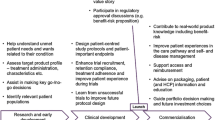
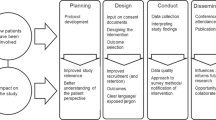
Abstract
Objective
To identify the elements necessary for successful collaboration between patient groups and academic and industry sponsors of clinical trials, in order to develop recommendations for best practices for effective patient group engagement.
Methods
In-depth interviews, informed by a previously reported survey, were conducted to identify the fundamentals of successful patient group engagement. Thirty-two respondents from 3 sectors participated: patient groups, academic researchers, and industry. The findings were presented to a multistakeholder group of experts in January 2015. The expert group came to consensus on a set of actionable recommendations for best practices for patient groups and research sponsors.
Results
Interview respondents acknowledged that not all patient groups are created equal in terms of what they can contribute to a clinical trial. The most important elements for effective patient group engagement include establishing meaningful partnerships, demonstrating mutual benefits, and collaborating as partners from the planning stage forward. Although there is a growing appreciation by sponsors about the benefits of patient group engagement, there remains some resistance and some uncertainty about how best to engage. Barriers include mismatched expectations and a perception that patient groups lack scientific sophistication and that “wishful thinking” may cloud their recommendations.
Conclusions
Patient groups are developing diverse skillsets and acquiring assets to leverage in order to become collaborators with industry and academia on clinical trials. Growing numbers of research sponsors across the clinical trials enterprise are recognizing the benefits of continuous and meaningful patient group engagement, but there are still mindsets to change, and stakeholders need further guidance on operationalizing a new model of clinical trial conduct.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
US Food and Drug Administration. Patient-focused drug development. http://www.fda.gov/ForIndustry/UserFees/PrescriptionDrugUserFee/ucm347317.htm. Accessed June 17, 2015.
Albarracin D, Gillette JC, Earl AN, Glasman LR, Durantini MR, Ho MH. A test of major assumptions about behavior change: a comprehensive look at the effects of passive and active HIV-prevention interventions since the beginning of the epidemic. Psychol Bull. 2005;131:856–897.
Parsons S, Starling B, Mullan-Jensen C, Tham S-G, Warner K, Wever K. What the public knows and wants to know about medicines research and development: a survey of the general public in six European countries. BMJ Open. 2015;5.
Pushparajah DS, Geissler J, Westergaard N. EUPATI: collaboration between patients, academia and industry to champion the informed patient in the research and development of medicines. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=0ahUKEwifr5TQkZjUAhXGKCYKHaBjA1UQFggkMAA&url=https%3A%2F%2Fwww.eupati.eu%2Fwp-content%2Fuploads%2F2016%2F09%2F2015-07-Journal-Med-Dev-Sciences-EUPATI-Collaboration-between-patients-academia-industry_EN.pdf&usg=AFQjCNGlYRt2IlhBs758wOmeL-N5974yYg&sig2=hIwnuY4eKDavpzP0ncA7kg&cad=rja. Accessed May 30, 2017.
Hoos A, Anderson J, Boutin M, et al. Partnering with patients in the development and lifecycle of medicines. Therapeutic Innovation & Regulatory Science. 2015;49:929–939.
Pavitt S. EUPATI: an initiative to provide expertise in patient advocacy and in medicines development processes. https://www.eupati.eu/wp-content/uploads/2016/09/2013-09-Regulatory-Rapporteur-September-EUPATI_EN.pdf. Accessed May 30, 2017.
Clinical Trials Transformation Initiative. CTTI Membership. https://www.ctti-clinicaltrials.org/membership. Accessed November 3, 2016.
Smith SK, Selig W, Harker M, et al. Patient engagement practices in clinical research among patient groups, industry, and academia in the United States: a survey. PLoS One. 2015;10:e0140232.
Clinical Trials Transformation Initiative. Patient groups and clinical trials. Meeting materials. http://www.ctti-clinicaltrials.org/what-we-do/ctti-projects/patient-groups/expert-meeting. Accessed November 2, 2016.
Clinical Trials Transformation Initiative. Effective engagement with patient groups around clinical trials. Overview and deliverables. http://www.ctti-clinicaltrials.org/what-we-do/projects/patient-groups/products. Accessed November 2, 2016.
Clinical Trials Transformation Initiative. CTTI recommendations: effective engagement with patient groups around clinical trials. Recommendations document. https://www.ctti-clinicaltrials.org/files/pgctrecs.pdf. Accessed March 28, 2016.
Borup G, Bach KF, Schmiegelow M, Wallach-Kildemoes H, Bjerrum OJ, Westergaard N. A paradigm shift towards patient involvement in medicines development and regulatory science. Therapeutic Innovation & Regulatory Science. 2016;50:304–311.
Parsons S, Starling B, Mullan-Jensen C, Tham SG, Warner K, Wever K. What do pharmaceutical industry professionals in Europe believe about involving patients and the public in research and development of medicines? A qualitative interview study. BMJ Open. 2016;6:e008928.
Levitan B, Getz K, Eisenstein E, et al. Assessing the financial value of patient engagement: a quantitative approach from CTTI’s Patient Groups and Clinical Trials project. Therapeutic Innovation & Regulatory Science. In press.
Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Lancet. 2009;374:86–89.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bloom, D., Beetsch, J., Harker, M. et al. The Rules of Engagement: CTTI Recommendations for Successful Collaborations Between Sponsors and Patient Groups Around Clinical Trials. Ther Innov Regul Sci 52, 206–213 (2018). https://doi.org/10.1177/2168479017720247
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1177/2168479017720247