Boustani M, Neufeld KJ, Lee HB, Maclullich AM, Brayne C: The epidemiology of delirium: challenges and opportunities for population studies. Am J Geriatr Psychiatry. 2013, 21 (12): 1173-1189.
Eriksson I, Gustafson Y, Fagerstrom L, Olofsson B: Urinary tract infection in very old women is associated with delirium. Int Psychogeriatr. 2011, 23 (3): 496-502.
Brayne C, McCracken C, Matthews FE: Cohort profile: the Medical Research Council Cognitive Function and Ageing Study (CFAS). Int J Epidemiol. 2006, 35 (5): 1140-1145.
Copeland JR, Dewey ME, Griffiths-Jones HM: A computerized psychiatric diagnostic system and case nomenclature for elderly subjects: GMS and AGECAT. Psychol Med. 1986, 16 (1): 89-99.
Kay DW, Dewey ME, McKeith IG, O'Cuill M, McCracken C, Fairbairn AF, Harrison R, Illing JC, Hrisos S: Do experienced diagnosticians agree about the diagnosis of dementia from survey data? The effects of informants’ reports and interviewers’ vignettes. Int J Geriatr Psychiatry. 1998, 13 (12): 852-862.
Daviglus ML, Plassman BL, Pirzada A, Bell CC, Bowen PE, Burke JR, Connolly ES, Dunbar-Jacob JM, Granieri EC, McGarry K, Patel D, Trevisan M, Williams JW: Risk factors and preventive interventions for Alzheimer disease: state of the science. Arch Neurol. 2011, 68 (9): 1185-1190.
Brayne C, Davis D: Making Alzheimer's and dementia research fit for populations. Lancet. 2012, 380 (9851): 1441-1443.
Yip AG, Brayne C, Matthews FE: Risk factors for incident dementia in England and Wales: The Medical Research Council Cognitive Function and Ageing Study. A population-based nested case-control study. Age Ageing. 2006, 35 (2): 154-160.
Matthews F, Brayne C: The incidence of dementia in England and Wales: findings from the five identical sites of the MRC CFA Study. PLoS Med. 2005, 2 (8): e193-
Matthews FE, Arthur A, Barnes LE, Bond J, Jagger C, Robinson L, Brayne C: A two-decade comparison of prevalence of dementia in individuals aged 65 years and older from three geographical areas of England: results of the Cognitive Function and Ageing Study I and II. Lancet. 2013, 382 (9902): 1405-1412.
Rahkonen T, Eloniemi-Sulkava U, Halonen P, Verkkoniemi A, Niinisto L, Notkola IL, Sulkava R: Delirium in the non-demented oldest old in the general population: risk factors and prognosis. Int J Geriatr Psychiatry. 2001, 16 (4): 415-421.
Davis DH, Muniz Terrera G, Keage H, Rahkonen T, Oinas M, Matthews FE, Cunningham C, Polvikoski T, Sulkava R, Maclullich AM, Brayne C: Delirium is a strong risk factor for dementia in the oldest-old: a population-based cohort study. Brain. 2012, 135 (9): 2809-2816.
Pre-publication history
The pre-publication history for this paper can be accessed here:http://www.biomedcentral.com/1471-2318/14/87/prepub
Acknowledgements and funding
We are indebted to the local general practitioners and their staff for the support and assistance. We warmly thank the interviewers for their efforts, and volunteers who allowed us to practice on them. Thanks are especially due to the residents of East Cambridgeshire, Liverpool, Ynys Mon, Dwyfor, Newcastle upon Tyne, Nottingham and Oxford for their continuing participation in the study.
The MRC CFA Study is supported by major awards from the Medical Research Council (grant number G9901400) and the Department of Health. DD is supported by the Wellcome Trust as a research training fellow (WT090661).
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
DD and JC devised the algorithm. DD undertook the statistical analysis with assistance from BS and FM. LB conducted and co-ordinated the fieldwork for CFAS. AM and DM provided expertise on syndromal and subsyndromal delirium phenomenology. CB contributed to the conception and design of the study, and is a principal investigator on CFAS, along with FM. All authors read and approved the final manuscript.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Davis, D.H., Barnes, L.E., Stephan, B.C. et al. The descriptive epidemiology of delirium symptoms in a large population-based cohort study: results from the Medical Research Council Cognitive Function and Ageing Study (MRC CFAS). BMC Geriatr 14, 87 (2014). https://doi.org/10.1186/1471-2318-14-87
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1471-2318-14-87